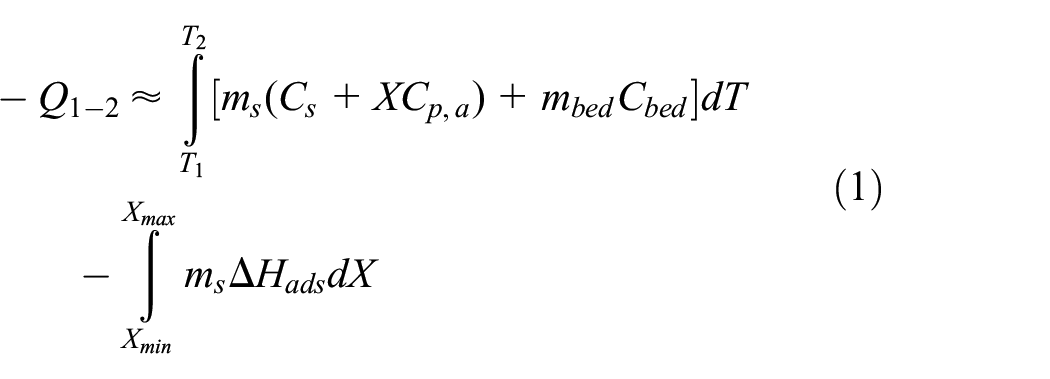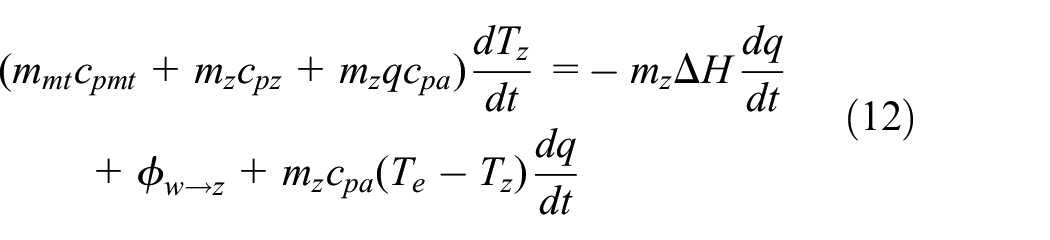Abstract
Adsorption heat pumps (AHPs) have gained noticeable attention in recent years. The review discusses significant research in the domain of adsorption heat pumps focusing on heating applications, the working principle, adsorption equilibrium and kinetics, the design of adsorbent beds, physical models, operating parameters, and physical and thermodynamic characteristics of adsorbents. A comparison is presented among AHPs and traditional heating technologies and systems for AHPs that certain market participants have created. The review focuses on the most appropriate conditions for each measurement technique and the constraints of the modeling processes that are an essential element for the comprehensive performance evaluation of adsorption cooling units. For adsorption bed applications, it is suggested to use various bed designs and heat exchanger structures. Moreover, significant literature gaps and constraints in designing AHPs for heating applications are identified and analyzed. The heat and mass transfer resistance should be reduced by developing novel adsorbent materials and an improved interface among the duct wall and the bed, where heat transfer fluid circulates to increase adsorption heat pump system performance. Furthermore, optimum operational modes for the intended application might significantly enhance system performance.
Introduction
Transition in the global economy, rapid urbanization, technological innovations, demographic shifts, ecological degradation, and the depletion of natural resources have been recognized as the vital driving factors reshaping the world, affecting living practices, and business strategies. 1 To alleviate the scarcity of resources and climatic variations, researchers are developing technologies, and governmental institutions are implementing strategies aligned with environmental and energetic issues. After a decade of breakneck technological breakthroughs in the field of energy, an unprecedented disruption was brought to the energy sector in 2020. 2 The total energy demand depreciates by 7% due to COVID-19, which has caused decreased economic activity. It will take late 2021 for the energy demand to reach back to the pre-COVID-19 levels. Gas endures the increment of global energy demand share in the next 10–15 years and peaks in the late 2030s. 2
Notwithstanding this, the demand for gas is 5% higher in 2050 than the current level. Oil demand follows a projected peak till 2050, exhibiting a 10% substantial decrement, mainly driven by increased electrification of automobiles. From 2019 to 2050, the demand for coal falls to approximately 40%, owing mainly to the global phase-out of coal plants in the power sector. 2 Consequently, it is critical to implement technology that can turn affordable environmental energy into usable heat, reducing the cost of thermal comfort, thereby minimizing global energy and environmental impacts. This study reviews key research areas of adsorption heat pumps, as given in Figure 1.

An overview of major themes analyzed and addressed in this study.
Currently, vapor-compression systems (VCS) are regarded as one of the competent heating systems satisfying consumer demands. Nevertheless, non-renewable energy resources are the primary sources through which electrical energy is derived. In turn, VCS is powered and employs the sinister fluorocarbon (FC) refrigerants (possessing ozone depletion potential) and produces drawbacks such as vibration and noise.3–5 Figure 2 shows the implementations of heat pump systems out of which the closed-cycle compression heat pumps are widely used ones, available in a large variety of sizes for various uses. Therefore, other technologies such as adsorption-based systems seek researchers’ attention due to their advantages over VCS.6–8 In recent years, adsorption heat pumps (AHPs) have received much attention because of their numerous benefits and various attributes, including low electricity requirement for operation, no moving parts, and common environmental effects. 9

Implementation of heat pump principle along with its drive power, drivers, type of compressors, and type of cycles.
Furthermore, since there are no moving parts for the working fluid circulation, adsorption-based thermal compressors need less maintenance than traditional mechanical compressors. 10 Besides that, AHPs have been criticized for being more costly, large, and bulky than VCS. 11 In 1976, Tchernev 12 showed the use of AHP for the heating process for domestic hot water and space for the first time. Ever since, considerable studies have been undertaken to enhance their coefficient of performance, working at reduced temperatures of desorption, and providing intermittent cooling and heating notwithstanding this capacity for AHPs to integrate energy and environmental efficiency with heating industry demands, several literature reviews on adsorption-based heating are presented.4,7,13,14 Instead, some reviews are mainly based on cooling applications.15–24
Adsorption rate, improvisation of COP, and desorption temperatures are some of the most researched topics at the material stage of finding new or refining the previous working pairs. Besides, the implementation of more reliable and effective computational models, heat and mass transfer optimization via operating conditions, and adsorbent bed design have been investigated. 12 Conversely, there are a few studies available on AHP systems considering all of their components and aspects. The current literature review summarizes adsorption heat pumps primarily focused on employment for heating. Instead of repeating the existing studies in the area, the critical goal is to analyze the bridges that need to be constructed between heat exchanger geometries, operating conditions, cycle features, working pairs, bed configuration, adsorption kinetics, and remaining device components. Therefore, significant challenges are identified, solution concepts are discussed, and directions for future system development are set, analyzing the existing gaps, control strategies, components design, and integrating AHPs with other systems. Additionally, this paper systemizes literature data of various complex AHP systems, their working principles, the kinetics of adsorbent-adsorbate working pair, physical models in different perspectives (integration of different studies) to identify significant achievements aiding as a guide for researchers in terms of R&D in this domain.
Adsorption heat pump systems
Working principle
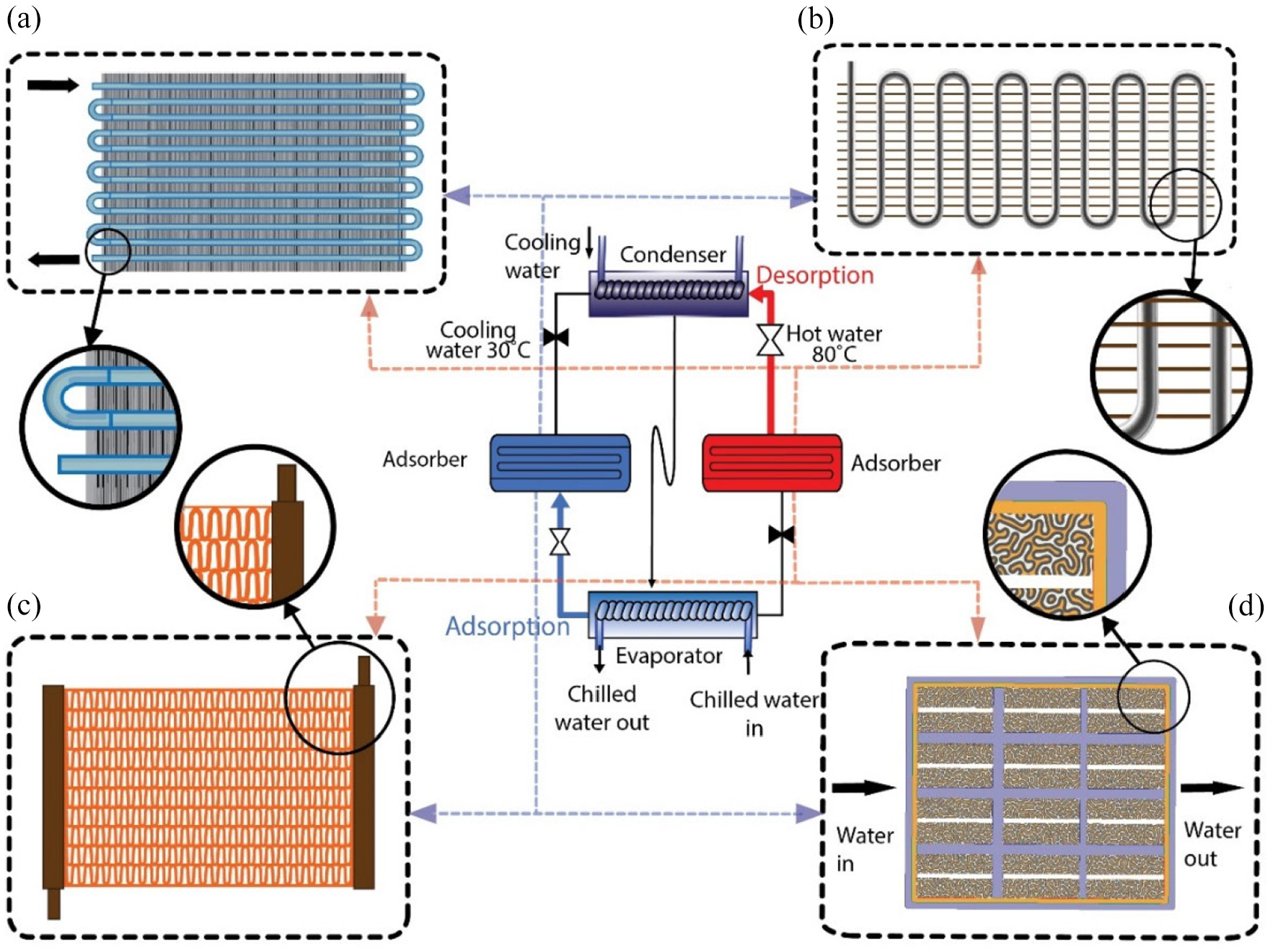
Heat pumps perform an operation of collecting heat from a reduced temperature level and transferring this to a temperature with elevated levels, all while being operated by a tertiary energy supply. 25 The AHPs working principles may be comprehended by considering the comparatively basic situation example of a single bed unit, that involves an adsorbent heat exchanger (AHEx), a collection of valves, an evaporator, and a condenser with an expansion system (V). Figure 3 depicts, there are four stages of the adsorption heating cycle: isobaric adsorption (from 1 to 2), isosteric heating (from 2 to 3), isobaric desorption (from 3 to 4), and isosteric cooling (from 4 to 1).4,9,26 Figure 3 portrays an optimal period and demonstrates the pressure (P) over four successive cycles.9,27 Almost every loop works in the following manner.9,27

Schematics of four phases of an adsorption heating cycle with the simple depiction of the fundamental AHP.
