Abstract
Alkali sulfates and chlorides may result in rapid corrosion attack of the materials during the process of biomass-fired power generation. The 9-mm-thick 12Cr1MoV superheater tube in biomass-fired boiler was cut into samples by wire cutting. Hot corrosion behaviors of the materials in molten NaCl–50% KCl, KCl–50% K2SO4, NaCl–50% Na2SO4, and Na2SO4–50% K2SO4 at 923 K were investigated by the weight change kinetics, scanning electron microscopy, as well as energy-dispersive spectroscopy. The weight gain curve was studied by electronic balance after each experiment cycle at 12, 24, 48, and 60 h. Compared to the microstructures of the corrosion surface and cross section of each material, concentrated pores and cracks were observed on the samples in molten NaCl–KCl, indicating that chlorides induce severest corrosion by active oxidation of chlorine in all cases. There is an extensive decrease in corrosion rates when mixing chlorides and sulfates together, where active oxidation was inhibited by the sulfates. It is interesting to note that sodium (Na) is not as active and detrimental as contents of potassium (K) to the corrosion of 12Cr1MoV steels, which results in the slightest corrosion effects by NaCl–Na2SO4 both on the surface and cross section of the specimens. Therefore, appropriate adjustment on the mixtures of chlorides and sulfates helps to improve the corrosion resistance of 12Cr1MoV.
Keywords
Introduction
12Cr1MoV steel is one of the most utilized high-temperature alloy components widely applied as heat pipe materials in biomass-fired power plant. The low alloying elements content elements makes the steel more economical. Good strength, toughness, and ductility can be obtained by solution strengthening and dispersion strengthening caused by elements’ formation in matrix.1,2 It has been further proved that the steel has a relatively high heat conductivity and good heat shock resistance which enable the material to work at high temperatures for long duration. 3
In the process of thermal power generation, low-grade fuels containing high concentrations of S, Cl, K, and Na are usually used in biomass-fired power generation. During the combustion process, volatile alkali sulfates and chloride vapors from the flue gas stream are easy to combine with other ash constituents, and these low melting point mixtures are condensed to form molten deposits. The molten deposits may cause rapid material damage due to oxidation, sulfidation, chloridation, and even hot corrosion, and the corrosion rate of the steel was accelerated with the increase in the temperature. The alkali molten deposits are the main corrosion factors of heat tubes in biomass-fired boiler.4–7 Sulfates are easy to generate in the reaction of alkali metal elements and gaseous SO3 in the high-temperature oxidation atmosphere, which may react with Fe2O3 and lead to a destruction of oxide film. The diffusion of ions through sulfates is faster than that in oxide, which may result in accelerated attack.8,9 On the other hand, active oxidation of chlorine occurs on the steel surface without protection, which accelerates the oxidation rate of the materials. Chlorine is deleterious to heat tubes in biomass combustion boiler.10,11
The damage due to molten salts’ corrosion of Cr–Mo–V materials has been taken into consideration. The depths of internal attack are more severe and oxide films become easier to spall off with the increase in NaCl in mixed sodium salt conditions. 12 Van Lith et al. 13 studied corrosion in biomass combustion and showed that potassium chloride (KCl) increased the corrosion rate of superheater tube steels. The presence of chlorides allows a salt phase to exist at the metal–scale interface; some investigations have proved that the mixed chlorides lead to a fast corrosion rate.14,15 The corrosion effect induced by molten chlorides is much stronger than by molten sulfates. The presence of sulfur in the form of sulfates caused internal sulfidation of the alloy beneath the external oxide layer, but only small amount of sulfides were formed in corrosion scales. 16 Skrifvars et al. 17 analyzed the corrosion of superheater steel under alkali salt deposits and pointed that neither pure potassium sulfate nor sodium sulfate caused any corrosion of steels when the temperature is lower than T0 (melting point of salts), while the steel was corroded by chlorine-containing sulfate salts even if the temperature is below T0.
Based on the extensive literatures, the studies of molten salts’ corrosion properties on 12Cr1MoV steel are relatively scanty for the reason that most specimens were tested in the gaseous conditions and focused on the reaction between molten chlorides and sulfates, and the detailed relationship between molten sodium–potassium salt mixtures has not been studied extensively. Based on the fact, the objective of this investigation is to study corrosion behaviors of 12Cr1MoV steels in several typical mixed molten alkalis at the working temperature of biomass boiler. Corrosion behavior between different molten alkali mixtures was discussed by corrosion kinetics. The microstructures of the specimens were observed and analyzed by scanning electron microscopy (SEM) equipped with energy-dispersive spectroscopy (EDS).
Experimental procedures
The 12Cr1MoV superheater tube from biomass-fired boiler was cut into 9 mm × 9 mm × 9 mm cube specimens for molten alkali salts’ corrosion experiments by wire electrical discharge machining. The chemical composition of the alloy is shown in Table 1.
Chemical composition of 12Cr1MoV steels (wt%).

