Abstract
Objectives
To assess the performance of dynamic or 4D CT in characterizing endoleaks in advanced endovascular aortic repair (branched and fenestrated) when other modalities fail to fully characterize the leak, most often conventional CTA.
Methods
Retrospective review of 13 patients from 2008 to 2021 who underwent 16 4D CTs to characterize endoleaks in branched and fenestrated endovascular aortic repair (FB-EVAR). The 4D CTs were performed covering up to 16 cm of the z-axis, with anywhere between 10 and 40 iterations performed every 2 s. These settings were adjusted depending on graft characteristics and type of endoleak suspected. The scans were assessed for their ability to detect the endoleak (sensitivity), and further to characterize the endoleak by type and subtype (specificity).
Results
Overall sensitivity in 16 scans for endoleak detection was 100%. There was a specificity of 87.5% for determining the type of endoleak (14/16). These results included two studies that were inconclusive and repeated due to technical difficulties. In patients where a specific subtype was not established, the leak was localized to the appropriate target vessel. Average dose for the 4D CT was 4724 mGy*cm (1108–11069), with the outlining higher dose scans secondary to higher iterations in those scans.
Conclusions
4D CT is a useful adjunctive tool in FB-EVAR surveillance with excellent sensitivity and specificity in characterizing endoleaks. This allows for accurate localization of leaks, which is critical for management planning.
Introduction
Endoleak classification for fenestrated endovascular aortic repair.

Accurate detection of endoleaks is crucial in determining longer term management strategies; however, characterization of endoleaks is more challenging in fenestrated and branched endovascular aortic repair (FB-EVAR) with multiple potential sources as described above.
Imaging modalities for surveillance after aortic endovascular repair include plain radiography, ultrasound (with or without contrast), CT angiography (classically unenhanced, arterial and delayed phase images), MR angiography, and conventional angiography.5,6 CT angiography is the mainstay of surveillance as images are easily reproducible, and the aorta and branches are very well visualized. 7 Conventional triphasic CT provides high sensitivity and specificity for endoleak detection, approximately 85% and 95%, respectively. 8 Ultrasound is an alternative surveillance option and can be augmented with contrast enhanced ultrasound (CEUS); however, operator dependence, availability, and identifying/interrogating branches/fenestrations can be challenging. Therefore, in circumstances of difficult to detect endoleaks, CEUS can be a good screening tool, but the traditional “gold standard” has been triphasic CT. Conventional angiography can be used, and treatment can be offered in the same session; however, in complex branched/fenestrated cases, interrogating different endoleak sources can be very challenging and often requires different vascular access routes for diverse angles of visceral branches. It is important therefore to establish the specific endoleak location and type to best guide therapy and minimize the risk of therapy-related complications. The timing of contrast enhanced phases in conventional CTA provides only two discrete time points for assessment. Although this is sufficient to detect and characterize many endoleaks, the exact type of early and large leaks may remain ambiguous. Further, the location of endoleak can be nebulous in complex branched/fenestrated repairs with close proximity of the visceral branches to each other. For these reasons, dynamic (or four dimensional) CTA was proposed to provide the ability to follow the contrast bolus in a limited field of view at multiple time points (often a few seconds apart), which has allowed better specificity for endoleak type and location in diagnostically challenging situations. 9 It has been shown to provide excellent specificity in cases of difficult to diagnose endoleaks and even decreased overall radiation and contrast load when re-intervention is eventually performed. 10 To our knowledge, there are no specific studies that have evaluated the performance of 4D CT specifically for complex branched/fenestrated repairs.
The purpose of this study was to summarize our experience with the use of 4D or dynamic CTA for assessing endoleaks, with a specific focus on those performed in complex aortic endovascular repairs where the type and/or source of leak was uncertain using other methods of assessment.
