Abstract
Thermally driven adsorption-based atmospheric water harvesting (AWH) is becoming an emerging technology to provide potable water. In this regard, various adsorbent materials including solid (MOFs, silica-gels, and zeolites), liquid (CaCl2 and LiCl), and composite adsorbents are explored in the literature. This study reviews recent advancements in adsorbent materials based on their water production capacity at different conditions that is air temperature and relative humidity (RH). The MOF of type MIL-101(Cr) shows water production capacity of 3.10 L/m2/day at RH ranging from 10% to 40%. Similarly, Zr-MOF-808 possesses water production capacity of 8.60 L/m2/day at RH more than 50%. Among the studied silica-gels, mesoporous silica-gel shows highest water production capacity that is 1.30 L/m2/day at RH ranging from 10% to 40%. The zeolite yielded water production capacity of 0.94 L/m2/day at RH ranging from 10% to 40%. On the other hand, liquid adsorbents like CaCl2 + cloth, K-LiCl showed water production capacity of 3.02 L/m2/day, and 2.9 g/g/day, respectively at RH of about 70%. Composite adsorbent modified with binary salts and functionalized carbon nanotubes resulted water production capacity of 5.60 g/g at RH of about 35%. The study will be useful to identify the energy-efficient adsorbent for the development of sustainable AWH device.
Keywords
Introduction
Freshwater scarcity is an immense problem nowadays. Every developed or developing country is facing the dilemma of freshwater unavailability. About 2.2 billion people do not have access to water while 4.2 billion people do not have a clean water source and sanitation facilities. 1 The problem has caused the deaths of about 3.4 lac children under the age of 5 due to diseases such as diarrhea, typhoid, and cholera. 2 Figure 1 shows the water supply of the world in the year 2040. The water flow across far distinct lands becomes the cause of floods and drought leading to the deficiency of fresh water in much-needed areas. The disastrous nature of human schemes and lack of humidity in the atmosphere is the major source of the water crisis. 3 Soon, half of Earth’s population will have to face the unavailability of freshwater 4 and these water stress conditions will cause wars and disputes around the globe .

Water supply prediction in the year 2040 taken from Aqueduct Water Risk Atlas. 5
According to the United States Bureau of Reclamation, Earth has about 3% freshwater, out of which 2.5% is trapped in glaciers, ice caps, atmosphere, soil, and contaminated or deep below the Earth’s surface that cannot be accessed easily and the remaining 0.5% is the freshwater available. 6 Many attempts and experiments have been made to reserve the essential natural resource and make it readily available. Scientists have developed plants for liquid water harvest, such as water desalination plants,7,8 groundwater harvesting plants, and rainwater harvesting plants. Water desalination plant provides freshwater on a large scale, however, the equipment used and the energy consumption is quite costly. Moreover, the residuary of the processed freshwater is wastewater with a high concentration of salts which is not healthy for sea life. 9
About 13,000 km3 of freshwater is in the atmosphere, out of which 98% is in the vapor phase, and 2% is in the condensed phase. This proportion is close to all the freshwater present on the surface or underground; excluding glaciers and ice caps. 10 The harvesting of water vapors from the atmosphere is complex thermodynamically, as the water vapors are meant to liquefy into fluid involving a large amount of heat release. 10 In this regard, researchers introduced the atmospheric water harvesting techniques. Some of those techniques include fog harvesting,11–13 dew water harvesting,10,14 bioinspired water harvesting,15–18 vapor compression cycle,19,20 heating, ventilation, and air-conditioning,21–23 evaporator coils, 24 hybrid air-conditioning/humidification-dehumidification system,25–27 thermoelectric cooling,28–32 radiative cooling condenser,33,34 selective membrane,35,36 and adsorption based water harvesting.37–42 Among all these technologies, adsorption-based atmospheric water harvesting (AWH) technology has gained attention in recent years because of its ability to extract the enough amount of freshwater using minimum amount of energy. Furthermore, this technology is possible in in dry and desert regions having lowest relative humidity (RH) by utilizing renewable energy options like solar, wind, and low-grade biomass. In research, solar energy has been inspected the most to desorb the moisture from the adsorbent (i.e. desiccant).37,43 Figure 2 shows the schematic representation of working principle for the adsorption-based atmospheric water harvesting. Initially, the adsorbent is exposed to ambient conditions to adsorb the water vapors owing to its hydrophilicity. After adsorption, the system is covered and the adsorbent is heated, and due to increase in temperature hydrophilicity of the adsorbent decreases, thereby desorption of water vapors take place. The closed system’s partial pressure slightly increases due to water vapor desorption. Finally, water vapors develop in condenser as a result of heat exchange with a clod source at a temperature below the system’s dewpoint. Once the water vapors have grown large enough, gravity pulls them out of the condenser and directed toward collection tank.

Schematic representation of working principle for the adsorption-based atmospheric water harvesting technology reprinted form. 44
These technologies are used efficiently in places where the climate is rather hot, dry, and has low RH. 44 Scientists have introduced countless desiccant-based technologies to fulfill the needs of freshwater in such areas. Some of these technologies that are worth mentioning are: desiccant wheels,42,45,46 adsorption chillers, 47 adsorption pipes,39,48 glass pyramids.49–52 Adsorbents have shown great potential in harvesting water from the air. They are cost-effective, easily available, and efficient to use. The use of hygroscopic salts, metal-organic frameworks, and compound desiccants has been discussed and focused of this review. The study also covers the progress and development of harvesting technologies to deliver the importance of air-water harvesting technologies.
