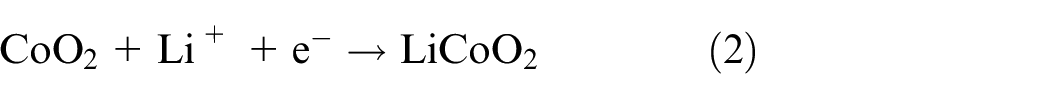Abstract
Lithium-ion batteries have enabled electric vehicles to achieve a foothold in the automobile market. Due to an increasing environmental consciousness, electric vehicles are expected to take a larger portion of the market, with the ultimate goal of supplanting traditional vehicles. However, the involved costs, sustainability, and technical limitations of lithium-ion batteries do create substantial obstacles to this goal. Therefore, this article aims at presenting magnesium-ion batteries as a potential replacement for lithium-ion batteries. Though still under development, magnesium-ion batteries show promise in achieving similar volumetric and specific capacities to lithium-ion batteries. Additionally, magnesium is substantially more abundant than lithium, allowing for the batteries to be cheaper and more sustainable. Numerous technical challenges related to cathode and electrolyte selection are yet to be solved for magnesium-ion batteries. This paper discusses the current state-of-the-art of magnesium-ion batteries with a particular emphasis on the material selection. Although, current research indicates that sulfur-based cathodes coupled with a (HMDS)2Mg-based electrolyte shows substantial promise, other options could allow for a better performing battery. This paper addresses the challenges (materials and costs) and benefits associated with developing these batteries. When overcoming these challenges, magnesium-ion batteries are posed to be a groundbreaking technology potentially revolutionizing the vehicle industry.
Introduction
It is well accepted that the usage of gasoline and internal combustion engines to power automobiles is not sustainable. When cars burn gasoline, they are not only depleting valuable finite resources, but also causing harmful pollution to the environment. It is reported that one third of U.S. air pollution is a direct result of motor vehicle use, 1 and cars are responsible for 75% of carbon monoxide contamination in the air. 2 This in mind, there has been a global effort toward incorporating more environmentally friendly transportation techniques into society.
The main innovation supporting this effort is the electric vehicle (EV), which has been a work-in-progress since the 1830s. Over the past two centuries, several factors have contributed to the fluctuating popularity of electric cars, but it was not until the 1990s that new environmental regulations sparked modern interest in this alternative. 3 Since then, models such as the 1996 GM EV1, the 2000 Toyota Prius, the 2008 Tesla Roadster, and the 2010 Nissan Leaf have prompted widespread application and increased recognition. Today’s most popular EV is the 2020 Tesla Model 3, which is the first vehicle to outsell its gasoline-run counterparts. 4 The transition from gasoline to electric vehicles has been proven to downsize the amount of pollution being emitted into the atmosphere. Indeed, the average EV produces approximately only a third of the global warming pollution of a similar gasoline-powered vehicles.5,6
The widespread success of the EV is based on the underlying battery chemistry. As the EV market was growing, it had to leverage available rechargeable battery technologies. For instance, the Tesla Model S used 7100 Panasonic 18,650 batteries, which is the common battery used in devices ranging from laptops to e-cigarettes. The recent success of EV is tied to the high energy densities and power capacities achieved from lithium-ion batteries. As EVs usage is likely to take over a large portion of the automobile market, it can now lead the battery development efforts. One battery chemistry in particular, magnesium-ion, would offer future EVs numerous advantages over lithium-ion.
Background
Electric vehicles
Generally speaking, EVs can be classified into three main categories: hybrid electric vehicles (HEVs), plug-in hybrid electric vehicles (PHEVs), and battery electric vehicles (BEVs). These three alternatives have a decreasing reliance on an internal combustion engine and an increased reliance on grid power. HEVs are dually powered by electricity and gasoline, requiring only gas tank refills, and no electric plugs. The battery is recharged by the car’s regenerative braking system. PHEVs also require gas refills, but the battery can be recharged both through the car’s braking system and external sources of electricity. Finally, BEVs are comprised of only a rechargeable battery, which requires plugging into an external electrical energy source. 7 Since an increasing amount of grid power comes from renewable sources, the BEV is more environmentally friendly than the PHEV, which, in turn, is more environmentally friendly than the HEV. Additionally, BEVs use cheaper fuel (electricity vs gasoline), require less maintenance, score their owner a tax credit as high as $7500, and hold the privilege of cruising in the HOV lane at any time. 8
However, owning a BEV does come with downsides. First and foremost, the initial investment is not always the most affordable, since the prices of new BEVs range from $30,000 to $80,000+. 8 One of the most economic BEVs, the 2020 Tesla Model 3 starts at $35,000. In addition, an electric-powered transportation system is not necessarily the most convenient. Rather than being able to spend less than 10 min at the nearest gas station, EVs require hours of charging time. After that, a full battery can last an average of 200–400 miles, with the Model 3 driving up to 250 miles on one full charge. 9 This range degrades with battery usage and with cold weather resulting in ranges substantially shorter than their gasoline counterparts.
