Abstract
In this study, new microencapsulated phase change material, with organic composite phase change as the core material and polymethylmethacrylate as the shell, was prepared by in situ interfacial polymerization reaction. The surface morphology of the formed microcapsule was observed by scanning electron microscopy, and its phase change temperature and phase change heat were measured by differential scanning calorimetry. The results of this study indicate that the mean diameter is in the range 2–3 µm, with a uniform size distribution, and the phase change temperature and enthalpy of microencapsulated phase change material are of 26.2°C and 139.68 J/g, respectively. The prepared microcapsule was mixed with ordinary paint in a 1:1 ratio for performing the comparative tests in two rooms with the same dimension and settings. The results indicated that with the introduction of a 2- to 3-mm-thick coating containing microencapsulated phase change material the temperature amplitude decreased by 5°C–6°C. The power monitoring results for the period of the highest temperature in 1 day indicated that the start-up time of air conditioner significantly decreased, thus saving about 26% electric consumption.
Introduction
Phase change material (PCM) is a type of highly efficient energy storage materials. Making use of the unique characteristics of PCMs to coordinate the time and intensity mismatches of energy supply and demand is an economically applicable approach. Energy storage and temperature control can be achieved by the application of PCMs to building materials in the form of composite enveloping. The composite structure not only can decrease the indoor temperature fluctuation and improve the comfort degree but also can reduce the self-weight of building structure and save the air conditioning and heating cost.
Although PCMs have significant advantages, pure PCMs also have some shortcomings in direct applications. Inorganic compounds such as salt hydrates and metals and organic compounds such as paraffin waxes, fatty alcohols, fatty acids, and their mixtures have been applied as solid–liquid PCMs.1,2 During the phase transition, pure PCMs would show deformation and uneven dispersion in the used material matrix, as well as causing the environmental pollution. The microencapsulated phase change material (MEPCM) can overcome these shortcomings. It is a composite PCM with a core–shell structure, which is the solid–liquid PCM particle within a stable layer, as evidenced by the microencapsulation technology.
In recent years, many researchers had carried out a series of studies on the MEPCM. Alkan et al.3,4 prepared a series of MEPCM with polymethylmethacrylate (PMMA) shell and characterized their properties. Wang et al. 5 prepared the paraffin/PMMA MEPCM by polymerization catalyzed by ultraviolet (UV) light. In order to lucubrating the effect of preparation methods on the size of microcapsule, Wang et al. 6 prepared a variety of MEPCM with PMMA shell and analyzed the effects of various factors on the size of microcapsule. Ma et al. 7 introduced the copolymer of methyl methacrylate and divinylbenzene as the shell material and measured the thermal properties of the prepared MEPCM. The shell thickness was in the range 85–250 µm. Salaun et al. 8 prepared MEPCM with a shell of melamine–formaldehyde resin through in situ polymerization. Su et al. 9 used methanol to modify the melamine–formaldehyde resin and found that the prepared microcapsules have better durability. Lone et al. 10 prepared the n-octadecane/polyurea MEPCMs using the tubular microfluidic method. The microcapsule size ranged from 35 to 500 µm and could be controlled. Fe3O4 nanoparticles were added into the microcapsule for increasing the thermal conductivity. Fang et al. 11 prepared the microcapsule containing n-tetradecane with a shell of urea–formaldehyde resin and analyzed the effect of cross-linker on the microencapsulation efficiency. Through suspension polymerization method, Sanchez-Silva et al. 12 prepared the MEPCM with a polystyrene shell and studied the effects of different suspension stabilizers on the surface morphology, size, and thermal properties of microcapsule. Zhang et al.13,14 and Chen et al. 15 prepared the inorganic MEPCM with an outer shell of silica. The results indicated that the silica shell improved the heat transfer performance and thermal stability of the PCM.
Within the range of temperature fluctuation, PCMs can absorb, store, and release a large amount of latent heat; therefore, they can adjust to the indoor temperature and save energy consumption. 16 So far, a large number of literatures17–20 has reported the applications of PCMs in energy-saving buildings. Microcapsule technology can protect the PCM. The size of microcapsule is small, usually between some microns to a few 100 microns, and the particle size decreased with increasing surface/volume ratio, which improved heat transfer performance. 21 After doping with MEPCM, the bricks, walls, and ceilings can directly prevent the overheating phenomena in the summer and can make use of solar energy to heat the room in the winter. In addition, MEPCM can be used in refrigeration or heating equipment to achieve the peak load shifting.
