Abstract
In response to the difficulties in machining cemented carbide, this paper proposes a new approach to electrochemical milling of cemented carbide. A composite rotating tool cathode for electrochemical milling is designed, and the electric field simulation calculation is conducted for the electrochemical milling process. The electric field results show that as the tool cathode continues to penetrate, the machining area of electrochemical milling continues to increase, and the current density in the machining gap increases. After the tool cathode enters the semicircle, if the processing area of electrochemical milling remains unchanged, the amount of material removed per unit time remains unchanged, and the current density also remains stable. At the same time, orthogonal experiments and process parameter optimization were conducted on the electrochemical milling of cemented carbide side edges. The results showed that the maximum material removal was achieved under the process parameters of processing voltage 14 V, feed speed 10 mm/min, spindle speed 3000 r/min, and duty cycle of 70%. Based on the optimized process parameters of side-edge electrochemical milling, full edge electrochemical milling of experiment was carried out. When the feed rate is 0.3 mm/min, the surface of cemented carbide electrochemical milling is relatively flat and has a roughness of 0.389 μm.
Introduction
Cemented carbide is widely used in industrial production because of its high hardness, high strength, and excellent wear and corrosion resistance. However, cemented carbide is a brittle material. In the traditional machining process, it is affected by stress, high temperature, and corrosive medium, causing formation of machining defects such as stress concentration, cracking, and burn. As a result, if the cemented carbide workpiece is defected, use requirements cannot be met. In order to solve this problem, electrochemical mechanical compound machining methods have received great attention in cemented carbide machining. NC ECM integrates the advantages of NC flexibility, high efficiency of ECM, and high precision of mechanical grinding. A rotating cathode made of a metal fund–rigid sand composite with internal spraying function is used as a tool. Emery plays the role of insulation, ensuring an ECM gap and scraping off the anode passivation film of the workpiece. In the process of machining, the composite cathode is connected to the negative pole of the DC pulse power supply, and the workpiece is connected to the positive pole of the DC pulse power supply. Electrolyte is sprayed between the composite cathode and the workpiece. The composite cathode performs NC-generating movements relative to the workpiece, and complex parts are machined based on the principle of electrochemical mechanical grinding. By replacing different composite cathodes, NC Electrochemical grinding and NC electrochemical grinding/milling can be implemented.1,2 It has been found that passivation occurs increasingly at higher voltages and it is removed almost instantaneously at higher speed leading to higher current density and MRR. The surface finish is not very much dependent on grinding speed. Gupta et al. reported the modeling and analysis of turbulent flow in the inter-electrode gap of electrochemical grinding to show the performance of erosion in the material removal rate (MRR) of an aluminum–alumina composite. The results showed that the shear stress increased with the rotational speed of the grinding wheel for a fixed inter-electrode gap, and the same was found for the shear force. The MRR by the erosion process was very small, and it increased with the rotational speed. 3 A mathematical model based on the finite element method was established by Fan et al. to study the electrochemical grinding process. The results showed that the electrochemical characteristics could be well predicted for the performance of electrochemical grinding in different electrolytes. 4 Rahi and Dubey studied the effect of voltage on quality characteristics during electrochemical grinding of Al-SiC-Gr HMMCs. The results revealed that the MRR increased with the applied voltage. 5 Based on a simulation of a gas-liquid two-phase flow field, the effects of the grinding head rotational speed and superimposed linear and circular translational movements on the flow velocity and void fraction were investigated by Wang et al. The fluctuations of electrolyte flow velocity and void fraction in the machining area were small when the circular translational movement was superimposed, and a synergistic effect of dissolution and grinding was noted. 