Abstract
The aim of this article is to study the effect of depth of phase change material over the absorber surface of an integrated collector-storage type flat plate solar water heater. Flat plate solar water heaters are extensively used all over the world to utilize the natural source of solar energy. In order to utilize the solar energy during off-sunshine hours, it is inevitable to store and retain solar thermal energy as long as possible. Here, phase change material is not used for heat storage, but to minimize losses during day and night time only. The depth of phase change material over a fixed depth of water in a solar thermal collector is an important geometric parameter that influences the maximum temperature rise during peak solar irradiation and hence the losses. From the results of the studies for different masses of paraffin wax phase change material layers, the optimum depth corresponding to the maximum heat gain till evening is found to be 2 mm, and the heat retention till the next day morning is found to be 4 mm.
Keywords
Introduction
Heat storage is considered as a serious option for saving energy these days. Heat storage makes it possible to use heat energy at the time of demand. Solar water heating for residential and industrial purposes is the best alternate to conserve conventional energy sources, thereby high-grade electrical energy and fossil fuels can be conserved due to the increase in their prices. A conventional solar water heater is costly, bigger and occupies more space because of copper heat absorber tubes and separate insulated thermal storage unit, but retains heat during night time better. On the other hand, integrated collector storage–type solar water heater is compact and cost-effective when compared to conventional flat plate solar water heaters (FPSWHs), gains heat directly and quicker than a conventional solar water heater, but loses heat to the surrounding faster because of the difficulty that a permanent insulation cannot be provided over the absorber surface. Providing a transparent insulation can improve the efficiency and temperature achieved, but its cost, durability and availability need to be clarified. Elimination of copper tubes and providing a floating absorber surface made of galvanized iron (GI) sheet which is directly in contact with water can minimize cost. Phase change materials (PCMs) can be used in solar water heaters to enhance heat storage and discharge of heat at a later period when required. Providing a layer of PCM and optimizing the depth of PCM are the parameters that can be used to enhance the performance of FPSWH.
Mohsen et al. 1 conducted experiments on compact solar water heater for water depths of 5, 10 and 15 cm and concluded 10 cm as the optimum water depth. Single glazing showed better water temperature rise and double glazing retained heat better. 1 Sridhar and Reddy 2 conducted experiments on cuboidal solar integrated collector storage system for depths of 2, 5, 8 and 12 cm and inclination angles of 10°, 20°, 30°, and 50°. The average heat transfer coefficient at the bottom surface of the absorber plate is 20% higher for depth of 12 cm as compared to the 2 cm depth of cuboidal section, after 2 h of heating.
Agbo and Unachukwu 3 concluded that the loss coefficient decreases with increase in the gap between the absorber plate and the glass cover. A gap width ≥5 cm is recommended for optimum loss coefficient. 3 HP Garg et al. 4 reported that the storage potential of built-in storage–type solar water heater with transparent insulation is higher than that of a system with moveable insulation. The decrease in transmittance of the transparent insulation more than offsets its better insulating property during the sunshine hours.
Tarhan et al. 5 studied the temperature distribution in a trapezoidal built-in storage–type solar water heater with and without PCMs. Lauric acid, stored in a storage unit that was also used as a baffle plate, considerably reduced the peak temperatures during the trials, but had small effects on the dip temperatures. Therefore, lauric acid can be used to stabilize the temperature and reduce the necessary volume of the water tank during the day. It cannot be very effective to retain the water temperature during the night. Myristic acid, stored in a storage unit that was also used as an absorbing plate, was more effective to retain the water temperatures during the night since it solidified at 51°C–52°C water temperature and acted as a thermal barrier. However, the optimum thickness of PCM should be determined experimentally or by computer simulation.
Talmatsky and Kribus 6 compared the performance of a storage tank with PCM to a standard tank without PCM, with realistic environmental conditions and typical end user requirements imposed. Annual simulations were carried out for different sites; load profiles different PCM volume fractions and different kinds of PCM. The results were contrary to the expectations that the use of PCM in storage tank does not yield significant benefit in energy provided to the end user. PCM used was arranged in small cylindrical containers. However, it was suggested that optimizing the geometry of PCM might be beneficial.
