Abstract
Microbial solidified sand effectively enhances the strength of the soil, but it will cause brittle failure. In order to reduce the impact of microbial solidification sand brittleness, an improved method for adding carbon fiber to microbial solidified sand is proposed. The qualitative analysis was based on unconfined compressive strength test, calcium carbonate content determination, and penetration test. The results show that the addition of fiber in the microbial solidified sand can significantly increase the unconfined compressive strength of the sample. The unconfined compressive strength of the sample increases first and then decreases with the increase of fiber addition. The addition of fibers during the soil process enhances the toughness of the specimen and causes plastic damage during the failure of the specimen. Based on the analysis of the microstructure of the sample, the effect of fiber bundles on the strength characteristics of the sample is discussed when the fiber content is higher than the optimal fiber content. The addition of carbon fiber to microbial solidified sand can greatly improve the strength of the sample and increase the toughness, which plays a positive role in improving the safety and stability of the project.
Introduction
Microbial induced calcium carbonate deposition (MICP) is a relatively new soil improvement and reinforcement technology in the field of geotechnical soils in recent years. This technology utilizes the microbial mineralization process—microbes promote urea hydrolysis by secreting urease and calcium carbonate to form calcium carbonate, thereby cementing sand to achieve solidified sand. In the past, traditional soil reinforcement techniques such as pre-compaction consolidation and chemical grouting often have shortcomings such as high energy consumption, large pollution, and long construction period. Most of the slurry in chemical grouting will cause soil pollution. 1 The technology of MICP has the advantages of abundant resources, clear mechanism, small environmental pollution, and excellent performance. 2
Mitchell and Santamarina 3 discussed the application value and potential of microbial solidification technology in geotechnical engineering. DeJong et al. 4 showed that MICP technology can improve the mechanical properties of sand. Van Paassen et al. 5 applied large-scale in situ tests to show that this technique can improve the bearing capacity and stiffness of soil. Park 6 studied the effects of different content of polyvinyl alcohol (PVA) fibers on the strength of microbial solidified sand.
Chunxiang Qian and colleagues7,8 elaborated on the application of MICP technology and microbial cement; Zhang and Xiaohui Cheng 9 studied the dynamic characteristics and numerical simulation of MICP liquefied sand foundation; Mingjuan Cui et al. 10 studied the effect of particle size on the solidification effect of microorganisms and analyzed the influence of particle size on the mechanical properties of MICP-improved soil; and Shihua Liang et al. 11 studied the fiber-to-microbial curing. The influence of the sand strength of different particle sizes indicates that the pores between the fibers and the particles affect the formation of “effectively deposited calcium.”
Most of the current research directions focus on how to improve the effective deposition of calcium carbonate, how to improve the strength of the soil after curing of the MICP and how to carry out large-scale soil improvement. 12 However, there are few studies on the ductility and toughness of sand after curing of MICP. Although the sand solidified by microorganisms can reach a high strength, it mainly manifests as brittle failure of the material, that is, the strength is instantaneously lost after the damage, and there is a safety hazard problem in use. How to enhance the toughness of MICP solidified soil becomes an indispensable research direction. Recent studies have shown that the inclusion of discrete short-staple fibers in the soil can effectively increase the strength and toughness of the soil, causing plastic damage during the destruction of the specimen. Compared with conventionally used polypropylene fibers, carbon fibers have the following advantages: low density; high axial tensile strength and modulus; no creep; good fatigue resistance; high temperature resistance; insoluble in organic solvents, acids, and alkalis; no swelling; outstanding corrosion resistance; and so on. Compared with traditional glass fiber, carbon fiber has a Young’s modulus of more than three times; compared with Kevlar fiber, Young’s modulus is about twice. 13 Microbial solidified sand reinforcement is mainly used for polypropylene fiber,14,15 but the field of carbon fiber reinforcement is still blank. Therefore, it is necessary to study the mechanical properties of microbial solidified sand by different carbon fiber reinforcements. Combined with unconfined compressive strength test, calcium carbonate content determination, permeation test, and microstructure observation, the strength characteristics of carbon fiber–reinforced microbial solidified sand were preliminarily investigated.
Experimental program
Experiment material
This test used Xiamen ISO standard sand, and its mechanical properties are shown in Table 1. The fibers used in the test were carbon fiber short filaments having a length of 10 mm, and the mechanical properties thereof are shown in Table 2.
Basic physical and mechanical properties of sand.