Fundamentals of water vapor adsorption
Adsorption is the process of capturing gas or liquid molecules on the surface of solid. The molecules get attached to the surfaces of adsorbents rather than diffusing them. The detachment of these molecules from the adsorbent surface is called desorption. Figure 3 represents the water vapor adsorption mechanisms in adsorption-based atmospheric water harvesting. Adsorption is classified into two types: chemisorption and physisorption.43,53 Chemisorption involves strong forces like hydrogen bonding as binding sites for the adsorbates with an enthalpy change of 80–400 kJ/mol. Physisorption involves weak forces like van der Waal’s for the adsorbate to attach itself to the adsorbent and enthalpy of less than 20 kJ/mol. 53
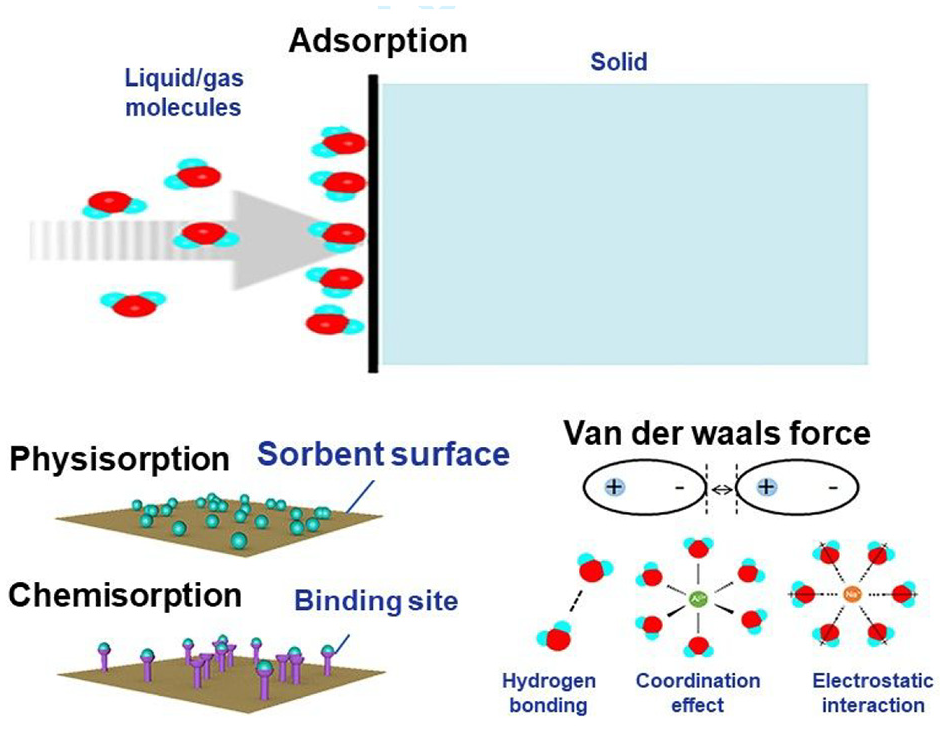
Water vapor adsorption mechanisms in adsorption-based atmospheric water harvesting, reproduced from Zhou et al. 54 Further permission related to the material excerpted should be directed to the ACS https://pubs.acs.org/doi/10.1021/acsmaterialslett.0c00130.
Depending on the change of enthalpy, many adsorption isotherms models have been introduced to present the processes involved in physisorption and chemisorption.55,56 Langmuir isotherm was introduced for adsorption on monolayer surface 57 while Freundlich isotherm was introduced as an empirical equation for adsorption on a heterogeneous surface 58 and Brunauer-Emmett-Teller (BET) model was introduced for adsorption on multilayer surface. 59 Simulation of the kinetic models helps in a better understanding of the processes involved in adsorption. The linear driving force (LDF) model is a common and simple mathematical model for gaseous state adsorbates.60,61 The pseudo-first-order and pseudo-second-order kinetic equations were used widely for better understanding of adsorption kinetics as well.62–64
The solid adsorbents used in the adsorption-based AWH technology include metal-organic frameworks (MOFs) silica gel, zeolite, and liquid adsorbents include calcium chloride (CaCl2), lithium chloride (LiCl), and composite adsorbents.65–67 These adsorbents are used in the AWH technologies based on the sorption systems discussed above. Higher adsorption rates can be achieved by altering the structure and operation of these systems. Some of these innovations regarding water production capacity will also be explored in this study.
Solid adsorbents
Metal-organic frameworks (MOFs)
Among various metal-organic frameworks (MOFs), the MOF-801 [Zr6O4(OH)4(fumarate)6] has been employed relatively more times. The AWH device was designed and demonstrated by using MOF-801 which was able to produce about 2.8 Lwater/kgMOF/day at a RH of about 20% without the need for any external energy source other than natural sunlight. 68 Another study was also conducted by Kim et al. 40 in Tempe, Arizona, USA having arid climate with RH of 10%–40%, thermal efficiency of about 14% in the day, and the sub-zero dew point at night. It was estimated that about 2.5 Lwater/kgMOF/day can be obtained by using MOF-801 with this air-cooled air-water harvesting technology. 40 Similarly, the fluidized form of the MOF-801 for the AWH was also studied by Terzis et al. 69 The experiment involved the adsorption-desorption cycles in two different RH of 18% and 39%, 26 and 36 min time intervals with moisture uptake capacity of MOF-801 to be about 80%. About 0.33 L/kgMOF/h water was collected with 40 cycles/day, at 22°C and 18% RH while about 0.52 L/kgMOF/h water was collected with 55 cycles/day, at 23.5°C and 39% RH. 69 Fathieh et al. 70 reported a prototype to extract water from desert air using MOF-801. About 1.2 kg of the MOF-801 can produce 100 gwater/kgMOF/day-and-night cycle using ambient cooling and solar energy. The prototype was also used to extract water from aluminum-based MOF-303 that gave about 175 gwater/kgMOF. This proved to be efficient where the dew point can even reach sub-zero. Likewise, a study was demonstrated by Hanikel et al. 71 that can provide a continuous supply of harvested water using MOF-303 under the outdoor conditions of the Mojave desert (10% RH and 27°C temperature). 71 The MOF-303 was able to surpass other commercially available sorbents as it gives a higher productivity rate at the desert conditions. The harvesting device can provide about 1.3 L/kgMOF/day under indoor conditions (32% RH and 27°C temperature) and about 0.7 L/kgMOF/day.
A mesoporous-MOF CoCl2BTDD was reported by Rieth et al. 72 that was able to capture moisture 82% by its weight at <30% RH. 72 The mesoporous MOF was able to harvest water of about 0.82 gwater/gMOF under desert climatic conditions. The mesoporous MOF also has the uptake limit of reversibility due to its pore diameter being greater than the critical diameter of sorbents. Yilmaz et al. 73 reported a polymer-MOF which was a hybrid of hydro-active polymers and MOFs which produced continuous and undisturbed sorption and desorption phenomenon. 73 The experiment was demonstrated for 1440 h straight and produced about 6 gwater/gMOF/day of water at RH of 90%. This provided a 95% efficiency of total liquid delivered and 71% efficiency of the autonomous liquid delivered.