The specimens were ground with silicon carbide abrasive papers and then polished to obtain a mirror finish. The specimens for corrosion experiments were coated with the molten mixtures of KCl–50% NaCl, Na2SO4–50% K2SO4, KCl–50% K2SO4, and NaCl–50% Na2SO4 (wt%) separately at room temperature, and then they were sent into a box-type furnace and heated to 923 K. The samples were weighed (±0.1 mg) and measured (±0.01 mm), and a total of 50 g of alkali salts’ mixtures were coated on the surface of the samples and dried at 473 K for 2 h before the experiment. The mixtures were heated in a muffle furnace where the samples were corroded under air atmosphere. During the corrosion processes, the weight change of all the specimens was measured at the time of 12, 24, 48, and 60 h using the AUX120 electronic balance with an accuracy of 1 mg. After the corrosion processes, the etched sections of the samples which were inlaid by cold setting method were ground by a precision polishing machine. The morphology of the corrosion surface and cross sections was scanned by JSM-6360LV scanning electron microscope (SEM) operated at 20 kV. The contents of the corrosive products and characterization of the cross-sectional scales were analyzed by EDS.
Results
Corrosion kinetics of the samples in molten alkali salts’ mixtures
The corrosion kinetics of the specimens in different molten mixtures at 923 K is shown in Figure 1. Figure 1(a) is the weight change versus time. Figure 1(b) shows the depth of the corrosion products versus time, which indicates the degree of molten salts’ corrosion.

Corrosion kinetics of 12Cr1MoV: (a) mass gain versus time and (b) corrosion depth versus time.
It can be concluded that the weight gain of all the samples increases at preliminary stage, while the alloy with chloride mixtures revealed the highest weight change as well as the depth of the corrosion products. After 12 h, the weight of the specimens in four different corrosion conditions is all decreasing. As shown in Figure 1(b), the depth of the corrosion products is increasing in the whole corrosion process. The corrosion depth in molten NaCl–KCl is higher than the other three kinds of mixtures, and in molten Na2SO4–K2SO4 the corrosion depth is the lowest. It can be inferred that molten chloride causes severe corrosion on the surface of the samples than sulfates in high-temperature environment. When compared with the depth of the corrosion products in KCl–K2SO4 and NaCl–Na2SO4, molten sylvite is more active than sodium salts to react with the surface of the specimens. From these research results, the sample in molten NaCl–Na2SO4 shows the slightest corrosion on the oxide film, and the corrosion depth is much less than the sample in molten NaCl–KCl.
Photomicrographs
The surface morphology of the mixed molten corrosion specimens at 923 K after 60 h is shown in Figure 2.

