Abstract
Biodiesel, a biodegradable, highly oxygenated and renewable energy resource, is produced by esterification of vegetable oils. This work focuses on using electrolytic separation and emulsification to produce purer biodiesel having high cetane index of 61.4. Vegetable oil mixture is used as feedstock. Maximum production yield is 84%. The decrease in engine peak torque was minimum for B5 at 1.94% while maximum decrease was for B20. B5 show a slight increase in power while B20 and B50 show significant drop. For all test speeds, B50 shows higher efficiency than all test fuels however the BSFC was significantly higher than diesel until 88.8% of the maximum engine speed. The maximum increase in brake thermal efficiency for B5 is found to be 2.09% which is 7.9% more than diesel at 2000 rpm. A significant increase of maximum 3.719% in brake specific fuel consumption (BSFC) is observed. Maximum reduction in CO emissions is 53.3% for B50 at 2250 rpm accompanied with a maximum average drop in HCs of 74.4%. The variation in the NOx is insignificant. B5 is found to be the most effective blend for both maintaining the engine performance and improving the engine emissions.
Introduction
Fossil fuel reserves are depleting and over the years their prices have been on a steady rise. Moreover, most countries depend upon import of fossil fuels for their energy sector which puts tremendous strain on their economy. 1 Furthermore, the energy extraction from fossil fuels releases environmental pollutants and greenhouse gasses (GHGs) such as carbon monoxides (CO), oxides of nitrogen (NOx) and sulfur (SOx), particulate matter (PM), and unburnt hydrocarbons (HCs); which contribute towards global warming, acid rains, smog, and other forms of air pollution as well as health issues.2–4 Countries need to shift their dependence from fossil fuels and invest in sustainable and renewable sources of energy.
For a country like Pakistan, biodiesel seems to be a viable option as it would reduce dependency on imported oil with the added benefit of improved vehicular emissions. Biodiesel or fatty acid alkyl esters (FAAE) is a renewable biofuel with high oxygen content, low to zero sulfur content, and an eco-friendly behavior.5,6 Biodiesel is derived from triglycerides (TGs) found in vegetable oils and animal fats.
Triglycerides (oils and fats) are combustible and have been found to improve engine performance and emission6,7 but direct use results in several problems due to TGs high viscosity, poor ignition quality, fuel filter clogging, poor atomization, and production of cycloalkanes on combustion. 8 When converted to alkyl esters, the thermophysical properties are relatively improved. Although TGs can be converted to biofuels by pyrolysis, 9 emulsification, and dilution, 10 the most effective and reasonable for Pakistan is transesterification.
During transesterification, a triglyceride molecule is broken down to glycerol and its constituent fatty acids in the presence of a catalyst. The glycerol is removed while the fatty acids are converted alkyl esters in the presence of an alcohol. 11 Each triglyceride molecule has three fatty acid chains bonded to it. These fatty acids have different percentages in different feedstock, so produce a mixture of alkyl esters with different compositions and varying thermophysical properties. 12 The reaction equation is shown in Figure 1 adapted from Marchetti et al. 11