As presented in Figure 3, the isobaric adsorption (from 1 to 2) and the evaporator and bed are attached, which gains heat from the atmosphere. If the temperature of the bed drops on the porous adsorbent, the adsorption of vapor occurs, and the device generates heat, which can be employed for various purposes. At pressure Pevap, it can prevail to the point, till maximum (equilibrium) concentration of adsorbent is achieved. Veritably, although this is unlikely to be the case, the phase could be disrupted if the temperature differential among the adsorbent bed and the heat transfer fluid (HTF) is no longer sufficient. 28 The adsorbent bed production in this stage can be estimated using the equation (1)4,29:
Similarly, the heating and bed isolation takes place in the isoteric heating stage (2 → 3), and the bed pressure elevates from Pevap → Pcond, as shown in Figure 3 due to desorption of some part of the adsorbate. The amount of heat emitted during this process is determined as follows (equation (2))4,29:
The isobaric desorption (3 → 4) begins, as presented in Figure 3 when the adsorbent bed is exposed to the condenser, and Pcond is attained. At this stage, for the promotion of adsorbent regeneration, heat is needed. The adsorbate desorption (W decreases from W3 to W4) occurs when bed temperature elevates (from T3 to T4), and adsorbate condenses while releasing the latent heat, that can be used for multiple applications. The heat that really should be delivered to the bed during this process is given by equation (3)4,29:
Eventually, in the isosteric cooling stage, the separation and bed cooling occur, that is from 4 to 1 (Figure 3), which results in the decrease of pressure since a vapor gets partly adsorbed; mostly sensible heat is emitted. Similarly, another cycle starts as the bed pressure (Pevap) is reached, by reconnecting the evaporator and the adsorbent bed.4,9,30 The heat extracted from the bed under isosteric cooling is given as (equation (4
The condenser and the evaporator operate according to vapor-liquid equilibrium (VLE) conditions, where the flow rate of HTF and inlet temperature affect the heat exchanger. In practice, each AHP cycle can be considered a two-stage process in which the bed is provided the heat or the bed releases heat. The AHP unit often matches with the isosteric heating’s beginning step, as shown in point 2 of Figure 3.
5
The process from 1 to 4 shows the heat supplied and pumped out in every cycle of the AHP unit, with the help of arrows of various colors, representing different temperature ranges of AHPs. Although fuel for the evaporation process is provided during the reduced temperature, regeneration of the adsorbent bed necessitates a heat supply that is at an elevated temperature, and usable heat is emitted at a mid-temperature range. The amount of heat provided at high and low temperatures (for regeneration, bed heating, and evaporation) correlates to the heat emitted at a moderate T stage (by the bed and the condenser through isobaric adsorption and isosteric cooling).
31
The greater the cycle’s temperature level (
Δxmax from equation (5) represents the gap in the maximum water content of zeolite xmax at evaporation pressure and minimum adsorption temperature and the minimum water content xmin at condensation pressure and maximum process temperature. These process points cannot be attained due to the requisite temperature gradient among the adsorber, heating system, and between the gas burner and the desorber. Dx, which is the difference in the water content of zeolite at the end of the adsorption phase xads and the end of the desorption phase xdes, describes the quantity of water that can be evaporated under actual scenarios. 32
In one module throughout the heat pumping cycle, a gas burner supplies heat to the heat transfer circuit of the adsorber/desorber to increase zeolite temperature and vacuum vessel pressure in the desorption phase. Liquification of water in the condenser occurs. The heat emitted causes the condenser temperature to elevate. As the condenser temperature surpasses, the temperature can be transmitted via usable heat (theat). This stage is halted as the highest process temperature reaches around 200°C and the burner is turned off. Similarly, a decrease in the temperature starts the adsorption phase – the zeolite and evaporator temperatures and pressure decrease. The ambient heat may be provided in heat pumps when the evaporation temperature stays below ambient temperature (tcool), the evaporated water is adsorbed onto zeolites, and the heating system can be supplied with the adsorption heat. When all the adsorbate evaporates, the current phase stops, and the next phase starts. 32
Parameters affecting performance
Adsorption heat pumps are usually tested based on their coefficient of performance (COP) and specific cooling or heating capacity (SCP/SHP). For cooling requirements, COPc (equation (6)) is defined as the ratio between the heat of evaporation to the heat provided to the bed. For heating, COPh (equation (7)) is defined as the ratio of the heat of the adsorbent bed released during adsorption phases and isosteric cooling (Q4–1 + Q1–2), including the condensation heat Qc, as well as the heat used for desorption and isosteric heating (Q2–3 + Q3–4)4,29:
The ratio of proper heat, the cycle time and adsorbent mass gives SHP (equation (8)). The SCP (equation (9)) is computed by the heat of evaporation over the cycle time and adsorbent mass. The specific cooling and heating power is one of the distinguishing features. The specific heating/cooling capacity is a feature that is strongly reliant on the system design, providing analysis of various system configurations and designs4,29:
According to Cabeza and Schossig, 33 a skilled assessment of the efficiency of AHPs for real-world implementations necessitates a consistent specification of key performance indicators (KPIs). It provides a comparative analysis of system efficiencies employing variable energy sources. The primary energy ratio (PER) is the ratio of total useful energy (Quseful) and the total primary energy inputs. The following formula (equation (10)) can be used to measure PER:
where E, F are the electrical energy and the fuel energy inputs, respectively, and the fp,fuel is the primary energy conversion factor for fuel, and fp,elect is the primary energy conversion factor for electricity. A thermal COP is often determined excluding electrical energy because this contribution demanded by AHPs may be overlooked (equation (7)). 31
Effect of operating conditions on the system performance
The operating conditions which have a substantial impact on the performance of AHPs include temperatures of evaporation (Tevap), condensation (Tcond), minimum bed temperature, and bed regeneration (T4).34–36 The T2 and Tcond depend on the useful heat’s temperature and should be analogous for delivering the condenser and the bed heat at a comparable T level.14,37,38 The basis of Tevap is the temperature (
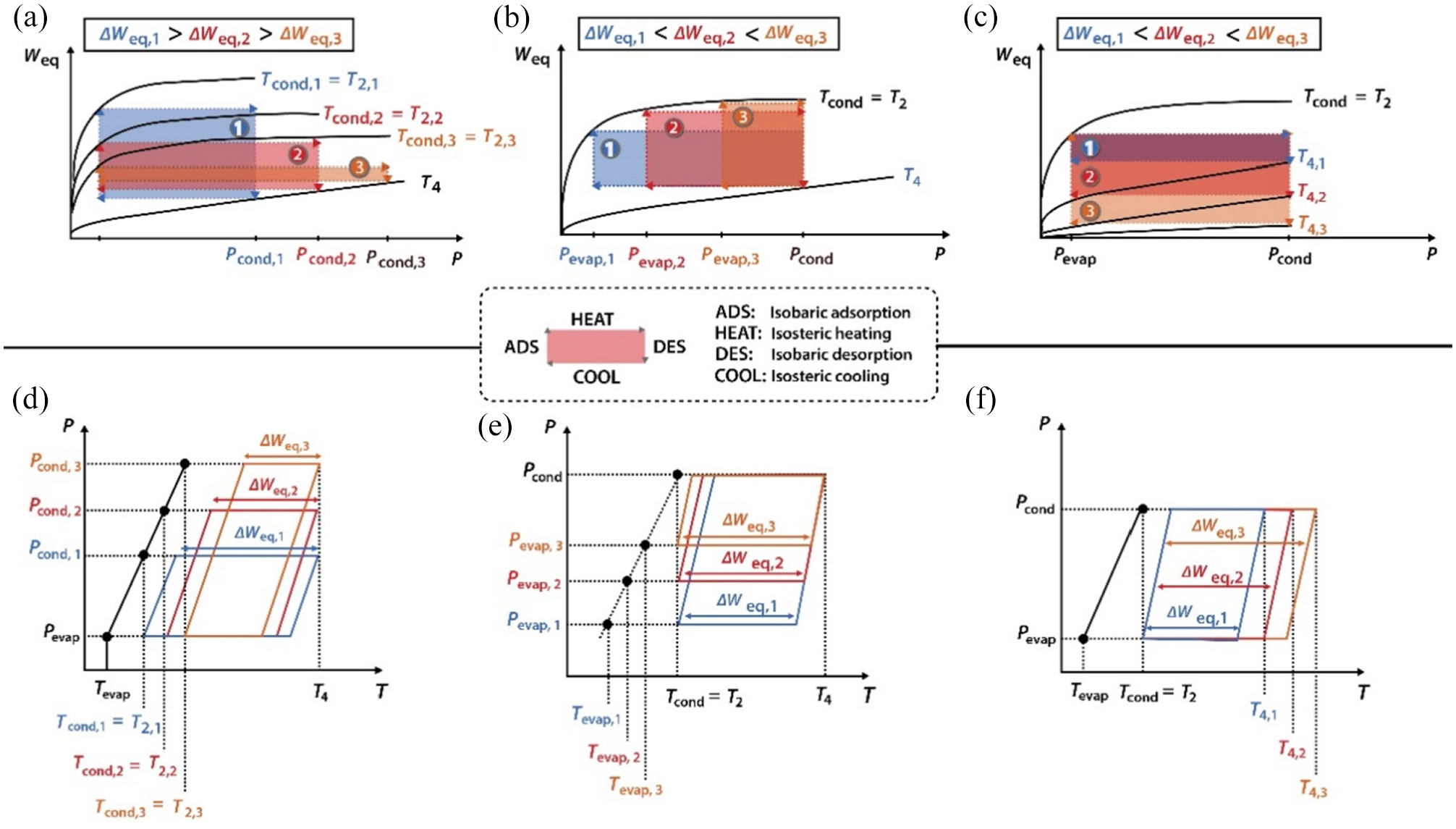
Impact of increasing: (a and d) Tcond from Tcond,1 to Tcond,3; Pevap from Pevap,1 to Pevap,3; and T4 from T4,1 to T4,3, using (a–c) type I isotherms and (d–f) ideal P versus T plots. The equilibrium adsorption cycles are represented by colored rectangles (case 1: blue; case 2: red, case 3: orange), reproduced from Pinheiro et al. 1
The effect of minimum bed and condenser temperatures is that with the increase in the condenser and minimum bed temperatures, the adsorption and desorption stages are constrained. As presented in Figure 4(a), a reduction in ΔWeq from ΔWeq,1 → ΔWeq,3 occurs when Tcond is increased from Tcond,1 to Tcond,3 when T4 and Tevap are kept constant. As the Tcond increases from Tcond,1 to Tcond,3, the adsorption cycles become increasingly constrained (Figure 4(d)) and result in a uniform drop in the performance of AHP. This phenomenon occurs in successive cycles for domestic hot water (DHW) production, such as the setpoint value from ambient air temperature being defined by the consumer.
The bed regeneration temperature and evaporation are associated with each other, as shown in Figure 4(b). The increase in Pevap from Pevap,1 to Pevap,3, when T4 and Tevap are kept constant, increases the ΔWeq. Consequently, an increased Weq was attained after the adsorption phase (Figure 4(b) and (e)). Furthermore, an increase in the ΔWeq and the adsorbent regeneration occurs when T4 is increased, as shown in Figure 4(c) and (f).
Mathematical investigation of AHPs
The mathematical modeling development and its subsequent investigation and validation is a crucial phase. Therefore, the comparative analysis and theoretical results or actual experimental values have been incorporated where possible. The presentation of solutions of various models has been made because the majority of the models are expected to be addressed through numerical methods. The major conclusions drawn from the mathematical models were also defined, including the adsorbent-adsorbate pair’s physical properties.