Shukla 7 proposed an FPSWH with PCM lining walls which had an efficiency of 45%. Prakash et al. 8 suggested that the use PCM underneath the tank stores and discharges heat during off-sunshine hours but exhibited poor heat transfer characteristics. Rabin et al. 9 designed an integrated solar collector storage system based on salt hydrate. He used a salt hydrate eutectic mixture (48% CaCl2, 4.5% KCl, 0.4% NaCl and 47.1% H2O) approximately 2.59–3.45 times greater than that of conventional solar water heater. For a shallow rectangular vessel with a high-surface-area-to-volume ratio, the incident insolation has a small depth of water to heat up. However, a store with a large exposed surface area will also lose substantial amounts of heat by convection and long-wave radiation during normal conditions and will cool down significantly by radiation losses to the night sky.
Mettawee and Assassa 10 conducted experiments on a right triangular-shaped solar water heater with PCM for latent heat storage with an effective absorber surface area of 0.2 m 2 , having a 12 mm diameter water pipe at its centre. The propagation of melting during charging and solidification during discharge was studied. The effect of mass flow rate on useful heat gain was studied. The experimental results showed that in the charging process, the average heat transfer coefficient increases sharply with increasing the molten layer thickness, as the natural convection grows strong. In the discharge process, the useful heat gain was found to increase as the water mass flow rate increases.
Kurklu et al. 11 designed and developed a solar collector, with solar aperture area of 1.44 m 2 , consisting of two adjoining sections one filled with water and the other with a PCM with a melting and freezing range of about 45°C–50°C, that is, paraffin wax. Covering the collector surface with an insulation blanket at a time when the water temperature was at its maximum improved the energy conservation of the water significantly. The instantaneous thermal efficiency values were between about 22% and 80%. This solar collector was much advantageous over the traditional solar hot water collectors in Turkey in terms of total system weight and the cost in particular.
Enibe 12 conducted experiments on single-glazed flat plate collector of area of 1.503 m 2 integrated with paraffin-type PCM energy storage in slender rectangular channels at the bottom of absorber plate. The space between each module pair serves as an air heating chamber, the chambers being connected to common air inlet and outlet header manifold. The predicted results were compared with the experimental results. The maximum predicted cumulative useful and overall efficiencies of the system are within the ranges 2.5%–13% and 7.5%–18%, respectively.
A comprehensive study is essential to investigate the performance of integrated collector storage–type FPSWH over a wide range of PCM depth data to compare and evolve the optimum depth of PCM for better heat transfer characteristics. Varieties of PCMs are available for latent heat storage and discharge. Paraffin wax, lauric acid, myristic acid and so on are some of the PCMs used in earlier researches. Paraffin wax is available over a wider range of temperature. Lauric acid has a melting temperature of 49°C and latent heat of fusion of 178 kJ/kg. Myristic acid has a melting temperature of 58°C and latent heat of fusion of 199 kJ/kg. Paraffin wax with a melting temperature of 50°C and latent heat of fusion of 189 kJ/kg is selected, because its melting temperature enables quick melting of PCM, maintains a lesser mean temperature of absorber surface, to reduce loss coefficient, and solidifies during night time below 50°C forming a low thermal conductivity insulation layer of solid, to reduce losses during night time, and also because of cheap and easy availability. The novel idea of finding the optimum depth of paraffin wax PCM for maximum heat gain till evening and that for maximum heat retention till the next day morning was acquired from Tarhan et al. 5 that showed the way to the originality of this research work.
Experimental set-up
A schematic diagram of the experimental set-up is shown in Figure 1. The compact solar water heater model is of dimension 0.5 m × 0.5 m × 0.18 m made of mild steel sheet, filled with water to a desired depth. The insulation provided is polyurethane of 0.05 m thickness with an outer layer of 0.017 m thick plywood to minimize convection heat losses to the surrounding. A single glazing of 4 mm thickness is provided on the top. A floating absorber plate made of GI sheet of 20 gauge thickness in the shape of a square tray of dimension 0.5 m × 0.5 m × 0.03 m is used. The floating absorber plate is in 100% contact with the entire water surface. The emissivity and absorptivity of the glazing cover are taken as 0.1, and the transmissivity of the glazing cover is taken as 0.9. The emissivity of the absorber plate painted with black enamel paint is taken as 0.8. Resistance thermal detectors (RTDs) with an accuracy of ±0.3°C are positioned at the top, middle and bottom layers of water in the integrated collector storage–type movable flat plate solar water heaters (ICSMFPSWH).

Cross-sectional view of three integrated collector storage–type movable flat plate solar water heaters.