Siliva et al. 74 studied the MOF MIL-125(Ti)_NH2 experimentally and results were evaluated with other sorbents. The results showed a water harvesting rate of 320 L/day/tons, which were about 0.35 gwater/gMOF/day when the temperature for regeneration was kept between 9.85°C and 99.85°C. 74 Trapani et al. 75 studied several MOFs and compared with other porous sorbents when the adsorption conditions were 40% RH and 25°C temperature while the desorption conditions were 10% RH and 60°C temperature. The study reported that the MOFs HSO3-UiO-66 and UiO-66 showed the highest water harvesting capacity of 0.061 gwater/gMOF and 0.054 gwater/gMOF . 75 In a similar study by Logan et al., 76 different hydrolytically stable and diverse structured MOFs were investigated. It was found that the Zr-MOF-808 provided about 8.66 gwater/gMOF/day. 76
Another MOF series, MIL-101(Cr) was explored along with other adsorbents (silica gel and zeolites) under different spatial and climatic factors. The results presented that the MOF produced maximum amount of water collection that is 3.1 L/m2/day. 77 Xu et al. 78 studied composite hygroscopic salt LiCl@MIL-101(Cr). The results showed water harvesting of 0.45–0.7 kgwater/kgsorbent. 78 By summarizing the studied literature Figure 4 compares water production capacity (L/kg/day) by various MOFs at RH of 10%–40% and Figure 5 compares the water production capacity (L/kg/day) by various MOFs at RH > 50%. Table 1 shows the operating conditions, water production capacity, structures, and harvesting devices employed MOFs found in the literature.


Summary of various MOFs adsorbents based on operating conditions, water production capacity, and structures with adsorption-based AWH devices.

Silica gel
The adsorption of water vapors using silica gel is common. Many harvesting devices have been developed to use this property of the adsorbent for the AWH. For instance, desiccant wheels are being used not just for cooling purposes but also to collect water from the air. Milani et al. 42 developed the synthetic model in TRNSYS and validated with experimental data of solar assisted desiccant dehumidification system. The model validated the experimental data of the system and produced 52 L of water in 1.3 weeks. This model was run for different climatic conditions (Sydney, Abu Dhabi, and London) to explore annual water production. The annual water production was maximum against the climatic conditions of Abu Dhabi as compared to Sydney and London. 42 Heidari et al. 45 investigated desiccant wheel based evaporative cooling system to harvest water from the air. The system was able to capture moisture from the exhaust air and provide about 590 L/week, while the evaporative cooling system used 296 L/week, thereby excess of 289 L/week was available for domestic usage. In addition, the system performance was explored using dynamic hourly simulation in TRNSYS by taking vapor compression based cooling and dehumidification system as a reference. The results showed that the electricity consumption of the proposed system by achieving maximum COP (i.e. 1.53) was lower than the reference system (1.2). 45 Similarly, Tu and Hwang 46 investigated the multi-stage desiccant wheels for the atmospheric water harvesting. The air was humidified through multi-stage desiccant wheels and then evaporate water vapors to condense for harvesting of water. The system performance showed that the water harvesting rate and water harvesting efficiency increased to about 32.5 kg/h and 1.26 kg kW/h. 46
Another experimental and predictive study 79 was performed to investigate the performance of a double-slope half-cylindrical basin solar still (DS-HCBSS) for extracting the water under Egyptian conditions. The DS-HCBSS consists of four longitudinal fins, which along with the basin liner of solar still were covered by silica gel having thickness of 1.5 cm. The DS-HCBSS with silica gel gives productivity and efficiency of about 76% and 15% when longitudinal fins were used and 166% and 35% when longitudinal fins with gravels were used. The overall productivity of silica gel was 400 mL/m2. The productivity of the system under ambient temperature and solar irradiance was predicted by a Neuro-fuzzy Inference System and validated good with experimental results. 79 In a study, 80 a solar concentrator having surface area of 1.54 m2 was used to desorb water from the already soaked silica gel. The water production rate was determined as 155 mL/day. Similarly, the study 81 found about 142 mL/day of water was collected from silica gel when it was left at a constant ambient temperature of 31.35°C. 81 The study 82 used silica gel which was kept in a solar glass desiccant box with sand as a host material. The theoretical study of the system gave a water production of were 600 mL/kgsilica gel/day and the experiment gave a water production of 160 mL/day which were significantly different. 82 In a similar study, 83 when orange silica gel was investigated with a solar recuperation system in Coimbatore, India it gave freshwater production of 98 mL/day at an 80% RH. 83
Sleiti et al. 84 designed, built, and tested an indoor water harvesting device using silica gel which was exposed to radiant flux ranging from 509 to 556 W/m2. The experiment was conducted under the conditions of RH 30%–60% and 25°C temperature. The device produced about 159 g/kgsilica gel in a cycle of 12 h. It was also concluded that the device could provide about 800 mL of water/ day with an efficiency of 50% and that increasing RH can increase adsorption-desorption rates. Mulchandani et al. 85 indicated that the use of solar energy for desorption is an efficient but time-consuming way of generating harvested water from adsorbent material. Therefore, photothermal nanomaterials like gold cubes and rods, and carbon black were applied to silica gel. This step increased the temperature of the adsorbent surface, thus improving the desorption rate and increase in the number of capture cycles of water vapor. The desiccants were tested at different RH during adsorption and under 1-sun simulated solar irradiation. The result showed that the desiccant coated with 5 wt% carbon black can provide about 0.47 gwater/gsilica gel/12 h at 40% RH through >10 adsorption-desorption cycles/day. In a study, 77 it was proved that the amount of water collection from desiccants differs throughout the year. During the fall and winter seasons, desiccants produced the least water such as zero to 5 L/m2 while during the spring to the summer season, desiccants produced the highest amount of water of 4–8 L/m2 in which silica gel contributed about 1.3 L/m2/day. 77 By summarizing the studied literature, a comparison of the water production capacity of various silica gels is shown in Figure 6. Table 2 shows the operating conditions, water production capacity, structures, and harvesting devices employed silica gel found in the literature

Summary of silica gels, activated alumina, molecular sieve, and zeolites based on operating conditions, water production capacity, and structures with adsorption-based AWH devices.