Adsorption equilibrium
Four equations are widely used related to adsorption equilibrium: Freundlich equation, Dubinin equations (typically Dubinin-Astakhov equation), Toth equation, and Langmuir equation. Hassan et al. 41 have presented a thorough review through the theory and equations of adsorption equilibrium. Another work on the experimental adsorption data for the AQSOA zeolite-like adsorbent and equation predictions comparison was performed. 42 The International Union of Pure and Applied Chemistry (IUPAC) 43 classifies equilibrium isotherms into six forms (I–VI types), depicted in Figure 5. Hysteresis can be observed in Type IV and Type V isotherms represented through the red curve for adsorption and the blue curve for desorption. An S-shaped isotherm is advantageous for AHP applications since a large portion of adsorbate absorption occurs at reduced relative pressures. 44 Many other simulation models for adsorption kinetics use the LDF (linear driving force) technique, which is a feasible approach for portraying the growth of adsorption uptake in time.45,46

Classification of adsorption isotherms according to IUPAC. 47
Numerical methods and physical models
Thermodynamic models
The model centered on thermodynamic evaluation is the basic model between the several models, wherein heat transmission characteristics are not considered. Therefore, it is only possible to run first and second-law studies in the model, which may be employed to identify the maximum effectiveness limitations. The first law analysis delivers a decent response to a cycle’s performance expectations, and the second law investigates the efficiency. 48 Cacciola and Restuccia 49 suggested a thermodynamic model derived from experimental evidence for regenerative and simple cycles. An analysis was performed to assess the efficiency of adsorption systems in a cooling processor or a heat pump and the helpful heat/cold produced per kilogram of adsorbent throughout a complete cycle. In total, there were three adsorbent/adsorbate pairs of regenerative cycles, for example, AC-35 activated carbon/methanol, 13X zeolite-water, and 4A zeolite-water, that were compared. The research demonstrates that zeolite/water is ideal for creating devices for employment in the household. Christoph 50 researched the efficiency shortcomings of solar cooling adsorption cycles and developed a simple model focused on a preliminary assessment of the thermodynamic process. Shelton et al. 51 presented a study of the thermal wave adsorption cycle based on several presuppositions. The adsorption characteristics and temperature throughout the bed function were given as square waves to give reasonable and limited wavelengths. Hence, the model has been proved to produce solid first-order results.
Researchers further expanded by considering the adsorbent bed with a more reasonable ramp wave shape. 52 Instead of the simpler square wave model, the ramp wave model gives a reasonable representation of the adsorption performance, allowing for a more accurate estimation of system performance. The findings showed that the heating COP on a thermal basis was 1.6 and that the performance was somewhat unresponsive to the heat input/output temperatures of the cycle. Finally, Gui and Wang 53 presented a functional model for a heat regeneration adsorption air-conditioning system. The three-heat-reservoir cycle corresponds to the conditions are: (1) Elevated temperature reservoir (indicated by the temperature of the heater, Th) for generator power supply (Qh); (2) The low-temperature reservoir (represented with fan coil supply temperature, Tf) produces the latent heat cooling power (QF); (3) Heat sink (in the ambient temperature, T0) removes the heat generated in both condensers and adsorbers. In the model mentioned earlier, the main governing equations emanate from the laws of thermodynamics, the heat leakage in the cycle has been overlooked, and adsorption has been in balance. A prototype of a natural cycle was developed, and several tests were undertaken. The actual thermodynamic cycle was discovered to be nearly similar to the ideal cycle on the PTX graph. The impact of Tf and Th on SCP and COP have also been evaluated among the practical and experimental thermodynamic model findings, and the concordance was determined to be successful.
Lumped parameters models
Some specific assumptions are applied commonly to facilitate the study depending on lumped parameters; (i) there is a uniform temperature within the adsorbent; (ii) uniform adsorption of refrigerant occurs in the adsorber; and (iii) at thermodynamic equilibrium, both solid and gas phases exist. The thermal resistance and mass transfer resistance of the adsorbent are disregarded in these models. One of the features of such models is that the adsorbed phase varies with time generally as a dynamic model. The model generally consists of three main equations: energy equilibrium, mass equilibrium, and the adsorption equilibrium equation. Throughout the desorption process, the generalized statement is presented of an energy balance equation (11) for the adsorbent bed 54 :
Consequently, throughout the adsorption process, the energy equation (12) is obtained as:
The mass balance of adsorbate was provided in the model proposed by Sakoda and Suzuki 46 as follows (equation (13):
The term on the left-hand side bracket of equations (11) and (12) comprises the sensible heat of the metallic elements of the adsorber, completely regenerated adsorbed bed, and the adsorbed phase. The first right-hand side term of equations (equation (11)) and (equation (12)) is the heat due to desorption and adsorption. The character w → z indicates heat from the heating or refrigerating medium. Owing to the movement of coolant vapor, the third component on the right-hand side of equation (12) is sensible heat, where “mae” in equation (13) is the evaporator adsorbate.
Douss and Meunier 55 proposed the predictive dynamical model. It employed a primary lumped model parameter and considered that in the adsorber, evaporator, and condenser every component was homogenous in the system. Thus, the model was suitably confined to waterfall heat exchangers and film evaporators. However, there were also certain numerical instabilities, especially in short periods. The two-reactor heat adsorption pump was numerically simulated by Cacciola et al., 56 given the heat regeneration. Two critical assumptions were made: to facilitate the analysis (a) each reactor had a uniform temperature and (b) a balanced adsorption operation. The adsorber, condenser, and evaporator’s energy balance equations were constructed based on the efficiency of different components. The equations have been changed and numerically solved to a non-dimensional form. At different times, the convergence of the numerical approach was validated. The model was evaluated using the adsorption pair of zeolites and the technical characteristics available. 57 Douss’s findings were consistent with the projected changes of temperatures of the adsorbent bed, condenser, and evaporator. 57 It was also experimentally validated, 58 and the software was an effective tool for the design of adsorption heat pumps and for the maximum performance to be optimized in component sizes.54,58
The models presented by Wu et al. 59 were similar to what Sami et al. 60 have suggested. A new heat exchanger tube and shell-type were used as adsorber in their investigation, but its impacts were not analyzed. Instead, an experimental investigation has been conducted depending on the experimental parameters reproduced by the suggested model. In the simulation, various operating variables were optimized according to experimental data, including cycle duration, the heat source temperature, cooling water, and the adsorber total heat transference coefficient. On the contrary, Sami’s model 60 calculated the heat transfer coefficients correspondence for smooth tubes. The findings therefore correlated closely with the computation of dynamic simulation data in the research of Wu et al. 59
A double bed silica gel-water adsorption chiller was presented by Saha et al. 61 as a numerical simulation comparable to that of Sakoda. 46 By applying the Freundlich Equation with the experimental data of the supplier, the adsorption balancing equation was achieved. The simulation was carried out to validate the working temperature effect, discharge, and adsorption-desorption cycle periods. It was shown to be the most crucial parameter of COP of the system for the operating temperature associated with water discharge. In addition, it was discovered that cycle times were less quantitatively impactful but still quite essential in terms of quality. Critoph 62 developed and patented a novel, ongoing rotational cooling adsorption system. The technology is comparable to the Llobet and Goetz 63 rotary systems. The adsorbent bed within the tube as a closed segment of the module in the rotational system was suggested by Critoph 62 which includes a generator and a receptor/condenser/evaporator. The coolant in the module was comparable to the coolant in the heat pipe. A laboratory-sized prototype device was manufactured and validated. As mentioned earlier, the efficiency of the system, comprising of 32 modules, was forecasted by applying basic governing equations. Research development on improving this system has been proposed and uncovering the optimal constraints of the notion utilizing second law analysis.
Heat and mass transfer model
Simulation-based heat transfer and mass transfer are particularly relevant among many models found in the field as they offer an interpretation of the adsorber dynamics. A model for heat and mass transmission was developed, demonstrating that the temperature or weight of adsorbate fluctuates with time and space. For the appropriate/optimal design of adsorption systems, it is therefore vital. The models may be categorized into single, two- and three-dimensional models based on the adsorber geometry. The variations in the simplification of assumptions, the numerical resolution method, the design, and the purpose often exist across different models employed for the adsorption cooling system simulation.
The heat and mass transmission of solid adsorbents to adsorbent beds generally includes four primary governing equations: energy balancing, mass conservation equation, momentum equation, and adsorbent-adsorbate system state equation. Generally, the completeness and nonlinearity of these heat-connected models restrict the feasibility of an analytical approach. Therefore, numerical approaches are ultimately the only possible solution for the modeling of adsorbent bed dynamics. Several approaches employed provide the technique of final differences,64,65 finite volume, 66 and the technique of finite element. 67 The finite difference approach is commonly used due to its brevity, effectiveness, and easiness of different start and limiting conditions. Some assumptions have been made that may be divided into four categories to ease numerical analyses, involving heat and mass transfer: (i) thermodynamic condition; (ii) heat and mass transfer; (iii) evaporator, heat source, and condenser; (iv) thermophysical and material characteristics.
To evaluate the heat transmission of a solid, fixed bed adsorbent, Guilleminot et al. 64 devised a uniform pressure model. The approach disregards mass diffusion resistances yet incorporates heat diffusion resistance into consideration via two coefficients: the adsorbing bed’s thermal conductivity and the thermal transmission coefficient among the fins and the adsorbent bed. Boubakri et al. 68 performed the comparable modification of the Guilleminot et al. 64 models to predict the operational efficacy of an adsorbent solar-powered ice system and conducted experimental verification. The classification procedure utilized by Guilleminot et al. 64 was employed for three unknown heat transfer coefficients related to condensers, evaporators, and ice tanks. This universal model also determined the ice production limit employing an adsorbent collector-condenser system: daily ice production (D.I.P.) may approach around 115 kg/m2 of collector and the COP of around 19%. An expansion of Zheng et al.’s model may be regarded as a standard numerical model69,70 that Sun et al. 69 could first offer. This model was utilized to study several features of heat pumps for thermal wave regeneration. The adsorbers have high-temperature gradients: the energy transmission to the adsorber during the heating phase is considerably greater than that provided by the heat supply. Pons and Feng 70 also utilized the previous model to study the influence on the adsorption cooling cycles of the number of transfer units (NTU) and the dimensionless output fluid temperature using thermal regeneration.
Szarzynski et al. 71 utilized this model to study various internal vapor transfers to study the adsorber to lessen the internal irreversibility of adsorption cycles. Firstly, adiabatic direct pressure recovery with a condenser instead of pressure variations with heat transfer; secondly, adiabatic of inner vapor recovery among adsorbers; thirdly, the division of the adsorber into specific chambers. 64 The one-dimensional model of Hajji and Khalloufi 72 was nearly similar to Guilleminot et al., 64 with some adjustments. By taking on the continuous fluid heating/cooling temperature, researchers have improved the model. The definite difference equations were computed employing an explicit technique and confirmed utilizing the average values of the apparent heat and heat-conductivity comparison of the projected temperature gradient with the analytical formulation.
The parametric study of this two-dimensional model demonstrated that the sorption kinetics might be considerably improved by lowering the distance among the fins and the contact resistance of the metal adsorbent contact. Therefore, thin fins and inexpensive metals have been advocated for minimizing the operating cost and weights. Sward et al. 73 suggested a model wherein heat transfer travels axially and thermal contact with the bed for thermal wave adsorption thermal pumping cycles. The exponential performance was expected to be provided via local balance.