The experiments were conducted simultaneously at a time on a given day with three geometrically similar FPSWH models as described above with different depths of PCM over the absorber plate and constant 10 cm depth of water underneath the absorber plate. The distance between the absorber plate and the glass cover was maintained 5 cm or greater for optimum loss coefficient as referred by Agbo and Unachukwu. 3 For distances above 5 cm, the variation in loss coefficient was negligible for variation in distance. Hence, for depth variation by a centimetre makes no significant difference in loss coefficient.
Figure 2(a) shows the photograph of the experimental set-up consisting of three geometrically similar ICSMFPSWH models, fabricated for conducting experiments with different water depths with same solar insolation input. Radiation sensor and RTD were connected to a 16-channel universal data acquisition system. The process logger shown in Figure 2(b) accepts the input of RTD, thermocouple, voltage and current pulse. The logger is suitable for sensing signals of temperature, pressure, humidity, CO2, level, flow, r/min, voltage and current. In all the three ICSMFPSWH models, with different depths of PCM, the temperatures at the bottom of water were labelled T1, T2 and T3, at the middle of water were labelled T4, T5 and T6 and the top of water were labelled T7, T8 and T9, respectively. T1–T9 were located at the mid-point of the water storage tank. The ambient temperature was sensed by thermocouple labelled T10, and radiation data (I) in W/m2 were sensed by a solarimeter shown in Figure 2(b) and recorded by connecting to channel 11 of data logger, every minute for a period of 24 h from 6:00 a.m. on a given day to 6.00 a.m. the next day. The average water temperatures Twm1, Twm2 and Twm3 were calculated by calculating the mean of the temperatures measured at the bottom, middle and top of water in storage tank in the three models, respectively.

(a) Photograph of three geometrically similar ICSMFPSWH models, temperature sensor cables, radiation sensor and data acquisition system. (b) Photograph of radiation sensor and data acquisition system.
Measuring equipments and their characteristics
A 16-channel universal data acquisition system was used to record the temperature and radiation measurements periodically. The process logger accepts the input of RTD, thermocouple, voltage, current and pulse. The logger is suitable for sensing signals of temperature, pressure, humidity, CO2, level, flow, revolutions/min, voltage and current.
Solar radiation sensor ‘Pyra 300V1’ model was used to measure global radiation, the point of measurement of both the direct and diffuse solar irradiance. The sensor’s transducer, which converts incident radiation to electric current, is a silicon photodiode with wide spectral response. From the sensor’s output voltage, the console calculates and displays solar irradiance. The output is 0–5 V direct current (DC) for 0–1800 W/m2 range. Its operating temperature range is −40°C to +65°C and range is 0–1400 W/m2.
PT100 sensors were used to sense the temperature at different locations. PT100 sensors can be used to sense temperatures up to approximately 250°C.
Effect of uncertainties and errors on the parameters
The uncertainty of the 16-channel universal data logger equipment specified by the supplier is 0.15% of full scale deflection (FSD). The uncertainty of the solar radiation sensor is ±2% of FSD. The accuracy of Pt100 sensors is ±0.3°C. In order to nullify the error in temperature measurements, all thermocouples were kept at a single point in atmosphere and tested to sense the ambient temperature.
A series of temperatures were measured using RTD sensors and a digital thermometer. Relative uncertainty is calculated using the difference between the temperature sensed by RTD and that by digital thermometer divided by the temperature sensed by digital thermometer. The average value of uncertainty of temperature measurement is ±4%. Each channel is brought to calibration mode and the fractional variations if any were nullified in the data logger so that the sensed parameters are reliable and accurate, and the effects of uncertainties are negligible.
Results and discussion
Variation in water temperature for a fixed water depth of 10 cm and for different PCM masses, ambient temperatures and solar insolation
Figures 3–8 show the variation in the mean water temperature for different masses of PCM, ambient temperatures and radiation data on the respective dates of experiment conducted. It was observed from the graphs that the mean water temperature variations agree with the changes in radiation data due to clear sky or sudden cloud covers. The ambient temperature was found to drop during short rain showers and then increase gradually later. Increasing the mass of PCM and hence the depth of PCM offer greater resistance to heat transfer during both day and night time. The radiation for melting the PCM is greater than that could be transferred to water for heating it. On a day with low average radiation, the model with higher mass of PCM could gain lesser heat since PCM remains solid for greater day period and hence resisting heat transfer. On the other hand, on a day with high average radiation, the model with PCM mass melted quickly, and hence heat transfer to water improved. PCM melts and maintains a low absorber plate temperature thereby reducing the loss coefficient during day time. During night time, the water temperature decreases because heat loss takes place by both convection and radiation mode to the surrounding. Once the temperature drops below the melting point of PCM, that is, below 50°C, PCM mass solidifies. Since the thermal conductivity of paraffin wax is low, it offers resistance heat transfer thereby reducing the heat loss to the surrounding from then onwards.