Transesterification reaction.
The reaction catalyst can be acidic, basic, enzymatic, or non-ionic. 13 Any mono-alcohol can be used in the reaction but methanol is preferred due to high yield. Ethanol offers complete independence from fossil fuels; however, ethyl esters have low pour and cloud point, and the density difference between ethyl esters, glycerol and water is insignificant making separation and purification difficult. 14 The triglycerides can be of plant, animal or fugal sources.15,16 Although different yield is obtained from different sources under similar conditions it is not the defining feature for feedstock selection.
The yield of the reaction also depends upon the ratio of triglycerides to alcohol used. Stoichiometric calculations recommend a molar ratio of 1:3. However, due to reaction reversibility, excess alcohol is added to drive the reaction to completion. If needed, glycerol, a process by-product can be further processed to form ethyl and methyl alcohols and even hydrogen.
The general idea for use of biodiesel in diesel engine is that the emissions improve significantly at a slight expense of performance. Sanjid et al. 17 reported that most researchers found insignificant changes in engine torque, power, and efficiency regardless of the feedstock used. However, BSFC was found to increase in most researches due to reduced calorific value of biodiesel blends. Adaileh and Alqdah 18 conducted a study on a diesel engine using waste oil biodiesel in 5% and 20% blends. An increase in power was recorded for all blends, with B20 giving the maximum increase. However, a decrease in the brake specific energy consumption (BSEC) was recorded. B5 showed insignificant change while a large decrease in BSEC for B20 was observed. Can 19 used waste oil biodiesel’s 5% and 10% blends and reported an increase in brake specific fuel consumption (BSFC) and decrease in brake thermal efficiency (BTE). Asokan et al. 20 also reported a reduction in BTE. Dueso et al. 21 conducted a study a 1.487 L diesel engine using 10% and 100% sunflower oil biodiesel (SB10 and SB100). Brake power was reduced for both SB10 and SB100. A 7.3% decrease in brake thermal efficiency (BTE) was recorded for SB100 while a 0.1%–0.9% increase in BTE was recorded for BTE. Hajlari et al. 22 tested castor oil ethyl ester 5% and 20% blends on a 3.78 L, four-cylinder diesel engine and reported minor variation in power and BSFC.
As for studies focused on emissions from biodiesel operated diesel engine, Tesfa et al. 23 using rapeseed oil biodiesel (ROB) and corn oil biodiesel (COB) found that for 100% biodiesel, there was a 17% reduction in CO2, 27% increase in NOx, 28% reduction in HCs, and 28% reduction in CO for virgin oil biodiesels. Tesfa also conducted emission test on rapeseed oil biodiesel blends and found B50 to give least CO2 emissions. Adaileh and Alqdah 18 reported that with increasing percentage of biodiesel, a decrease in CO was recorded due to increased oxygen content but HCs, and NOx emissions increased. Can 19 also reported increase in NOx for waste oil biodiesel. Sanjid et al. 17 reported that most researchers found that the CO and HC emissions were found to be reduced as compared to diesel for all blends with the percentage decrease having a direct relation with the percentage of biodiesel. They also reported a decrease in smoke number and exhaust gas temperature. Devarajan et al. 24 reported a significant decrease in CO and HC emissions with an increase in NOx for all test conditions when cashew nut shell oil biodiesel (CNSB) was used in a 4.2 kW single cylinder engine. However, Hajlari et al. 22 reported a reduction in NOx while Asokan et al. 20 found no significant change in NOx. The effects of biodiesel on the performance show litter conformity except for the reduction in power and torque. However, the reduction of carbon monoxide and unburnt hydrocarbons, and increase in brake specific fuel consumption are the common results.
Furthermore, the effects of biodiesel on the performance and emission characteristics of a diesel engine hugely depend upon the feedstock used, the method of production and purification, and on the alcohol used. However, in general, biodiesel, when used in CI engines, whether as alternative fuel or a fuel additive, can be expected to improve the engine’s emissions with insignificant changes in engine performance except for increased fuel consumption. Reduced CO, HCs, PM, and SOx emissions can be achieved. This is mainly due to the high oxygen content of biodiesel. However, NOx can be expected to increase because of increased combustion chamber temperature due to better combustion. Although biodiesel is found to be a green fuel, it has a lower heat release rate when used in engines as found by Sinha and Agrawal, 25 which results in a decreased torque output. Furthermore, the engine brake specific fuel consumption (BSFC) increases. However, if the biodiesel is pure enough, it could result in a significant increase in efficiency such that the BSFC curve becomes equal diesel.
This work produces biodiesel from a feedstock containing a mixture of virgin sunflower oil, canola oil, and soybean oil consisting mainly of oleic and linoleic acids with small amounts of palmitic acid. The biodiesel is purified by a novel method and then tested in 5%, 20%, and 50% blends with diesel on a 2.49 L, four-stroke, three-cylinder, 46 hp Perkins AD3.152 diesel engine manufactured by Millat Tractor Ltd. Pakistan. This engine is used to power Millat 240 Tractors widely used in commercial, agricultural, industrial, and transportation sectors of Pakistan.
Methodology
Biodiesel synthesis
In this article, biodiesel is produced by a combination of several techniques available in the literature26–29 to produce a pure sample with highest yield. The chemicals and materials used in the production are,
Vegetable oil
Methanol
Potassium hydroxide
Distilled water
Sodium chloride
The procedure followed for this paper is mentioned below:
A 0.5 M solution of potassium hydroxide (KOH) is made in methanol and added to vegetable oil in the ratio 1:5. Forty-seven grams of powdered KOH were dissolved in 2 L of 97% pure methanol and added to 10 L of vegetable oil. With continuous stirring, the mixture is heated to 60°C and the temperature is maintained for 1 h. The mixture is then allowed to cool and settle for 24–36 h. This settling time ensures that the transesterification is complete and glycerol separates out. To enhance the speed and efficiency of glycerol separation, electrolytic separation is used, in which metal plates connected to AC terminals are dipped into the mixture. The enhancement of separation speed depends on the voltage and the purity of biodiesel depends upon the time. Settled glycerol is removed from the bottom. If glycerol is present in biodiesel it would result in improper combustion in the engine, clog the fuel filter and damage the fuel storage tank.
In the next step, biodiesel is washed to remove potassium hydroxide and other impurities. Distilled water is added with vigorous stirring and an emulsified solution is formed. For this amount of initial reactants, 2 L distilled water may be required. The emulsion is then separated by addition of hot sodium chloride solutions and then the mixture allowed to separate.
Layers form again with pure biodiesel on top and sodium chloride, potassium hydroxide, and soapy solution at the bottom. Biodiesel obtained is then dried to get rid of any excess water and unreacted methanol present in it by heating to 100°C, maintaining this temperature for 1 h.
Thermophysical properties of biodiesel
The biodiesel produced in the lab scale apparatus was tested according to test method of ASTM D93 standard for flash point, ASTM D2500 standard for cloud point, ASTM D97 standard for pour point, ASTM D240 standard for calorific value, ASTM D86 standard for distillation which was required for ASTM D4737 standard to obtain calculated cetane index (CCI). The results of the tests conducted are shown in Table 1.
Comparison of biodiesel and diesel properties.