6 Inner-jet electrochemical grinding of GH4169 alloy was described by Li et al. The maximum grinding wheel feed rate and the maximum MRR increased the applied voltage (10–25 V) when the other machining conditions were fixed. 7 Goswami et al. conducted orthogonal experiments of electrochemical grinding on Al2O3/Al composites and studied the effects of electrolyte concentration, machining voltage, machining depth, and electrolyte flow rate on the MRR, surface quality, and grinding force, It is observed that metal removal rate (g/min) is maximum at a concentration of 30 g/l, 20 V power supply, 0.04 mm depth of cut, and flow rate of 0.3 l/s. From the analysis of surface roughness data it was observed that concentration, applied voltage, and depth of cut are the three important process parameters and to obtain the best surface finish, the following parametric combination must be used: a concentration of 30 g/l, 20 V power supply, 0.04 mm depth of cut, and flow rate of 0.3 l/s. 8 Tehrani and Atkinson studied the “overcut” problem in electrochemical grinding through an experiment of electrochemical grinding die steel and 304 stainless steel with the grinding wheel. They pointed out that the “overcut” could be effectively reduced by using pulse power supply and by adjusting the duty cycle, under finishing conditions appropriate to obtaining mechanical abrasive contribution at the final workpiece surface, a repeatability within 0.010 mm is possible. Repeatability deteriorates to 0.018 mm when electrochemical dissolution is the dominant contributor to metal removal. 9 Ge et al. studied the material removal mechanism in electrochemical grinding. They have suggested that the material removal is completed by electrochemical anodic dissolution, abrasive grinding, and stray corrosion. 10 Electrochemical milling is a continuous dynamic machining process coupled with multiple physical fields, such as electric, flow, and temperature fields, milling, and so on. It has its special hydrodynamic and electrochemical dissolution characteristics. Such a machining process involves many factors and is complex, and the machining accuracy and machining stability need to be further studied. We address this literature gap by carrying out a numerical simulation on the cathode structure of the designed cemented carbide electrochemical milling tool. The distribution of the electric field of full-edge electrochemical milling was explored. Furthermore, side-edge and full-edge electrochemical milling process tests were performed, the machining process of cemented carbide electrochemical milling was investigated, and high surface quality of cemented carbide electrochemical milling was obtained.
Theory and numerical simulation
Principle of electrochemical milling of cemented carbide
Tool cathode plays an important role in improving the machining accuracy of electrochemical milling, a smooth surface is observed for the micro-tool fabricated by rotating the tool inside the workpiece’s hole, as proper flushing of debris particles takes place due to tool rotation. 11 It is mainly composed of an emery coating adhered to a standard-size cylindrical metal rod. When the full-edge electrochemical milling process is performed, the whole tool cathode edge cuts into cemented carbide, as shown in Figure 1. When the cemented carbide is connected with the tool cathode, the tool cathode uses the side edge to process it so as mill the plane. In the electrochemical machining process, the cemented carbide is the anode, and the bar with emery is the tool cathode. The electrolyte is sprayed to the tool cathode by external spraying and ejected from the surface of the cemented carbide. The gap between the tool cathode and the cemented carbide is the machining gap. Through the flow of electrolyte, a disturbed flow field is formed in the machining gap between the tool cathode and the cemented carbide. Under the action of electric field, a part of the tool cathode with emery forms an electric field with the cemented carbide. At this time, the cemented carbide loses electrons and dissolves in the electrolyte. Thus, the cemented carbide is removed. At the same time, the tool cathode gets electrons, and a reduction reaction, accompanied by black substances attached to the emery, is performed. This method can be used for processing of groove structures or large-cutting-depth processing. It has large one-time processing depth, removes the material efficiently, and has slow feed speed. It is mainly aimed at pre-processing of materials that are difficult to process.