Variation in Twm1 without PCM, Twm2 with 400 g of PCM and Twm3 with 800 g of PCM on 30 July 2014.
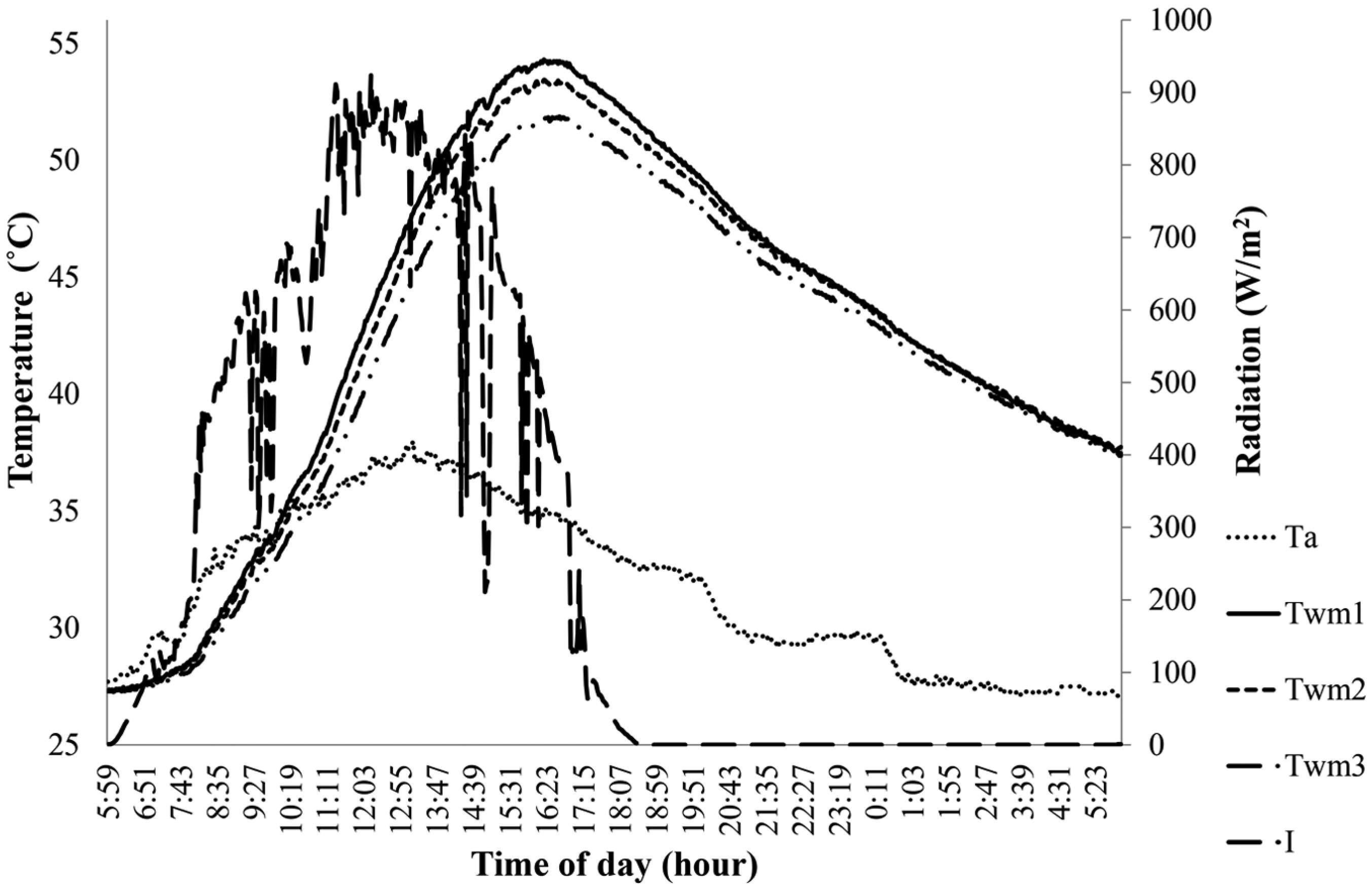
Variation in Twm1 without PCM, Twm2 with 700 g of PCM and Twm3 with 1100 g of PCM on 5 August 2014.

