Abstract
Electrolytic in-process dressing grinding holds tremendous promise for ultra-precision grinding on hard and brittle materials. Besides oxide films on the grinding wheels, it is important to study the content generation and transformation of α-Fe2O3 inside the oxide film and its effect on polishing process. In this study, X-ray diffractometer measurement was performed in electrolytic in-process dressing grinding with different grinding wheels in order to investigate the content and transformation procedure of α-Fe2O3. The non-abrasive iron-bonded wheels showed significantly improved polishing performance with better surface quality.
Introduction
Electrolytic in-process dressing (ELID) grinding is an efficient polishing process due to its ability of generating oxide film surface on grinding wheels. Oxide films on ELID grinding wheel surface, investigated since the late 1990s, are functional for absorbing vibration, moderating impact, separating the abrasive grains, and polishing the workpiece. The oxide film consists of all kinds of iron oxides, such as α-Fe2O3, β-Fe2O3, and γ-Fe2O3. 1 The most excellent polishing agent among iron oxides is α-Fe2O3. Unstable iron oxides convert into α-Fe2O3 under certain circumstances, 2 which is used to precisely polish optical glass and silicon chip. Early reports and results show that the majority content of oxide film is all kinds of iron oxides. Conventional mechanical tests on oxide films including rigidity, elastic modulus, and contact stiffness are valuable.3–5 ELID grinding has been considered as a compounding process of grinding, grounding, and polishing by oxide films. 6 We have reported a test on ELID-grinded nano-ceramic material by oxide film polishing process and proved that the friction coefficient of nano-ceramic decreased with grinding depth and feeding speed increased. 7 We have focused on process parameters of ELID grinding that recognize the grinding depth factor on the adhesiveness of the oxide films. 8 In addition, Kersschot et al.1,9 have studied several process parameters such as electric double layer, electric resistance, growth speed of oxide films, including provided power, electrolyte, speed of wheel, and abrasive grain. ELID grinding has offered a way to obtain better process stability and material removal rate by comparing with traditional grinding depth.10,11 Yu et al. 12 used ELID grinding to increase the ground surface quality for difficult-cut material SiCp/Al. Zhao et al. 13 studied grinding influence of speed, applied load on surface finish, and material removal rate of ELID-grinded SiC bearing. As a result, very few studies on the basic mechanism of ELID process generated oxide film surface, the consequent polishing performance on surface finish accuracy and quality have been reported.
Therefore, the major objective of this study is to understand the formation and transformation procedure of α-Fe2O3 in the oxide film surface. X-ray diffraction (XRD) measurement is a reliable tool for the measurement of α-Fe2O3. The influence factors of α-Fe2O3 and its polishing behavior is also discussed.
Experimental setup and procedure
Powder XRD measurements were performed on a Max-RC-typed diffractometer (Rigaku, Tokyo, Japan) with Cu-Ka radiation from 20° to 80° at a scanning speed of 6°/min. ELID grinding was carried out using iron-based diamond abrasive wheels on an MM7120A surface grinder. The grinding wheel speed was maintained 1500 r/min and the feed rate was fixed to 10 m/min. Grinding depth was set to be 0.001, 0.003, 0.005, and 0.01 mm. And the spindle static stiffness was 100 N/μm. Diamond grinding wheels were of grain sizes 40, 10, and 1.5 μm (grit size: W40, W10, and W1.5), and the mass concentrations were 100%, 50%, and 25%, respectively. A high-frequency pulse was supplied from the ELID mirror surface grinding power supply (HDMD-V). The amplitude of the applied voltage was 60–120 V and the current was set from 0 to 50 A. The pulse width was supplied to 1–99 μs and the inter-pulse was 1–99 μs. ELID grinding liquid HDMY-V model was supplied at a dilution of 1:50. ZrO2 ceramic workpiece of 200-nm grain size (hardness of HRA87 and toughness of 12.2 MPa·m1/2) was used for experiments.
First of all, the iron-based wheels W1.5, W10, and W40 were used to conduct ELID grinding experiments after 30-min pre-electrolysis treatment by MM7120 surface grinding machine, which coincides with electrolysis dressing unit. All values of the parameters are mentioned in section “Experimental setup and procedure.” Grinding was stopped when reddish-brown oxide films were observed evenly adhering onto the surface of the wheels. The oxide films were scraped away from the wheels and collected up, as shown in Figure 1.

Oxide film powder scraped away from the grinding wheels.