Experimental engine and test bed
Performance and emission parameters for neat diesel and biodiesel-diesel blends were compared experimentally. The technical specifications of test engine are mentioned in Table 2. The engine and test bed are shown in Figure 2.
Engine specifications and Equipment.


Engine mounted on test bed.
Results and discussion
Blend properties
The properties of the blends used in the experimentation are given in Table 3. The values were calculated theoretically and then confirmed experimentally before employing them in the calculation of results. B5 is chosen to test engine response if biodiesel is used only as a fuel enhancer additive and not a replacement. Moreover, B5 is fuel with thermophysical properties similar to diesel but with a small amount of in built oxygen.
Biodiesel blend properties.

B20 is chosen as a comparative fuel percentage as much of the previous literature used B20.
B50 is chosen to test engine response if biodiesel in later stages is to be used as a replacement. Moreover, B50 is fuel mixture where the thermophysical properties of the blend are an average of diesel and biodiesel.
Biodiesel purity
The biodiesel produced from the use of described method is found to be purer and cleaner than other methods in which similar feedstock was used. It has a significantly high calculated cetane index (CCI) of 61.4 as compared to the CCI or Cetane number of mentioned in a study by Canakci. 30 High cetane number suggests lower percentage of unsaturation studied by Gopinath et al. 31 further implying an improved oxidation stability. Moreover, the blends of this biodiesel will tend to have reduced ignition delay in accordance with the findings of Killol et al. 32 However, the density of biodiesel is found to be 40 kg/m3 higher than the standard limit of 900 kg/m3 while the flash point is significantly lower than the minimum standard limit mainly due to the presence of methanol in the biodiesel. Furthermore, during the distillation of the biodiesel, it was found that 95% of the volume was recovered leaving behind very small amounts of residue. This residue could mainly consist of long chain hydrocarbons and aromatic compounds. The fact that most of the biodiesel was recovered as the distillate indicates that it had very low amount of impurities. Thus, we can state that the biodiesel produced from this method is found to be of good quality. Very small amounts of methanol present in the biodiesel would assist combustion inside engine.
Performance analysis
Torque
Figure 3 shows the torque curve of the test engine run on different fuel blends. The torque decreases with increase in biodiesel concentration; a verification of previous research. Furthermore, the decrease in torque at low engine speed is significantly higher as compared to high engine speeds. The peak torque is obtained at 1400 rpm.