Variation in Twm1 without PCM, Twm2 with 700 g of PCM and Twm3 with 1400 g of PCM on 8 August 2014.

Variation in Twm1 without PCM, Twm2 with 800 g of PCM and Twm3 with 1400 g of PCM on 12 August 2014.

Variation in Twm1 without PCM, Twm2 with 800 g of PCM and Twm3 with 1600 g of PCM on 16 August 2014.
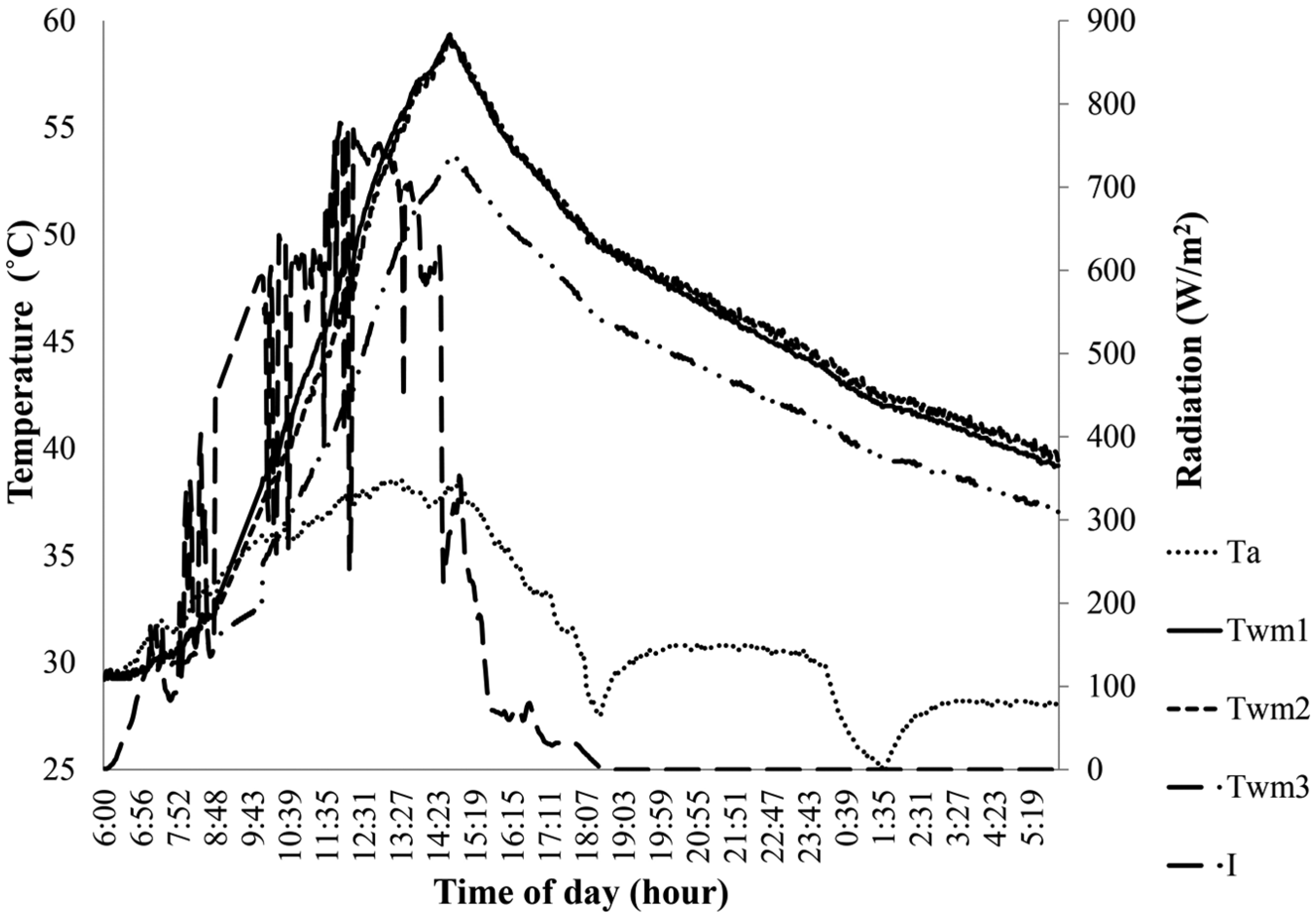
Variation in Twm1 without PCM, Twm2 with 900 g of PCM and Twm3 with 1700 g of PCM on 20 August 2014.