The material balance f(z) for every portion of the bed included an adsorbed phase term, a gas-phase term, and a flow term as follows (equation (14)):
The energy balance comprises the four terms stated earlier, including a heat flow term via the bed walls (Jb).
In which g(z) in equation (15), input and outlet valves represent the energy fluxes from and into the bed. The preceding equations have been transformed into dimensional, differentiated, and inserted in the ordinary differential equations into multiple separate cells of a specified height, and the equation set was numerically included in the Gear’s process using the LSODE software. 73 The findings revealed an influence on the layout of the temperature and loading fronts of the entrance/outflow valve inside the bed.
Design and operation of AHPs
Various research have been conducted and are reported in the literature to consider the AHPs operation and design. According to the literature review conducted, and based on the operating conditions, performance indicators, and the adsorber heat exchanger (AHEx) geometries of the AHP systems, it has been observed that around half of the reported research literature evaluates the performance of AHP based on simulation and modeling or is theoretical. In addition, several studies involve experimental investigation, which involves AHP prototypes and a combination of experimental and practical approaches.
Adsorbent-adsorbate working pairs
Significant research has been conducted and continues to be carried out to classify and evaluate adsorbent-adsorbate working pairs, usually under standard operating conditions.3,4,13,30,74–77 Water is perhaps the most used adsorbate, but ethanol, methanol, and ammonia are also used. Since they have low vaporization temperatures, methanol and ethanol are widely employed for refrigeration applications. 13 Since ammonia has high specific heating power during the adsorption phase due to high pressure, thus it is widely employed with activated carbon adsorbents, but it has the disadvantage of being toxic and emits a strong odor accidental leakage. 13 Activated carbons, zeolites, zeolite-like AIPOs (aluminophosphates), silica gels, and SAPOs (silico-aluminophosphates) are common adsorbent materials for AHPs. One of the most critical aspects of a durable adsorption heat pump is the adsorbent-adsorbate pair selection. While several experiments on working pairs have been performed, most of them focus solely on the kinetics and outputs of the adsorbate and adsorbent materials, overlooking their practicability for a functional or marketable framework. Numerous working pairs with outstanding adsorption potential are currently being presented; even then, the majority of these have poor hydrothermal stability or exhibit hysteresis due to material maturing through some cycles. One other consideration includes market readiness, and while some adsorbent materials exhibit strong, hydrothermal durability, absorption capability, and prolonged life cycle time.
Zeolites
Though zeolites can be paired with various refrigerants, water is by far one of the most frequent. Zeolites, including silica gel, were the most studied adsorbents and have been used in experimental systems. The zeolites’ water absorption ability and adsorption heat are both significantly higher. Therefore, the employment of zeolites in adsorption heat pumps as adsorbent material at high desorption temperatures (200°C–300°C for natural zeolites) is severely restricted. Several advances in lowering the desorption temperature have been carried over the recent years.75,78
Silica gel
In recent years, silica gel has been regarded as a conventional water adsorbent on which a vast number of studies have been conducted, and it has seen widespread use in numerous adsorption systems. The low cost, low regeneration temperatures (60°C–100°C), and durability in potential uses are advantages of silica gel adsorbents. 75 Unfortunately, at elevated relative pressures, the majority of water adsorption occurs. Many approaches have been developed to overcome this issue, including reducing the pore size and incrementing the number of silanol groups on the surface. However, mesoporous silica, including aluminum or other metals, has the highest water adsorption capability. 78
Activated carbons
Activated carbons find extensively use as adsorbates of ammonia and methanol. They are found in powder, lightweight, granular, monolithic carbon, and fiber forms. The temperature levels for activated carbons vary greatly depending on the adsorbate utilized and their type. Tamainot-Telto et al. 79 presented in-depth research on the activated carbons for adsorption refrigeration applications.
AIPO and SAPO
Aluminophosphates (AIPOs) and silico-aluminophosphates (SAPOs), like zeolite materials, have a high capacity to absorb water and the ability to be employed with reduced temperatures of desorption, that is, (60°C–100°C). The S-shaped isotherms are possessed by these materials, which means that they have a strong potential for water exchange at reduced temperature variations.75,78,80 On a commercial level, two innovative adsorbent materials were developed by Mitsubishi Plastic Inc. named AQSOA®-FAM-Z02 and AQSOA®-FAM-Z01.80,81 Both materials have desorption temperatures of 60°C–90°C and seem like a reliable alternative for various applications such as heating.
Metal-organic frameworks (MOFs)
The porous metal-organic frameworks are a new set of water adsorbents due to the exhibition of the distinct inability of their hydrophilic and structural properties. 38 Even though MOFs have high methanol, ethanol, and water, adsorption capability under typical operational conditions, deterioration over time, high cost, and hydro-thermal volatility remain significant barriers. 75 Nonetheless, research conducted by Henninger et al. 82 shows that MOFs are significantly necessary materials for potential use as adsorbents. Hysteresis conducts and hydrothermal stability must be enhanced so that these materials can be employed on experimental systems. New technologies are investigated and developed every day, and some of them, such as MOFs (Metal-Organic Frameworks) and composite adsorbents, show great potential for potential applications.9,38,82–87 However, even though the adsorbents depict high performance and uptake capacities, they are also expensive, hydrothermally unstable, and degrade over time, which are significant barriers. 75
Consequently, they are hard to fabricate on a broad scale, rendering them unsuitable for specific applications requiring large-scale manufacturing. Some recent literature includes an overview of adsorption material research.4,13 Screening of potential working pairs was conducted by Boman et al. 80 for AHPs possessing transport and thermodynamic properties. Additionally, an exhaustive quantity (around 100) of comparisons of adsorbent-adsorbate pairs, along with the uptake equations of working pairs, are investigated and cited, acting as a valuable database for future study. 80
Composite adsorbents
The formation of composite adsorbents is generally occurred by the insertion of inorganic salts in the porous matrices. The adsorbent-adsorbate can be paired such as ammonia, ethanol, methanol, and water.75,88 Among them, several materials that exhibit relatively reduced desorption temperature, that is, 70°C–120°C, and possess a large intake of water, that is, 0.75 kg/kg, based on mesoporous silica paired with LiBr (SWS-2L) and CaCl2 (SWS-1L).89,90 The composite SWS-8L permits a reduced desorption temperature of 75°C–80°C with water uptake of 0.2 kg/kg 90 . Several experiments on the use of ethanol and methanol as adsorbates can be found in the literature.91–93
Heating performances
It is worthwhile considering the variation of SHP and COP values mentioned in the publications for adsorption heating purposes over the last several years. Figures 6 to 9 depict this evolution by differentiating the working pairs, cycle characteristics, and work domain for different cases. For example, commercial AHPs have COPs in the region of 1.30–1.50 when zeolite/water is used or 1.40–1.60 when silica gel/water is used. 38

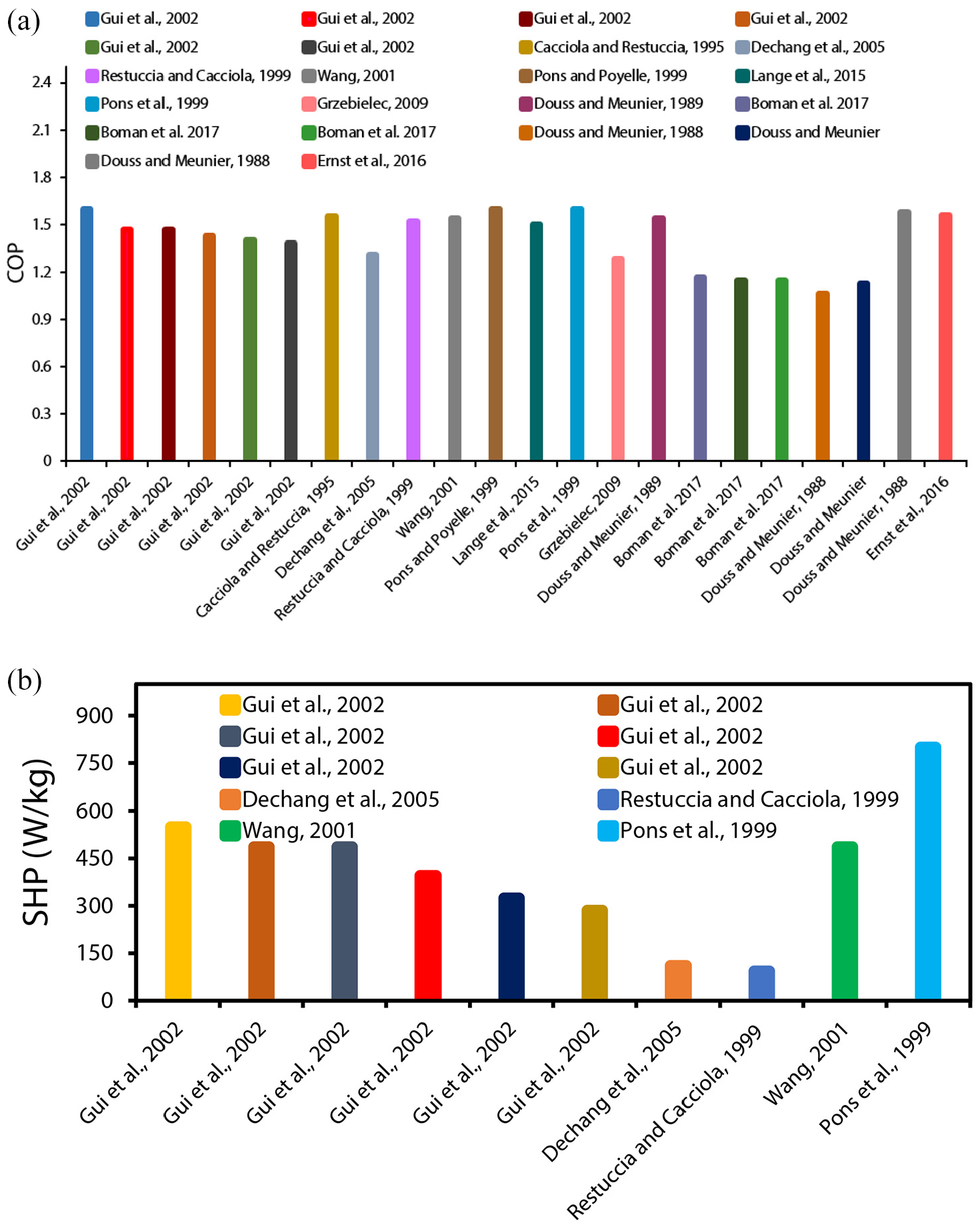
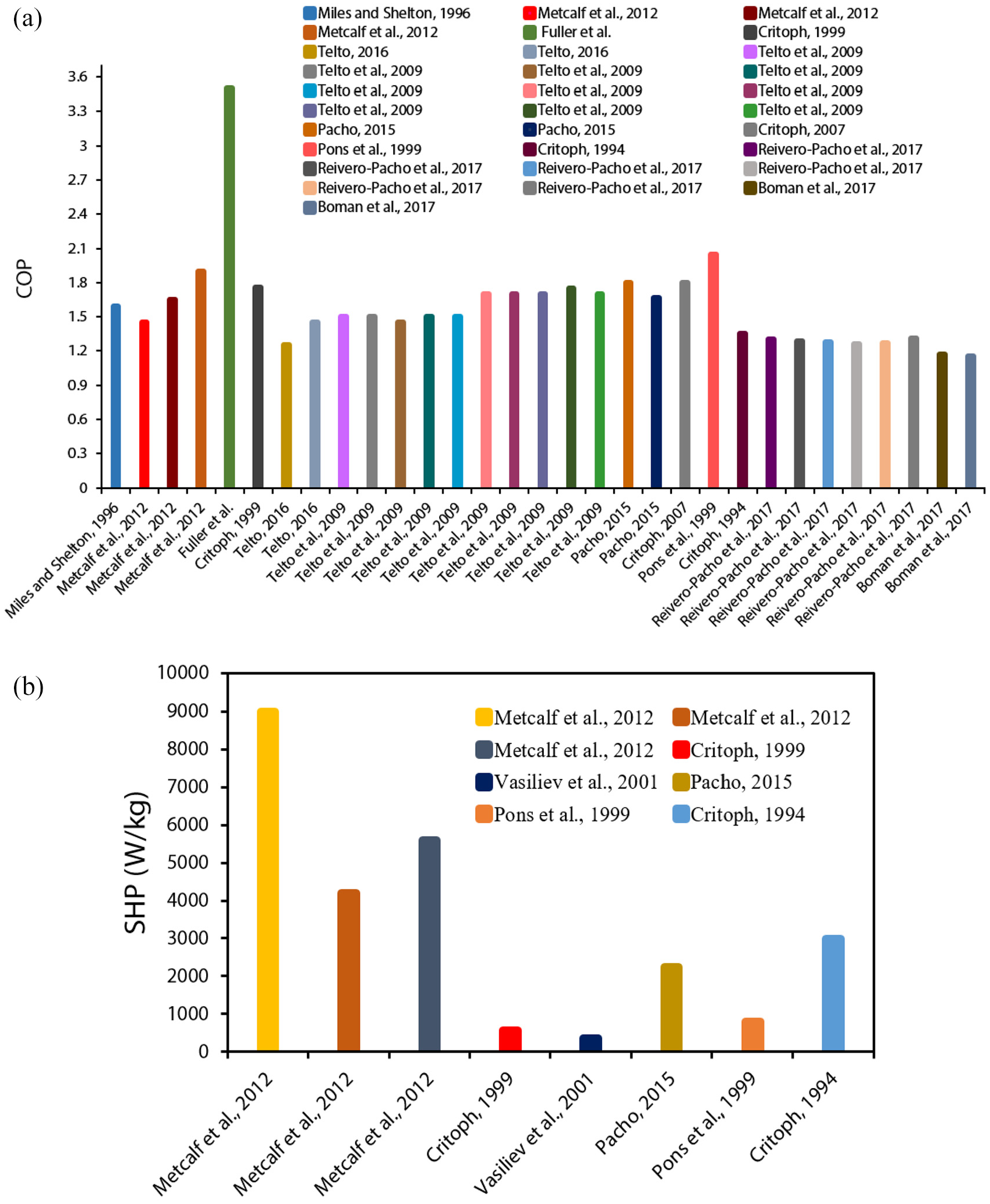

An investigation by Freni et al. 75 stated that the suitable COP for heating pumping applications is >1.55, which was following the suggestion of Shelton et al., 52 which was 1.50. The COP values discussed in Figures 6(a), 7(a), 8(a) and 9(a) assist as references. Contrary to this, for the cost advantage of AHPs, the minimum SHP is found to be ca. 1000 W/kg, 120 whereas the SHP is 1600 W/kg for Viessmann AHP, which is regarded as a remarkable reference case in the coating technology of adsorbents. The SHP values discussed in Figures 6(b), 7(b), 8(b) and 9(b) assist as references. The difference between the experimental and theoretical values of COP on an average is 18%, where theoretical values outperform experimental values. The highest COPs having the values of 1.30–1.60 reported in experiments are within the market range. 38 In 2016, by employing silicoaluminophosphate SAPO-34/water working pair, the COP of single-bed AHP was 1.4083 whereas, in 2011, AQSOA® FAM-Z02/water working pair was employed and for single-bed prototype, the reported COP was 1.47. 115
