Abstract
Purpose of the Research
Immune thrombotic thrombocytopenic purpura (TTP) is a rare, life-threatening condition with long-term impacts on survivors’ health-related quality of life (HRQoL). A top HRQoL concern among TTP survivors is cognitive dysfunction. However, the specific cognitive aspects affected by TTP remain poorly understood. This gap limits the ability to assess cognitive outcomes in clinical trials and hinders the development of targeted interventions. To address this gap, we designed THINK-TTP – a qualitative study designed to identify and characterize cognitive dysfunction experienced by patients with TTP through interviews with patients and patient-observer dyads.
Major Findings
This protocol describes a multi-site, descriptive, qualitative study of cognitive dysfunction using semi-structured interviews of 24 patient and close observer dyads (48 interviews total). This study is the first step in selection of appropriate clinical outcome assessments that are specific to cognitive dysfunction experienced after a TTP diagnosis. Additional data sources include standardized questionnaires, clinical data, and performance-based cognitive tasks.
Conclusions
A qualitative study protocol describing procedures for eliciting and documenting patient and observer perspectives on cognitive function facilitates rigor and transparency and enhances clinical trial readiness. After completion, this study will generate foundational evidence to support cognitive function measures tailored to the experiences of people with TTP in clinical trials.
Keywords
Background
Immune thrombotic thrombocytopenic purpura (TTP) is a rare blood disorder with an estimated prevalence of 10 per million (Nuñez Zuno & Khaddour, 2025). Antibody-mediated inactivation of ADAMTS13 (A Disintegrin And Metalloproteinase with ThromboSpondin type 1 motif, member 13) leads to build up of large von Willebrand multimers causing a microvascular hemolytic anemia. This can cause significant tissue damage including neurologic and renal injury, and without treatment, TTP carries a mortality rate of up to 90% (Amorosi & Ultmann, 1966; Chaturvedi et al., 2015; Cox, 1966; Deford et al., 2013; Ridolfi & Bell, 1981) TTP treatment advances have dramatically decreased mortality. Recent mortality estimates are as low as 7.4% at 6 months (Jacobs et al., 2025). As a result, attention has shifted from decreasing mortality towards improving health-related quality of life (HrQoL).
In focus groups, most TTP survivors (76%) identified their most significant HrQoL concern as the impact of TTP on their cognitive function (Joly et al., 2019; Lewis et al., 2009). TTP-associated cognitive dysfunction may be driven by vascular injury. Therefore, it may be modulated by current and emerging therapies such as caplacizumab, and recombinant ADAMTS13. However, the pre- and post-treatment impacts of these therapies on TTP-associated cognitive function have not and cannot be assessed as primary endpoints in clinical trials due to a lack of evidence of fit-for-purpose Clinical Outcomes Assessments (COAs) (Onwuemene et al., Forthcoming) for TTP.
One of the key components needed to establish a fit-for-purpose COA is a clearly defined meaningful aspect of health that is important to patients (FDA, 2025). Cognitive function is a broad construct encompassing multiple inter-related domains such as object recognition, motor skills, attention and concentration, memory, executive functioning, processing speed, and verbal skills (Harvey, 2019). Individuals with TTP have described cognitive function impacts as short-term memory loss, feeling “scattered”, and having disorganized thoughts (Kelley et al., 2023). Despite anecdotal characterizations of TTP-associated cognitive dysfunction, to our knowledge, no targeted studies have systematically and comprehensively documented the specific cognitive dysfunction experienced by people diagnosed with TTP. Lack of this specific information is a barrier to systematic, comprehensive measurement of this meaningful aspect of health.
Another component of evidence needed to support the use of a COA as a primary endpoint in clinical trials for assessing cognitive function is evidence of who is in the best position to report on the patients’ cognitive function. Options include the patient themselves, observers of the patients (e.g., parents, friends, siblings, children), or clinicians. Assessing cognitive function using only patient report may be biased because, depending on the cognitive dysfunction severity, individuals with TTP may not be fully aware of the cognitive function changes they experience (De Wit et al., 2024). Observers and clinicians can report on observed signs but may not be able to “see” all of the patients’ cognitive function challenges. Although clinicians may include clinical judgement in their report of the patients’ cognitive function, it is limited to what the patient tells them and what they can observe during short clinic visits. A fourth option is performance measures – standardized tests of specific aspects of cognitive function. In ensuring a fit-for-purpose COA for cognitive function in individuals with TTP, a rationale for which reporter (or reporters) would be most accurate in reporting on TTP is critical.