Materials and methods
This retrospective study was approved by the Institutional Review Board at University Health Network. Patients who underwent complex branched and/or fenestrated endovascular repair between 2008 and 2021 were reviewed (264 patients). Cross referencing was performed against a Montage™ database search of radiological reports containing the keywords “4D,” “dynamic,” and “endoleak.” All patients who underwent a 4D CT were included. Patients for whom 4D CT was recommended and not performed, and patients who underwent 4D CT for standard EVAR were excluded.
4D CT technique
Standard dynamic CTA specifications.
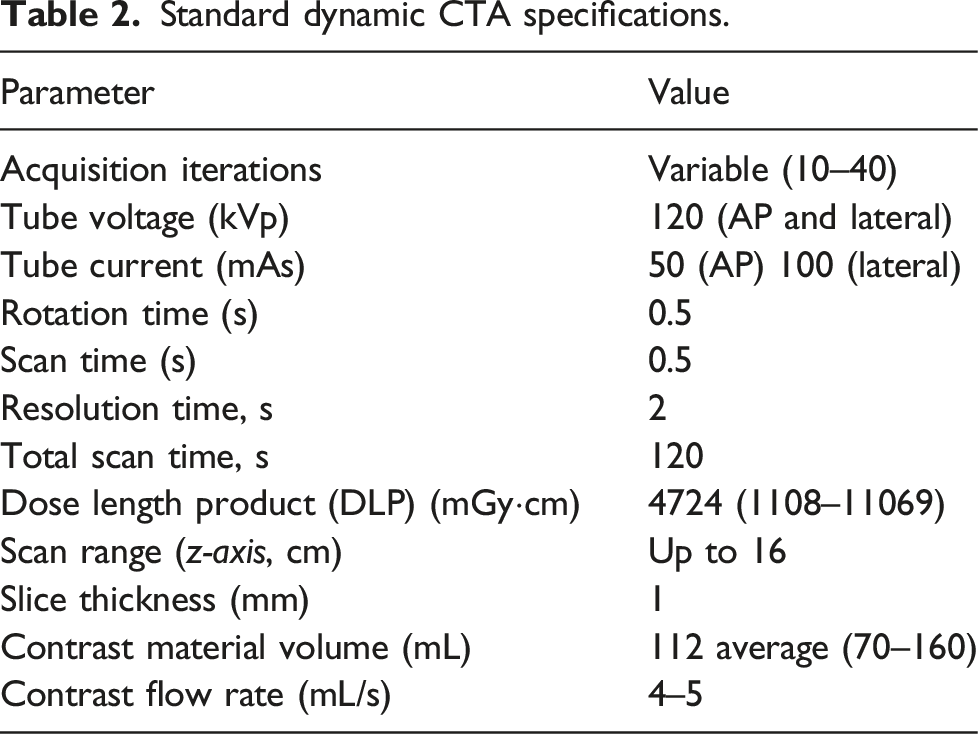
Post-processing was completed on the Aquilion One console and was also transferred to a dedicated post-processing workstation for creation of 4D digital subtraction CTA images as well as 3D and thin-section maximum-intensity projection cine loops.
The images and reports were reviewed using the Coral PACS system (as well as reconstruction algorithms) and the clinical history for the aforementioned patients was reviewed to establish if additional intervention was performed, as well as the outcome.
Results
Patient characteristics.