Basic physical and mechanical properties of carbon fiber.

Sample preparation
Bacteria and medium
The bacteria used in this experiment were Sporosarcina pasteurii, purchased from DSM, the Netherlands, under the number DSM33. The components for preparing the liquid medium were sodium hydroxide 2 g/L, ammonium sulfate 10 g/L, and yeast 20 g/L. First, the bacteria were sterilized at 121°C for 20 min using a high-temperature sterilizer, and the bacteria required for the test were added under aseptic conditions, and cultured in a constant temperature shaking incubator at 30°C and 200 r/min for 24–48 h. 16 Finally, the OD600 of the bacterial liquid was 2.20 measured by a spectrophotometer, and the enzyme activity measured by a conductivity meter was about 4.75 ms/(cm min). 17
Nutrient preparation
The nutrient solution of this test has a composition of 0.5 mol/L CaCl2 and 0.5 mol/L urea. The operation steps of adding nutrient solution in this experiment are three-round grouting, grouting six times per round, and adding 40 mL of nutrient solution to each sample. The bacteria solution was first added once per round of grouting, and the first nutrient solution was added at intervals of 2 h. The interval of nutrient solution was 5 h. The method of adding the bacterial liquid and the nutrient solution is repeated to ensure more calcium carbonate and uniformity, and the samples are kept consistent.
Sample preparation
The prepared sand column has a height of 80 mm and a diameter of 39.1 mm. When preparing the sample, a polyvinyl chloride (PVC) film is wrapped between the mold and the sand column to release the mold after the sand column is formed. Each sand column was filled with 170 g of sand and uniformly mixed with fibers having mass fractions of 0.0%, 0.1%, 0.2%, 0.3%, and 0.4%, respectively, using the SM950 mixer as shown in Figure 1 to uniformly mix the sand and carbon fiber during sample preparation and then added to the mold. Three parallel samples were prepared for each set of samples. The molded sample is shown in Figure 2 below.

SM950 mixer.

Photo of sample in private mold.
Results
Unconfined compressive strength test
Microbial solidified sand mainly enhances the strength of the soil by inducing calcium carbonate to form loose sand to enhance the strength of the soil. The unconfined compressive strength test can directly show the effect of solidification of the sample. The test was carried out using a liquid crystal automatic pressure testing machine (YAW-S300, Jinan HengRuiJin Testing Machine Company) with a loading rate of 1.0 mm/min to the sample failure, and the peak compressive strength was taken as the unconfined compressive strength of the sample. 18 The unconfined compressive strength is shown in Figure 3.