Activated alumina
Activated alumina contains a sequence of nonequilibrium forms of partially hydroxylated alumina oxide. Kumar and Yadav 82 investigated theoretically and experimentally solar powered glass box having desiccant for the atmospheric water harvesting. The theoretical results showed about 28 mL/kgactivated alumina/day, while the experimental results showed 20 mL/gactivated alumina/day of water. 82 Similar study 80 investigated the atmospheric water harvesting using Scheffler reflector using with 1 kg activated alumina. According to the results, water collection was about 38 mL/day of water. 80 Likewise, experimental study 81 to harvest water from the atmosphere with solar concentrator gave a water production rate of 100 mL/kgactivated alumina/day. 81 Figure 7 shows comparison of water production capacity by activated alumina used in literature for the atmospheric water harvesting. Table 2 shows summary of operating conditions, water production capacity, and harvesting devices employed activated alumina for the atmospheric water harvesting.
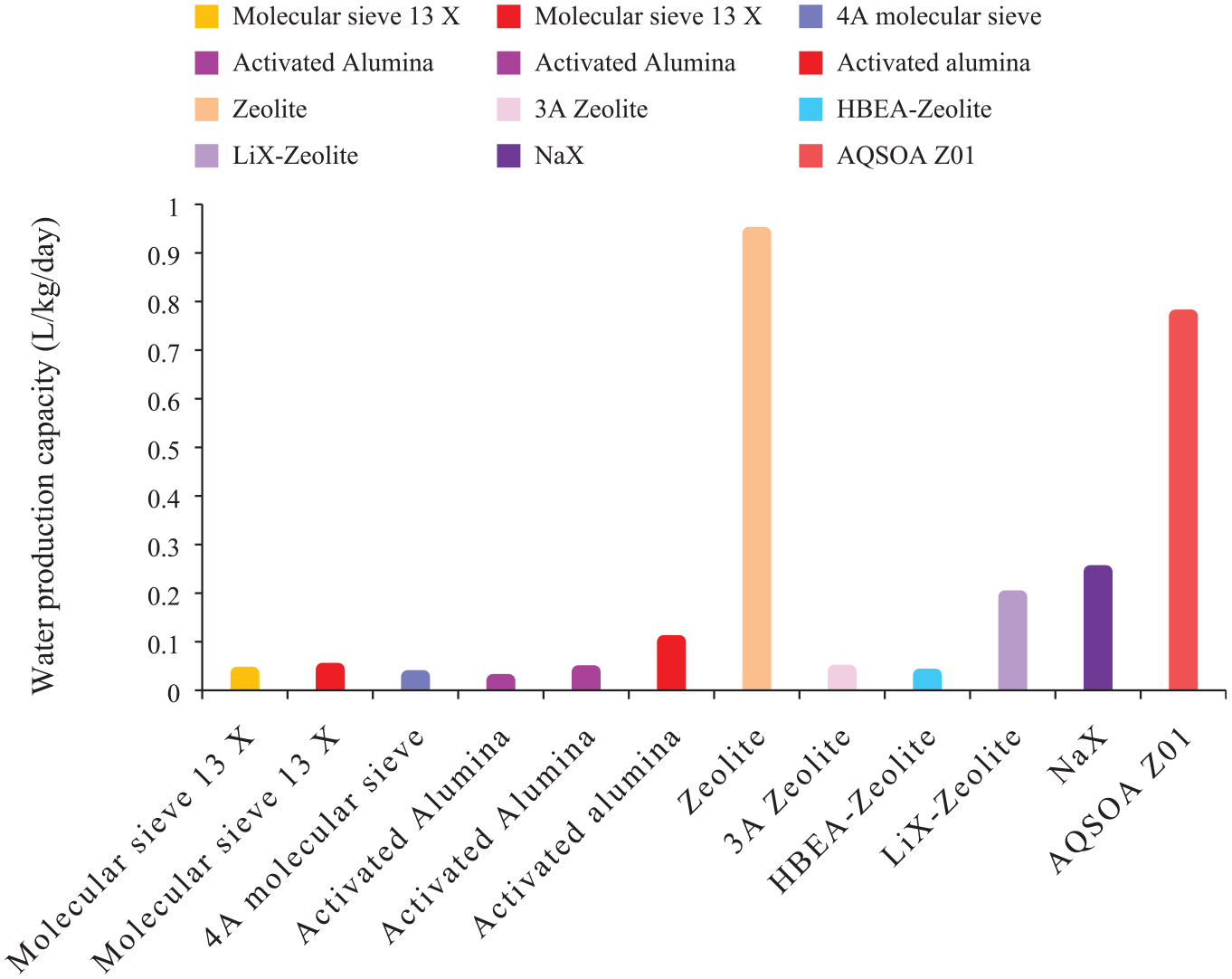
Molecular sieve
Different types of molecular sieve have been explored in literature for atmospheric water harvesting. Tarpani et al. 75 conducted experimental study using 4A molecular sieve for atmospheric water harvesting and compared with other adsorbents like activated alumina, activated clay under the same climatic conditions. The 4A molecular sieve gave a water production of 0.028 gwater/gmolecular sieve. 75 Srivastava and Yadav 80 investigated molecular sieve 13X with Scheffler reflector to explore water production from atmosphere by considering theoretical and experimental analysis. The theoretical results gave about 60 mL/kgmolecular sieve 13X/day of water and experimental results gave about 35 mL/kgmolecular sieve 13X/day of water. 80 A similar experimental study 82 with a solar concentrator for solar radiation 1 kg of molecular sieve 13X produced 43 mL/day of water. Figure 7 shows comparison of water production capacity by molecular sieve used in literature for the atmospheric water harvesting. Table 2 shows summary of operating conditions, water production capacity, and harvesting devices employed molecular sieve for the atmospheric water harvesting
Zeolite
In a study, 75 the porous ability of modified zeolite sorbents 3A zeolite and HBEA-zeolite (H form dealuminated zeolite) to harvest atmospheric water was determined under the desert climatic conditions of the Sahara. The HBEA zeolite produce water about 28 mg/gHBEA zeolite while the 3A zeolite gave modest production. Zeolite can provide a constant amount of water production. It was proved by the study 77 that zeolite can produced water about 0.94 L/m2/day. 77 In another study, 86 zeolite was modified with ion exchange to improve its sorption properties and was tested using different physicochemical methods. It was found that two of the ion-exchanged zeolite materials, LiX-Zeolite and NaX, gave a storage density of 576 and 864 kJ/L and a sorption capacity of 0.192 and 0.244 gwater/gzeolite, respectively. 86 A dual-stage AWH device was introduced that provides a high concentration of water by maintaining the temperature difference between the two stages as well as retrieving the latent heat of condensation in the top stage. 87 The study considered commercial zeolite AQSOA Z01 as the adsorbent material and sunlight was used for desorbing the water. About 0.77 L/m2/day of water was obtained and it was concluded that it can achieve high-performance efficiency with some modifications. 87 Figure 7 shows comparison of water production capacity by zeolites used in literature for the atmospheric water harvesting. It is clearly shown that the productivity rate of zeolite is higher than the other adsorbents mentioned in the Figure 7. Table 2 shows summary of operating conditions (i.e. atmospheric conditions), water production capacity, and harvesting devices employed zeolites for the atmospheric water harvesting.