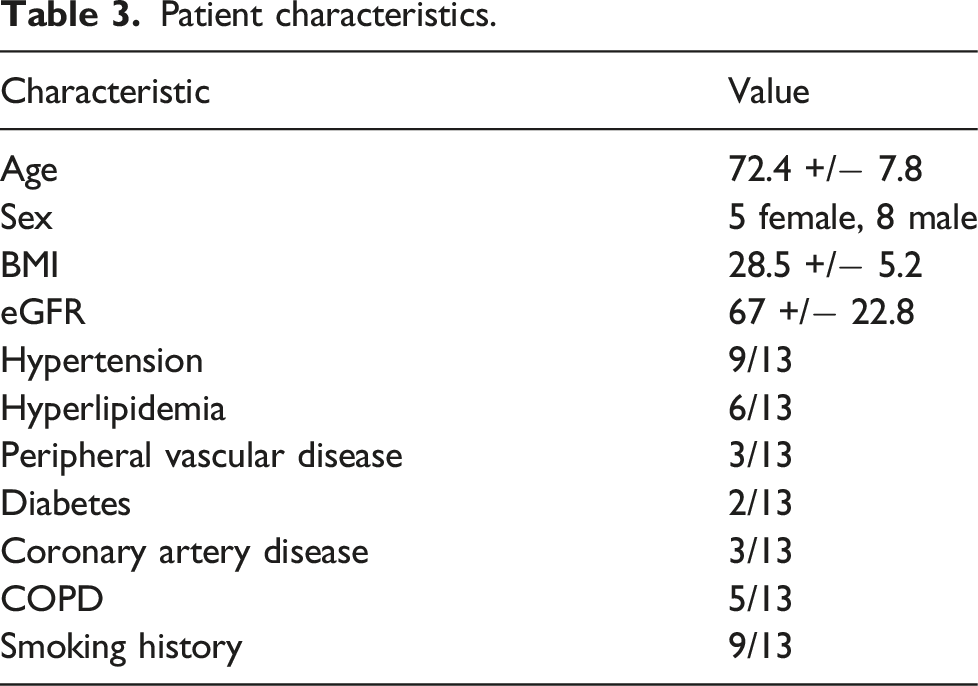
The endovascular aortic repairs performed in these 13 patients had a mean of 1.6 branches and 2.1 fenestrations.
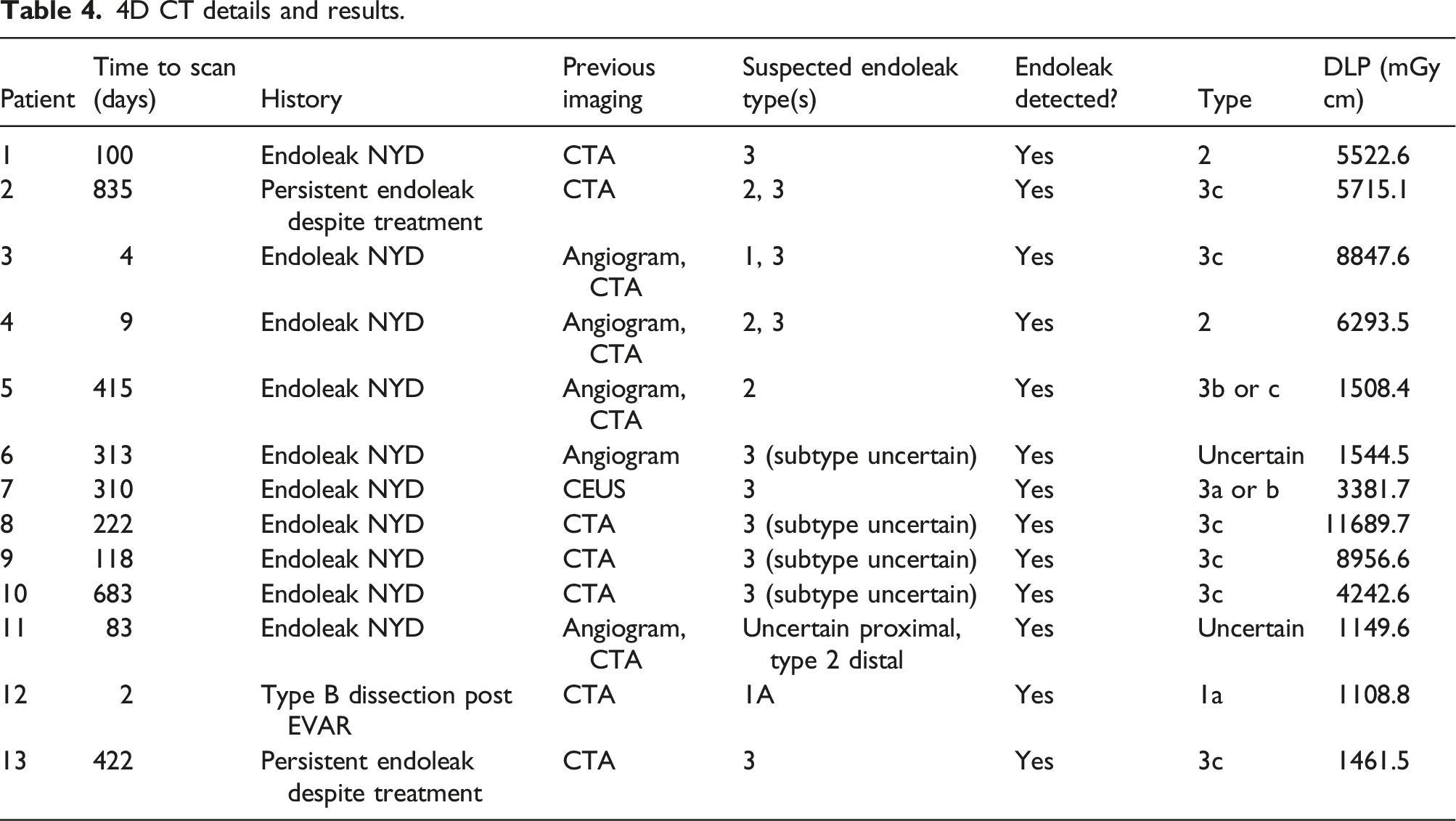
4D CT details and results.
Patients with additional 4D CT.