SEM micrograph and EDS analysis of 12Cr1MoV corrosion surface in molten (a) NaCl/KCl, (b) KCl/K2SO4, (c) NaCl/Na2SO4, (d) Na2SO4/K2SO4, (e) EDS spectra a1, (f) EDS spectra b1, (g) EDS spectra c1, (h) EDS spectra c2, and (i) EDS spectra d1.
As shown in Figure 2(a), the steel surface is severely corroded by molten NaCl–KCl and the outer oxide film has been peeled off, which is detrimental to the alloy because it may decrease the corrosion resistance of the alloy. The result of EDS on Spectra.a1 (Figure 2(e)) shows that abundant Fe, O, and a small amount of Cl were detected. From the atom percentage of Fe and O, it can be inferred that the corrosion products mainly consist of Fe2O3. Figure 2(b) shows the surface microstructure and EDS detection of the specimen in molten KCl–K2SO4. Compared with Figure 2(a), the pore structures on the surface are smaller and the corrosion products are more compact. According to EDS detection (Figure 2(f)), Fe, Cr, O, and S were detected, which were induced by K2SO4. Figure 2(c) shows the corrosive surface in molten NaCl–Na2SO4, the granular deposits were observed on the upper left of the figure, and abundant Fe, O, and Cl were detected on Spectra.c2 (Figure 2(h)), which are extremely similar to the products of pure NaCl at 873.15 K. 18 The compact and thick products without obvious defected structures were also observed in Figure 2(c). From Figure 2(g), Fe, Cr, O, and S were all found, but there is almost no Cl on this area. The surface of the specimen in molten NaCl–Na2SO4 is totally different from that in KCl–K2SO4; thus, this is the cause relating to potassium (K) or sodium (Na). The morphology of the surface in Na2SO4–K2SO4 is shown in Figure 2(d), and the compact and hard corrosion products without pores were observed. Fe and S are the main elements on the surface, both of which are higher than O by Figure 2(i) EDS analysis. It can be inferred that there is a different corrosion mechanism induced by molten sulfates and chlorides.
Cross sections of the corroded samples were used to examine the scale density, adherence to the metal, and the nature of metal–scale interface. The SEM micrograph of the cross section of the 12Cr1MoV alloy corroded by molten NaCl–50% KCl at 923 K and the corresponding EDS spectra of Fe, O, Cr, Mn, and Cl are shown in Figure 3.

SEM micrograph and EDS spectra of 12Cr1MoV cross section in molten NaCl–50% KCl.
The evident cracks which inclined to separate the substrate are observed below the corrosion surface (as shown in Figure 3). EDS analysis (Spectra.1) shows a large spread of O and Cl in this area and also indicates that the layer has been severely corroded. A large amount of Fe, O, and Cl was also detected in Spectra.2. In this case, it can be inferred that the depth of the corrosion layer is approximately 600 µm where the thickness of the oxide film is not included. Numerous pores were found on the loose layer, which are detrimental to the alloy because the pores destruct the integrity of the whole structure. The pore structures are mainly caused by the penetrating gas which is induced by the reaction of molten salts and substrate elements in the inner side of the samples. Figure 4 shows the microstructures and EDS spectra of cross section of 12Cr1MoV steel corroded by the molten KCl–50% K2SO4.

SEM micrograph and EDS spectra of 12Cr1MoV cross section in molten KCl–50% K2SO4.
Compared with the samples in molten chloride mixtures, the destructive structures such as cracks have not been observed on the corrosion layer. However, clear grains and grain boundaries indicate that the feature of the intergranular attack is more pronounced as the content of K2SO4 increased. This result is similar to others’ research that grains and grain boundaries were also proved to be corroded by Na2SO4.12,19 Fe and O are the most abundant elements in Figure 4 Spectra.1, and S and Cl were also observed on the oxide films of the sample. With the increasing depth of corrosion, the effects induced by molten salts gradually weakened. Meanwhile, when the sulfates and chlorides mixed together, the active oxidation by Cl will be inhibited, that is why the Cl and S cannot be found as shown in Spectra.2 and Spectra.3. The average depth of the corrosion layer is about 170 µm which is extensively lower than in chloride mixtures.
The micrograph and EDS spectra of cross section of the corrosive specimen in NaCl–50% Na2SO4 are shown in Figure 5. It has been reported that NaCl shows a typical uniform internal attack, 12 while no deep corrosion layers were observed under the corrosion surface for the reason that a part of the corrosion products is peeled off by the strong corrosion of molten salts.

