Abstract
Metal-organic frameworks (MOFs) have been synthesized using several elements, including the rare-earth. Research on luminescent diketones as an efficient alternative for the generation of OLEDs has increased in recent years, however, the evaluation of parameters such as morphology or excess of the cheating agent is not completely studied. In this work, the synthesis of thenoyltrifluoroacetone (TTA) combined with europium at the different chelating agent and rare earth ratio is presented, founding a change in morphology directed related with optical performance, structural analysis was performed using x-ray diffraction (XRD), and was observed a monoclinic structure in Eu 15% and Eu 20% samples. Morphological analysis was done with scanning electron microscopy (SEM). The TTA5Eu sample presented an irregular morphology with nanometric particles embedded, the TTA15Eu had a morphology of agglomerated particles, and TTA20Eu sample the morphology changes radically to an angular morphology. Nanowires were present in the sample of 25% Eu3+. The chemical properties were determined using Fourier transform infrared spectroscopy (FTIR), and the optical properties were measured by photoluminescence (FL). The results showed vibration bands and stretching of the thiophene group, without apparent change due to the chelation of the Eu3+.
Keywords
Introduction
Metal–organic frameworks (MOFs) have emerged as new porous material platforms used in potential applications in technology emergent. MOFs are organic–inorganic hybrid crystalline porous compounds consisting of inorganic metallic clusters, that are connected by organic linker molecules.
1
Using different metal centers and ligands are applied to produce MOFs, which results in a suitable flexibility for modifying physical and chemical features. There is a correlation between the coordination geometry and the dimensionality: size or hardness of the metal center and electronic configuration affect the resulting topology of MOF.2,3 Over the last years, MOFs have emerged as a new promising porous material in the areas of gas sorption and separation,
4
as platform for immobilizing molecular catalysts on conductive substrates,5–8 water treatment,
9
storage,
10
antibacterial and antobiofilm
10
and light emitting diodes (LEDs).
11
Rare earth doped materials are very useful nowadays, because the characteristics that provide the lanthanides to the material can confer new applications as optoelectronics devices, fiber amplifiers, solid state laser,
12
solar cells,
13
and luminescence.
14
Studies of the photoluminescence of the europium metalorganic frameworks have increased over the last decade, due to their high luminescence that make these systems a promising for areas of potential application such as optical markers, optical sensors, and fluorometric tests and other clinical applications.
15
These complexes can be seen as molecular light-conversion devices that exploit the antenna effect; proposed by Crosby et al.
16
in 1962 and that has been confirmed by various authors, such as Ronghu Wu and Su
17
in 2001,18–21 stablish that the luminescent metalorganic frameworks are efficient in their absorption sequences and their energy and emission transfer to the rare earth, which involve a reduction of the organic compound emission and a increasing of the rare earth emission.17,22,23 In other hand, it is well established that 2-thenoyltrifluoroacetone (TTA) chelate agent, can sensitize a large number of lanthanide ions, for example, Nd3+, Sm3+, Eu3+, Tb3+, Tm3+, Yb3+, Er3+, and others, and these complexes (Nd3+, Sm3+, Eu3+, Tm3+, Tb3+, Er3+) produce a visible luminescence, depending on the nature of the lanthanide ion24,25 but research on the effect of morphology and other parameters is lacking. Mostly a particle in form of fibers and nanorods were obtained.26–30 Therefore, organic complexes of photoactive lanthanides, like the macrocyclic ligands of europium
Materials and methods
Organic synthesis of TTA: Eu3+
For the synthesis of TTA: Eu3+ at 25 mol%, the precursors, europium nitrate 99.9% and sodium bicarbonate 99.7%, were purchased from Sigma Aldrich; in addition, distilled water and ethanol 99.8% were used solvents, without further purification. In a typical synthesis, 5.72 mmol of thenoyltrifluoroacetone was dissolved in 42 ml of ethanol and kept under vigorous agitation at room temperature for 40 min. In another flask, europium nitrate was dissolved in 2.2 ml of deionized water, then 18.6 mmol of sodium bicarbonate were added, and the solution was stirred for 30 min, after which, the product of the first reaction (TTA dissolved in ethanol) was added to the second drip reaction and agitated for 24 h. Next, the solution was dried to 80°C for 24 h, and the powder consisting of TTA: Eu3+, with a Eu3+ concentration of 25 mol%.
In order to increase the luminescent intensity of the TTA organic matrix, an excess of the chelating agent was added to fix the europium composition at 5%, 10%, 15%, 20% (TTA5Eu, TTA10Eu, TTA15Eu, TTA20Eu). Structural characterization by x-ray diffraction (XRD) using a Bruker eco D8 Advance with radiation at Kα1 (λ = 0.15406 nm) was performed at each stage, at diffraction angles (2θ) of 20–80 [and a step of 0.02 s−1]. For optical photoluminescence (FL) analysis, a Hitachi spectrofluorometer, model F-7000 (5 J-0003) at a wavelength of 254 nm was used. The morphological analysis was carried out using the SEM technique, supported by a SEM apparatus (JEOL DS-500). Chemical analyses were performed with Fourier transform infrared spectroscopy (FTIR), using the KBr splicing technique with Perkin Elmer Spectrum 65 apparatus in a range of 4000–400 cm−1.
Results and discussions
Structural and morphological analysis
The x-ray diffraction results obtained from the (TTA5Eu, TTA10Eu, TTA15Eu, TTA20Eu) samples are shown in Figure 1; the XRD pattern for TTA without Eu3+ is included for purposes of comparison. Some increases in intensity can be observed in the TTA15Eu and TTA20Eu samples. With the information obtained, Rietveld refinement was employed using the P21/c model with the initial values of the network parameters indicated in Table 1. The crystalline structure of the samples TTA5Eu, TTA10Eu, TTA15Eu, TTA20Eu were calculated using the Rietveld method to obtain a suitable adjustment for a monoclinic structure (