Despite these shortcomings, EVs comprised 2.5% of new vehicles that were sold globally in 2020. The market share from EVs is expected to grow significantly with an expected market share of 32% by 2030. Much of this growth is seen in three regions – China, Europe, and the United States. Past 2030, the growth of EVs is likely to decrease, as other regions lack the wealth to develop the infrastructure necessary to support EVs. These infrastructure changes, including charging stations and increased electricity capacity, will require multi-billion-dollar capital investments, which is not feasible for numerous countries. Even with the infrastructure changes, the high costs of EVs will limit their usage in poorer countries. Unfortunately, to make a long-term, environmental impact, EVs will need to phase out gasoline-powered vehicles. With the current limitations and costs of the EV, this is not possible.
Lithium-ion batteries
The success and challenges of EVs resides in its battery technology. In particular, the increased popularity of EVs correlates with the development of lithium-ion batteries. Lithium-ion batteries were first invented in 1985 and are now used in various devices such as computers, cell phones, and cars. Lithium-ion batteries offer high energy densities, high power outputs, and low discharge rates when compared to other rechargeable batteries. Prior to lithium-ion, the main battery chemistry was nickel-cadmium, which had an energy density of 70 WHr/kg. However, lithium-ion batteries can readily achieve twice that energy density. 10 Additionally, lithium-ion batteries only lose 1.5%–2% of their charge in a month, 11 whereas nickel-cadmium batteries would lose about 20% for the same time. 10
However, some significant shortcomings and bottlenecks are present in the use of lithium-ion batteries. First, with prolonged usage, they tend to form dendrites, which can cause overheating resulting in fires or explosions. 12 Second, these batteries are quite expensive, costing around 40% more than nickel-cadmium batteries.10,11 This cost will increase with the demand caused from the large battery packs required for EVs. Additionally, the United States Geological Survey estimates that the total remaining lithium supply on earth is only 80 million tons, 13 with much of that supply unattainable. The current annual demand for lithium is only 50,000 tons, which is expected to double by 2024. 14 The price of lithium will skyrocket with this increased demand, further increasing the price of electric vehicles past what would allow for wide-scale adoption. This will continue to rise with the demand for EVs and consumer products. Moreover, as EVs replace other applications in the automotive industry (e.g. shipping, trucking), the bottleneck will be the lithium-ion batteries. Indeed, though higher than nickel-cadmium, the energy density, and power density from lithium-ion will be less than what is necessary to support these needs.
Battery chemistry
Any battery is comprised of the following three components: the anode, the electrolyte, and the cathode. Batteries are powered by an electrochemical redox reaction, which takes place as ions flow from the cathode to the anode through a material known as the electrolyte. 12 In lithium-ion batteries, the cathode is typically a lithium-cobalt oxide, the anode consists of lithium ions in graphite, and the electrolyte is most often a liquid of salts containing lithium.
The battery functions through a series of chemical reactions, with oxidation occurring at the anode and reduction taking place at the cathode. 15 The oxidation step at the anode is given by equation (1) and involves the lithium ions splitting from the graphite, resulting in a lithium-ion, and a free electron.
Equation (2) gives the associated reduction reaction at the anode, where the lithium ion and electron bond with the cobalt oxide.
Equations (1) and (2) can be combined to show the full reaction, as shown in equation (3). Note that the full reaction is bidirectional, such that when discharging it moves from left to right and when charging it moves from right to left.12,16
Researchers have been exploring alternative metals for new battery chemistries that can potentially replace lithium-ion batteries. First, they must identify what element can replace lithium in equation (3). Typically, this element must be readily able to lose electrons, such that they are typically on the left-hand side of the periodic table.