Engine torque curve; BXX, where B stands for biodiesel and XX denotes the percentage of biodiesel.
There is minor change in torque at low biodiesel percentage while there is a significant change for higher percentages. An average percentage decrease of 1.10%, 5.85%, and 6.25%, for B5, B20, and B50 is observed while the peak torque is observed to have decreased by 1.94%, 6.43%, and 6.51%, respectively. This decrease attributed to the low calorific value of the blends as compared to diesel. Although better combustion is achieved as a result of high oxygen content in biodiesel but the combustion process is slow due to higher density, higher viscosity, and low volatility of biodiesel. 33 Moreover, B5, 5% biodiesel blend, overlaps the diesel curve at speeds over 1800 rpm with only a minor average decrease below this speed. As for B20 and B50 the reduction is significantly high. Bari 33 also observed a decrease in torque to be less at high speeds and more at low speeds similar to these results. It is attributed to proper fuel mixing at higher swirl of air produced by the engine at high speed. Furthermore, the decrease is found to be non-linear with the percentage of biodiesel.
Power
Figure 4 shows the variation in engine power with engine speed for various biodiesel blends. Engine power shows similar trends of average decrease as torque. This is because of the reduced calorific value of the biodiesel diesel mixture. This decrease in power is relatively constant for B20 and B50, however, B5 follows the diesel curve beyond 1800 rpm while below this speed the decrease in power is relatively small. The maximum power obtained is at the engines rated speed and max test speed of 2250 rpm for all blends. For B5 the maximum power is only slightly greater than the maximum power for diesel, about 0.16%. For B20 and B50, the maximum power drops by 6.87% and 6.98%, respectively. This decrease in power has been recorded by other researchers for other types of biodiesel feedstocks used.19,28,34,35

Engine power curve; BXX, where B stands for biodiesel and XX denotes the percentage of biodiesel.
Efficiency
Figure 5 shows the changes in engine efficiency at test engine speeds for various biodiesel diesel blends. B50 shows significantly higher efficiency than diesel and other blends for all engine test speeds while B5 and B20 show higher efficiency than diesel only at higher speed. This is an indication of a cyclic behavior of efficiency with biodiesel percentage as quoted by a few authors.
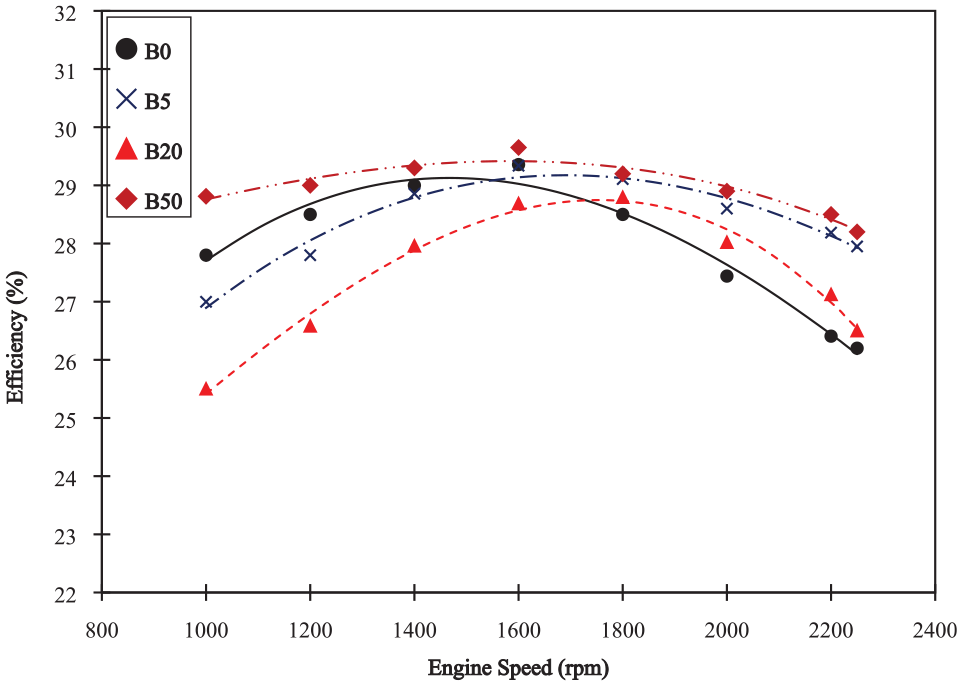
Engine efficiency; BXX, where B stands for biodiesel and XX denotes the percentage of biodiesel.
At lower speeds, B5 shows very small decrease in efficiency when compared with diesel. However, the efficiency increases with increase in speed. The efficiency becomes greater than diesel. This may be due to the increased oxygen content and better mixing with relatively similar properties to diesel. Also, the CCI of biodiesel is 61.4 as compared to diesel’s 45.2. This means that the biodiesel has high cetane number that is the ignition delay is short, thus giving us an increase in efficiency. 6 For B20, the efficiency is lower than diesel at lower speeds but it increases after 1800 rpm and becomes almost equal to diesel. For B50, the efficiency remains higher than diesel for the entire range of test speeds opposite to an almost constant efficiency observed by Wang et al. 36 This indicates, that there exists a biodiesel percentage between 0% and 50% for which the efficiency would be the lowest. More oxygen content is the reason for this B50 trend. However, the results of B5 and B20 corresponds directly to Bari. 33 Furthermore, the maximum efficiency points for B5 and B20 shifts to the right. This means that at low percentage blends the engine is fit for high speed uses.
The increase in efficiency for B50 is an important result, however when it is taken in correspondence to the brake specific fuel consumption it becomes somewhat insignificant. Due to the decrease in power, more fuel is required as compared to diesel. The increase in efficiency reduces fuel consumption only after 2000 rpm, 88.8% of the maximum speed, 400 rpm beyond the peak efficiency as seen in Figure 6.