TTA sample XRD patterns at various molar proportions of Eu 3 +.
Dimensions of the crystalline structure TTA: Eu3+.

Refined parameters for the TTA: Eu3+ samples.


Monoclinic crystalline structure (
The morphologies obtained from the luminescent powders of TTA: Eu3+, with different proportions of Eu3+, are shown in Figure 3(a)–(d). In the TTA5Eu sample, an irregular morphology is seen with incrustations of nanometric particles dispersed evenly throughout the sample. In the TTA15Eu sample, a rounded morphology of agglomerated particles appears. In the TTA20Eu sample, the morphology changes radically with 5% increases in the amount of Eu3+, showing an angular morphology. In the sample consisting of 25% Eu3+, the morphology that presents in the form of nanowires with a diameter of approximately 6 nm and an average length of 4 μm (see Figure 3), this morphology is also reported by Medina-Velazquez et al. 41 as the typical morphology obtained for the MOF.

Morphologies obtained from the TTA: Eu3+, with different proportions of Eu3+: (a) 10%, (b) 15%, (c) 20% and (d) 25%.
Infrared spectroscopic analysis (FTIR)
Identification of the functional groups present in the TTA: Eu3+ luminescent powders was performed using the Fourier the infrared transform (FTIR) technique, supported by the KBr pelleting technique. Figure 4 shows the FTIR spectra obtained from the TTA: Eu3+ luminescent powders in molar proportions of Eu3+ at 5%, 10%, 15%, 20%, and 25%. In the figure, vibration bands of (OH) can be seen at 3400 cm−1, which can be attributed to the water absorbed by the KBr during the measurement, and vibrational bands associated with the (CH)2 group are visible at 2972 cm−1. In the TTA sample, an absorption band attributable to C=O at 1600 cm−1 is seen. It shifts to the right as the molar ratio of Eu3+ is increased, due to a strong interaction between the oxygen atoms from TTA and Eu3+; this indicates that the Eu3+ ion coordinates through the oxygen atoms that form the chelation ring. Vibrational bands from C=C appear at 1541 cm−1 and increase their relative intensity as the ratio of the Eu3+ increases up to 10 mol% and then reduce their intensity by 5% of Eu3+. Finally, vibration bands and stretching of the thiophene group are visible at 1138 cm−1 and at about 1295 cm−1, respectively, with no apparent change due to the chelation of the Eu3+.