Liquid adsorbents
Calcium chloride
An inclined solar collector device was investigated to extract water using calcium chloride (CaCl2) and sand bed. 88 The experiment was performed with device inclination angles of 25°, 20°, and 15° and the collector was able to collect 1.2 L/m2/day of water. The studies proved that the system efficiency increase on an inclination angle of 25°. Similarly, a glass pyramid solar collector system in different climatic conditions was also investigated. 49 Two sorbent beds were prepared one with CaCl2 soaked cloth and another with CaCl2 and saw wood. The beds were placed in two systems and left open throughout the night while during the day, they were closed and the adsorbed water was allowed to condense. The water collection rate was 2.5 L/m2/day at a 60% RH and 29°C. It was also found that the soaked cloth had a 5% more productivity rate than saw wood. In another study, a compact system of a non-conventional method for harvesting atmospheric water was introduced. 89 The main focus of the study was to determine the operation and efficiency of the system. The study used a packed tower with two columns of different concentrations of CaCl2 soaked cloth beds and the temperature was kept between 17°C and 50°C. It was concluded that the initial and final desiccant concentration, regeneration temperature, absorption airstream velocity, and cycle time affect the system efficiency. Gad et al. 90 used CaCl2 soaked in a corrugated cloth surface in a water harvesting device driven by solar energy. 90 A mathematical model was also prepared to compare with the experimental results of the device. The air was allowed to flow through the cloth at night and the water collected from the device during the day resulted in 1.5 L/m2/day of water.
The performance of a tubular solar still (TSS) was investigated for the first time in the low humid region having 12% RH of Hail city, Saudi Arabia. 91 The TSS contained a CaCl2 concentrated black cloth bed and a small fan to pass night air in the tube. The experiment was performed at the natural, 0.5, 1, 3, and 4 m/s air speed. The fans were removed and the vapors were allowed to condense using solar energy. The water collection rates of natural and 4 m/s air speed were 230 and 467 mL/m2 day and the cost per liters for both air speed was $ 0.4/L and $ 0.2/L, respectively. Similarly, the TSS with a solar concentrator for atmospheric water harvesting was also proposed. 92 At night, the TSS was opened to absorb the moisture from the air, and at sunrise, it got closed and placed in the parabolic concentrator for regeneration. The system produced 0.51 L/kgCaCl2.
A study for atmospheric water harvesting potential was presented in Hail city, Saudi Arabia. 93 The study utilized trapezoidal prism solar still and a black cotton cloth saturated with CaCl2 solution was filled inside its shelves. This prism was exposed to the atmosphere at night and moisture is absorbed by the desiccant cloth, then during the day, the prism is closed for the regeneration process and the condensed water is collected in a bottle. The total yield produced by this system is 1.06 L/m2 day with a solar energy consumption of 22.96 MJ/L. Another similar study was presented in which a fiberglass trapezoidal prism solar collector was used. 50 The system was operated for different weather conditions and different desiccant carriers that is cloth with CaCl2 and sand with CaCl2. The study let moisture be adsorbed during the night and got regenerated and condensed during the day. The system showed a water production and efficiency of 2320 gm/m2/day and 29.3% for cloth with CaCl2, and 1235 gm/m2/day and 17.76% for sand with CaCl2. In another study, the use of 30% concentrated CaCl2 fabricated on a sandy surface in a solar-based water harvesting unit was presented. 94 The experiment was performed in the city of Taif, Saudi Arabia where the climatic conditions were dry. The device was left in the atmosphere overnight and was covered with a glass layer on next day. The water gets condensed on the glass surface and about 1 L/m2 of water was collected. A foldable solar-powered system was introduced in Mansoura city of Egypt. 95 The system contained a black cloth soaked in a CaCl2 solution absorber and a polyvinyl chloride (PVC) cover for the sun rays to transmit energy. At night, the apparatus was unfolded and attached to a telescopic stick. During the day, it was mounted and isolated from the atmosphere. The temperature rise caused evaporation in the absorber and the evaporated water was condensed and collected giving a water production rate of 750 g/day. In another experiment, a solar concentrator was used to harvest water from the atmosphere using CaCl2 with sand as the liquid adsorbent. 96 The study reported a water collection of 115 mL/day from the experiment.