The following details show the difference in the mean water temperature after 12 h among the three models with different masses of PCM over the absorber surface. Figure 3 shows that the model with 400 g of PCM has 0.1°C greater than that without PCM and 1.2°C greater than that with 800 g of PCM. Figure 4 shows that the model without PCM has a mean water temperature 0.9°C greater than that with 700 g of PCM and 2.1°C greater than that with 1100 g of PCM. Figure 5 shows that the model with 700 g of PCM has a mean water temperature of 0.5°C greater than that without PCM and 1.3°C greater than that with 1400 g of PCM. Figure 6 shows that the model with 800 g of PCM has 0.8°C greater than that without PCM and 2.1°C greater than that with 1400 g of PCM. Figure 7 shows that the model without PCM has the same temperature as that with 800 g of PCM and 1.9°C greater than that with 1600 g of PCM. Figure 8 shows that the model with 900 g of PCM has the same temperature with that without PCM and 3.5°C greater than that with 1700 g of PCM.
The following details show the difference in the mean water temperature after 24 h among the three models with different masses of PCM over the absorber surface. Figure 3 shows that the model with 800 g of PCM has 0.6°C greater than that with 400 g of PCM and 0.2°C greater than that without PCM. Figure 4 shows that the model with 700 g of PCM has 0.3°C greater than that without PCM and 0.4°C greater than that with 1100 g of PCM. Figure 5 shows that the model with 700 g of PCM has 0.7°C greater than that without PCM and 0.1°C greater than that with 1400 g of PCM. Figure 6 shows that the model with 800 g of PCM has 1.3°C greater than that without PCM and 0.1°C greater than that with 1400 g of PCM. Figure 7 shows that the model with 800 g of PCM has 1.2°C greater than that without PCM and 1.8°C greater than that with 1600 g of PCM. Figure 8 shows that the model with 900 g of PCM has 0.4°C greater than that without PCM and 2.8°C greater than with 1700 g of PCM.
Variation in water temperature at 18:00 h for different depths of PCM and constant water depth of 10 cm
From Figure 9, it was observed that the water temperature in the evening decreases with an increase in PCM mass with curves of negative slope. It was observed that for most of the cases with PCM mass below 600 g, the slope of the trend line is positive, and for those above 600 g, the slope of the trend line is negative showing inversion in heat gain.
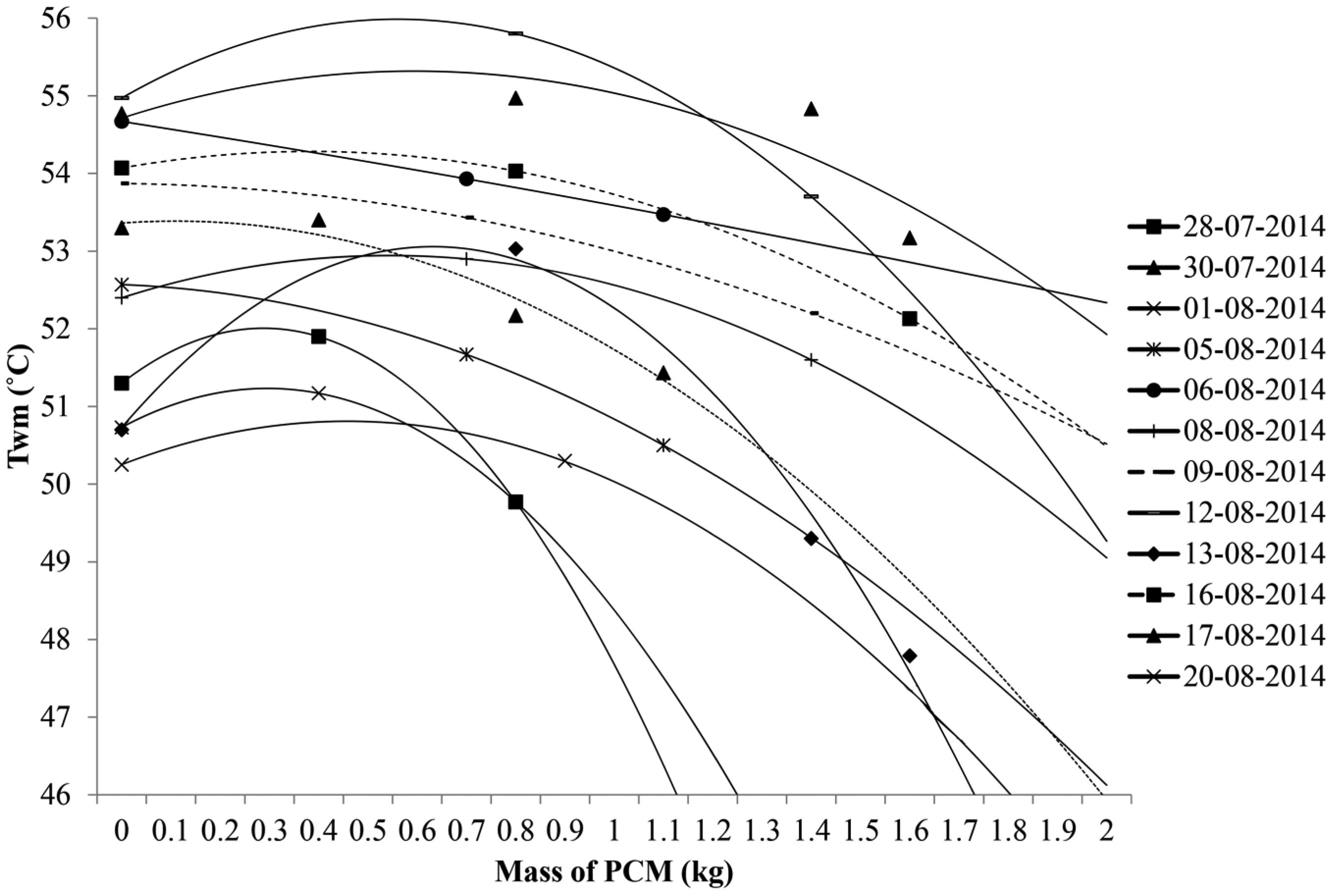
Variation in mean temperature of water in the evening for different PCM masses on different days.