Overview of magnesium-ion battery
Scientists have investigated numerous metals to replace lithium in batteries. These elements include sodium, potassium, aluminum, zinc, and calcium. 17 Though several of these metals showed promise, magnesium came out as having many of the properties that would make for an attractive replacement for lithium. 18
One of the most promising characteristics of magnesium is that it is divalent, such that the oxidation step (equation (1)) would result in two electrons and a Mg2+ ion. The divalent nature of magnesium results in a high specific capacity and volumetric energy density. 18 In particular, the theoretical volumetric capacity of a magnesium-ion battery is 3833 mAh/mL, which nearly doubles the volumetric capacity of lithium (2062 mAh/mL), as shown in Figure 1. 16 Note that these values are the theoretical maximum values and in practicality, lithium-ion batteries have a volumetric capacity less than 200 mAh/mL. However, the higher theoretical volumetric capacity indicates an increased likelihood for a higher practical value.

Comparison of the theoretical volumetric and specific capacity for different anode materials (adopted with permission from Liu et al. 19 ).
Since magnesium is heavier than lithium, the battery will naturally be heavier for a given energy capacity. Indeed, the theoretical energy density of a magnesium-ion battery is 2205 mAh/g compared to 3861 mAh/g for lithium-ion. However, in practicality, lithium-ion batteries are achieving less than 150 mAh/g. Early tests have shown that with a sulfur cathode, a magnesium-ion battery can achieve 1000 mAh/g. 20 Given that most EVs are space and weight constrained, the use of magnesium-ion batteries could potentially increase the range of the vehicle.
In addition to the increased energy capacities, magnesium-ion batteries have numerous other advantages over lithium-ion. First, magnesium does not tend to form dendrites, resolving the safety issues associated with lithium-ion batteries. As such, a magnesium-ion battery can last substantially longer than a lithium-ion battery. Additionally, magnesium-ion batteries can be charged faster since lithium-ion batteries charge times are constrained to avoid dendrite formation. Magnesium is also reported to be the eighth most abundant element on earth’s crust, 21 eliminating the depletion risk, and granting a much cheaper product. Moreover, magnesium is safer than lithium. Since magnesium does not form toxic compounds, manufacturing magnesium-ion batteries would be more cost-effective and environmentally friendly than lithium-ion batteries. Thus, the transition from lithium to magnesium will provide the opportunity to store energy more efficiently at a lower cost.
Challenges with magnesium-ion
Despite substantial work pointing toward the feasibility of magnesium-ion batteries, several challenges still remain. These challenges primarily relate to the definition of the right cathode-electrolyte configuration to match the magnesium anode that will deliver a high voltage and high capacity battery. 18 For the cathode, those utilized in lithium-ion batteries have been proven to provide low storage capability when in reaction with magnesium ions due to magnesium’s high charge density. The stronger electrostatic forces present between the magnesium ions and their surrounding anions cause slow kinetics of ion diffusion. Consequently, the use of conventional cathode materials results in high voltage hysteresis and low capacity. 18 In terms of the electrolyte, the main selection criteria involved is to find a material that is chemically and electrochemically compatible with the magnesium anode, the respective cathode, and the other parts of the battery. Common electrolytes utilized in lithium-ion batteries, most often carbonates, cause dysfunctionality in magnesium ion systems by forming an impermeable layer of deposited electrolytes, which disables magnesium ion transfer. 22
Extensive research is underway to establish ideal material combinations for high-quality magnesium-ion batteries. Figure 2 displays the wide array of materials that have been evaluated for the anode, cathode, and electrolyte. The goal is to resolve the main two issues associated with this development, which are the battery’s proneness to the formation of a passivation layer, and its tendency to experience sluggish kinetics. Defining the proper electrode and electrolyte combination will end up in an applicable product capable to compete with lithium-ion batteries.