Critoph 106 in 2007 investigated the COP of a two-bed, activated carbon (AC)/ammonia-based, forced convection AHP prototype, where COP was found to be 1.80. Furthermore, in 2005, another four-bed prototype using a heat recovery cycle was reported with a COP of 1.31 using the activated carbon (AC)/ammonia working pair. 107 In contrast to AHP industry statistics before 2012, no significant changes in creative COPs have been recorded since that time. Perhaps this is primarily due to the small adsorption ability of the investigated adsorbents. Attempts are being taken to produce new materials with excellent adsorption capacities to increase the efficiency of AHPs. 82 The single-bed AHPs, which employ water and novel metal-organic frameworks (MOFs) as adsorbate, have been found to report theoretical COPs greater than 1.55. 38 The analysis of AHPs using COP (Figures 6(a), 7(a), 8(a) and 9(a)) tend to be more discussed in the research than the evaluation using SHP. In theoretical cases, SHP values were on average 35% greater than in practical applications. In advanced cycles, for AC/methanol and silica gel/water pairs, the experimental SHPs of up to 500 W/kg were recorded until 2007, with the adsorbents specifically in a loose grain configuration.105,115,121
In a study, coatings were presented and found that a single-bed AHP with AQSOA® FAM-Z02 binder-based coating for water adsorbate obtained a maximum SHP of 2200 W/kg. 122 In 2016, SAPO-34 coating was prepared through direct crystallization, where water was used as adsorbate, and the SHP higher than 3200 W/kg was recorded. 123 An impact of adsorbent configuration on experimental SHP was demonstrated by contrasting the (zeolite or zeotype)/water working pairs based on single-bed AHPs, and it was found that: loose grains (240 W/kg (zeolite/water in 2001)) < binder-based coatings (2200 W/kg < directly synthesized coatings (3200 W/kg)). Coatings were found to have a 10-fold rise in SHP as compared to lose grains. Consequently, another research presented that zeolite coating technology has allowed attaining the SHPs in the thousands, which is around 10 times greater than random zeolite pellets among finned tubes. Consequently, transitioning from traditional pellets/grains to novel adsorbent configurations is critical for improving cost-competitiveness and adsorption heat pump’s SHP. Furthermore, improving the efficiency of AHPs requires the production of materials with optimized adsorbent configurations and enhanced adsorption capacities for effective heat transfer.
Adsorbent bed configurations
The adsorber bed configurations vary in their packing techniques, including finned tube, coated tube, spiral plate, plate-finned, fin plate, flat pipe, and tube, elaborated by Li et al. 124 Similarly, shell packing was employed by Pinheiro et al. 3 The significant impact of various configurations is the variable metal to adsorbent mass ratios, heat losses to the atmosphere, adsorbent-metal wall contact areas, and the area of adsorbent being exposed to the adsorbate. Consequently, a significant amount of effort must be expended to choose the right adsorber arrangement, which can also vary depending on the physical properties, adsorbent-adsorbate working pair, and adsorber metal thermodynamics. Nonetheless, the HTF travels the adsorbent material within shell configuration, while in the tube configuration, the adsorbent material is wrapped all over the tube, minimizing heat losses so heat could only be transmitted to the adsorbent material from the tube. The finned heat exchangers have a smaller adsorbent-adsorbate contact area and a greater adsorbent-metal contact area and, consequently, a higher mass ratio of adsorbent-metal. The adsorber bed design process should include the following essential factors: a maximum amount of heat transfer should be ensured among the heat transfer fluid and the adsorbent material, adsorbent surface-exposed should be maximum to the adsorbate, the metal-adsorbent mass ratio should be minimized, an excellent thermal interaction among the metal and the adsorbent bed should be assured, and the overall design should be kept straightforward that it can be developed by using the vacuum technology elements. Choosing the correct arrangement and constructing the appropriate adsorber is a complex process that necessitates additional research to achieve the best combination between all these factors. 29
The configurations of the adsorbent bed being commonly employed can be classified into four types: binder-based coatings, consolidated beds, directly synthesized coatings, and loose grains.
125
For loose grains, fibers, pellets, powder, and adsorbent grains are inserted in the heat exchanger without any binders. Whereas for consolidated beds, the pore-forming additive and the binder are compacted along with the adsorbent to ensure the formation of a dense compressed layer on the wall of HEx. The preparation of binder-based coatings is performed by combining binder and an adsorbent, and then the composite material can be served on heat exchangers using dip coating or spraying techniques. Directly synthesized coatings are created by depositing adsorbent directly upon the HEx walls.
125
Considerable attention (approximately 80% of the published studies) has been received by loose grains configuration, probably owing to its simplicity and being relatively inexpensive. All of the other remaining configurations account for below 10% of the findings. Table 1 presents a quantitative comparison of the four-bed configurations, representing the bed thickness (based explicitly on research for silica gels, zeolites, and zeotypes (i.e. SAPO-34, AQSOA® FAM-Z02) where water acts as adsorbate), heat transfer coefficient on the adsorbent side (hads), the thermal conductivity of the adsorbent bed (λads). It also includes the qualitative comparison based on mechanical resistance, pore structure stability, bed permeability, and industrial factors. Improvements in the AHEx total heat transfer coefficient (U) benefit heating efficiency, for which higher λad and hads, and smaller δ are advantageous, conferring to equation (16
Comparison of the adsorbent configurations for AHPs in terms of λads, hads, δ, bed permeability, stability issues, and industrial application features. 1

where Afluid denotes heat exchanger area on the fluid side, Aads represents heat exchanger area on the adsorbent side, and hfluid is the convective heat transfer coefficient on the fluid side. Then, equation (16
The λads values for the beds comprised of loose grains/pellets or binder-based coatings, in the tenths of W/m K have been reported, as shown in Table 1. For example, for the equivalent powdered bed, the λads = 0.11 W/m K and AQSOA binder-based coating λads = 0.36 W/m K 126 ; for zeolites, zeo-types, and silica gel, λads in the range 0.10–0.20 W/m K.26,32,120 Restuccia et al. 26 identified a binder-based coating with λads = 0.30 W/m K. λads values for condensed beds and specifically synthesized coatings were nearly two orders of magnitude higher than for binder-based coatings and loose grains/pellets. For loose grains, the values for convective heat transfer coefficients (hads) are reported in the range of 70–120 W/m K, 133 but the most commonly used values are 10–50 W/m K. 133 For the other configurations, the convective heat transfer coefficients are greater than 1000 W/m K, such as, for directly synthesized SAPO-34 coatings on aluminum fibers, binder-based coating of silica gel, and consolidated 13X zeolite, the hads are >1000 W/m K 125 for former, and up to 3000 W/m K125,134 for the later ones, respectively. Such findings are helpful for improved interaction among the AHEx metal and adsorbent surface while consolidated beds or coatings are used rather than loose grains. In terms of stability, direct synthesis’s loose grains coatings and configuration are typically not a major concern. According to Freni et al., 125 direct zeolite crystallization produces enhanced coating stability. The reduced mechanical resistance 135 possible compression of pores due to the binder, and the hazard of volatile compound outgassing from natural binders (which impacts pressure and can result in total failure) of binder-based coatings are disadvantages that must still be addressed to obtain sustainable products. 125 There can be a negative effect of large quantities of additives or binders on the performance of AHP because they can cause pore occlusion, which further leads to the reduction in the adsorption capacity. 125 Contrastingly, in the binder-bed coating, a typically thicker layer is attained, which restrain the mass transfer, and at the interface in coating and the HEX, thermal resistance is exhibited, decreasing the efficiency of heat transfer. Nonetheless, the attained performance for the binder-based technique is indeed very satisfactory, as evidenced by comparative research published in some of the scientific publications,136,137 wherein the authors performed corresponding experimental studies on both the loose and coated grain configurations, using the identical heat exchanger equipment.