Results
XRD pattern of the oxide film on W40 wheel is shown in Figure 2. The patterns show that there are several types of iron oxides and hydroxides, including bivalent and trivalent iron. It could be remarked that there are also α-Fe2O3, β-Fe2O3, γ-Fe2O3, FeO, Fe(OH)2, and Fe+3O(OH) in the oxide film. The evolutionary process of iron oxides was also checked from value obtained from this pattern. As grinding processes, the surface layer of the oxide film transformed to γ-Fe2O3, β-Fe2O3, and α-Fe2O3·H2O, eventually. Under the continuous action of grinding heat in the grinding zone, iron oxides then lost crystalliferous water and transformed into α-Fe2O3. And there are still other intermediate valence states underneath the wheel surface. The surface layer of oxide film scraped might confound with the base layer. Hence all types of valence-state iron oxides can be found in XRD results. The XRD pattern showed some heterogeneous components except the above-mentioned iron oxides and hydroxides because the components of the wheels consist of iron, copper, tin, nickel, and graphite. It may result confused data in this case. To solve this problem, three evident pairs of diffraction peaks of α-Fe2O3 were marked in Figure 2.

XRD pattern of oxide film on W40 grinding wheel.
Discussion
Effect of grinding temperature on transformation of α-Fe2O3
The content and transition of α-Fe2O3 in oxide films with different temperatures were investigated by variable-temperature XRD, as shown in Figure 3. It was found that the diffraction peaks of α-Fe2O3 were first observed at grinding temperature of 300°C and the transition of α-Fe2O3 was almost completed at 600°C. The diffraction peaks of α-Fe2O3 in oxide films were observed at 24.15°, 33.16°, 35.61°, 40.86°, 49.93°, 54.04°, 62.40°, and 64.45°, respectively, where these diffraction peaks matched with standard XRD patterns of α-Fe2O3. The results of XRD measurements show that Fe oxides in the oxide film converted to α-Fe2O3 at grinding temperature of 600°C.
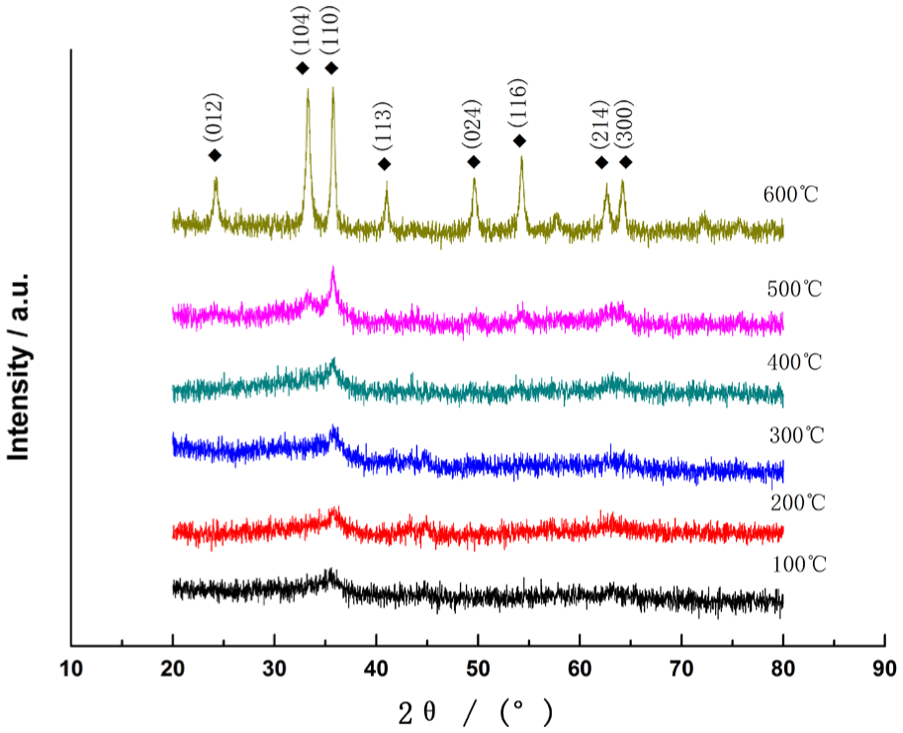
Variable-temperature XRD measurements of the oxide films on grinding wheel.
The formation and transformation processes of α-Fe2O3 were matured by increasing the grinding temperature. More Fe oxides progressively converted to α-Fe2O3 under the action of grinding heat. In ultra-precision grinding, especially in final polishing process, the wheel stops electrolytic dressing and is performing to and fro grinding with no cutting depth. The efficiency of wheel polishing depends on the surface oxide film, which transformed to α-Fe2O3 under repetitive squeezing and friction action with high grinding heat, to improve surface quality by ELID grinding.