The absence of fit-for-purpose COAs presents a barrier to TTP clinical trial readiness. Therefore, the first step toward identifying or developing fit-for-purpose COAs measures is a comprehensive understanding of (1) the specific cognitive dysfunctions experienced by individuals with TTP and (2) a suitable reporter type (e.g., patient, observer, clinician, performance test). To describe TTP-associated cognitive dysfunctions and align them with suitable COA types, this study will use qualitative concept elicitation interviews with patient-observer dyads (Patrick et al., 2011a).
This protocol describes procedures for eliciting and documenting cognitive function experiences from the perspectives of patient and observers. Publishing this qualitative protocol facilitates rigor and transparency of the study and enhances clinical trial readiness. After completion, this study will generate foundational evidence to support fit-for-purpose measurement of cognitive function tailored to the experiences of people with TTP in clinical trials.
Theoretical Framework
We will describe the facets of cognitive function experienced by people with TTP using Harvey’s cognitive function framework (Harvey, 2019). This framework, which extensively describes the facets of cognitive function that can be impacted by any condition, will aid us in identifying only the impacted cognitive domains as described by people with TTP and their observers (Figure 1). Cognitive function framework (Harvey, 2019)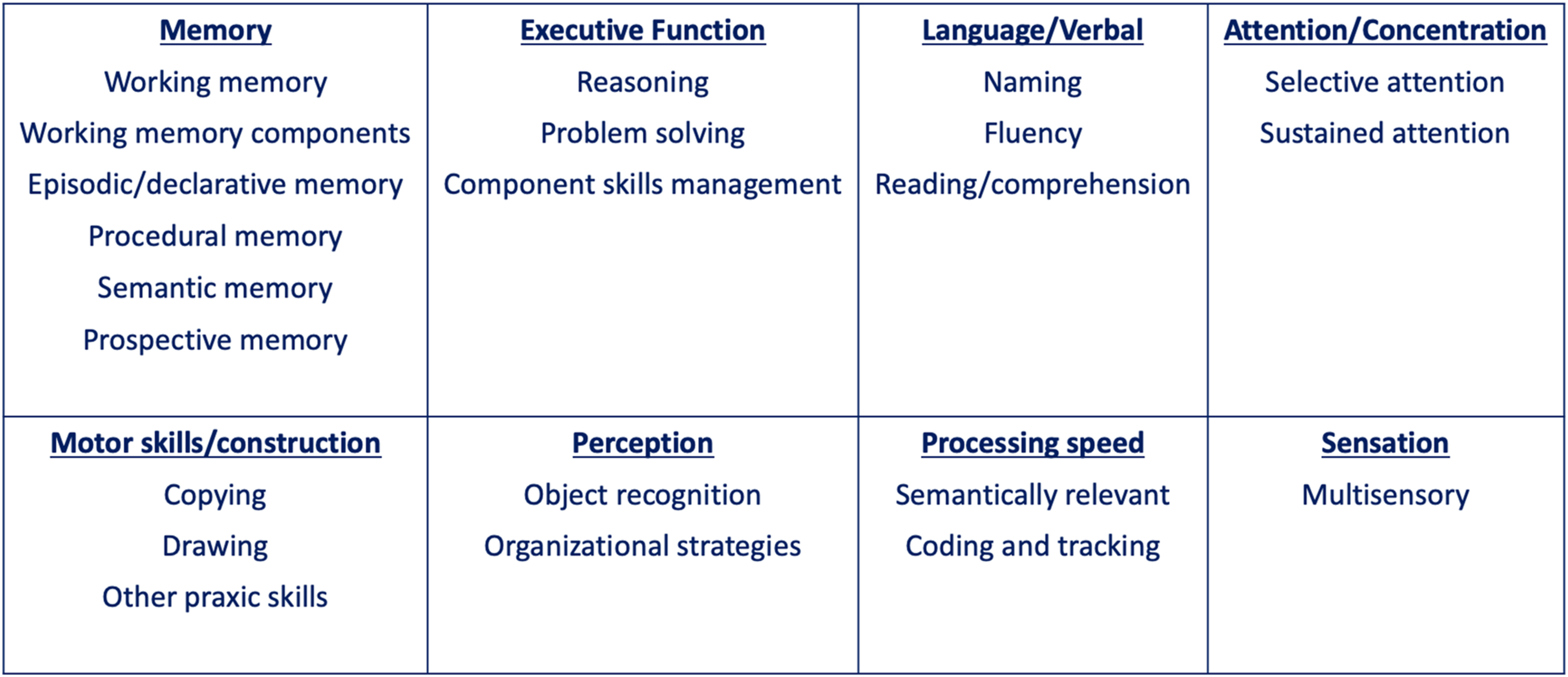
Study Aim
The aim of this study is to characterize cognitive dysfunctions experienced by patients with TTP by interviewing dyads of patients and their observers.