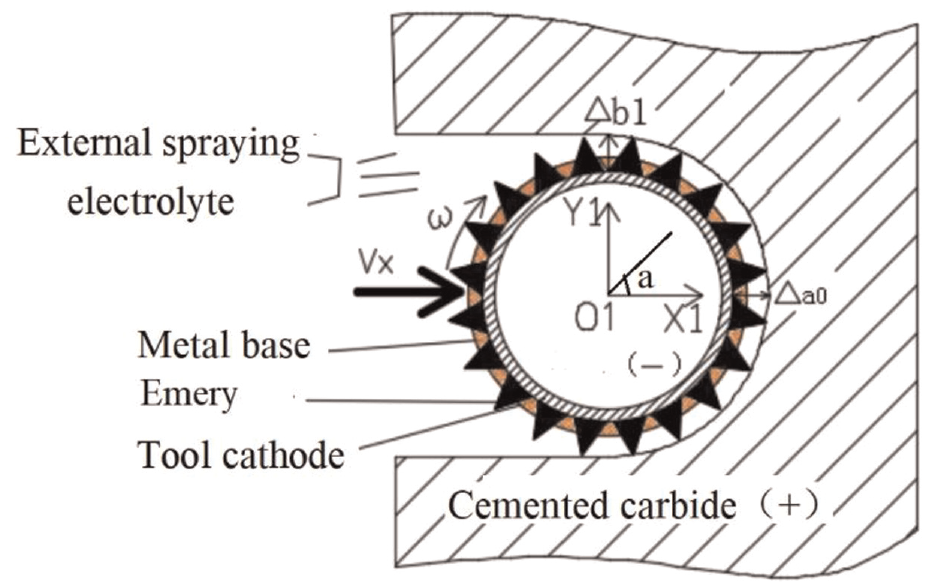
Principle of electrochemical milling of cemented carbide.
According to the balance gap theory of electrochemical machining, the balance gap in the feed direction is △a0 (α = 0) basic equation:
where η is the current efficiency in processing, ω is the volume electrochemical equivalent (mm3/A min), κ is the conductivity of electrolyte (Ω mm), U is the processing voltage (V), δE is the sum of the cathodic and anodic polarization potential values, and Vx is the electrolysis speed (mm/min).
Assuming that the conductivity, electrolyte temperature, and bubble rate remain unchanged for a certain period of time, equation (1) can be optimized and differentiated into the numerical model of electrochemical milling:
where R is the cathode radius of the tool and α is the tool cathode arbitrarily filet.
Numerical simulation of electrochemical milling of cemented carbide
Electrochemical milling is a dynamic machining process coupled with multiple physical fields. Because there are many influencing factors and the relationship between them is complex, it is very difficult to establish the theoretical expression. Even if the relationship expression is obtained, the analytical solution cannot be obtained. Therefore, many researchers explore the processing rules through numerical simulation, shorten the process development cycle and reduce costs.12,13 Based on this, this paper expects to obtain the law of electric field change through the dynamic simulation of the whole process of carbide electrochemical milling, which provides theoretical support for carbide electrochemical milling process.
Analysis model
According to the structural design requirements of cemented carbide electrochemical milling, the established analysis model is shown in Figure 2. The total length of the tool cathode is 72.6 mm, radius is 4.1 mm, and length of the embedded emery part is 26.7 mm. The electric field distribution during full-edge electrochemical milling is mainly considered to judge whether the material removal is uniform in the process of electrochemical milling. Assuming that the electric field analysis is linear, the electric field is proportional to the applied voltage, and the conductivity distribution of the electrolyte in the machining gap is uniform, and the electrolyte temperature does not change with the power line. When the analysis model is established, the influence of abrasive particles on the electric field distribution on the cathode surface is simplified (when the abrasive particle diameter is less than 100 µm). The mesh division of the finite element model adopts a triangular plane mesh, which is more uniform for the main analysis area, no appearance of defective mesh and element.
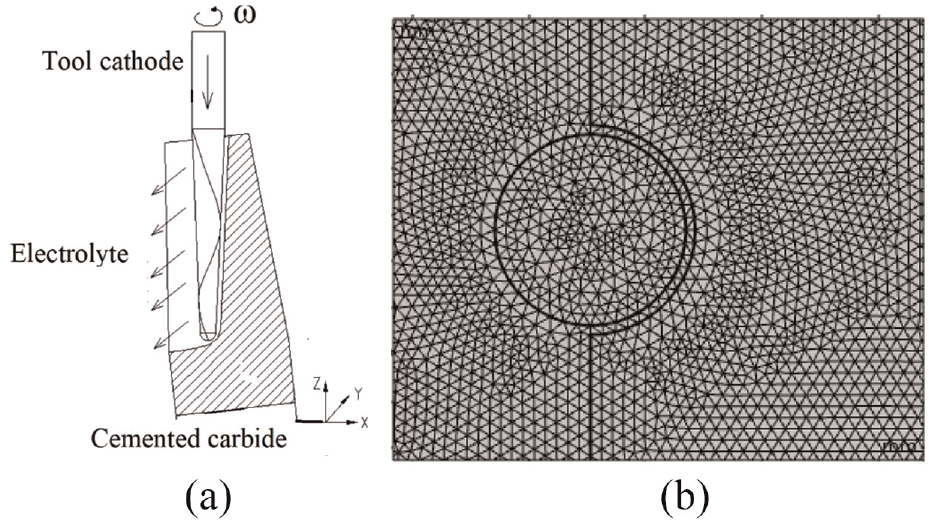
Analysis model of electrochemical milling of cemented carbide: (a) processing principle and (b) finite element model.