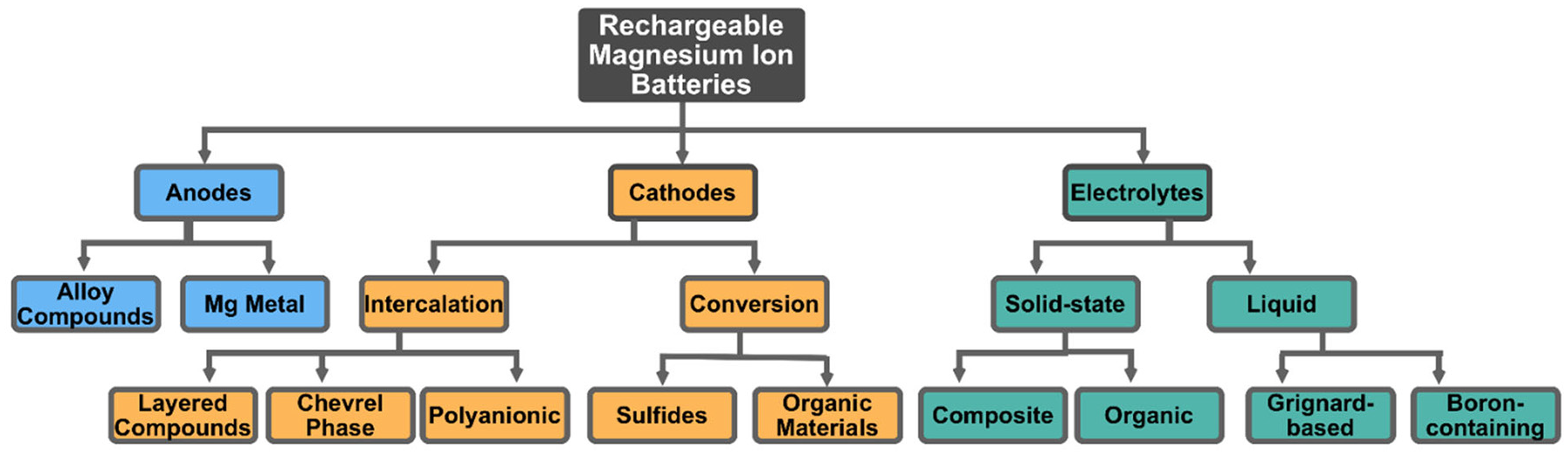
Summary of involved key materials for rechargeable magnesium-ion batteries with respect to their anode and cathodes materials as well as potential electrolytes (adopted with permission from Liu et al. 19 ).
Anode selection
The first matter to address is the anode. Main materials considered for this portion are metallic magnesium, alloy-based, and carbon-based materials. Although using magnesium as a direct anode is a viable option, other techniques are contemplated for the purpose of mitigating the passivation layer that is commonly formed due to anode reaction with the electrolyte. For instance, metallic magnesium with a micro-nano structure could help reduce polarization and potentially decrease the thickness of the films. 23 Furthermore, insertion-type anodes, which are synthesized by alloying, are also considered as a potential solution to the formation of impermeable layers. Moreover, alloys containing bismuth or antimony are named as practical candidates for anode materials based on their theoretically low diffusion barriers of 0.67 and 0.43 eV, respectively. 19 However, it can be argued that such utilization of alloys would hinder the high energy density that magnesium ion batteries are predicted to portray. 24 Thereby, directly applying a metallic magnesium anode seems to be the most reasonable option.
Cathode selection
A great challenge is to establish a cathode to oppose the anode. An ideal cathode material has high operating voltage, high specific capacity, reversibility, fast kinetics of ion transfer, and cycling stability. Potential cathode materials for magnesium-ion batteries can be divided into two groups, intercalation-type and conversion-type cathodes. Generally, intercalation-type cathodes tend to maintain a stable structure throughout the reaction, resulting in good cycling stability. These cathodes can be further classified into three different types: layered materials, Chevrel phase materials, and polyanionic compounds. Although Chevrel phase materials and layered materials are widely focused on, they exhibit harmful interlayer spacing and slow diffusion of Mg2+ ions. For polyanionic compounds, good structural stability, and high voltage are delivered, but their electrical conductivity is poor. 19
Conversion-type cathodes are proven to significantly outweigh the storage capabilities presented by intercalation-type cathodes. These cathodes mainly include sulfides and organic materials. Battery systems involving a magnesium anode and sulfur cathode have been favored due to their impressive electrochemical performance yielding a theoretical volumetric energy density of up to 3200 Wh/L. A number of different materials have been tested for use as the cathode to include graphene-based sulfur composites, cobalt sulfide, copper sulfide, ultra-thin nanoporous nickel sulfides, titanium sulfides, selenium sulfides, and molybdenum chalcogenides. 25 These potential cathodes materials are able to achieve high energy capacity, high voltage, or high cyclability, but none are able to achieve all three. 26 Of these cathode materials, sulfur has emerged as the preferred option for cathode material, having demonstrated high cell energy density and cyclic stability. 17 Additionally, Gao et al. were able to achieve 1000 mAhr/g for 30 cycles with a sulfur cathode, 20 achieving a higher energy capacity than is possible with lithium-ion batteries.