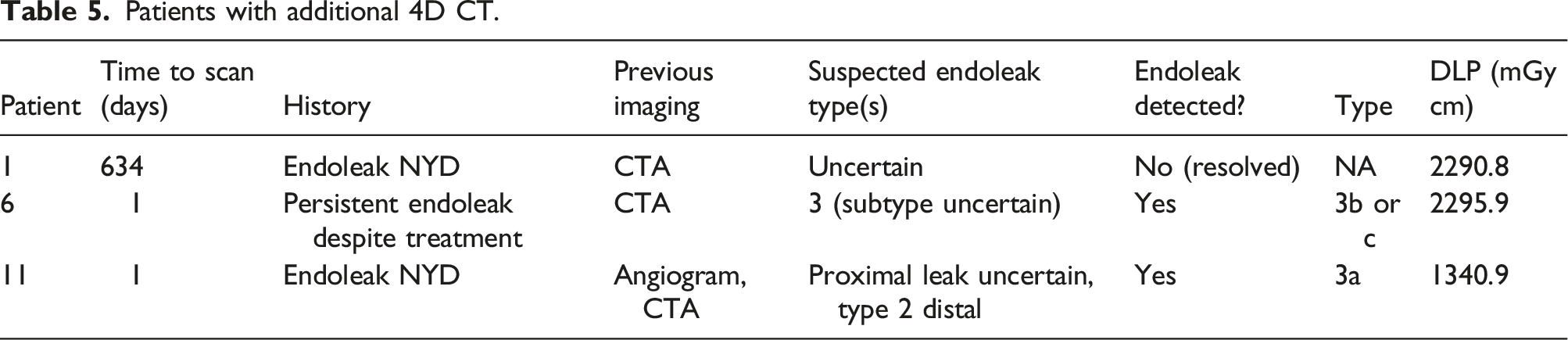
Two patients with additional 4DCT underwent subsequent re-intervention. One patient in this cohort had an endoleak that resolved spontaneously. Therefore, no endoleak was detected in the 4D CT.