Electric field distribution of electrochemical milling
The current density distribution of milling electrochemical machining obtained under the condition of a machining voltage of 16 V and an initial gap of 0.5 mm is shown in Figure 3. It can be seen from Figure 3(a) that when the tool cathode is close to the cemented carbide, no large machining current is formed between them. Only a small machining current is generated in the area where the cemented carbide is closest to the tool cathode. As the tool cathode continues to cut into the cemented carbide, the relative processing area of the cemented carbide and tool cathode increases, and the amount of removed material increases. As shown in Figure 3(b), the current density value in the processing gap remains uniform, and the current density decreases at the cutting in and out of the tool cathode processing edge. When the cutting amount reaches the cathode radius of the tool, the machining area reaches the maximum (semicircle), as shown in Figure 3(c). At this time, the current density in the machining gap is 8 × 108 A/m2, when the tool cathode is completely cut in. As shown in Figure 5(d), the processing current density remains unchanged. It can be seen from the whole process that, with the continuous cutting of the tool cathode, the area of cemented carbide milling electrochemical machining increases, and the current density in the machining gap increases. When the tool cathode cuts into a one-half diameter, the area of cemented carbide milling electrochemical machining remains unchanged, the amount of removed cemented carbide material per unit time remains unchanged, and the current density remains stable.

Current density distribution in milling electrochemical machining: (a) current density distribution when the tool cathode is not cut in, (b) current density distribution when the tool cathode cuts into a one-fourth diameter, (c) current density distribution when the tool cathode cuts into a one-half diameter, and (d) current density distribution when the tool cathode is fully cut in.
Experimental
Experimental device of cemented carbide electrochemical milling
Based on the numerical analysis results of cemented carbide electrochemical milling, the cathode and tooling of electrochemical grinding and milling tool were designed and manufactured. The outer surface of the tool cathode contained emery. Subsequently, a experiment device was built. As shown in Figure 4, this experiment used a self modified and built five axis CNC electrochemical machining machine tool. The workbench of the machine tool is made of marble, which is made of insulating material. The four axis and five axis turntables of the machine tool are placed on the marble workbench. The workpiece is clamped on the C-axis workbench using flat pliers, while the cathode is clamped on the spindle of the machine tool. From this, it can be seen that the workpiece and cathode are mutually insulated, meeting the requirements of electrochemical machining. The tool cathode adopts the carbon brush conduction mode. That is, a conductive seat is equipped under the main shaft, an annular distributed carbon brush is set in the conductive seat, and the carbon brush is in contact with the rotating tool cathode.

Experimental device of cemented carbide electrochemical milling.
Process experiment of cemented carbide electrochemical milling
Orthogonal experimental design
To study the influence of machining parameters on cemented carbide side-edge electrochemical milling, four groups of process parameters, including machining voltage, feed speed, spindle speed, and duty cycle, were selected and studied by an orthogonal test method. The results of the orthogonal test are shown in Table 1. A NaNO3 electrolyte with a mass fraction of 8% is selected as the electrolyte, the electrolyte temperature is 30°C, the initial thickness of cemented carbide is 20.912 mm. During actual machining, the cathode diameter is 8 mm. During each experiment, it is hoped to conduct experiments based on the set combination of electrolytic milling parameters to obtain the width and depth of the groove obtained during milling. According to the basic principles of electrochemical machining, when the experimental parameters change, the machining results also change. In order to obtain the influence of various parameters on the milling results, first, based on a single factor experiment. The limit values during cathode electrochemical milling with a diameter of 8 mm were measured, and the selected values for the cathode experiment were within the range of the limit values. In this experiment, if the cutting depth is set to 6 mm each time, the processing area is 125.6 mm2.