Effect of the particle sizes on transformation of α-Fe2O3
XRD diffraction patterns of three different particle sizes (W40, W10, and W1.5) were also checked, which is shown in Figure 4. And three pairs of α-Fe2O3 main peaks obtained from this measurement were distributed at 33.16°, 35.61°, 49.93°, 54.04°, 62.40°, and 64.45°. The diffracted intensity increased gradually, whereas the particle size of the wheels decreased. This variation of particle size caused more oxide films involved in grinding process and therefore transformed more α-Fe2O3 in the oxide films.

XRD measurement of W40, W10, and W1.5 particle sizes.
Mass concentration of the wheels
Variations in diffracted intensity of α-Fe2O3 within the oxide films were measured under different mass concentration of the wheels (100%, 50%, and 25%) plotted in Figure 4. The diffracted intensity value obtained from this measurement increased with decreasing mass concentration. From this figure, it can be stated that volume fraction of the abrasive particles decreased with decreasing mass concentration. The more the volume fraction of the binder, the more the iron-based binder involved in electrochemical reaction during ELID grinding processes. The formation and subsequent transformation of α-Fe2O3 in the oxide films gradually matured. The wheels exhibited increasing amount of α-Fe2O3 with less mass concentration, consequently.
In common, the ultra-precision grinding processes with finer particle size showed better grinding performance. The less mass concentration of the wheels resulted in smaller particle size to avoid the wheel block up. Thus, compared to other conditions, only less mass concentration (25% in this case) of the wheel exhibited its suitability to ultra-precision grinding, which is identical to the general knowledge in ultra-precision machining.
The component content of Fe oxides in the wheels
Our measurements were carried out using three grinding wheels of same chemical composition. So the results could not explain the effect of formula on the formation of α-Fe2O3. However, according to ELID electrochemistry mechanism, increasing Fe element can directly influence the content of Fe oxides in the oxide films of grinding wheels. Then, these Fe oxides transform to α-Fe2O3 eventually by the high grinding heat. So with higher Fe element content, more α-Fe2O3 can be generated, which is concluded by theoretical analysis. In addition, the formation of α-Fe2O3 is also distributed with different characteristics with respect to the wheel’s condition. Sintering process, adhesion, and combination properties of grinding wheels should be taken into consideration to increase Fe’s content in the bonder.
Correlation of α-Fe2O3’s content with polishing effect
As an abrasive, α-Fe2O3 is second in grinding effect only to cerium-based polishing powder. The formation and transformation of α-Fe2O3 in the oxide film is the fundamental cause how the oxide films work on grinding and polishing procedures. The transformation of α-Fe2O3 tends to complete while the oxide films operating the electrochemical reaction due to the grinding heat per grinding pass. It could be observed that the content of α-Fe2O3 in the oxide films gradually increased and eventually formed an abrasive film with finer grain size onto the surface of the grinding wheel, as shown in Figures 5 and 6. The grain size of α-Fe2O3 obtained from our measurement was only 5–10 nm. Nano-scaled α-Fe2O3 has a minor effect on polishing process and lower removal ability. Some α-Fe2O3 abrasive grains tended to gather via cluster state due to ease of reactivity and formed membrane structure adhered to the grinding wheel surface. Therefore, it can be concluded that polishing ability of the oxide films on the abrasive wheel surface is a cooperative phenomenon with many α-Fe2O3 grains in membrane structure.

Grinding wheel surface generated with α-Fe2O3 measured by TEM: α-Fe2O3 grains size.

Grinding wheel surface generated with α-Fe2O3 measured by TEM: membrane structure of α-Fe2O3.
The oxide film surface was easy to be stroked off during coarse grinding and semi-finishing grinding processes due to its small hardness and weak adhesive bond with the wheels. Thus, the grinding and polishing effect of the oxide films is estimated to be valid only in ultra-precision grinding. The reason is that the oxide film is far thicker than the grinding depth in ultra-precision grinding, and there is no direct contact between the workpiece and the abrasive grains with the oxide films separated in between.
The nano-scaled grits and the hardness of α-Fe2O3 showed considerably better polishing performance (improved surface quality with no big surface damage). On the other hand, the oxide films with abrasive particles increase the potential of secondary scratch on precision finished surface, which leads to increase in quantity of substandard products.
Non-abrasive iron-bonded grinding wheel
The traditional iron-bonded grinding wheel has lower α-Fe2O3 content after electrolysis by ELID grinding. In addition, the diamond grits in the oxide film is responsible for poor surface quality with some deep scratches. Thus, traditional iron-bonded grinding wheels are considered to have limited effect on polishing performance.