Significant pressure reductions can result in grain beds based on the size of particles. However, the problem is usually not as severe as heat transfer constraints (low λads and hads).125,138 Because of the shortage of information in the literature (Table 1) shows the bed permeability qualitative comparisons. Nevertheless, for the adsorbent beds’ permeability, the favored values are more than 10–12 m2,123 but the estimates stated in the research are in the range of 10−14 to 10−8 m 2 for the beds employing methanol, water, or ammonia as adsorbates. 139 The loose grains replacement by directly synthesized coatings allows for an increase in λads and hads by 1–2 orders of magnitude and a considerable reduction by a factor of around 100 in δ.
Conversely, it results in a reduced mads = mmetal ratio, which reduces COP. 125 To make the materials marketable, the industrialization of product features is of utmost importance, and to enable cheaper production, the loose grains beds are inarguably one of the basic configurations. The heat exchanger consists of adsorbent particles wrapped and enclosed with a grid. 140 However, the adsorbent structure in consolidated layers is limited to basic geometries such as cylindrical and planar, limiting the application in complex and actual HEx. 125 Furthermore, directly synthesized coatings may necessitate costly and time-consuming production practices. For example, in SAPOs, the necessary conditions for autoclaves are 200°C and 20 bar.
Consequently, for industrialization, the production of simplified and less expensive preparation procedures is favorable. 125 For heat exchanger production complexity and expenses, binder-based coatings fall among directly synthesized coatings and loose grains because they can be manufactured in mild reaction conditions in serial production lines and therefore are simple to scale up and incorporate. 125 It was presented by Schicktanz et al. 140 that dip coating can be employed for the formulation of binder-based coatings, which involves the metal substrate immersion in an organic binder (such as resins) mixed active powder liquid solution or an inorganic binder (such as aluminum hydroxide clays). For the removal of surplus solvent, it is thermally treated. In this way, by adjusting the dipping velocity and liquid solution’s viscosity, the coating thickness can be varied from 1 × 10−4 to 10 × 10−4 m. For the consolidated beds, the reduced bed permeability is a significant disadvantage. 140 It has been observed through current findings that research on configuration for AHPs appears to be mainly concentrated on AC/ammonia working pairs.
Similarly, a parametric study was presented by Kowsari et al. 141 in which using the numerical model, heat exchanger (HEx) was employed as an adsorbent bed, and validation was performed with an experimental setup. Using variable pitches and fin heights, the trapezoidal and rectangular bed geometries were investigated, and then a comparison was made based on metal mass, COPc, VSCP, SCP, and adsorbent mass. While contrasted to a comparable arrangement for the trapezoidal geometry, the rectangular geometry demonstrated impressive SCP.
Earlier studies have shown that higher metal mass results in reduced COPc and that a strong adsorbent thickness results in a low SCP. Consequently, a dynamic model was presented by Graf et al. 142 that was experimentally optimized to estimate the COP and SCP of adsorption heat pumps. On a demonstrative adsorber component, gravimetric large temperature jump (G-LTJ) experiments were carried out for the calibration of the adsorber model, which is being used to predict COP and SCP of AHP. The evaluation of the model was made for a minor adsorber component, resulting in a faster analysis of operating and design parameters comprising operating temperatures, fin geometry, and cycle times. San et al. 143 and Frazzica et al. 77 demonstrated the AHP systems testing, and design based on earlier single. Such methodologies are strictly experimental and offer reliable device features for a provided experimental setting. Conversely, such experimental methods are restricted to the applied configuration, making a parametric study of the whole structure challenging because the whole setup will have to be demolished and rebuilt to adjust specific parameters.
According to the reported literature, it has been found that the binder-based coatings appear to provide reasonable exchange among performance drivers such as hads, δ, λads, bed permeability including manufacturing characteristics, and enhancements stability can increase their market value. This configuration has produced a slew of patent literature. Sauer et al. 144 reported the development of coatings that do not expel gases over the required 15-year period of AHPs and, are constituted of an inorganic colloidal binder such as colloidal aluminum oxides or hydroxides and colloidal silicon oxides for binding the adsorbent particles and thermally conductive inorganic fibers (such as carbon nanotubes, carbon fibers, glass fibers) to provide mechanical stability and elasticity. Nevertheless, experimental work on loose grain beds seems to be that when particles with diameters below 5 × 10−4 m and more than 1000 m 2 /m3 area/volume heat exchangers are used, this configuration can produce adequate efficiency. 140 The production of coated AHExs that allow for the scaling-up of coating procedures and compact designs continue to be well-known R&D’s main concerns in the AHP field. 11
Adsorbent heat exchanger (AHEx) geometries
The primary purpose has always been to attain relatively portable and lightweight systems by decreasing the weight of the adsorbers, that are the most important determinant from the perspective of dynamic optimization. 11 Adsorbent optimization entails determining the optimal balance between adsorbent material and HEx. Consequently, it is commonly known as the adsorbent HEX (Ad-HEX) optimization issue. 145 Hybrid systems, operation controlling, bed improvements, and adsorbents, including ADCs, are increasingly the topic of investigation. 146 Different heat exchangers in conjunction with different adsorbent placement methods result in various bedforms, such as consolidated bed, coated bed, and granular bed. 146
AHP adsorbent bed geometries are classified into four types: extended surface (including fiber plate and finned plate geometries), tubular (e.g. hairpin, spiral), plate-type (e.g. lamella heat exchangers) both with as well as without fins (annular or longitudinal). The geometrical classification is premised on the broad categorization of heat exchangers based on construction elements given by Shah et al. 147 ; however, finned tubes are incorporated in a specific area of tubular with fins. Figure 10 depicts the literature research on AHEx geometries and systematically studied pairings, where only works with experimental domains or theoretical and experimental domains were included in computing the percentages presented.

Adsorption heat exchanger geometries studied for AHPs (tubular with and without fins), extended surface (e.g. finned plate and fiber plate), plate-type (e.g. lamella heat exchangers), and percentage of works that adopted each geometry, along with information of the respective investigated adsorbent/adsorbate pairs. Again, only research focusing on experimental or theoretical and experimental work was considered to calculate the percentages.
The total heat transfer coefficient (U), heat exchanger size, and the ratio mads/mmetal are essential elements in AHEx performance 136 (Table 2). The most often used AHEx geometries for adsorbent/water pairings are extended surface heat exchangers and tubular with fins, as shown in Figure 10. Finned tubes have been reported to have heat transfer area per total volume of heat exchanger (or surface area density) values of up to 3300 m 2 /m3, whereas finned plate heat exchangers (extended surface type) can potentially reach ca – 5900 m 2 /m3, 147 enabling further efficient configurations. Table 2 depicts average readings of U, mads/VAHEx (an indicator of compatibility), and mads/mmetal for the two designs employing loose grains or coating structures: finned tubes/loose grains, 94 finned tubes/coatings, 122 extended surface/loose grains, 136 or extended surface/coatings. 148 Employing coated finned tubes rather than loose grains raised U by about 4.5 times, nearing 106 W/m2 K, resulting in higher hads and λads, and lowered δ (Table 1)).
Dawoud et al. 148 revealed that the SHPs for loose pellets in the finned tube AHEx were smaller than a fourth of the tested utilizing coatings. Thus, when employing loose grains rather than coatings, a more significant amount of adsorbent can be incorporated per unit volume of heat exchanger (mads/VAHEx) regardless of shape (Table 2). The ratio mads/VAHEx for coatings in extended surface, and finned tube AHExs is in the limit of 40–390 kg/m3, 148 and 100–200 kg/m3 149 (the corresponding typical values are 167 and 149 kg/m3). Generally, extended surface heat exchangers have had the greatest mads/VAHEx values; for loose grains, the value is (567 kg/m3) 150 for finned tubes the value is 315 kg/m398 for coatings, it is 388 kg/m3, 151 and the value for loose grains and coatings is 203 kg/m3, 149 respectively.
The findings imply that by employing an expanded surface geometry, relatively efficient AHExs may be developed. Consequently, a reasonable balance among the different variables is required to build efficient, small, lightweight, and large surface area AHExs, a research emphasis in the domain of AHP components. 11
Fin-tube heat exchanger
Fin-tube heat exchangers are straightforward designs centered on straight tubes with perpendicular fins. A finned tube has the potential to be utilized as a single straight line linked in parallel, curved into a coil with straight finned components, or formed into a u-tube with fins (Figure 11(a)). Fins can be rectangular or circular. Fin-tube HEX may be converted into an adsorption bed by covering the area between the fins using loose adsorbent granules and mesh enclosing to hold the adsorbent granules in place. It is also feasible to wrap the top of a heat exchanger with a coat composed of adsorbent granules and a binder. Poyelle et al. 152 used zeolite and fit-tube HEX to evaluate grained and cemented beds. The chiller was indeed a two-bed adsorption chiller, including mass and heat recovery. It was discovered that SCC might rise threefold to 97 W/kg and that mass transfer and heat transfer resistances in bed seemed to impact both COP and SCC substantially. Restuccia et al. 153 reported analogous findings during chiller studies using the material SWS-1L as adsorbent. Fins used in this kind of HEX may have varying geometries.
Schematic representation of a conventional adsorption heat pump, 129 (a) fin-tube heat exchanger, (b) plate fin-tube heat exchanger, (c) finned flat-tube heat exchanger, and (d) modular adsorbent bed.
A thorough examination of the effects of typical geometric characteristics on chiller efficiency was carried out. 154 Raising fin length necessitates more excellent heat for regeneration, yet it has been shown that the COP value grows simultaneously. Thus, the increased cooling power outnumbers the increased heating needs. According to the findings, utilizing circular fins to improve SCC value is advised, mainly when a low-cost heat source exists. A COP of 0.54 and 73 W/kgads of SCC was attained in experimental testing 143 of a bed with HEX packed with adsorbent particles wrapped with mesh. These findings can be pretty encouraging, especially given the ease of bed assembly.
In 2015, Sharafian et al. 155 investigated an identical bed structure specialized to an automobile A/C system. Rectangular plates have been used instead of convectional ribs on a u-tube. This was discovered that when employing loose grains, the fin interval should be more than the adsorbent particle size, while the overall HEX length stays the same. Li et al. 134 investigated silica gel in laminated bed designs. Hydroxyethylcellulose (HEC) was chosen among various binders, including bentonite, epoxy resin, and polyvinyl alcohol. Combining binder and powdered silica gel allowed for forming a 0.7 mm width of solid layer on the heat exchanger area while not affecting adsorption capacities. Caglar 156 exhibited in a numerical simulation, including its effects of fin design specifications on chiller with thermal wave cycle performance, found that increasing fin width will not enhance temperature distribution of adsorbent. The fin spacing (fs) and fin height (fh) analyses revealed that an increase in the fin length has a detrimental influence on heat transfer applications in the adsorption bed. Decreasing fin distance has been advantageous. However, it also reduces adsorbent weight, which reduces cooling capacity.