SEM micrograph and EDS spectra of 12Cr1MoV cross section in molten NaCl–50% Na2SO4.
In Figure 5 Spectra.1, a large amount of detected Fe, O, Cr, and Mn indicates a severe corrosion of the layer. Spectra.2 is the substrate inferred from the elements’ distribution. The presence of NaCl with different mass ratios in the mixtures of NaCl–Na2SO4 has a greater corrosion impact on high chromium content alloys. 20 The addition of NaCl in Na2SO4 coatings can cause the cracking of protective Cr2O3 layers and increase the amount of sulfur incorporated into the substrate, accelerating the corrosion of alloys. However, the material used for the experiment is a kind of low-alloy steel. With a relatively low ratio of alloy elements, destruction mechanisms can be different from previous literature. Meanwhile, the depth of the layer corroded by molten NaCl–50% Na2SO4 is much lower than that by molten KCl–K2SO4. Combined with the corrosion kinetics, it can be further inferred that there is a big difference between corrosion effects by K and Na. Figure 6 shows the cross-sectional image and the corresponding EDS analysis of 12Cr1MoV corroded by Na2SO4–K2SO4 mixtures at 923 K.

SEM micrograph and EDS spectra of 12Cr1MoV cross section in molten Na2SO4–50% K2SO4.
In Figure 6, cracks and scattered pores with different sizes were observed on the corrosion layer. The area near the corrosion surface (Figure 6, Spectra.1) was thoroughly corroded, but S was still not detected. The corrosion effect in Spectra.2 is slighter and Figure 6 Spectra.3 is from the substrate area, from which the average depth of the corrosion layer was observed to be about 500 µm. Corrosive grains and grain boundaries were found in the corroded layer, especially at the bottom of the layer, meaning the intergranular attack, through the molten sulfate mixtures’ corrosion.
Discussion
Generally, molten salts’ corrosion can be simplified as two steps: oxidation of the specimen and then dissolution of the protective oxide films by molten salt deposits after a basic reaction; 21 similar study results were obtained by Fryburgh et al. 22 in IN-738 in molten Na2SO4. This well explains the phenomenon of the kinetics in Figure 1(a). When the corrosion layer separates from the substrate, Fe and Cr in the corrosion products will react with the mixtures and accelerate the dissolution into molten salts. In this process, the mass of the sample decreases rapidly as time goes on. From the comprehensive results of corrosion kinetics, SEM micrographs, and EDS analysis, it can be concluded that the mixtures of NaCl–KCl generate the highest rate and depth of corrosion. Cracks and concentrated pores extensively deform the structure of the materials and result in the obvious separation of layer areas and substrate. The corrosion depth of the specimen in Na2SO4–K2SO4 is approximately 500 µm which shows that molten sulfate can also generate severe corrosion while the effect is weaker than molten chlorides. The microstructure of the layer is quite different between these specimens in the other two different corrosion environments. The layer is compact and there is no obvious separation. Corrosion by molten sulfates is partial to intergranular corrosion while the molten chlorides induce internal destruction. However, when the chlorides and sulfates were mixed together, both corrosion effects and weight loose decreased no matter in the mixtures of sodium salts or sylvite. NaCl–Na2SO4 shows the lowest corrosion rate from all the samples. It is concluded that there is strongest destruction on 12Cr1MoV steels in molten chlorides, and molten sulfates take the second place. For the conditions provided, a possible reason why molten chlorides induce the highest corrosion rate is the active oxidation of chlorine. When the oxide scale on the steel surface has no protective character and function, chloride contaminants may strongly accelerate its oxidation rate. Based on Grabke et al.’s 23 study, the main reactions involved in the active oxidation mechanisms of 12Cr1MoV steels can be summarized as a cycling reaction (M = K, Na)
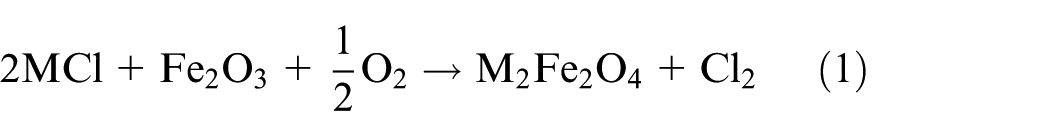
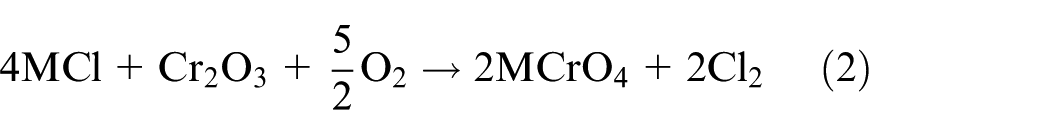