In a study, 97 the author experimented with a solar glass desiccant box-type system with floral foam cubes impregnated with CaCl2 of different concentrations. A total of six samples were taken with varying concentrations of CaCl2. The maximum production of water of about 0.35 mL/cm3/day from the sample of 37% concentrated CaCl2 fabricating floral foam, marking an efficiency of 76.44%. 97 In a similar experiment, CaCl2/saw wood was also used to harvest water in SGDBS. 52 Three different types of concentrations of CaCl2 were used in the experiment and compared. The 60% concentration CaCl2 composite sorbent produced about 500 mL/m2/day water. 52 Similarly, another newly synthesized CaCl2/Vermiculite/Saw wood composite sorbent was used in the same system. 98 The system collected water about 195 mL/kgsorbent/day. In another study, 99 composite desiccants with four matrixes and salt CaCl2 stated as SCCA (silica gel with 30% CaCl2 solution), ASCA (activated carbon fiber with 30% CaCl2 solution), ECA (natural graphite treated with sulfuric acid mixed with CaCl2 at 3:2), and ESCA (mixed with LiCl at 3:2) were developed. The purpose of the research was to increase the uptake capacity, decrease the desorption temperature, modify the optimal structure, and textural properties of the sorbent. The sorbents gave the water production of 0.51 gwater/gsorbent for ASCA, 0.18 gwater/gsorbent for SCCA, 0.14 gwater/gsorbent for ESCA, and 0.07 gwater/gsorbent for ECA at 20% RH and 77°C temperature. Similarly, another experiment was performed to evaluate the performance of CaCl2 with ACF felt and silica gel. The ACF felt was fabricated with viscose-based fibers. The characteristics of the host matrix, water crystallization, and quality of impregnation of CaCl2 were studied and tested using different techniques. Different characteristics of the sorbents ACF-CaCl2 and silica gel-CaCl2 were also studied and compared. 100 It was found that ACF with a 30% concentration of CaCl2 namely ACF30 gave the highest water uptake capacity of 1.7 gwater/gsorbent that was 3× more than the silica gel composite. Wang et al. 101 introduced the interfacial solar heating that was based on GO-aerogel, a salt-resistant. The aerogel allowed the liquid desiccants to be used in atmospheric water harvesters with ease and more efficiently. The experiment was performed using CaCl2 as 50 wt% solution adsorbent and it resulted in a collection of 2.89 kg/m2/day water at 70% RH with an efficiency of 66.9%. 101 Later on, two types of water harvesters were also designed; one open-type and the second semi-open-type. Both used about 2.25 and 40.8 kg of activated carbon fiber combined with CaCl2 and produced about 0.32 and 9 kg of water, respectively. 102
A solar collector for the AWH was presented in a study. 51 The operation conditions of the device like initial solution mass, initial concentration of desiccant, and host material, were studied to evaluate their effect on the characteristics of system performance like efficiency, productivity, and evaporation rate. The study investigated the system performance under the weather conditions of spring and summer in Egypt and recorded the amount of water collection as 3.02 L/m2/day. In another study, 103 a potable apparatus was proposed which uses CaCl2 as desiccant solution. In the experimental unit, solar heat energy was used as a heat source. At night, the water gets absorbed by the double-faced absorber which is covered by the desiccant material. In the daytime, the absorbed water gets evaporated when exposed to solar radiation. The vapor condenses and is collected in a bottle. The results showed water production of 0.3295–0.6310 kg/m2/day. 103
Different modifications of the CaCl2 proved to be efficient in adsorption, water production, and operation of the harvesting devices. Some of those modified and synthesized adsorbents are discussed herein. The use of ACF as a host matrix for CaCl2 was proposed in three-phase sorption. 104 The host material was fabricated with CaCl2 and LiCl in different concentrations and proportions. The composite sorbent ALiCa30 (at 3:1) was synthesized by impregnating ACF into a 30 wt% solution of LiCl and CaCl2 (three portion LiCl and one portion CaCl2). The desiccant gave a water uptake of 2.98 gwater/gsorbent at 90% RH and 25°C. Similarly, polyacrylamides (PAM) were fabricated with carbon nanotubes (CNT) to make hydrogel which was then freeze-dried and then impregnated with CaCl2 to make the required water sorbent. 105 The material was reuseable and could maintain its form even after absorbing moisture. When the experiment was performed, the prototype gave water about 20 g/35 g of PAM-CNT-CaCl2 in only 2.5 h. In another study, 106 2 mm size spherical beads of CaCl2 fabricated into an alginate-derived matrix were presented. The material was able to provide water of about 0.66 gwater/cm3 of the bulk material. 106 Ji et al. 107 synthesized a composite adsorbent using mesoporous crystalline material MCM-41 and impregnated it with CaCl2. The sorbent had a low desorption temperature of 80°C. and a sorption capacity of 1.75 kgwater/kgsorbent. 107 The above-mentioned devices, atmospheric conditions, and their water production capacity is shown in Table 3 and a comparison of water production capacity by these liquid adsorbents is shown in Figure 8.
Summary of liquid and composite adsorbents based on operating conditions, water production capacity, and structures with adsorption-based AWH devices.


Lithium chloride
Lithuim chloride (LiCl) has been used with a host material instead of directly using it for water adsorption. The use of MOFs, ACF, and sand as host materials has proved to be effective in moisture harvesting. Srivastava and Yadav 96 investigated water production when the sand was used as a host material, LiCl was able to produce about 0.09 kg/kgsorbent/day of water. 96 Similarly, when LiCl was impregnated into MOFs, it resulted in water production of about 0.45–0.7 kg/kgsorbent/day. 78 A nano vapor sorbent LiCl (HCS-LiCl) was introduced which was obtained by the immersion of HCS in the LiCl solution, vacuum filtration, and 2 h of oven drying the sorbent. 108 The sorbent could catch 100% of atmospheric water per its weight below an RH of 60% in just 3 h. Furthermore, it could allow the water to desorb in 30 min in the presence of 1 kW/m2 sunlight. An AWH device experimented with the nano sorbent provided about 1.6 kg of water/kg of sorbent in three adsorptions and desorptions processes. Similarly, in a study, the binary salt of LiCl, ACF, and MgSO4 was used in a prototype harvester. 109 The ACF and MgSO4 were injected mainly to keep LiCl stable and prevent its leakage from the sorbent. The sorbent showed an uptake capacity of 0.78 gwater/cm3 in terms of volume and 2.29 gwater/gsorbent in terms of mass. When the desiccant was tested in a prototype it produced about 0.92 gwater/gsorbent of water at 35% RH in an arid climate. 109
A theoretical framework and its scalable prototype were introduced and experiments were performed by synthesizes ACF-LiCl sorbent. The prototype produced about 38.5 kgwater/day of water and consuming about 7.2 MJheat/kgwater. It was also claimed that the prototype is a universal water-harvesting device. 110 In another study, 104 different concentrations of the ACF synthesized with LiCl and CaCl2 were prepared for three-phase solid-liquid sorption. The experiment showed that the 30% wt concentration of the sorbent proved to be the best with a sorption capacity of 2.98 gwater/gsorbent. 104 Similarly, use of the ACF felt modified by silica and soaked with LiCl was reported in study. 111 Among the sorbents of different compositions, the AS5Li30 having a composition of ACFF, 5 wt% silica gel, and 30 wt% of LiCl, had water capturing capacity of about 2.1 gwater/gsorbent at 70% RH. The result showed that the sorbent can desorb about 1.41 gwater/gsorbent at 75% RH and under 25°C temperature.