Including the two repeated 4D CTs due to technical issues, the overall sensitivity was 100% for detecting endoleak (i.e., the 4D CT was able to confirm the presence of an endoleak) and specificity of 87.5% for determining the specific type of endoleak (14/16). Not all scans of patients with Type 1 and 3 endoleaks (13 scans) were able to establish the correct subtype, 8/13 (61.5%). Despite this, it is important to note that 2 of these unsuccessful scans were those associated with technical difficulties. Also, the remaining 3 scans were able to localize the leak to a specific branch, which effectively guided management.
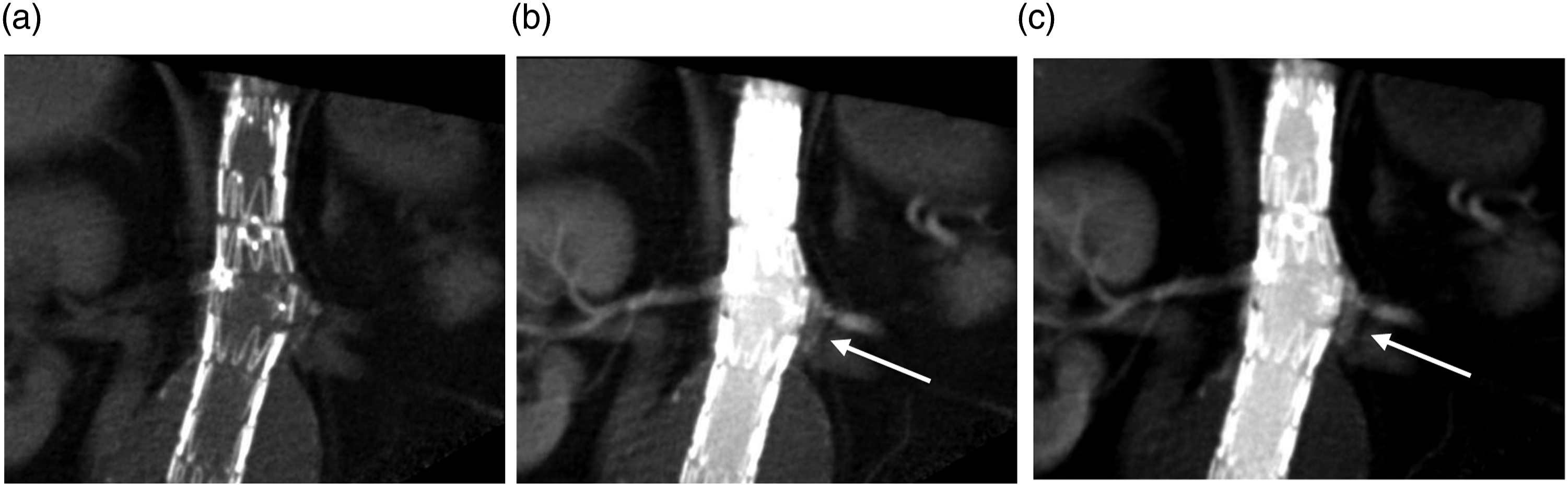