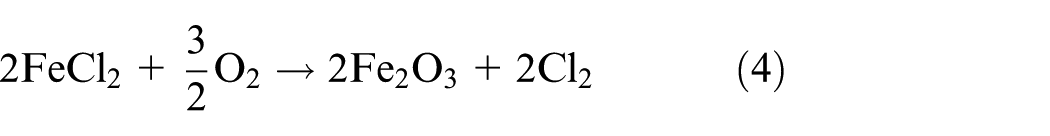
According to equations (1)–(4), diffused Cl2 has the greatest driving force to break through the surface and corrosion layer, where concentrate cracks and pores were formed after the penetration of Cl2. After the reaction in equation (4), the generated Fe2O3 reacts with the molten chlorides (MCl) again. Comparing with the mechanism of molten chloride mixtures, the high-temperature sulfate corrosion will be taken place due to the reactions of equations (5) and (6).19,24 Meanwhile, gaseous SO3 is easily generated in high temperature after oxidation of SO42− in equation (7), 19 which can dissolve the oxide films at the metal interface, as shown in equation (8) 24




If there is no protection of the oxide film, Fe and Cr in the substrate will be exposed to the mixtures. The molten sulfates are easy to penetrate into the alloy and the low melting point metal sulfates may be generated, which may lead to further corrosion on the 12Cr1MoV steels. As known in previous study, there is an extensive decrease in corrosion when chlorides and sulfates are mixed together. The reactions are described in equations (9) and (10). With participation of the reaction product SO2, the sulfate can generate from chloride in the reaction equation (10)


The changed mixtures in equation (9) can also react with Fe2O3 as shown in equation (5), making the adherence of stable and compact Fe2(SO4)3 on the corrosion surface, as shown in Figures 5 and 6, which hinder the corrosion effects by molten chlorides. The corrosion rates of the specimens are also lower in this process. Most Cl in the reaction is distributed on the surface and Cl2 will diffuse out of the sample. Once Cl2 cannot permeate into the inner side of the specimen, the corrosion destruct will become slight. This is why Cl and S were observed on the surface while it could not be detected on the corrosion layer.
Conclusion
The corrosion behaviors of 12Cr1MoV steel in mixed molten alkalis were investigated in this study, and the following conclusions can be drawn:
Based on the study of weight change and corrosion depth after 60 h of corrosion in four kinds of different mixed molten alkalis, the corrosion rate of the 12Cr1MoV specimens is the following order: NaCl–50% KCl > K2SO4–50% Na2SO4 > KCl–50% K2SO4 > NaCl–50% Na2SO4.
Molten chlorine salts are the main corrosion influence factors in high-temperature corrosion comparing with the mixtures of NaCl–KCl and Na2SO4–K2SO4. The presence of active oxidation by molten chlorides induces cracks and pores on the corrosion layer, which is more serious than molten sulfates.
Intergranular corrosion in molten sulfates’ mixtures is more obvious, and the corrosion induced by molten KCl–K2SO4 is lower than NaCl–Na2SO4.
Footnotes
Academic Editor: Yongming Liu
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51275058) and College Innovation Platform Project of Hunan Province (13K052).