Factor levels of the orthogonal experiment.

Orthogonal experimental data
Cemented carbide electrochemical milling was performed for each group of process parameters. The results of the orthogonal experiment are shown in Table 2. The average current of cemented carbide electrochemical milling was obtained through the experimental records. The removal amount of cemented carbide is achieved through the weighing method, using a weighing device as shown in the following figure. Before each processing, the total weight of the workpiece is recorded. After processing the workpiece according to the preset processing parameters, it is weighed again and the data is recorded. The weight after processing is subtracted from the weight before processing to obtain the removal weight for this processing.
Data of the orthogonal experiment.

Results and discussion
Analysis of cemented carbide side-edge electrochemical milling
The range analysis was carried out on the orthogonal experimental data of cemented carbide side-edge electrochemical milling, as shown in Table 3. It can be seen that the primary and secondary relationships affecting the amount of removed material are duty cycle > feed speed > voltage > spindle speed. It can be seen from the 16 groups of the experimental data that the processing effects of Groups 4, 6, 7, 9, and 14 are relatively good. The removed amount in these five groups is greater than the depth value of each cutting. The largest removed amount (0.102 mm) belongs to Group 4, whereas the least removed amount (0.031 mm) belongs to Group 1.
Range analysis of cemented carbide electrochemical milling.

The appropriate process parameters of electrochemical milling have a very important influence on the performance of the electrochemical milling parts. The research results indicate that the optimal process parameters can be found through experimental design, achieving high geometric and machining accuracy.14,15 According to the range analysis results, the relationship between the machining process parameters of cemented carbide electrochemical milling and the removed amount is shown in Figure 5. It can be observed that the removed amount first increases and then decreases with increasing machining voltage. When the machining voltage is 14 V, the removed amount is largest (0.083 mm). With increasing feed speed, the removed amount first decreases and then increases. When the feed speed is 10 mm/min, the removed amount is largest (0.088 mm). With increasing spindle speed, the removed amount first decreases and then increases. When the spindle speed is 3000 and 6000 r/min, the removed amount is basically the same (0.083 mm). Considering the safety and stability of cemented carbide electrochemical milling, it is appropriate to select a spindle speed of 3000 r/min. With increasing duty cycle, the removed amount first decreases and then increases. When the duty cycle is smaller, the power input cycle is largest, processing time is longer, processing is more sufficient, and removed amount is larger. Thus, it is better to select 70% duty cycle for processing. In summary, optimum parameters are a voltage of 14 V, feed speed of 10 mm/min, spindle speed of 3000 r/min, and duty cycle of 70% for the full-edge electrochemical milling experiment.

Relationship between process parameters and removed amount of cemented carbide electrochemical milling: (a) relationship between processing voltage and removed amount, (b) relationship between feed rate and removed amount, (c) relationship between spindle speed and removed amount, and (d) relationship between duty cycle and removed amount.
Analysis of cemented carbide full-edge electrochemical milling
Based on the optimized process parameters of side-edge electrochemical milling, full-edge electrochemical milling of cemented carbide was carried out. Under the condition that other process parameters remain unchanged, a test was carried out by changing the feed speed to 0.1, 0.2, and 0.3 mm/min. The experiment results are shown in Figure 6.