Schossig et al. 22 used 6-mm thick plasterboard with 40% MEPCM as a material in the internal wall and carried out comparative test. The results showed that compared with the room using ordinary plasterboard, the time of temperature above 28°C was decreased and above 22°C was increased meaning the temperature at night is higher. Cabeza et al. 23 constructed a room whose south wall, west wall, and panels were the concrete slabs containing MEPCM. Respectively, the doped PCM exhibited a phase change temperature and enthalpy of 26°C and 110 kJ/kg. The on-site test results showed that the test room had a higher thermal inertia and lower maximum indoor temperature than the control room with ordinary concrete slabs. Kuznik and Virgone 24 prepared a composite wallboard consisting of phase change copolymer material and tested its properties under real climate. The results showed that the phase change wallboard can help reducing the maximum indoor temperature by 4.2°C, and the attenuation factor of indoor temperature get up to 0.7. The results also indicated that adding PCM was conducive to the natural convection of the indoor air and avoided the excessive accumulation of heat. Ahmad et al. 25 studied the effect of the thickness of phase change wallboard on its properties. As the thickness of wallboard was <20 mm, the temperature fluctuation cannot be distinctly weakened, indicating that the effect of PCM was not obvious. Gowreesunker and Tassou 26 studied the characteristics of PCM and concluded that the phase change of wallboard can control the room temperature in the summer. The results showed that compared with the traditional plasterboard, the phase change of wallboard can reduce the maximum indoor temperature by 3°C. The results also indicated that in order to optimize the performance of wallboard, the PCM could be better used in the upper part of the wallboard or ceiling.
Recently, although several types of phase change microcapsules have been utilized in the building energy-saving field, Due to some restrictions, such as preparation method, decreasing the surface/volume ratio, and overall heat transfer performance, the size of the used phase change microcapsules was all in the range 7–20 µm. In contrast, the MEPCM was mixed with concrete to form composite wall for the application in energy saving of building usually. In these occasions, the content of microencapsulated multi-PCM was strictly restrained and complicated operation results in a relatively poor heat transfer between MEPCM and the indoor space. In order to solve these problems, 2–3 µm phase change microcapsule with organic composite PCM as the core and PMMA as the shell was prepared by interfacial polymerization reaction. The surface morphology, phase change temperature, and latent heat were studied by small-scale tests. Later, the coating material was prepared by the MEPCM and painting in a ratio of 1:1. The comparative test was carried out in two identical rooms (both 2.5 × 2.5 × 3.6 m3). One was painted with the coating material containing prepared coating material, and the other was painted the coating without MEPCM. The thermal stability of the MEPCM coating layer and the energy-saving performance of room painted with MEPCM were analyzed.
Methods
The experimental study included two parts: performance test of MEPCM and energy-saving comparative test of coating material containing MEPCM.
Preparation and performance test of MEPCM
The test raw materials included organic composite PCM, PMMA, azodiisobutyronitrile (AIBN), ethyl alcohol, and polyethylene glycol octylphenol ether (emulsifier, OP-10).
The experimental instruments contain the following:
Constant-temperature water bath (HH-4; Changzhou Zhibo Rui Instrument Manufacturing Co., Ltd) with a temperature range of room temperature to 100°C and a temperature control accuracy of ±0.5°C. The instrument had digital display, accurate and automatic temperature control.
Electronic balance (RS232; Shanghai Shunyu Hengping Scientific Instrument Co., Ltd) with a measuring range 0.5–2100 g and a scale interval of 0.01 g.
Differential scanning calorimeter (DSC) (DSC-200L; Nanjing Institute of Electrical and Mechanical Technology) with a measuring range and temperature range ∼0 ± 500 mW and −100°C to 600°C, respectively. The heating rate is in the range 1°C–80°C/min with a temperature resolution of 0.1°C and temperature fluctuation of ±0.1°C.
Vacuum drying oven (DWZ-6050; Kunshan Dongwang Precision Instrument Co., Ltd) with a temperature range 10°C–200°C.
A certain amount of PMMA and AIBN at a ratio of 3:1 was dissolved in cyclohexane and mixed evenly. Under constant stirring, the solution was slowly added into the water phase with a certain amount of emulsifier (OP-10). After 300 revolutions/7 min stirring for 30 min, the mixture was fully emulsified into a uniform oil/water (O/W) emulsion. After the stirring speed decreased to 150 r/min, the PMMA solution was added dropwise to the emulsion for initiating polymerization. After dropwise addition, the temperature was raised to 70°C and incubated for 2 h, and the microcapsules were obtained by filtration and vacuum drying process.
Energy-saving test of coating material containing MEPCM
The prepared phase change microcapsules and Nippon paint were mixed at the ratio of 1:1. For comparative tests, the inner parts of west, east, south, and north walls in one room with dimensions of 2.5 × 2.5 × 3.2 m 3 were painted with the mixed coating material, and the other room was painted with an ordinary coating material. The building envelope of the experimental rooms is internal vacuum with 50-mm thickness, and the thermal conductivity is 0.004 W/m K. The window is low-emissivity (LOW-E) glass with thermal conductivity of 1.0 W/m K. The thermal disturbance in the rooms is the person and computers. The final thickness of coating was ∼3 mm, as shown in Figure 1. A total of 31 T-type thermocouple temperature sensors were installed on the walls, and data were collected by the computer’s collecting module. Both rooms were equipped with 1.5 kW hanging-type air conditioner. The start-up time and power consumption of these two air conditioners were recorded simultaneously. The experimental room is shown in Figure 1.