Furthermore, researchers are exploring the use of organic materials for cathode due to their easy chemical manipulation and low cost. 19 Initial research has found that quinone-based organic compounds are able to achieve similar performance to sulfur. There is the added potential that with molecular design and synthetic approaches, that even better performance can be realized. 18
Electrolyte selection
The electrolyte of the battery plays an important role of transporting ions between the electrodes as shown in Figure 3. The ideal electrolyte for a magnesium ion battery should have low corrosiveness, a wide electrochemical window, high ionic conductivity, and reversible dissolution/deposition of magnesium. It should also exhibit necessary safety properties of low volatility, low flammability/toxicity, and thermal stability. 27

Schematic illustration of an ideal electrolyte behavior for rechargeable magnesium-ion batteries (adopted with permission from Liu et al. 19 ).
Magnesium-ion battery electrolytes can be classified into liquid- and solid-state electrolytes. Liquid electrolytes can be further divided into three main categories: Grignard-based electrolytes, boron-containing electrolytes, and (HMDS)2Mg-based electrolytes. 28 Grignard-based electrolytes have the ability to prevent the formation of a passivation layer and achieve reversible magnesium transfer, as shown in Figure 4.29,30 With a general chemical formula of RMgX (where R is an alkyl or aryl, and X is Cl, Br, or other halides), the properties of the electrolyte lie in the R group. This implies that different manipulations of that formula will provide different results. For instance, an electrolyte system was synthesized, in which an organic Mg salt, ROMgCl, reacted with AlCl3, and was able to show promising ionic conductivity and anodic stability. One technique used is the addition of ionic liquid additives to the electrolyte for the purpose of enhancing the conductivity and stability. 19 Importantly, many electrolytes containing chlorine show an unfavorable corrosive nature, which can be harmful to cell parts. 18 To account for this, boron-containing electrolytes have been studied as practical options, which reversibly transport ions without the corrosiveness of chlorine. 31 Moreover, (HMDS)2Mg-based electrolytes have been developed as feasible alternatives for the implementation of MgS batteries with non-nucleophilic properties, high anodic stability, and suitability for cathodes that deliver high-voltages such as sulfur. 28 To demonstrate, applying the lithium salt LiTFSI as an additive to form a (HMDS)2Mg-LiTFSI electrolyte for a magnesium anode and sulfur cathode exhibited an excellent capacity of 1000 mAh/g after several repeated cycles, as well as an energy density of 874 Wh/kg. 20 However, these electrolytes face an important drawback of being expensive.

Schematic illustration of the anode-electrolyte interfaces for both (a) lithium-ion and (b and c) magnesium-ion batteries. (d) The elimination of the impermeable layer for Mg can be achieved by the use of Grignard-based electrolytes (adopted from Mohtadi and Mizuno 30 ).
Moreover, solid-state electrolytes have recently gained popularity due to their safety performance, mechanical properties, high energy density, and wide voltage window. 32 They can be divided into three categories: inorganic, organic, and organic-inorganic composites. While most inorganic solid-state electrolytes have low ionic conductivity, some organic solid-state electrolytes are able to achieve very high conductivity, low polarization, and great cycling stability. Similarly, organic-inorganic composite solid-state electrolytes, which are composed of inorganic fillers and polymer electrolytes, present high Coulombic efficiency of Mg plating/stripping and high cycling stability. Overall, there is a need to develop the establishment of an inexpensive, non-corrosive liquid electrolyte with good safety and performance, with an alternative of instituting a suitable solid-state electrolyte which exhibits better safety, but still currently lacks compatibility. 19
The future of magnesium-ion batteries
Further research should be primarily focused on optimizing the key materials and material combinations discussed. Accordingly, it is necessary to design a reasonable cathode structure, which reduces polarization and speeds up ion diffusion kinetics. In addition, anode modifications must be done to help to eliminate passivation film formation. Finally, an acceptable and compatible electrolyte, which can achieve reversible transportation of magnesium ions without a corrosive character, must be developed.
Research conducted toward this direction has gained notable attention in recent years as shown in Figure 5. Figure 5 plots the number of unique scholarly articles about magnesium-ion batteries listed on Google Scholar. The published studies mainly address different types of anode, cathode, and electrolyte materials, while some of them show significant promise. 21 It is worth to point out that even with the steady increase in the number of scholarly works, publications dealing with magnesium-ion battery are still substantially less than the number of publications related lithium-ion batteries. In 2020, there were approximately 61,000 published scholarly works on lithium-ion batteries. 33

Number of published works about research dealing with magnesium-ion batteries from 2000 to 2020.