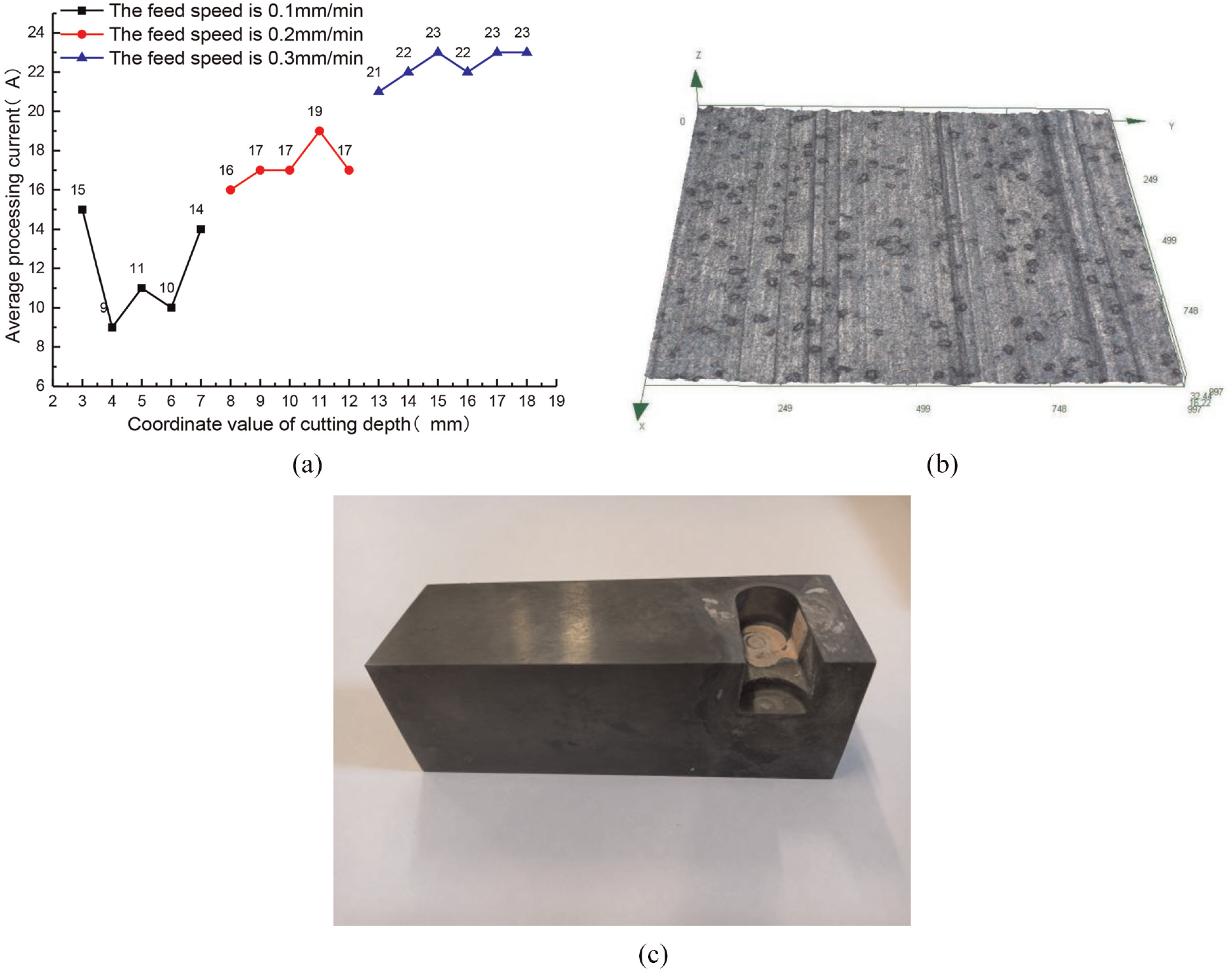
Full-edge cemented carbide electrochemical milling: (a) influence of feed speed on machining, (b) surface topography, and (c) processing samples.