Unconfined compression strength of samples with different fiber contents.
As can be seen from Figure 3, the unconfined strength of the fiber-free sample was only 0.23 MPa, which was the lowest in all samples. The unconfined compressive strength of the sample after adding the fiber is significantly higher than that of the non-added fiber sample. If the added fiber is 0.1%, 0.2%, 0.3%, and 0.4%, the corresponding unconfined compressive strengths are 0.39, 0.42, 0.37, and 0.35 MPa, and the strength improvement was 170%, 182%, 161%, and 152% of the unadded fibers, respectively. Generally speaking, with the increase of fiber content, the unconfined compressive strength of the sample increased at first, reached the maximum at 0.2%, and then began to decline. It can be seen that the addition of fibers during microbial solidification of the sand significantly improves the unconfined compressive strength of the sample.
Figure 4 is a graph showing the “bulging” of the sample during the unconfined compressive strength test. After the fiber is added to the sample, the fiber forms a three-dimensional (3D) network of reinforced structures in the soil (Figure 5), which limits the development of the failure surface and the gap and effectively improves the strength of the soil and enhances the toughness. This is mainly because after the sample is destroyed, the fiber crosses the crack like a “bridge” and can bear a certain tensile stress, which can effectively inhibit the further development of the crack and delay the overall damage of the sample. When the amount of fiber added is low, the reinforcing effect of the fiber is limited, and the effect of fiber reinforcement cannot be fully exerted. When the amount of fiber added is high, the phenomenon that the fibers are entangled into bundles is easy to occur during the preparation of the sample, which makes it difficult to form an effective calcium carbonate connection between the fiber and the sand sample, which affects the effect of microbial solidification. Therefore, in the actual engineering application, it is necessary to determine the optimum amount of fiber to be added, so as to obtain an optimum effect.

The sample expansion.

Carbon fiber forms a three-dimensional network in the sample.
Calcium carbonate content
Microbial solidified sand is mainly formed by the pores in the soil filled by calcium carbonate, which forms an effective cementation between the particles, thereby strengthening the strength of the soil. The content of calcium carbonate can reflect the effect of solidification from the side, and is an important indicator for judging the solidification effect of sand.
For the determination of calcium carbonate content, the un-crushed sample is first placed in a beaker and the mass m1 is called. To ensure the accuracy of the test results, the mass of each selected sand block is about 40 g. The sand was immersed in 1 mol/L hydrochloric acid solution, and sufficiently stirred until it was bubble-free and allowed to stand for 2 h. The completely reacted sand sample is rinsed with deionized water, and then placed in an oven for drying, and the mass of the sand sample after the beaker and the reaction is m2. Calcium carbonate content obtained by formula (1)
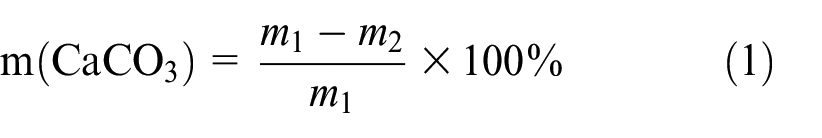
As shown in Figure 6, the sample without added fiber has a calcium carbonate content of 11.30%, and when the fiber addition amount is 0.1%, 0.2%, 0.3%, and 0.4%, it is 12.44%, 14.58%, 13.28%, and 13.18%, respectively. Overall, the fiber-added sample had higher calcium carbonate content than the un-fiber-added sample. When the fiber addition amount was 0.2%, the calcium carbonate content was the highest, which was positively correlated with the results of the unconfined compressive strength test. It can be seen that the content of calcium carbonate has a certain relationship with the effect of microbial solidified sand.

The average contents of CaCO3.
Penetration test
The permeability coefficient of each fiber addition amount is shown in Figure 7. It can be seen from the figure that the permeability coefficient of the added fiber sample is lower than that of the un-added fiber sample, and the permeability coefficient of the 0.2% fiber addition sample is 6.11 × 10–6 m/s, which is lower than the un-added fiber test. The permeability coefficient (1.14 × m/s) was reduced by about 46%.