Since three temperature points may not give an accurate curve fit, a fourth point has been added to few possible days of experimental data in developing the trend curves by normalizing the values of data where the variation in initial temperature data is less than 1°C and the average radiation data are less than 30 W/m2. Heat gain by water and hence temperature rise is directly proportional to the average useful radiation. Radiation heat gain is directly proportional to the fourth power of temperature in Kelvin. By nullifying the initial water temperature and calculating the proportionate change in temperature for small variation in radiation data, the mean water temperature is predicted for a given average radiation data and mass of PCM on a different day. The following data are added as fourth point in Figure 9: 51.4°C for 1100 g mass of PCM on 30 July 2014, 47.8°C for 1600 g mass of PCM on 13 August 2014, 55°C for 1400 g mass of PCM on 17 August 2014 and 47.3°C for 1600 g mass of PCM on 20 August 2014.
Variation in water temperature the next day morning for different depths of PCM and constant water depth of 10 cm
From Figure 10, it was observed that the water temperature the next day morning decreases with an increase in PCM mass in the negative slope region of the trend curves. It was observed that for most of the cases with PCM mass below 800 g, the slope of the trend line is positive, and for those above 800 g, the slope of the trend line is negative showing inversion in heat retention. The fourth point has been added to few possible days of experimental data in developing the trend curves. The following data are added as fourth point in Figure 10: 38.7°C for 1100 g of PCM mass on 30 July 2014, 37.2°C for 1700 g of PCM mass on 16 August 2014, 40°C for 1400 g of PCM mass on 17 August 2014 and 37.8°C for 1600 g of PCM mass on 20th August 2014. Although three curves showing completely opposite trend may be well within the error of the measurements, they are reported as recorded.

Variation in mean temperature of water the next day morning for different PCM masses on different days.
From Figures 9 and 10, trend line equations for different PCM mass combinations in both positive and negative slope regions alone were differentiated and equated to zero, to mathematically predict the average of the optimum depth of PCM depth for maximum heat gain in the evening and that for maximum heat retention till the next day morning.
Direct and quantitative comparisons of the present experimental results with earlier studies are restricted because of differences in sizes and configurations of solar water heaters and differences in climatic conditions and location in those studies. Hence, broad and qualitative comparisons were made.