Wang et al. 99 developed composite desiccants with four matrixes silica gel, consolidated ACF, ENG-TSA (expanded natural graphite treated with sulfuric acid), and consolidated ENG-TSA with salt LiCl, given the names as SCLI, ASLI, ELI, and ESLI. The development of sorbents depends on increasing their sorption uptake, modifying the optimal structure and textural properties, and decreasing the temperature for desorption. The desiccant ASLI gave the highest water production of 0.65 gwater/gsorbent at 20% RH and 77°C temperature. The other sorbents being 0.17 gwater/gsorbent for SCLI, 0.11 gwater/gsorbent for ESLI, and 0.06 gwater/gsorbent for ELI. An experiment was also performed with different conditions applying to the adsorption and desorption process in a semi-open system. 112 Flat plate and corrugated plates of the composite sorbent ACF modified with LiCl were developed and used. The device allowed to have a high heat transfer performance and mass transfer channels for the sorbent. For the adsorption process of sorbent, RH of 65%, 75%, and 85% were adopted and the amount of water obtained was 12.5, 13.6, and 14.7 kg, respectively. For the desorption process of sorbent, the temperature conditions were 60°C, 77°C, and 90°C, and the water collected was 0, 13.6, and 14.5 kg, respectively. The device gave the best results of adsorption at 23°C and RH 90%, extracting about 0.65 gwater/gsorbent at the desorption temperature of 70°C–80°C. Figure 9 shows a comparison of water production capacity by various LiCl adsorbents and operating conditions, water production capacity, and harvesting devices which employed LiCl are shown in Table 3.

Lithium bromide
The study 47 utilized lithium bromide (LiBr) in a solar cooling system to extract water and investigated along with the effect of different parameters (air volume ratio and solar flux) of the adsorption unit, performance of the system and water production rate. When the air volume ratio was 50% and solar flux was 800 W/m2, the water collection rate was 8 L/h and when the air volume ratio was 45% and solar flux was 935 W/m2, the water collection rate was 6.6 L/h. 47 In another experiment, 96 the LiBr was used with sand to adsorb moisture at night and a solar concentrator was used to desorb the water from the adsorbent. The experiment gave water about 73 mL/day. 96 Table 3 shows the water production capacity, harvesting devices that use lithium bromide as an adsorbent.
Composite adsorbents
The ACF has a frequent use as a host material in research as it proved to be efficient in stabilizing the adsorbent material. In a study, 100 the composite sorbents of ACF-CaCl2 and silica gel-CaCl2 were compared. The characteristics of the host material and sorbents, impregnation quality, and water crystallization of the liquid sorbent were presented. The composite sorbent of ACF30 (ACF felt+30% concentrated CaCl2) gave the best results of 1.7 gwater/gsorbent. 100 Similarly, a device was presented in the study 112 with flat and corrugated plates of composite sorbent ACF-LiCl and proved that at different conditions of adsorption and desorption, the amount of water harvested was different. During adsorption, the RH was kept at 65%, 75%, and 85% and the water collected was 12.5, 13.6, and 14.7 kg, respectively. During desorption, the temperature was kept at 60°C, 77°C, and 90°C and the water collected was 0, 13.6, and 14.5 kg, respectively. 112 It was concluded that the sorbent gave the highest uptake capacity at 23°C and RH 90% and about 0.65 gwater/gsorbent was collected at 70°C–80°C. In another study, 102 an open-type and improved semi-open-type sorption device were designed and investigated, both of which were operated by a solar energy collector of temperature 70°C–80°C. The open-type device used 2.25 kg of ACF-CaCl2 in a roll-up shape sorbent bed with a solar collector of 0.77 m2 and provided water about 0.32 kg. The improved semi-open-type device used 40.8 kg of ACF-LiCl in a flat corrugated shape sorbent bed with a solar collector of 4 m2 and provided about 9 kg of harvested water. The freshwater extracted was depend on the area of the solar collector, amount of sorbent, and capacity of mass transfer, which makes the semi-open-type devices to be more efficient. 102
The study 110 developed ACF-LiCl based water harvesting prototype and investigated water production of about 35.5 kgwater/day. 110 In another experiment, 111 LiCl was infused into the ACFF (LiCl@ACFF) and then modified by silica. The composite samples with varying concentrations of silica were prepared and experimented with, for the determination of different characteristics of the samples. The sample AS5Li30, consisting of a percentage weight of 5% of silica gel and 30% of LiCl, provided about 2.1 gwater/gsorbent water uptake at 25°C and RH of 70% and an evaluated value of 1.41 gwater/gsorbent at 25°C and RH of 75%. It was concluded that the AS5Li30 has a heat-storing capacity of 5.456 MJ/kg cycle stability and a low regeneration temperature of below 80°C, which is beneficial in using solar-driven water harvesting devices. Liu et al. 104 prepared ALiCa30 (at 3:1) composite sorbent using ACF with LiCl and CaCl2. 104 The composite sorbent, at 25°C and 90% RH, gave an uptake capacity of 2.98 gwater/gsorbent. Later on, Ejeian et al. 109 pointed out that the composite of LiCl and ACF makes a stable adsorbent but most of the salt is served in avoiding the leakage. The author introduced a composite salt of LiCl, ACF, and MgSO4 with enhanced capacity of water uptake and avoiding leakage. The addition of MgSO4 produced water about 0.78 g/cm3 by volume and 2.29 g/g of sorbent by mass. The AWH device gave a low intensity at high RH but it provided water of 0.92 g/g of sorbent when the RH was 35% in an arid climate. 109 Similarly, three types of ACF composites were presented that were used as sorbent hosts for three different hygroscopic salts 113. The performance of these desiccant composites of K-LiCl, K-CaCl2, and K-LiCO3 was analyzed and investigated in three-phase sorption and it was concluded that it increases the uptake capacity of the desiccants. The composite material K-LiCl was used in a proposed prototype and gave about 2.9 gwater/gsorbent/cycle at 70% RH.
The use of composite adsorbent material CaCl2/saw wood in a solar glass desiccant box-type system was investigated. 52 The concentration of CaCl2 determined the rate of production of water and about 500 mL/m2/day water was produced from the 60% concentration of CaCl2 in saw wood. The same system was used to investigate the performance of composite desiccant CaCl2/Vermiculite/Saw wood and about 195 mL/kgsorbent/day of water was collected. 98 Similarly, the composite sorbent CaCl2/floral foam with a 37% concentration of the liquid desiccant was presented for experiment. 97 The result gave an efficiency of 76.44% by providing about 0.35 mL/cm3/day of water. In another study, the authors experimented to generate water using a composite desiccant using a solar concentrator. 96 In the experiment, sand was used as a host material with three different composite desiccant materials that is, LiBr/sand, LiCl/sand, and CaCl2/sand. The result showed that a water production of 73, 90, and 115 mL/day for LiBr/sand, LiCl/sand, and CaCl2/sand, respectively.