The tested room with coating material containing MEPCM.
The relevant devices and instruments include the following:
Thermocouple. T-type thermocouple with a measuring temperature range of −200°C to 300°C and an accuracy of ±0.5°C. The thermocouple has high sensitivity and uniform electrode heating.
Data acquisition system for thermocouple temperature. The Remo-8018BL thermocouple module was used for acquiring data, and the data were stored in the access database through the RS-485 communication bus system. The data acquisition cycle can be set by researcher, and the data can be saved automatically. In the experiment, the acquisition interval was set as 3 s.
The instantaneous power and electricity were measured using a S350 smart power measuring instrument. The instrument parameters were set as follows: measuring accuracy, 1.0; temperature range, ∼10°C–60°C; voltage, ∼180–280 V/50 Hz; load current range, ∼0.01–16 A; active power range, 1–4000 W. The minimum resolution for voltage, current, and active power are 0.1 V, 0.01 A, and 0.1 W, respectively.
Results and discussion
Surface morphology of microcapsule
The microcapsules prepared by the reaction were dried at room temperature, followed by drying in a vacuum oven at 30°C for 24 h, and then used for the temperature control and temperature storage tests. The surface morphology of the dried microcapsules was observed by scanning electron microscopy (SEM). Figure 2 presents the surface morphology of the phase change microcapsule at a magnification of 2000 times.