Plate fin-tube heat exchanger
A HEX structure comprises finned tubes parallel to one another to form a flat surface, as shown in Figure 11(b). Plate fins positioned opposite to tubes increase the heat transfer surface. This approach is widely utilized, for example, in radiators and air coolers, HVAC condensers and evaporators, and car radiators. The basic design of that kind of heat exchanger potentially acts as an adsorption bed foundation. Hence, converting this type of heat exchanger into an adsorption bed is not complex owing to the simple access to gaps among fins and tubes. Additionally, there is also an option of building a coated bed. The benefit of such technologies is the ability to expand cooling capacity by stacking heat exchangers. Mesh holds particles in position in granular beds. 157
The study used a commercialized silica gel-water chiller. Thermal contacting resistance among the HEX interface and the adsorbent layer was shown to restrict the operating variables of the chiller. It was calculated that it accounted for 25% of the overall bed’s thermal resistance. Simultaneously, an issue with mass transfer in beds utilizing adsorbent with binder was discovered. A reported approach involves adhering to a thin layer of adsorbent and filling open regions with loose adsorbent particles. A mixed solution with a partly bonded adsorbent is simple to use. Increased HEX fin distance can overcome any technical challenges with their fabrication. It improves access to the HEX surface while does not affect operating parameters – doubling fs leads to a 17% loss in cooling capacity. 35
As a result, powdered metal additions can be used to lower thermal contact resistance The predicted enhancement in thermal conductivity of the adsorbent layer is a 1.5-fold rise at 15% copper or brass and a 2.4-fold rise at 15% aluminum. For the construction of coated beds, 158 recommends utilizing granules larger than that employed in beds with loose granules. However, the coated bed was studied, 128 where a significant COP of – 0.48 – was obtained at hot water temperature. Chen et al. 159 investigated plate fin tube HEX using circular fins rather than rectangular ones. The round fins occupied all or most of the accessible gaps among the tubes. However, some were not surrounded by fins utilized for mass distribution. Having arranged the silica gel layer annularly, increased heat transmission in the adsorbent layer is witnessed.
Finned flat-tube heat exchanger
The various plate fin tube versions of HEX, which employ flat tubes having corrugated fins rather than the standard design, could be used in ADCs.127,130 In addition, such HEX is utilized in the automobile market, such as evaporators for air conditioning. 127 Figure 11(c) depicts the graphical representation.
Rogala 160 used modeling to investigate the effect of flat-tube HEX shape on chiller operation by establishing the fin length and fin spacing limits with the best chiller performances. The numerical research demonstrated that a selection of optimum HEX design characteristics and grain size substantially impacts heat transfer rate and heat transfer surface area. At the same instant, the suggested building may be manufactured without significant technological issues. For example, since silica gel granules had a size of 0.3 mm, the COP and SCC increased by 3.7% and 6.3%, correspondingly.
Modular adsorbent beds
Aside from employing commonly accessible heat exchangers to develop an adsorption bed, additional suggested structures are specifically designed for this application. Mitra et al. 161 presented a U-tube-based approach. Adsorption beds of this sort have a basic structure, making it essential to load the adsorption bed with an adsorbent. Nevertheless, the SCP and COP values obtained by the chillers with varied builds varied. Modular buildings are becoming more common. Liu et al. 131 developed a system that includes water channels, mass transfer channels, and consecutive layers of silica gel channels. A range of 0.33–0.42 for COP was found for a hot water temperature of 70°C using a basic bed structure.
Modular architectures were also described in works121,162 which included a numerical analysis of the researchers’ concept. The original idea had rectangular silica gel modules positioned among hot and cooling hot water channels, with sufficient space dividing the modules to allow for working fluid vapor diffusion (Figure 11(d)). When contrasted to commercially available alternatives, a more excellent value of cooling capacity per adsorber volume ratio was obtained. Adsorption bed function requires the employment of a working fluid vapor channel. The second structure suggested that the silica gel units be placed on grooved plates. It possessed cooling/hot water passages carved out of the plate and located/placed under adsorbent modules. Water vapor dispersion was achieved using extra channels and interaction with the most extensive surface of the cuboidal silica gel unit.
Modular designs allow for simple device expansion to the desired cooling capacity. Breaking the adsorber into fewer components may enhance the cooling capacity to adsorber volume ratio, resulting in a reduced overall system. Except for traditional heat exchangers, these systems need substantial analysis and a novel manufacturing process for the heat mentioned above exchangers.
Applications and patents
Extensive research has been conducted on AHPs by many researchers, including Gui et al., 99 Wang, 105 Wang et al., 132 Lu et al., 163 Wang, 164 and Wang et al. 165 AHPs that use water as a refrigerant incorporate those produced by Vaillant and Viessmann, which are known to be significant players in the domain of AHPs for heating applications in the construction industry, primarily while water is employed as a refrigerant. Granted and filed patents by Viessmann and Vaillant are presented in Table 3. Some major Vaillant AHPs are discussed here. In 1998166 an AHP was proposed with simple construction and reduced structural complexity, having one vacuum container. It employed the perforated radiation shielding in the evaporator/condenser and AHEx, preventing heat transmission through radiation between the components, ensuring no loss of efficiency due to radiation. In 2000, certain other patents were presented167–169 improving heat transfer through a corrugated spiral-shaped tube. The use of an AHEx with a single layer of grains and expanded surface area among ribs allows for a quick and effective adsorption-desorption operation. Another patent of AHEx was presented with an increased T heat exchanger, an adsorption heating circuit, and for the adsorbent, refrigeration water was used. An increased discharge is provided to the water to attain efficiency improvement.
A list of significant patents by Vaillant and Viessmann filed and granted according to the order of their priority year, which is, for each invention, the year of filing for the first patent application.
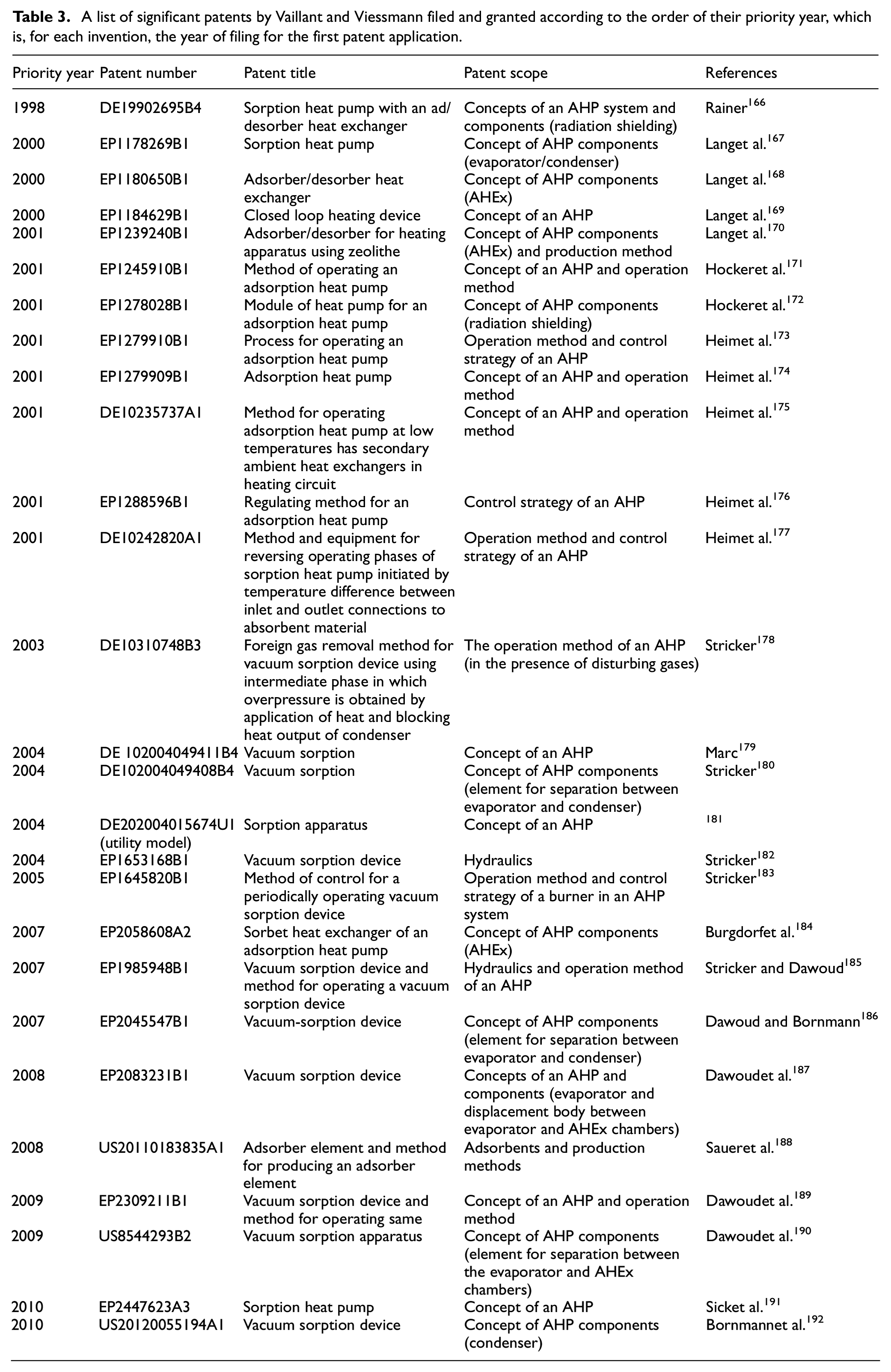
In 2001, some patents were demonstrated in the literature,170–177 which included the AHEx where zeolite pallets are tightly placed about a pipe by flanged rims and lamellas good heat transfer with the HTF. Similarly, the patents were presented with the adsorbent heat exchanger production method. Another AHP with ease of construction was stated to prevent heat losses in the switching stages. A few other radiation shielding variants for AHPs are identified (heat transfer through radiation from the AHEx to the condenser reduces condensation capacity (in the desorption stage), and heat transfer between the AHEx and the evaporator reduces the amount of heat removed from the setting (in adsorption stage)). A combination of an AHP and a burner can be used to (i) preheat the air going into the burner; (ii) heat the evaporator (especially when there is low air temperature); and (iii) heat the fluid in the customer heating circuit to get the most out of the heat in the exhaust gases. AHP is paired with a burner, under which a traditional heater provides heating while the AHP module is unable to work. A continuous-action cascade control system manages the complicated control of AHPs. The temperature of the consumer circuit is one of the control parameters, and the mass flow rates of the HTF in the AHEx circuit and the fuel and air fed to the burner are among the manipulated variables. The difference in T between the AHEx’s outlet and the inlet is regulated by a controller, which inverts the activity (from adsorption to desorption and vice versa) when the variations reach setpoint values.
Similarly, from 2003 to 2005,178,179,182,183,190,193 the reported patents included the invention related to a foreign gas extraction technique carried out periodically (e.g. every 500 h) and whenever disturbing gases (such as CO2, N2) are identified between the adsorption and desorption phases. It entails heating the AHEx while still simultaneously encouraging stresses on the condenser, where the vapor and foreign gases are discharged through a discharge device. To prevent condensation of the desorbed vapor in the evaporator rather than the condenser, a proper solution is established to ensure appropriate detachment between the evaporator and the condenser of a vacuum AHP (P is lower in the evaporator than in the condenser). A wall-mounted AHP is offered, along with a heat generator (burner for liquid or gaseous fuel) (prior art systems are floor-standing units). The discovery seeks to propose divided hydraulic circuits, such as an added heat exchanger between the AHEx and the customer heating circuits, which the AHEx circuit can circumvent. In an AHP, a burner gets hot in an HTF for adsorbent regeneration. Even during the desorption phase (
Furthermore, the patents from 2007 to 2010184–192 have specific properties, such as, an AHEx that allows effective desorption and adsorption by expanding the surface area of the AHEx and supporting channels for refrigerant flow. The AHEx and customer heating circuits are disconnected in the desorption stage and connected in the adsorption stage in a vacuum AHP. There is no extreme pressure in the AHEx circuit during the desorption stage, hence ensuring safe operation. Appropriate disintegration of a vacuum AHP’s evaporator and condenser (“ball closure element”) occurs. Pumping the refrigerant to the evaporator (helical tube), which leads to wettability or spraying the evaporator with refrigerant, improves the water evaporation in a vacuum AHP. A displacement body is employed between the AHEx and evaporator tanks. To prevent earlier art disadvantages, adsorbent coatings are formulated with 80%–85% adsorbent, inorganic binders, and fibers (degradation of organic substances, P increase in the AHP, incompatibility of binders with adsorbents). Metal coating methods include spraying, painting, and immersion application accompanied by a drying phase. The discovery helps improve hydraulic interconnectedness and function during the adsorption stage of a vacuum AHP compared to the previous research. A separating body ensures optimal isolation between the AHEx and evaporator chambers during the desorption stage. AHP system with a modular configuration that is easy to construct and fabricate and is well suited for large-scale production. A better vacuum sorption device with an AHEx enveloped by a condenser, specifics on the condenser characteristics, which enable good pressure stability and high heat transfer with a simplistic manufacturing process, are provided.