Break specific fuel consumption for B50 and diesel.
Emission analysis
NOx emission
NOx emissions are found to be increased as the improved combustion increases the combustion chamber temperature as clear from Figure 7. This is in agreement with previous researches.32,37 The increasing percentage of biodiesel increases the oxygen content of the fuel and thus, more NOx are released. Another fact that would increase the NOx emission is the presence of amines in the biodiesel. Biodiesel is produced from plant oil which naturally contains vitamins. Vitamins have bonded nitrogen in them. It is possible for the vitamins to slip into the biodiesel and not get completely separated. These amines when burnt inside the combustion chamber would certainly produce NOx which is the cause of increase in the NOx emissions. B5 and B50 give an average increase of 6% and 0.6% while B20 gives an average drop of 4.5%. The variation in the NOx is insignificant as compared to reduction in other exhaust pollutants.

Oxides of nitrogen: (a) variation with load at 2250 rpm and (b) variation with load at 1400 rpm.
Unburnt hydrocarbons
There is a significant drop in HC emissions for all load positions as observed in Figure 8. This complements previous researches which involved virgin oil biodiesels.6,16 At 2500 rpm, the average drop in HCs is 71.5%, 72.1%, and 74.4% for B5, B20, and B50. At 1400 rpm, the average drop is 58.1%, 65%, and 74.8%. This is again due the increased oxygen content in biodiesel and the greater the amount of biodiesel in the blend the better will be the combustion and thus, the amount of unburnt HCs emitted are significantly reduced. A mere 5% addition of biodiesel gives a significant drop. However, further increase in fuel percentage doesn’t significantly decrease the HC emissions, see Figure 8 for Unburnt Hydrocarbon emissions.

Hydrocarbon emissions: (a) variation with load at 2250 rpm and (b) variation with load at 1400 rpm.
Carbon monoxide
The line graph in Figure 9 illustrates the percentage of carbon monoxide in the exhaust of the test engine against various engine loads, at 1400 and 2250 rpm. The CO emission increase sharply with increase in engine load at both engine speeds. Overall, biodiesel blends give lower CO emission than B0 at most test locations. B5 gives the most reduction in CO at more than 6% while B20 and B50 shows an increase at 75% load position at 1.28% and 3.03%, respectively, see Figure 9 for CO Emissions.

Carbon monoxide emissions: (a) variation with load at 2250 rpm and (b) variation with load at 1400 rpm.
At full load position, CO emissions are reduced by 44.1%, 39.3%, and 53.3% for B5, B20, and B50 at 2250 rpm. At 1400 rpm, the reduction is 9.6%, 7.8%, and 12%. Although B50 does gives the most reduction at full load, B5 is the only blend that shows a decrease in CO at all test locations reaching a maximum reduction of 52%. This can be attributed to the mixing characteristics of B5. As the percentage of biodiesel is low, the blend behaves mostly like diesel, providing better air fuel mixture on one hand, and an increased oxygen content on the other. The other biodiesel blends, B20 and B50, do show similar reductions due to better combustion occurring as a result of increased oxygen content. But, as the increasing biodiesel percentage affects the physical properties of the fuel and its ability to form a better mixture in air, the reduction is insignificant. However, if the fuel injection nozzle design is varied with increasing percentage, better results can be achieved.
Smoke number
Figure 10 shows the filter smoke number (FSN) plotted against various engine loads at 1400 and 2250 rpm. Overall, the FSN increases, almost gradually, with increase in engine load for both engine speeds. At both test speeds, B5 almost overlaps the baseline diesel curve (B0). B20 and B50 remain lower than B0 at 1400 rpm while the opposite is observed at 2250 rpm. FSN is a measure of particulate matter present in the emissions of a diesel engine, mainly soot. Referring to the figure, it is seen that at lower engine speed there is a significant drop in FSN, at more than 50%, for both B20 and B50 at 10% load. With increase in load, the decrease in FSN for B20 is 0.08 while it is 0.78 for B50 when compared with diesel. At full load, the FSN for biodiesel mixtures is higher by more than 2%. At 2250 rpm, particulate matter emissions for B50 remain less than B0 by at most 6.5%. B5 shows a maximum increase in FSN of about 15%. Similarly, B20 shows an increase of 6%, 19%, and about 23%. At full load B20 gives the same reading as diesel, at 9.03.