The surface morphology of the phase change microcapsule.
In Figure 2, the microcapsules were regular spheres with a uniform particle size distribution. The surfaces of microcapsules were smooth and compact, with a mean diameter in the range ∼2–3 µm. Figure 2 also shows that only a small amount of resin remains around the microcapsules. Therefore, microcapsules do not adhere, resulting in better encapsulation efficiency and effect on the core material.
The higher encapsulation efficiency represents more PCM in the unit mass of microcapsule, resulting in higher latent heat and better heat storage and temperature control. The current encapsulation efficiency of the prepared microcapsule was is in the range ∼70%–85%, significantly enhancing the heat storage ability of MEPCM. Through the tests, the thermal parameters of MEPCM under different temperatures could be obtained as shown in Table 1.
Thermal parameters of MEPCM.

MEPCM: microencapsulated phase change material.
DSC characterization of phase change microcapsule
The phase change temperature and latent heat of phase change microcapsule were measured characterized by DSC, as shown in Figure 3. Figure 3(a) shows the DSC curve of the pure organic composite PCM, indicating that the phase change occurs in the temperature range 18.5°C–21.8°C, with a higher latent heat up to 206.6 J/g. Figure 3(b) also shows the DSC curve of the phase change microcapsule. Compared with the pure organic composite PCM, the phase change microcapsule, with organic composite PCM as the core material, exhibited the similar temperature variation process, and its phase transformation shifted in the temperature range 22.5°C–33.1°C.

DSC curves of micrographs: (a) pure organic composite PCM and (b) phase change microcapsule.
The heat storage amount per unit mass of microcapsule was distinguished from different mass ratios of the core and wall material. The coefficients of microcapsule are as follows: the mass ratio between the core and wall materials was 7:3, thus the phase change temperature and latent heat of microcapsule were 26.2°C and 139.86 J/g, respectively.
Temperature regulation analysis of MEPCM—the comparison for phase change wall and overall room
When the wall painted with the mixed MEPCM coating material, the heat transfer between the wall and the internal air becomes complex. When the internal temperature is higher, the core of the MEPCM would melt and absorb heat.
On the daytime, because of higher ambient temperature, the MEPCM absorbs and stores heat in the form of the heat transfer process through the wall. Therefore, the heat entering into the room becomes less, and the temperature fluctuation of the room decreased as well. Similarly, at night with lower temperature, the MEPCM undergoes phase change as well. The heat stored in the wall released, thus the indoor temperature fluctuation can be maintained within the limited range. The above analysis indicates that the MEPCM can manifest the energy storage effect of wall, diminish the fluctuation of heat flow between indoor and outdoor, and delay the acting time.
Figure 4 shows the 48-h temperature response curves for inner west wall with or without MEPCM change coating.

The temperature response of west wall.
As shown in Figure 4, the temperature fluctuation of the west inner wall painted with MEPCM was less than that without the MEPCM coating. The temperature difference between them reached up to 4.8°C. Because of the energy storage effect, MEPCM significantly improves and stabilizes the indoor temperature and endows the wall with a stronger ability to prevent heat entering into room during the daytime and prevent heat loss during the night.
Figure 5 presents the comparison of the mean air temperature in the two rooms with and without phase change microcapsule coating. As shown in Figure 5, the coating greatly enhanced the stability of the indoor air temperature, that is, the maximum temperature in the room painted with MEPCM was 6.8°C lower than that in the control room. However, the minimum temperature was 2.2°C higher than that in the control room. Compared with the control room, the time of reaching the maximum and minimum temperatures as 2.5 and 1.5 h, respectively, delayed in the room painted with MEPCM. The above results indicated that the application of the coating material containing MEPCM can improve the temperature condition in the room and enhance the indoor comfort degree.

The mean air temperature response between two rooms with and without MEPCM coating.
Analysis of energy-saving effect of coating material containing MEPCM
In order to investigate the energy-saving effect of coating material containing MEPCM; comparative analysis of the regularity of start-up time, running time, and shutdown time; and power consumption of air conditioners in the rooms with or without MEPCM coating at 10:00–16:00 h on 2 September 2016, the out air temperature during the tested period is shown in Table 2. The results are shown in Figures 6 and 7, and the temperature of indoor air conditioner was set as 25°C. Table 3 also gives the electricity consumption on 8 September and 10 September.
The average out air temperature during the tested period.