Methods
Study Design
The study is a descriptive, qualitative study designed to describe the experiences and perspectives of people diagnosed with TTP and observers. The study will be conducted within an interpretive/constructivist research paradigm (Kivunja & Kuyini, 2017; Patrick et al., 2011a; Patrick et al., 2011b). Study recruitment will use a multicenter approach at three performance sites affiliated with a clinical trials network – the Patient-Centered Outcomes Research Network (PCORnet®; Duke, Vanderbilt, and UT Southwestern) (Fleurence et al., 2014; Forrest et al., 2021). We will aim to conduct semi-structured, in-depth qualitative interviews with 24 patient-observer dyads (48 interviews total) or until saturation is achieved (Kerr et al., 2010). To frame the qualitative data in light of their disease course, the study will also collect background clinical data, as well as standardized patient- and observer-reported measures, and standardized discourse tasks.
Study Setting
To ensure geographic and institutional diversity, the study will recruit participants through three academic sites: Duke University (Durham, NC), Vanderbilt University Medical Center (Nashville, TN), University of Texas Southwestern Medical Center (Dallas, TX).
Sampling and Recruitment
Participants
Eligible patients will be aged ≥ 16 years and have a clinician-confirmed diagnosis of TTP. Patients will be excluded if they are diagnosed with hereditary TTP, do not speak English, or are unable to give informed consent. Eligible observers will be aged ≥ 18 years and have close contact (at least six hours a week) with a patient with TTP. Observers will be excluded if they do not speak English or are unable to give informed consent.
Recruitment
To ensure diverse recruitment, we will use purposive sampling (Patton, 2002). The primary sampling diversity objective is time from last acute TTP episode TTP operationalized as time since most-recent TTP treatment (less than 6 months vs ≥ 6 months). The team will also aim to track and seek diversity in other participant characteristics: age, gender, race, ethnicity, education. Patients who meet inclusion criteria will be nominated by site clinical team members. Patients will nominate their own observers.
To describe the study and determine level of patient/observer interest, Duke research staff will contact patients and observers from all three sites in person, or via phone, email, or electronic health portal message. For interested potential participants, study coordinators will complete a verbal eligibility screening. To aid in recruiting a balanced sample, they will also administer a demographic questionnaire during screening (Appendix 1). Eligible individuals who enable sampling diversity (e.g., TTP remission status, age, gender, race, ethnicity, education) will be consented and invited to schedule an interview. It is anticipated that cognitive function will be stable for patients within 4 weeks of their enrollment. For this reason, dyad interviews will be conducted within a 4-week window, with a goal to interview dyad participants within a shorter period of time: 1-2 weeks. This window will ensure that experiences and behaviors discussed in the dyad interviews are based on a similar timeframe. For patients, site investigators and research coordinators will enter historical clinical information into a secure REDCap electronic data tool (Harris et al., 2009, 2019).
Sample Size
As previously documented in other studies, little new information is gained after 12 interviews within a homogenous sample (Guest et al., 2006). As such, patients within six months of a TTP episode or treatment will be considered as one group, and patients more than six months from their last TTP episode will be considered another. It is anticipated that saturation will be met within 24 dyad interviews (24 patients and 24 observers). However, saturation will be monitored for cognitive function challenges using structured debriefing forms (see Data Collection); if saturation is achieved before we complete the total of 24 dyad interviews, we will stop interviews so as not to overburden potential participants.