Tarhan et al. 5 reported that the peak temperature achieved with myristic acid (as insulator) PCM at the top, lauric acid (as storage unit) and that without PCM were (61.5°C–66.6°C), (50.9°C–55.9°C) over those (59.9°C–65.1°C) without PCM. In our present studies, the peak temperatures achieved were 63.7°C, 60.2°C and 63.2°C, respectively, with 2 mm and 4 mm paraffin wax PCM and without PCM, respectively. For comparably similar melting temperature range of myristic acid and paraffin wax, the peak water temperatures achieved agree well although experiments were conducted on different dates, location and climate.
Prakash et al. 8 used 4 cm thick layer of paraffin wax metallic capsules at the bottom of solar water and reported that the water temperature with PCM was 5°C than that without PCM after 21:30 h. In our present studies, the water temperature was 0.3°C and 0.4°C higher with 2 mm and 4 mm thick layers of paraffin wax, respectively, at the top of absorber plate when compared with that without PCM since the objective of providing PCM here is not for latent heat storage.
The results of the study by Kurklu et al. 11 indicated that the water temperature exceeded 55°C with paraffin wax on the top during a typical day of high solar radiation and it was kept over 30°C during the whole night. The maximum water temperature achieved in this study was 63.7°C and was kept over 38°C during the whole night.
Variation in efficiency of ICSMFPSWH models with time
From Figure 11, it was observed that the efficiency increase is very steep at the beginning of the day due to the fact that the lesser the water temperature, the greater is the heat gained and hence efficiency. The rise and fall in efficiency data well agree with rise and fall in solar radiation due to clear sky or sudden cloud cover. During initial hours of the day, the model with 800 g of PCM absorbed more heat than the other two models for sensible and latent heat requirement. The absorber surface temperature of the model without PCM was greater than that of the model with 400 g of PCM. The model with 400 g of PCM was able to achieve maximum heat transfer to water because of its optimum PCM layer thickness. Hence, the efficiency of the ICSMFPSWH model with 400 g of PCM was greater than that without PCM, while that without PCM was greater than that with 800 g of PCM during initial hours.

Variation in efficiency of ICSMFPSWH model without PCM, with 400 g of PCM and with 800 g of PCM over the absorber surface on 1 August 2014.
The ICSMFPSWH model with 800 g of PCM takes more time to melt and achieved lesser water temperature than that with 400 g of PCM. Hence, during late hours of the day, the efficiency of the model with 800 g was greater than that with 400 g of PCM, while that with 400 g of PCM was greater than that without PCM.
Figure 12 shows the average efficiency of the system on different days with different masses of PCM over the absorber surface. The average efficiency of the systems for the day is 44.7%, 43.9% and 41.9% for the ICSMFPSWH models with 400 g of PCM, without PCM and with 800 g of PCM, respectively.

Variation in average day efficiency of ICSMFPSWH models with different masses of PCM over the absorber surface.
Chauhan and Agarwal 13 conducted experiments on a batch-type integrated collector storage system of FPSWH, with two black painted cylindrical absorber-cum-storage unit inside a flat plate solar collector. The average efficiency of system per day was reported to vary between 24.72% and 36.66%. The maximum efficiency reported was 70.34%. In our present studies, the average efficiency of system per day was reported to vary between 41.9% and 62.8%, which is in good agreement and well above the values reported early, since direct quantitative comparison is not possible due to the fact that the experiments were conducted on different location, time and of different specification.
Error analysis of experimental data is done using propagation of error rules to find the errors in the final results. Variance is the square of standard deviation. Variances are added on addition or subtraction. Relative variances are added on multiplication and division. Based on these error rules, the temperature difference varies by ±0.4°C, heat transfer calculated varies by ±42 kJ of energy and efficiency of the ICSMFPSWH models calculated varies by ±2%.
Conclusion
For effective retention of solar thermal energy, a layer of paraffin wax PCM provided on the top of the absorber plate reduces the mean temperature of the absorber plate and hence the losses during day time. Solidification of PCM during night time offers thermal resistance because of low thermal conductivity of solid PCM and hence reduces losses during night time. The optimum depth of PCM that corresponds to the maximum temperature and heat gain increases with an increase in average radiation on a given day. Similarly, the optimum depth of PCM decreases with an increase in the difference between the initial mean water temperature and initial ambient temperature. In general, it is found that a 2-mm-thick paraffin wax PCM with melting point around 50°C will gain maximum heat in 12 h day time and that with 4 mm thickness will retain maximum heat in 24 h, that is, the next day morning.
Footnotes
Appendix 1
Academic Editor: Jiin-Yuh Jang
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