The relationship between the feed speed and the average processing current is depicted in Figure 6(a). It can be seen that, after the composite cathode cuts into the workpiece, the current keeps fluctuating around a certain mean value at the same feed speed, and the mean value of the processing current is different at different feed speeds. Electrochemical milling is not a simple electrochemical machining, mechanical grinding, or milling process. Instead, it is a composite machining process that combines the advantages of electrochemical machining and mechanical grinding and milling. While removing materials with the help of electrochemical machining, it relies on cathode coating sand grinding to remove the passive film and maintain the continuity of processing. 16 Under the condition that all processing parameters remain unchanged, the processing current fluctuates within a certain mean value range, which indicates that the electrochemical milling process is a dynamic balance process. During the milling process, because the electrolyte is sprayed externally and the machining gap is small (about 0.15–0.3 mm), the electrolyte cannot fill the entire processing area only by spraying. To compensate for this, the composite cathode rotation promotes filling of the machining gap with the electrolyte. An uneven distribution of the electrolyte will change the machining current. In addition, cemented carbide is an alloy material made of a hard compound of refractory metal and bonding metal through a powder metallurgy process. When the bonding metal is electrochemically removed by electrochemical machining, the metal hard compound that is not fully involved in the electrochemical reaction will fall off from the substrate and enter the machining gap, resulting in spark short circuit between the cathode and the workpiece. As the rotating cathode takes the falling objects away from the machining gap, a single spark ends. Owing to the low machining voltage, the current fluctuation in the spark is small. When the feed speed is increased, the machining current increases. When other parameters remain unchanged, the machining gap decreases with increasing feed speed, the electric field strength and the current density between the composite cathode and the workpiece increase, and the MMR increases. The surface morphology of the sample machined by cemented carbide electrochemical milling is shown in Figure 6(b). It can be seen that the machined surface is relatively flat and the surface contains shallow grooves caused by the scraping effect of abrasive particles. Measurement of surface roughness is 0.389 μm using an OLYMPUS DSX110 ultra depth of field microscope. The processed samples are shown in Figure 6(c). Their surface meets the requirements of cemented carbide finishing.
Discussion of electrochemical milling cemented carbide
In the process of electrochemical milling of cemented carbide, the electrochemical anode reaction in the processing area is the process of passivation and scraping the passivation film alternately, that is, in the Sodium nitrate solution of the cemented carbide body, cobalt is first ionized to produce cobalt ions, which are immediately combined with Hydroxide ion ions in the solution to generate cobalt oxide with very low solubility, while the anodic oxidation of tungsten carbide is mainly due to strong oxidation, The process is that the Nitrous acid ion is first oxidized on the anode to produce Dinitrogen tetroxide, and then tungsten carbide is oxidized. The NO produced in the reaction is immediately oxidized into nitrogen dianitride due to the action of oxygen or oxygen atoms on the electrode, part of which is precipitated, and part of the solution is in the electrolyte to generate nitrite. According to the electrochemical reaction theory, cobalt combines with oxygen element to generate cobalt oxide with poor conductivity, which is adsorbed on the surface of cemented carbide substrate, slowing down the electrochemical reaction process gradually. The tungsten in tungsten carbide strongly combines with oxygen to generate tungsten trioxide and carbon monoxide. Tungsten trioxide will be further chemically dissolved in alkaline electrolyte to generate tungstate. Tungsten trioxide reacts with sodium hydroxide to generate Sodium tungstate and water. On the surface of the cathode, it is mainly the precipitation of electrochemical hydrogen gas.
The factors that affect the machining efficiency of electrochemical milling of cemented carbide include:
(1) The electrochemical equivalent of the material: the electrochemical equivalent of cobalt is 0.6465 g/A h, and the electrochemical equivalent of tungsten is 1.28 g/A h. According to Faraday’s law, the theoretical rate of metal removal per unit of electricity under the same conditions is directly related to the electrochemical equivalent of the material being processed. As a metal compound, cemented carbide contain multiple metal elements. The electrochemical equivalent of various metal components and impurities varies, and the speed of electrolytic removal varies, which is also one of the factors affecting surface roughness.
(2) Current density: The current density directly determines the rate of anodic dissolution. From the experimental results, it can be seen that an effective way to improve the rate of metal removal is to increase voltage, accelerate feed rate, reduce electrode gap, reduce electrolyte resistivity, and increase electrolyte temperature.
(3) Cathode size: The removal rate of the material is proportional to the conductive area. At the same current density, the more electricity passes through, the greater the metal removal rate per unit time. Therefore, increasing the processing area of the cathode and workpiece as much as possible, that is, increasing the cathode diameter and cutting depth, can effectively improve the material removal rate.