A typical sequence of imaging for a patient with a difficult to diagnose/characterize endoleak can be seen in Figures 1–2. Figure 1 demonstrates conventional CTA for a patient with a fenestrated abdominal aortic endograft (4 fenestrations) with an endoleak of unclear origin. Representative axial arterial phase images demonstrate contrast in the aneurysm sac near visceral vessels anterior to the main graft and below the renal fenestration; however, the exact source of the endoleak is unclear. Figure 2 demonstrates 4D CT images which clearly delineate the flow/progression of contrast directly inferior to the left renal fenestration clearly identifying the Type 3c endoleak. Attached video clips of the 4D CT further illustrate the endoleak. (a) Reconstruction of conventional CTA for 4 fenestrated endograft. (b and c) demonstrate representative axial images of the CTA with endoleak of unclear origin within the anterior and left aneurysm sac (white arrows). 4D CT with unenhanced (a) and early contrast enhanced phases (b and c) demonstrating early contrast enhancement below the left renal stent consistent with a Type 3c leak (white arrows).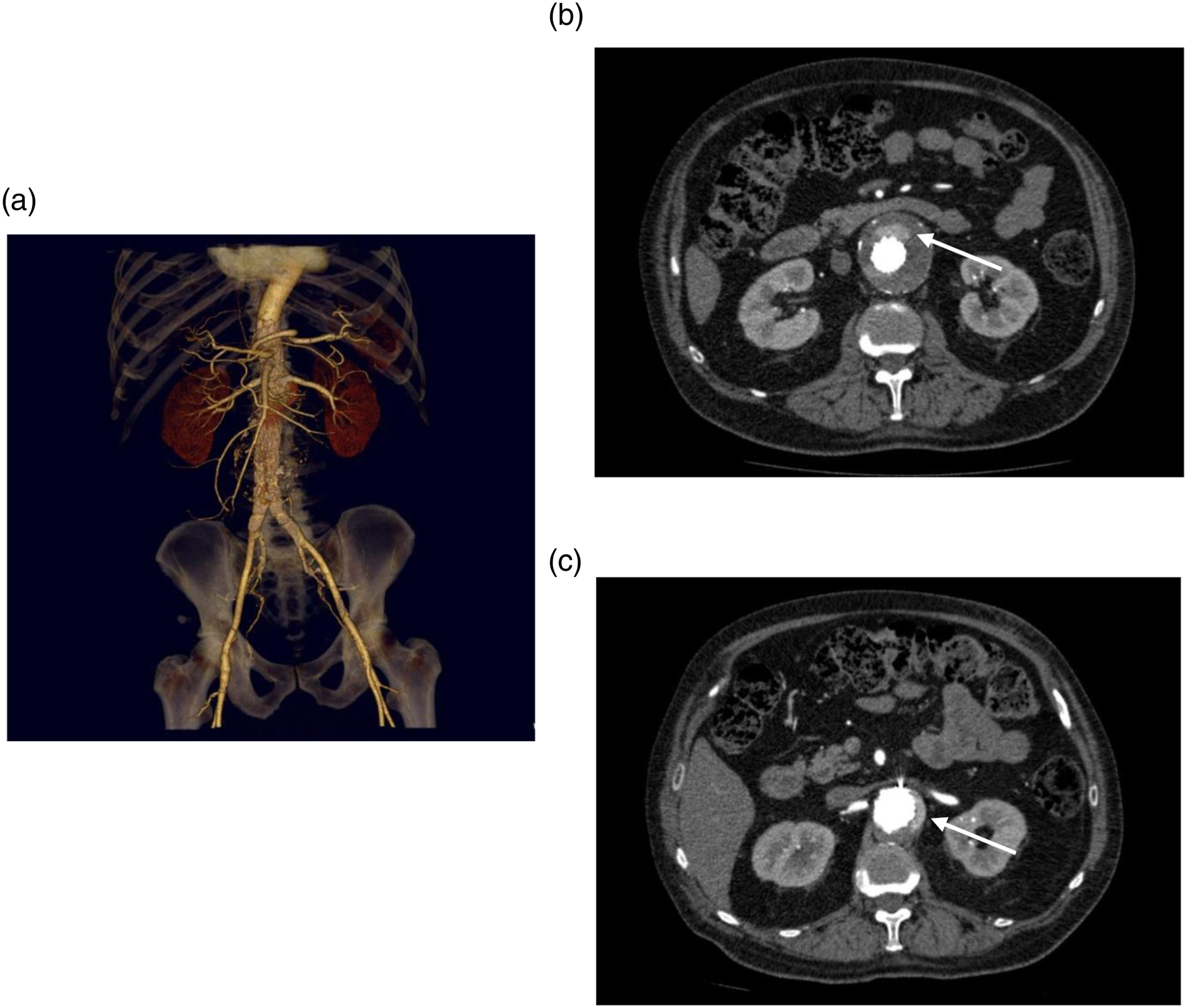
Discussion
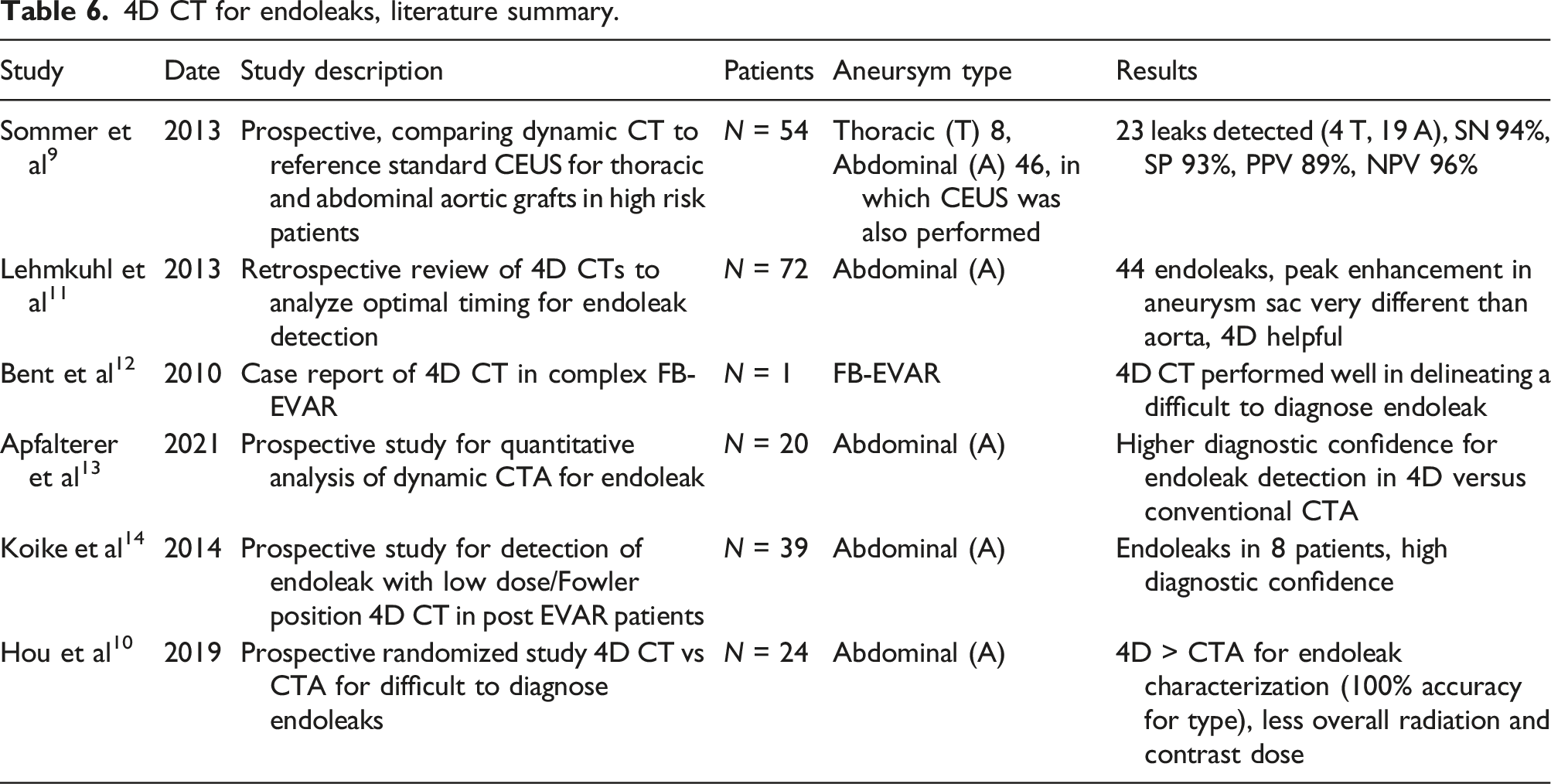
4D CT for endoleaks, literature summary.