In its current form, magnesium-ion batteries are still a fundamental research topic, with initial prototypes being built in labs around the world. Although showing great potential, there is still a long way to go before becoming commercially viable. Even once a company can prove that magnesium-ion batteries are commercially viable, they must cross the “valley of death,” a term associated with the massive cost associated with scaling a battery technology to a commercial level. 34 Many battery technologies, including variants on lithium-ion batteries, have failed to transition due to the immense cost involved. For magnesium-ion batteries to cross the “valley of death,” they must show immense promise to revolutionize energy storage in vehicle applications. The initial work has found that the increased volumetric charge density and safety have the potential for a company to receive the funding necessary to cross this hurdle.
Impact of Mg-ion batteries and concluding remarks
Though the pathway for magnesium-ion batteries is still long and arduous, they do represent a safer, more sustainable, higher capacity energy storage system for the future of EVs. The main source of criticism for EVs is related to their battery packs. Indeed, the lifecycle for a lithium-ion battery pack reveals a number of issues. First, the battery packs are made from lithium, which becomes an increasingly scarce and expensive resource. Upon mining the lithium, it must undergo numerous manufacturing processes to be made into a battery. These processes are energy intensive and partially hazardous. A large portion of the vehicle in turn becomes a battery pack, which can then explode in an accident. The high cost of this battery pack either limits the usage of electric cars to wealthy individuals or requires substantial government subsidies. The battery pack can only be used for a set number of years due to dendrite formation, which makes the battery pack unstable. Moreover, the battery must be disposed of, another energy intensive process with a non-trivial environmental impact.
Magnesium-ion batteries have the opportunity to improve on lithium-ion batteries on every phase of the lifecycle. First, magnesium is eight times more abundant than lithium on the earth’s crust. The relative abundance of magnesium versus lithium results in magnesium being a third the cost of lithium. This price difference will further increase as lithium becomes rarer. The manufacturing processes for a magnesium-ion battery is less energy intensive and releases less toxins than the equivalent processes for lithium-ion. Therefore, the battery pack can be substantially cheaper, allowing for EVs to be used by poorer nations and to remove government subsidies. The battery pack also does not form dendrites, allowing for the battery pack to last substantially longer.
Users of EVs with magnesium-ion batteries will notice other benefits in addition to the cost savings. The divalent chemistry of magnesium-ion batteries, as compared to the monovalent chemistry of lithium-ion batteries, allows for more magnesium-ion batteries in space-constrained vehicles. Since the vehicle will be able to hold more magnesium-ion batteries, the vehicle will be able to travel up to three times further on a full charge. 35 The inconvenience of running out of battery after a single round trip commute, or the arguably unaffordable down-payment, will no longer be issues, which hinder the widespread use of EVs. This will further prompt the growth of EV popularity, significantly reducing greenhouse gas emissions and other car pollution thereafter.
However, restrictions to this technology apply, and drawbacks are still present within the research of magnesium-ion batteries. For instance, in comparison to lithium-ion batteries, mg-ion batteries have a tendency to experience sluggish kinetics due to stronger electrostatic forces among the ions. Furthermore, the implementation of electrolytes commonly used in lithium-ion batteries often leads to the formation of an impermeable layer in magnesium-ion batteries, which impairs their proper function. Thus, it is apparent that the lithium-ion anode is more chemically compatible with conventional cathodes and electrolytes than the anode constituted of magnesium. Nonetheless, with the extended list of potential advantages in mind, the search for suitable materials continues.
Throughout these urgent times, society must implement strategies that will be helpful for the future of our planet. Researchers never stop working toward new goals that will achieve higher optimization and efficiency, while considering environmental effects. Batteries are the prime technology responsible for large-scale, sustainable energy storage. Manifesting the appropriate materials for a magnesium-ion battery system will ultimately result in a feasible product that is suitable to challenge its conventional lithium-ion counterpart. Many would agree that the EV industry is primed for a radical realization like this, which would alter the perception of the electric car altogether. The obstacles associated with these discoveries will eventually be overcome, and a high-performance magnesium ion battery will be established. Upon this, a new and improved rechargeable battery will be introduced, unlocking an even greater prospect for society and the world in general.
Footnotes
Handling Editor: James Baldwin
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A Rosenkranz gratefully acknowledges the financial support given by CONICYT-ANID within the project Fondecyt 11180121 as well as the VID of the University of Chile in the framework of “U-Inicia UI013/2018.” A. Rosenkranz gratefully acknowledges the financial support of the Chinese Academy of Sciences President’s International Fellowship Initiative (2020VEC0006).