(4) The mechanical factors of the cathode include the positive pressure of the cathode, the milling pressure on the tool surface, the rotation speed of the cathode, and the feed speed of the cathode, which all affect the material removal speed. The cathode milling pressure is high, the cutting speed of the workbench is fast, and the cathode speed is fast. The higher the degree of activation of the anode metal, the higher the speed of material electrolytic erosion. The feed speed is fast, and the machining gap between the cathode and the workpiece is smaller. The smaller the machining gap, the stronger the electric field, and the faster the workpiece is corroded; However, excessive pressure in electrochemical milling can cause abrasive wear and detachment, shortening the service life of the cathode; If the feed speed is too fast, the anode will not have enough time to react, causing the cathode to collide with the workpiece, causing a short circuit and burning the cathode, burning the workpiece, and ultimately reducing production efficiency.
The factors that affect the machining accuracy of electrochemical milling of cemented carbide include:
(1) The composition of the electrolyte, which directly affects the properties of the passivation film on the anode surface. If the generated passivation film has a loose structure and poor protection ability on the tool surface, it will cause severe stray corrosion and low machining accuracy. In order to obtain high-precision parts, a layer of dense and uniform structure should be formed on the surface of the workpiece during the machining process The anodic passivation film formed by passive electrolytes such as low valence oxide and Sodium nitrate with good protection performance is not easy to be damaged. When machining cemented carbide, the PH value of the electrolyte should be properly controlled. The oxide of hard alloy is easy to dissolve in alkaline solution. Therefore, in order to obtain a thicker anodic passivation film, it is better to use low PH electrolyte, generally PH = 7–9.
(2) Wear particle size and wear particle trajectory on cathode surface: cylindrical cathode, its motion trajectory is constant – a combination of Circular motion and linear motion. In the feed direction, the trajectory has a large repeatability. When the milling amount is large, it is easy to process a strip shaped gully shape, reducing the surface quality. Therefore, it can only reduce the size of wear particles, improve the electroplating density of diamond sand on cathode surface, so as to obtain a flat surface quality and improve processing accuracy.
(3) Composition of cemented carbide: As mentioned earlier, the problem is more serious when electrolytic milling cemented carbide due to different metal compositions and different dissolution rates. To solve this problem, the electrolyte is changed to a composite electrolyte dominated by Sodium nitrate.
(4) Mechanical factor: During the electrochemical milling process of hard alloys, the activation of the anode surface mainly relies on mechanical grinding, so the accuracy of machine tool movement, fixture accuracy, and cathode production must be improved. The finer the abrasive particle size, the more evenly the passivation film protruding from the processing area can be removed. On the other hand, it reduces the machining gap, accelerates the leveling speed, and is beneficial for improving surface quality. However, if the abrasive particle size is too fine and the machining gap is too small, it is easy to cause sparks and reduce surface quality. Generally, the abrasive particle size is between 100 mesh and 200 mesh.
Conclusion
(1) Cemented carbide electrochemical milling method integrates the advantages of high efficiency of NC flexible electrochemical machining and high precision of mechanical grinding, which can effectively avoid the machining defects such as cracking and burns caused by traditional machining, and provides a new way for cemented carbide machining.
(2) The cutting process of cemented carbide electrochemical milling is numerically simulated, and the variation law of electric field is obtained. It can be seen that with the continuous cutting of the tool cathode and the continuous increase of the machining area, the current density in the machining gap increases. After the tool cathode cuts into the semicircle, the machining area remains unchanged, so the material removal amount per unit time remains unchanged, and the current density remains stable.
(3) The orthogonal process test of four factors and four levels for electrochemical milling of cemented carbide side edges was carried out. The results showed that the optimal process parameters were under the conditions of processing voltage 14 V, feed speed 10 mm/min, spindle speed 3000 r/min, and duty cycle 70%.
(4) Based on the optimal process parameters of cemented carbide side edge electrochemical milling, the influence of different feed speeds on full edge electrochemical milling is studied, and the samples of cemented carbide full edge electrochemical milling are obtained. The measured surface is relatively flat, and its roughness Ra is 0.389 μm. It meets the requirements of cemented carbide finishing.
Footnotes
Handling Editor: Chenhui Liang
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of JiangSu Province (BK20211061), The Natural Science Foundation of the Jiangsu Higher Education Institutions of China (19KJA430005), The special fund project for transformation of scientific and technological achievements in Jiangsu Province (BA2021036).