Two patients required an additional 4D CT because of technical issues. In one of these patients the visceral branch responsible for the endoleak was not included in the field of view of the 4D CT, therefore the endoleak was detected however and the exact source was not certain. This was rectified with an additional 4D CT in a different field of view and the endoleak was fully characterized. In the second patient, the endoleak was early and the time delay used was too late to detect the early movement of contrast into the aneurysm sac, therefore specific characterization was not possible. This was rectified with an additional 4D CT and the delay from bolus injection to first acquisition was shorter, allowing detection and characterization of the endoleak. These two cases highlight the importance of planning in 4D CT. The z-axis field of view of this modality is limited to 16 cm with a 320 slice scanner. For more extensive branched/fenestrated repairs, coverage of every component of the endoprosthesis may be impossible. Therefore, an index of suspicion to the location of the endoleak is very helpful to guide the appropriate field of view. Appropriate timing of the delay between contrast injection and acquisition is determined after a test bolus, classically with a reference marker placed in the lumen of the graft. If the endoleak is early the contrast may traverse into the aneurysm sac before the lumen of the graft, leading to a time delay that captures the leak relatively late, making the source ambiguous. If an early endoleak is suspected it may be helpful to start intermittent acquisition earlier, although this should be balanced with radiation dose as earlier acquisition would also tend to lead to more iterations and higher radiation dose.
The major drawback of CTA is ionizing radiation, especially considering the need for lifetime surveillance. Conventional CTA is limited to three phases which limits the radiation dose. 4D CT however has considerably more radiation. Also, compared to other studies, radiation doses in our study were higher with significant variation from patient to patient. This is likely due to more iterations in our patient cohort compared to others. Therefore, 4D CT should only be used when necessary and the number of acquisitions performed during a 4D CT should be as few as possible to correctly characterize the endoleak.
Our study is limited to one tertiary care center with a limited number of 4D CTs performed (16 total), despite over 300 branched/fenestrated EVARS performed at this center. This demonstrates efficacy of conventional CTA in the surveillance of endovascular repair. In cases where an endoleak was not fully characterized (or persisted despite repair), 4D CT provided excellent sensitivity and specificity which was critical in planning for re-intervention.
Conclusion
4D CT is a useful adjunctive imaging modality for challenging or recurrent endoleaks in patients with complex endovascular aortic repairs. Care should be taken in planning the 4D CT to optimize the timing, reduce the radiation dose, and increase likelihood of identifying the source of the leak which guides further management.
Footnotes
Acknowledgements
To the dedicated collaborative team of vascular surgery and interventional radiology at University Health Network for the significant contribution to complex aortic aneurysm treatment and advancement in endovascular therapies, and medical imaging for the opportunity to customize diagnostic imaging modalities to optimize the detection and characterization of endoleaks, and thus optimize treatment.
Author Contributions
Tarulli, Massimo—Principal investigator, former interventional radiology fellow.Tan, KT—Staff interventional radiologist, involved in complex EVAR research at UHN, provided edits for the article.
Lindsay, Thomas—Staff vascular surgeon, involved in complex EVAR research at UHN, contributed to large dataset for complex EVAR, provided edits for the article.
Nasir, Daniel—Medical student, involved in complex EVAR database, which was used to analyze patients undergoing complex EVAR. Santiago, Sam—Technologist, UHN, involved in protocols and optimization of 4D CT, provided technical details regarding acquisition of 4D studies.
Jaberi, Arash—Staff interventional radiologist, involved in complex EVAR research at UHN, provided edits for the article.
Mafeld, Sebastian—Staff interventional radiologist, principal supervisor for this work, involved in complex EVAR research at UHN, provided edits for the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval from institutional research ethics board for complex EVAR.