In order to study the polishing mechanism of α-Fe2O3, non-abrasive-grain grinding wheel was fabricated with no diamond abrasive grains. We would like to investigate the fundamental principle in ELID ultra-precision grinding only on the effort of α-Fe2O3 in the oxide films. In our experiments, the transformation ratio from γ-Fe2O3 to α-Fe2O3 was increased properly to achieve precision polishing only by α-Fe2O3 in the oxide film.
ELID grinding processes were carried out using non-abrasive iron grinding wheel, W1.5, W10, and W40 wheels for nano-ceramics, nano-cemented carbide, and flat glass, individually. Figure 7 displays the variations of the surface roughness for nano ZrO2, nano-cemented carbide, and flat glass. For different grinding wheels, the surface roughness for these three workpieces decreased with the reduction of grain size of wheels. Compared with three wheels, the grain size of wheels nearly to non-abrasive can reach small surface roughness.
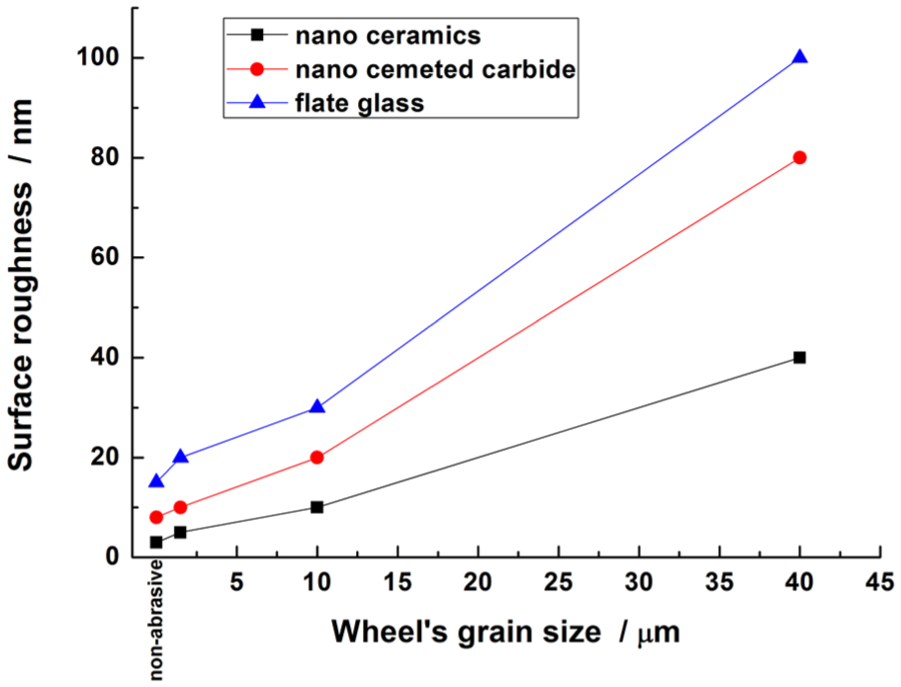
Surface roughness of three different wheels.
Conclusion
The ELID grinding was performed to understand the formation and transformation mechanisms of α-Fe2O3 in the oxide film and the consequent polishing performance. Experimental studies of the oxide film’s components for the grinding wheels have been conducted and discussed by XRD and transmission electron microscopy (TEM) measurements. These results were in agreement with the transformation mechanism of α-Fe2O3 that we have predicted.
The summarized results from this study are as follows:
The components of the oxide film on grinding wheels during ELID process have been investigated, and the formation and transformation of α-Fe2O3 is found.
The ELID grinding process at higher grinding temperature (nearly 600°C) produced more α-Fe2O3 in the oxide films of grinding wheels.
It could be observed that the grain size had certain influence on the content of α-Fe2O3 on the grinding wheel surface. The content of α-Fe2O3 increased with the grain size decreasing. The mass concentration also had certain influence on it, and the content of α-Fe2O3 increased with the decrease in mass concentration.
The mechanism of the oxide film working on polishing effect during ELID grinding is of the coordination behavior of numerous α-Fe2O3 particles which have formed membrane structure.
The non-abrasive iron-bonded grinding wheels showed better surface roughness compared to other grain size of wheels.
Footnotes
Handling Editor: Xichun Luo
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation, China (Grant No. 51475147) and by the key project of Henan Province Science and Technology Research (Grant No. 13A460341). Also funded by the Natural Science Foundation of the Jiangsu Province, China (Grant No. BK20150406) and the technical project of Nantong, China (Grant No. MS12016015).