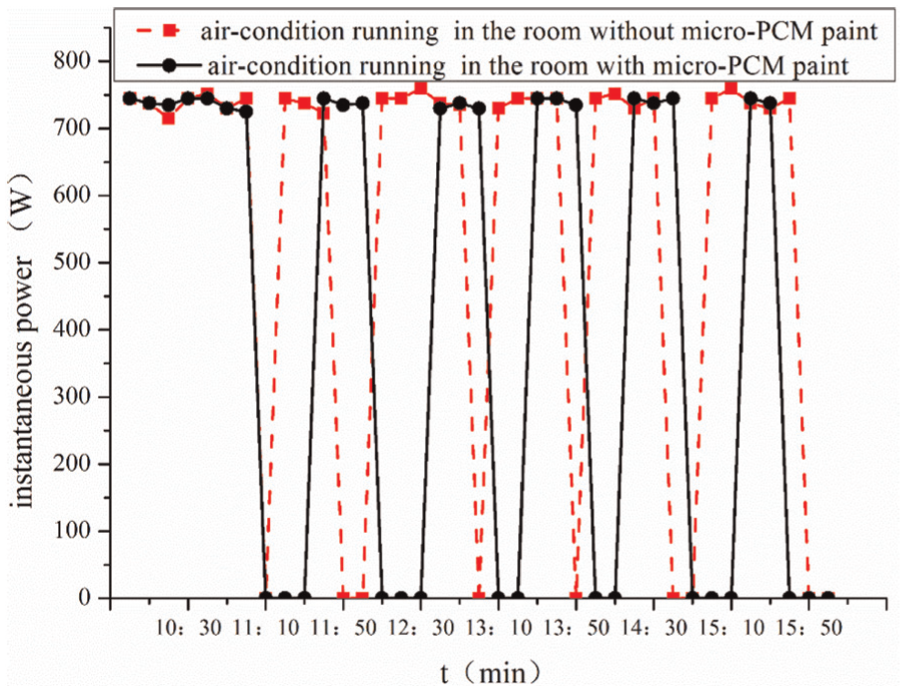
The instantaneous power of the air-condition between the room with micro-PCM and the room without micro-PCM.

The electricity consumption between the room with micro-PCM and the room without micro-PCM.
Electricity consumption on the 8 September and 10 September.

MEPCM: microencapsulated phase change material.
In Figure 6, the start-up time of the air conditioner in the room painted with MEPCM coating was shorter than that of the air conditioner in the room without such a coating. At noon, with the maximum temperature, the air conditioner in the room painted with MEPCM needed to run 30 min and had an intermittence of 20 min, while the air conditioner in the control room had to run 40 min and only had an intermittence of 10 min. The comparison indicates that MEPCM coating increases the indoor thermal inertia, shortens the start-up time of air conditioner, and reduces the energy consumption. Figure 7 shows the electricity consumption comparison for the two rooms in the above time period (10:00–16:00 h on 2 September 2016). Through 6-h running of the air conditioner, the power consumption of the control room was 3.52 kW h, whereas the power consumption of the room painted with MEPCM was 2.62 kW h with an energy saving of 26%. This could also be found in Table 3.
Conclusion
In this study, the phase change microcapsule with organic composite phase change as the core material and the diameter in the range 2–3 µm was synthesized by in situ interfacial polymerization. The phase change microcapsule was mixed with paint for performing the comparative test in two rooms with the dimensions of 2.5 × 2.5 × 3.2 m 3 and the conclusions of this study are as follows:
The phase change microcapsule had a phase change temperature of 26.2°C, consistent to the comfortable temperature of human body. Meanwhile, the phase change microcapsule had a relatively greater latent heat of 139.68 J/g. In addition, the microcapsule had a diameter in the range 2–3 µm and an encapsulation efficiency of ∼75%, appropriate for application in construction material.
The phase change microcapsule was mixed with paint at a ratio of 1:1 and then the comparative tests were carried out in two identical rooms. The results indicated that the temperature fluctuation in the room painted by coating material containing MEPCM was lower than that in the control room with ordinary painting, and the maximum temperature difference can reach up to 4.8°C. The maximum temperature in the room painted with MEPCM was 6.8°C lower than that in the control room, while the minimum temperature in the painted room was 2.2°C higher than that in the control room. For the time reaching the maximum and minimum temperatures, the room painted with MEPCM had 2.5 and 1.5 h delays than the control room, respectively. The aforementioned results indicate that the coating material containing MEPCM improves the thermal environment of the room.
Based on the analysis of the electrical consumptions of these two rooms, during the 6 h with the highest energy consumption in one day, the room painted with the coating material containing MEPCM exhibited an energy saving of 26%.
Footnotes
Academic Editor: Shuli Liu
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