The composite sorbent LiCl@MIL101(Cr) was reported that could capture about 0.77 g/g of water from the arid climate at RH of 30%, at the vapor pressure of 1.2 kPa, and a temperature of 30°C. 78 In the laboratory and outdoor atmosphere, the AWH device was able to provide about 0.45–0.7 kgwater/kgsorbent. The hygroscopic salt, confined in the MOF matrix proved to be highly efficient after the integration of multi-step 3-phase sorption processes. In another study, 2 mm sized spheres of composite sorbent Alg-CaCl2 were presented. 106 These synthesized beads can produce water about 0.66 g/cm3 of the sorbent at about 28°C temperature. It was concluded that the composite material is non-toxic, cheap, and easily available. Later on, in a similar study, sodium alginate was modified with binary salts, Li and Ca, and then functionalized carbon nanotubes (FCNTs) were fabricated into them. 114 The sorbent could give about 5.6 gwater/gsorbent. In a research, a fabricated sorbent CaCl2@Fe-Fc-HCPs with good stability was presented that was able to prevent the dissolving of CaCl2 with the water adsorbed and increased the uptake capacity of CaCl2. 115 The experimental result showed a water production rate of 2.685 gwater/gsorbent at 80% RH and 15°C. Similarly, silica gel-LiCl was modified with polyvinylpyrrolidone (PVP) to create a sorbent with high water-holding and water harvesting capacity. 116 The prototype built for silica gel-LiCl-PVP provided about 0.43 gwater/gsorbent.
A composite sorbent synthesized from MCM-41 and fabricated with CaCl2 was presented in study. 107 The composite adsorbent has a high sorption capacity of 1.75 kgwater/kgsorbent and a low desorption temperature of 80°C. In a study, polyacrylamides (PAM), carbon nanotubes (CNT), and CaCl2 were fabricated to make an efficient water hydrogel. 105 The material was cheap, reusable, and affordable. The prototype device used 35 g of the PAM-CNT-CaCl2 hydrogel and gave water about 20 g in 2.5 h, making it possible to supply about 3 kg of water for just $3.20. Similarly, Chen et al. 117 put forward a graphene nanocomposite foam of superelasticity, low density, and a stable structure with the ability to be compressed and portability. The adsorbent design was based on using solar energy as a regeneration component. At a 30% RH, the foam can provide about 0.23 gwater/gfoam and a 1.15 gwater/gfoam harvesting capacity on 90% RH. 117 Wang et al. 118 investigated the use of birnessite in the AWH. The material could adsorb water vapors to interlayers of the material at a low RH and on multi-layer, it could give condensed water at a high RH. At a water uptake capacity of 0.1 gwater/gbirnessite at 20% RH, the material can provide about 0.42 gwater/gbirnessite at a 23% RH and 11°C dew point temperature. 118 Similarly, several new composite desiccants of silica gel, ACF, and hygroscopic salts were developed. 99 At 20% RH and 77°C, the maximum water production ranged from 0.06 gwater/gsorbent to 0.65 gwater/gsorbent. Figure 10 shows a comparison of the water production of composite sorbents and Table 3 presents the harvesting devices which employed composite adsorbents for the atmospheric water harvesting.

Comparison of water production capacity by composite adsorbents. CaCl2 + MCM-41, 107 CaCl2 + saw wood + Vermiculite, 98 ALiCa30, 104 PAM + CNT + CaCl2, 105 LiCl + MgSO4 + ACF, 109 AS5Li30, 111 ACF30, 100 SC30, 100 graphene nanocomposite foam, 117 birnessite, 118 silica gel-LiCl-PVP, 116 binary/FCNT, 114 and CaCl2@Fe-Fc-HCPs. 115
Conclusions
Adsorption-based atmospheric water harvesting (AWH) is an emerging technology for obtaining potable water to undertake global freshwater scarcity. The present study conducted an overview on solid, liquid, and composites materials that employed in the adsorption-based AWH technology for potable water production. The solid adsorbents include metal-organic frameworks (MOFs), silica gel, zeolites while liquid adsorbents include calcium chloride (CaCl2), and lithium chloride (LiCl). These materials are reviewed for maximum water production capacity and associated operating relative humidity (RH) conditions. Different series of MOFs (i.e. zirconium, MIL, and polymer) were discussed to produce maximum potable water form the atmosphere. The MIL-101(Cr) was observed with maximum water production capacity of 3.1 L/m2/day at RH between 10% and 40%, while Zr-MOF-808 was observed with maximum water production capacity of 8.6 L/m2/day at RH of >50%. Likewise, various types of silica gel (i.e. blue, orange, mesoporous, and silica gel+carbon black) were discussed but mesoporous silica gel was observed with maximum water production capacity of 1.3 L/m2/day at RH between 10% and 40% as compared to other types of silica gels. The maximum water production capacity by zeolite was 0.94 L/m2/day at RH between 10% and 40%. Different CaCl2 adsorbents which include CaCl2+saw wood, CaCl2+sand, CaCl2+cloth, CaCl2+GO-aerogels were discussed but the CaCl2 + cloth was observed with maximum water production capacity of 3.02 L/m2/day at RH of ∼70%. Among various types of LiCl such as SCLI, ASLI, ELI, ACF+LiCl, HCS-LiCl, and K-LiCl, the maximum water production capacity was 2.9 g/g/day at RH of ∼70%. Compared with various composites (CaCl2+MCM-41, PAM+CNT+ CaCl2, ACF30, birnessite, CaCl2@FE-Fc-HCPs, CaCl2+saw wood+vermiculite, LiCl+MgSO4+ACF, SC30, silica gel-LiCl-PVP, ALiCa30, AS5Li30, graphene nanocomposite foam, and binary/FCNT), the Binary/FCNT (modified with binary salts and functionalized carbon nanotubes) shows maximum water production capacity of 5.6 g/g at RH of ∼35%. Therefore, the adsorption-based AWH is emerging as a possible and promising option for mitigating the global freshwater scarcity.
Footnotes
Handling Editor: Chenhui Liang
Authors’ Note
Uzair Sajjad is now affiliated to the Department of Energy and Refrigerating Air-Conditioning Engineering, National Taipei University of Technology, Taipei, Taiwan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was carried out in the Department of Agricultural Engineering, Bahauddin Zakariya University, Multan, Pakistan with the financial support of the BZU Director Research/ORIC grants awarded to Principal Investigator Dr. Muhammad Sultan.