Filter smoke number: (a) variation with load at 2250 rpm and (b) variation with load at 1400 rpm.
The decrease in FSN at 1400 rpm can be due to improved combustion. However, as B5 follows along B0, it can be said that low amounts of biodiesel don’t affect the Particulate Matter (PM) significantly. With increase in the biodiesel percentage, the drop is observed. FSN for biodiesel blends becoming either closer to the B0 curve or even surpassing it at higher loads at same speed can be attributed to the fact that excess fuel is being injected into the cylinder. The time available for the fuel to burn remains the same thus improper combustion takes place.
At high engine speed, the increase in FSN can be explained by the fact that the time required for the fuel to mix and combust is less. Also, it can be that some impurities in the biodiesel, such as glycerol or even small amounts of water present, are causing the growth in the PM.
Discussion
The biodiesel produced is pure and of good quality, although there is no significant improvement in yield. The CCI is improved, accompanied with a significantly lower pour point of −5.9°C and distillation residue. The calorific value is found to closer to diesel, at 41.6 MJ/kg and remarkably higher than the standard limit of minimum 35 MJ/kg. The biodiesel blend of B5 was the most effective in improving both the engine performance and engine emissions significantly. Larger percentages were found to be inefficient in making the engine characteristics better compared to effort required to produce the biodiesel. Not only does the B5 increases efficiency at comparable power of the engine, it further gives lesser rise in NOx emissions compared to B20 and B50. However, the FSN for B5 does follow the same pattern as B0.
Conclusion
In this work high quality biodiesel is produced having cetane index of 61.4 using electrolytic separation and emulsification. Biodiesel is found to improve CO and Unburnt HC emission and increase the NOx emissions in agreement to Devarajan et al. 24 The change in PM emission remain insignificant.
B5 is found to be the most effective blend for both maintaining the engine performance and improving the engine emissions. The power and torque output of B5 are similar to diesel while there is a significant decrease in BSFC accompanied with an increase in BTE at engine speeds higher than 1800 rpm. This means that B5 is an excellent fuel enhancer additive and should be used as such in high speed operations.
The maximum increase in BTE is found to be 1.75% which 6% more than diesel at 2250 rpm for B5. B5 also gives significantly improved emissions for CO and unburnt HCs. There is an increase in the NOx in the engine exhaust. Compared to lowered CO and HCs by over 50%, and maintained performance the increase in NOx is acceptable.
Increasing the percentage of biodiesel improves the emissions further but the adverse impacts on engine performance are greater. This is due to the deterioration of thermophysical properties of the blends in comparison to diesel. Torque and power output are reduced significantly while compared to B5 the reduction in emissions is very low. Increasing the biodiesel content in the blend reduces the calorific value of the blend thus, the BSFC increases.
Footnotes
Acknowledgements
We are grateful to Mr Sajid, Senior Manager, Engineering Dept, Millat Tractors Ltd, Pakistan and Mr Wajid, Manager Engine Test Bed, Engineering Dept, Millat Tractors Ltd, Pakistan, for providing us access to their research and development department and test engine for carrying out performance and emission analysis. We are also thankful to the Prof Dr Abdullah Khan, Institute of Chemical Engineering and Technology, University of Punjab, Lahore, Pakistan for providing us access to their Fuel Testing Laboratory for analysis of diesel and biodiesel fuels used in this experimental work.
Handling Editor: James Baldwin
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