The permeability coefficient of different fiber content.
Different fiber additions have different effects on the permeability coefficient of the sample: when the fiber addition amount is small, the preparation of the sample perfusion solution has less blocking effect on the bacteria, and the bacteria solution cannot be left in the sample. There is not much calcium carbonate produced, so the permeability coefficient drops less; when the fiber addition amount is large, the fibers added in the preparation sample are easily entangled into bundles, and the bacteria are difficult to enter into the fiber bundle, thereby reducing the survival of bacteria and the space for generating calcium carbonate, so that the calcium carbonate is reduced and the permeability coefficient is not significantly reduced.
Scanning electron microscopy
The structure of carbon fiber–reinforced microbial solidified sand was observed by scanning electron microscopy (SEM), as shown in Figure 8. Figure 8(a)–(c) is a microscopic structure in which the carbon fiber is magnified 500 times, 1000 times, and 1500 times, and Figure 8(d)–(f) is a microstructure of 0.2% carbon fiber added to the sample. In the condition that the fiber addition amount is 0.4% and the magnification is 100 times (Figure 8(g)), it can be seen that the fibers are entangled into bundles, and it is difficult for bacteria to enter the bundle body, and the survival and activity space of the bacteria are compressed, resulting in a test. The reduced calcium carbonate content reduces the unconfined compressive strength of the sample. Under the condition that the fiber addition amount is 0.4% and 190 times (Figure 8(h)), the fibers are denser, although a small amount of calcium carbonate is attached to the fibers, but there is no effective connection with the sand particles, which does not contribute well to the strength of the sample. Under the condition that the fiber addition amount is 0.4% and 1000 times magnification (Figure 8(i)), it can be seen that the calcium carbonate induced by the microorganism is attached to the surface of the sand particles in a large amount, and partially encloses the fiber, which provides a good fiber reinforcement. The site effectively increases the strength and toughness of the sample. The fiber is mainly present in the pores. As the amount of fiber added gradually increases, the surface area of the fiber gradually increases, and the area combined with calcium carbonate is also increasing. However, after a certain proportion, as the fibers increase, the fibers are entangled into bundles, and the effective area of the fibers and calcium carbonate is reduced, which is consistent with the results of the unconfined compressive strength test.

SEM images of bio-cemented sand with fiber reinforcement: (a) 500× (non-fiber), (b) 1000× (non-fiber), (c) 1500× (non-fiber), (d) 500× (adding 0.2% fiber), (e) 1000× (adding 0.2% fiber), (f) 1500× (adding 0.2% fiber), (g) 100× (adding 0.4% fiber), (h) 190× (adding 0.4% fiber), and (i) 1000× (adding 0.4% fiber).
Conclusion
Through the unconfined compressive strength test, calcium carbonate content, and penetration test, the strength characteristics of carbon fiber–reinforced microbial solidified sand were studied. The effect of fiber addition on the solidification effect of sand was analyzed. The fiber-reinforced structure was discussed in combination with the microstructure. Based on the mechanism, the following conclusions were obtained as follows:
The fiber added by the microbial solidified sand can form a 3D network of reinforced structures in the soil, limiting the development of the failure surface and the gap, effectively improving the strength of the soil and enhancing the toughness.
Different fiber additions have significant effects on the effect of microbial solidified sand. The unconfined compressive strength of the sample increases first and then decreases with the increase of the amount of added fiber. The optimum addition amount of carbon fiber added was 0.2%, which was 187% higher than that of the un-added fiber sample.
The calcium carbonate content was positively correlated with the overall trend of unconfined compressive strength; the permeability coefficient was negatively correlated with the overall trend of unconfined compressive strength. It is indicated that the unconfined compressive strength of the sample is related to these two parameters, but the specific quantitative relationship needs further study and determination.
As the amount of fiber added increases, the fibers tend to entangle each other into a bundle when preparing the sample, which hinders the entry of bacteria when the bacteria are poured, and reduces the space for the survival of bacteria and the formation of calcium carbonate. When the optimum fiber addition amount is exceeded, the unconfined compressive strength of the sample is decreased.
Footnotes
Handling Editor: James Baldwin
Data availability
All data, models, and code generated or used during the study appear in the submitted article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is financially supported by the Natural Science Foundation of Guangdong Province (grant no. 2017A030313293); the Guangzhou University Innovation and Strengthening School Project (grant no. 2016KTSCX108); Guangzhou science and technology project (grant no. 201804010250), Fujian provincial tunnel and Urban Underground Space Engineering Technology Research Center Open Research Project (grant no. 16FTUE02), and National Natural Science Foundation of China (NSFC) (Grant No. 51778159).