FTIR spectra for TTA at different molar proportions of Eu3+.
Photoluminescence analysis
The emission spectra for the samples TTA5Eu, TTA10Eu, TTA15Eu, TTA20Eu TTA25Eu are presented in Figure 5. An excitation wavelength of 254 nm was used. Luminescent powders of TTA with different chelating agent proportions were synthesized in order to modify their luminescence compared to TTA:Eu with a ratio of 85% of thenoyltrifluoroacetone and 25% Eu (TTA25Eu).
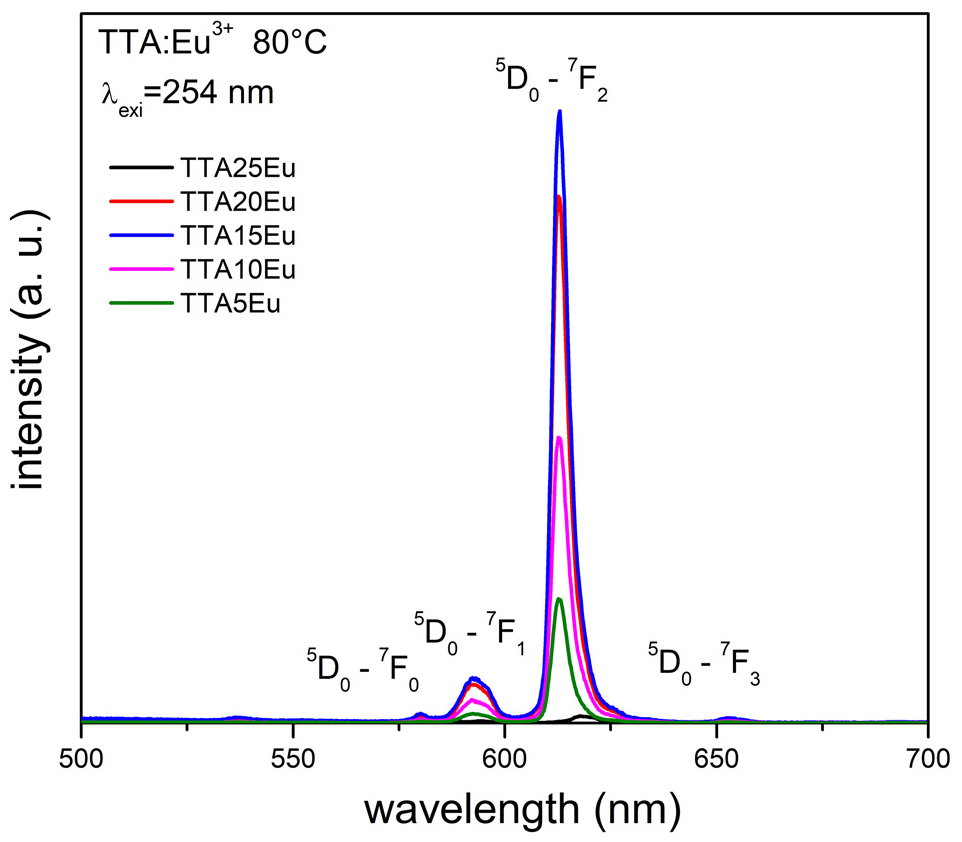
Transitions located between 570–585 nm,585–600 nm, and 610–630 nm.
Figure 6 shows the localized transitions between 570–585 nm, 585–600 nm, and 610–630 nm, which correspond to the 5D0→7Fj (J = 0–2) transitions. The highest luminescence intensity is found in the 5D0 transition to 7F2, which corresponds to the characteristic emission of the Eu3+ ion.

Emission spectra of the TTA: Eu3+ samples at different molar proportions of Eu3+.
The energy transition involved in the emission of TTA: Eu3+ obtained at an excitation wavelength of 254 nm starts with excitation of the S0 to S1 singlet, where the energy descends to a T1 state and passes to a characteristic energy state of Eu3+ to follow its descent from 5D0→ 7 Fj (j = 0,1, 2, and 3) radiatively, with its highest luminescence intensity at 612 nm. These results are similar at reported of the emission in the band of 530–630 nm which is a typical emission in Eu3+ within of MOF.15,19 The bands obtained by FTIR for TTA are congruent with the data by Ohwada 24 and Jiménez et al., 42 in this case both spectra are included in the same figure. As was mentioned before the morphology is like the analyzed in previous work and corresponding with the images of Dandekar et al., 28 with the difference in this work the nanowires are agglomerated. Finally, the red emission was comparable with the presented by Wanpeng et al., 43 although, int dopant this investigation a comparison between different concentration is available.
Figure 7 shows variations in the luminescence intensities of the samples TTA5Eu, TTA10Eu, TTA15Eu, and TTA20Eu. The highest intensity is seen with the TTA15Eu sample, followed by the TTA20Eu sample. The morphology obtained in the synthesis of TTA with Eu3+ at 25%, which consisted of nanowires with a diameter of approximately 6 nm and an average length of 4 μm, as well as its luminescence intensity, was completely modified when the molar proportion of Eu3+ was decreased, yielding rounded particles that agglomerated with an average size of 200 nm and favored the absorption of UV and emission.

Luminescence intensities of the samples TTA5Eu, TTA10Eu, TTA15Eu, and TTA20Eu.
Conclusion
The crystalline structure of the samples TTA5Eu, TTA10Eu, TTA15Eu, TTA20Eu were calculated using the Rietveld method to obtain a suitable adjustment for a monoclinic structure (
Footnotes
Acknowledgements
The authors also would like to thank Henry Jankiewicz for the editing work that he did for this paper and M. García Murillo for her assistance. And also for Maribel Pacheco and Mayra Salinas help.
Handling editor: Chenhui Liang
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CONACyT project 254280. The authors gratefully acknowledge the financial support of this work by the SIP-IPN projects 20211265 and 20211548 and Conacyt project A1-S-28234.