Significant results from the patent literature are outlined as (i) the reduction in design, manufacturing, installation efforts, and AHP component costs appear to be a significant problem (e.g. modular system designs are advantageous); (ii) When a low
Comparison of AHPs and traditional heating technologies
The relation of adsorption heat pumps to mechanical and absorption heat pumps seek to analyze where adsorption heat pumps are now and how they will evolve. For adsorption-based heat pumps, the COP values for zeolite-water, silica gel-water, and carbon-methanol pairs are found to be 0.8, 0.3–0.6, and 0.43 respectively. Similarly, for vapor compression and absorption-based heat pumps, the COP was found to be 3–4 and 0.7–1.1, respectively. 4 It can be observed that the vapor compression heat pumps have significantly greater COP values than thermally driven pumps. Of course, this is the primary benefit of mechanically driven heat pumps; nevertheless, thermally powered heat pumps have several additional benefits that compel the industry to use them.
Thermally powered heat pumps use waste heat or other types of thermal energy. An adsorption heat pump utilizing waste heat as a working source not only recovers waste energy but also provides a system with no operating costs. Thermal-driven heat pumps can be assisted with renewable energy sources, such as solar and geothermal energy. It must be noted that vapor compression heat pumps use electrical power, which is often generated by the heat emitted by fossil fuels. As a result of energy losses in power plants and during electrical power transfer, the mechanical heat pumps have a low primary energy efficiency than their COP values. 194 The real benefit of adsorption heat pumps in terms of practical use is that they do not require regular inspection for extended periods because they have no moving parts or, in advanced technologies, only several essential moving components. Moreover, the lifespan of an absorption heat pump is less than that of the adsorption heat pump due to salt corrosion.
The absorbent in the absorption system must be replaced every 4–5 years. Whereas adsorption heat pumps do not require adsorbent-adsorbate pairs to be changed for an incredibly long time. Adsorption heat pump systems do not use any corrosive chemical ingredients. As previously stated, vapor compression heat pumps emit hazardous greenhouse gases. Several refrigerants have been strictly prohibited, and others will be banned in the near future. Such mechanical heat pump issues boost the desirability of adsorption heat pump systems, making them competitive even with absorption heat pumps, another type of thermally powered heat pump. 194 Vaillant and Viessmann devised a modest capacity of up to 15 kW for gas-fired hybrid AHPs, including a gas condensing boiler to alternately operate a zeolite/water AHEx and satisfy maximum load demands that needed heat requirement surpasses the AHP module’s heating capacity. However, such devices have a COP almost equal to 1.35 for the water delivered at 308–313 K and cannot generate water beyond 328 K if employed for DHW generation and space heating. 7
Owing to the injection of environmental energy into the system (21% renewable energy share), gas-fired AHPs provide a reduction in primary energy consumption from 86 × 107 to 53 × 107 J/m2 (around 38%) as well as a 39% reduction in CO2 emissions when compared to non-condensing boilers (the predominant house heating system in Germany). Compared to gas-fired AHPs to vapor compression heat pumps (electricity-driven), the innovation has resulted in larger, more expensive products. The Viessmann AHP device has proportions of 0.600 ×0.595 ×1.875 m 3 , which comprises the AHP module and gas condensing boiler, although the DHW tank was still placed outdoors, the device cost 13,000 Euros in September 2017 (without VAT 7 ). Contrary to this, the IVT GEO 312C, a ground source electricity-driven heat pump with a similar power range and meant for DHW generation and space heating, has comparable dimensions of 0.600 ×0.660 ×1.800 m 3 but incorporates the heat pump module as well as a 190 l DHW storage tank within the device, and valued around 7500 € (excluding VAT) 195 in September 2017. Electrically powered heat pumps that are pretty efficient for providing DHW and space heating in houses have PER values of 1.40 and 1.60 (air-source and ground-coupled equipment, correspondingly), whereas thermal powered heat pumps have PER values ranging from 1.20 to 1.60. 11
Generally, gas-fired AHPs are a promising technology for lowering CO2 emissions and primary energy utilization in the heating industry. 7 Whereas primary energy ratios of thermally powered appliances have reached that of vapor compression technology, efforts must be made to produce lighter and more affordable systems to improve their share in the market. AHPs are highly costly and bulky for competing with conventional systems and have a low market presence than electrically powered heat pumps. 33 While sanitary hot water heat pumps have become the fastest-growing heat pump category in Europe, with exponential demand. There have been several reports of prototypes aimed toward this use. A hybrid system that included a water reservoir, a gas heater, and an AHP with two AQSOA® FAM-Z02/water adsorbent beds was investigated by Dong et al. 196 The gas boiler provides hot water for bed renewal (at 358 K) and supports extra heating of the water exiting the AHP (up to 343 K). A two and four-bed silica gel/water adsorbent AHP paired with a gas water heater was reported by Tajima et al. 116 and determined that two beds are more feasible. Ally and Sharma 197 presented a model and analysis of a home gas-fired adsorption heat pump water heater employing AC/ammonia, the results of which were below the aim, even though a significant competitive premium for the market launch was indicated.
AHPs development challenges and limitations
For society’s sustainable development, policies have been driven to address resource scarcity and overcome climate change. Adsorption heat pumps (AHPs) are growing in popularity in the building heating industry due to their environmental benefits over traditional techniques such as vapor compression systems. Over the years, various adsorbents for AHPs have been studied, ranging from traditional activated carbon and zeolites to MOFs, SAPOs, and CSPMs utilizing water, ammonia, and methanol as adsorbates. Despite this, quite a few other adsorbents are used primarily due to their generally restricted loading capacities, complex bed configurations, reliability problems, expense, and operation within a relatively constrained set of conditions, all of which are limiting the technology’s development. Environmental implications of heat pumps, low consumer interest, and inadequate knowledge of the costs can also affect the input rate of adoption of adsorption heat pumps. Inadequately educated specialists and a lack of expertise in the technology of heat pumps are also obstacles that must be addressed. Uncertainty in policies directed at decarbonizing the heating industry, legislative measures in some of these jurisdictions, and the lack of installation requirements, are among the factors limiting heat pump implementation. There also exists reasonable concern that heat pumps may pollute the atmosphere. There is an acknowledgment that heat pumps emit fewer GHGs than other fossil fuel heating methods, although this is dependent on geology, local climatic conditions, and nearby land use, which requires further investigation. Although heat pump implementation poses many challenges, the general result is that heat pumps can play a significant role in transforming low-carbon heating. The key shortcomings of an AHP system include: (i) reduced coefficient of performance (COP); (ii) challenging to supply heat at temperatures above 55°C; (iii) the materials employed in the systems such as welding, joints, and valves are supposed to be suitable and compatible under various operating, that is, vacuum; (iv) to achieve the necessary healing powers for most applications, a great demand for adsorbent materials is required to be fulfilled by a sufficient supply; (v) high regeneration temperatures are required for adsorbent-adsorbate working pairs capable of providing higher heating powers and a higher coefficient of performance; and (vi) several precautions must be taken by using regeneration temperatures higher than the boiling point of the heat transfer fluid due to its potential for high pressure within the pipes.
Future recommendations
Metal-organic frameworks have a great potential to increase the efficiency and extend the application of AHPs due to their beneficial chemical diversity and configurable adsorption characteristics, which must be investigated further to ensure cost competitiveness. In addition to the requisite research and development in the materials field and scale-up techniques, the investigation should be directed toward the advancement of control strategies and optimized components, such as (i) the smart and compact AHExs are developed by the use of coated extended surface heat exchangers having fiber plate and finned plate geometries which provides thermal power and high efficiency. The cost-effective and straightforward adsorbent configurations incorporated in production models, such as binder-based coatings, are preferred; (ii) advanced management strategies allow for adoption to variations in consumer needs and operational conditions while maintaining the optimum length of the cycle stages. AHP output objectives can include COP > 1.55and SHP > 1000 W/kg (adsorbent coatings are required for that), adsorption loading shift of at least 0.20 kg/kg, and cycle time less than 900 s. In 2009–2013, some domestic heating market competitors and giants built and launched low-capacity gas-fired AHPs with water as a refrigerant for DHW output and space heating, consisting of a zeolitic module and a gas condensing boiler. Such appliances need to be researched and developed further, as they are an innovative technology for reducing CO2 emissions and primary energy consumption in the heating industry. A review of a wide variety of patents revealed that designing specific sophisticated structures would most likely necessitate a 10-year R&D pathway. Thus far, AHPs built specifically for DHW development seem to be lacking in the industry. The commercialization of such AHPs can be supported by the invention of adsorbents that perform well at mild regeneration and high condensation temperatures, have a comprehensive market supply and are inexpensive. Combating such technical and economic constraints can enhance the attractiveness of AHPs involving electricity-driven heat pumps and even gas water heaters in the medium to long term as (a) costs of natural gas and electricity for domestic consumers appear to be rising; (b) sanitary hot water heat pumps have become the fastest-growing heat pump sector in Europe, with dual-digit growth; and (c) People are taking more responsibility and enlightening awareness of the ecological concerns faced by society.
Conclusions
The analysis and review of AHP systems have been presented with a focus on heating applications. The essential research in the field has been discussed, and the significant discrepancies in the literature have been identified and addressed. The fundamental subjects for analyzing an AHP were explored, including its working principle, physical properties, physical models, adsorbent material thermodynamics, kinetics, and adsorption equilibrium of traditional and new adsorbent-adsorbate working pairs, operating conditions, and adsorbent bed designing. Overall, the discussion served as a resource for studies in the field investigations and a reference for the new AHP research domain, outlining the challenges and work that remains to be studied for the development of a physical prototype and an integrated approach for an AHP framework. Notwithstanding all the research on new adsorbent-adsorbate working pairs, a very few of them are easily accessed, market-ready, hydrothermally stable, have ease of production, and have a long-life cycle. There is a need for the establishment of the relation among system-level and material-level research. Some common models include the LDF model and Darcy’s Law to account for the internal and external mass transfer resistances. However, there is no physical model in the literary works that consider the whole AHP system. Many numerical simulations and laboratory experiments do not provide sufficient information to evaluate new models. Furthermore, there is no evidence of simultaneous optimization of specifications, physical properties, adsorbent material thermodynamics, and working conditions in the literature. Therefore, a physical model development that characterizes the whole AHP device would be needed to fill the gap. Furthermore, adsorbent-adsorbate working pairs with low mass and heat transfer resistances that can be regenerated at lower temperatures will be suitable for most applications. Finally, new operating modes that can increase the overall efficiency of the AHP system are beneficial to attaining economical solutions for AHPs.
Footnotes
Appendix
Handling Editor: Chenhui Liang
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was carried out in the Department of Agricultural Engineering, Bahauddin Zakariya University, Multan-Pakistan. The authors acknowledge the support from Bahauddin Zakariya University under the research grant of ORIC Project# 2020-21 awarded to Principal Investigator Dr. Muhammad Sultan.