Interview Content
Interview Guides
The study team engaged in Iterative and collaborative development of the patient and observer interview guides.
Design and Development of Interview Guides
First, team members with expertise in qualitative research methods drafted the interview guides based on literature and discussions with clinicians while ensuring that key research questions could be answered (LM, TC). For additional feedback and refinement, draft interview guides were sent to the larger study team which included hematologists, a clinical pathologist, speech-language pathologist, and other qualitative researchers. Then, the interview guides were piloted with 3 patients and 2 observers. Pilot interviews were recorded, and key impressions about the interview efficiency and content were shared with study team members after each pilot interview. The interview guides were adjusted between pilot interviews.
Content of Interviews
Interview Concept Coverage

Patient and observer interview guides are presented in Appendix 2 and 3, respectively.
Data Collection
Data collection will include the following 7 data types: (1) qualitative recordings and transcripts; (2) debriefing forms; (3) saturation matrix; (4) background demographics/screening questionnaire; (5) background clinical data; (6) standardized questionnaires capturing self-reported and observer-reported cognitive change, the Cognitive Change Index; (7) standardized performance measures to assess cognitive function. The primary data for this study are the qualitative transcripts from patient and observer interviews. The debriefing forms and saturation matrix will be derived from qualitative interviews. These documents will be used to track thematic saturation. Data types 4-7 are intended to describe the study sample.
Qualitative Interview Recordings and Transcripts
Interviews will be recorded using Zoom or, if conducted over the phone, an encrypted recorder. Participants will consent to both video and audio interview recordings. Audio recordings will be orthographically transcribed by a professional. Audio and video recordings will be saved in a secure, encrypted, online folder. Transcripts will be de-identified and redacted. Participants will be provided a participant number; their names will not be associated with their data.
Debriefing Forms
Debriefing forms are structured interview notes used to capture participants’ responses, interviewer reflexivity, interviewer interpretation, and capture non-verbal responses. Interviewers will complete debriefing forms after each interview. Debriefing forms will be used to populate the saturation matrix one-by-one during data collection to keep track of high-level themes and saturation.
Saturation Matrix
Interviewers will also populate a saturation matrix. An Excel saturation matrix will be used to record types of cognitive dysfunctions endorsed by patients and their observers and track thematic saturation of these concepts. Additionally, the matrix will be used to initially compare and contrast the cognitive function difficulties expressed by patients vs observers. Key concepts from the matrix and the topics from the interview guides will be used to develop the initial codebook.
Demographic and Screening Questionnaire
The demographic and screening questionnaire (Appendix 1) is self-reported prior to enrollment to ensure a balanced sample.
Background Clinical Data
To describe the clinical status of patient participants, clinical data will be extracted from medical records. Data to be collected includes history of stroke, current medications, and recent lab values: hemoglobin and hematocrit, platelet counts, white blood cell counts, and ADAMSTS13 activity. Summary reports for brain imaging (CT or MRI), vitamin B12, and ferritin lab values will also be collected if patient participants have this information recorded in their electronic medical records.
Cognitive Change Index (CCI)
A modified version of the Cognitive Change Index (CCI) will be administered to patient and observer participants to describe overall impressions of cognitive change since patients’ TTP diagnoses (Rattanabannakit et al., 2016). The CCI includes 20 items assessing memory, executive function, and language. Scores range from 20 to 100, with higher scores indicating more cognitive changes. For the purposes of this study, the recall period of 5 years ago was changed to “since your TTP diagnosis” because this time period was found to be more salient to pilot interview participants. Participants will complete the CCI electronically via REDCap.
Discourse Performance Measures
At the end of the interview, patients will engage in standardized tasks designed to assess their cognitive function through their speech and language. The discourse performance measures include four tasks of the RHDBank Discourse Protocol (Appendix 2) (Minga et al., 2021): 1) a free speech sample modified for the diagnosis of TTP, 2) a descriptive discourse task [Cookie Theft (Goodglass et al., 2001)] 3) a narrative discourse task [Cat in the Tree (Nicholas & Brookshire, 1993)], and 4) the question production task [Unfamiliar Object Task (Minga et al., 2020)]. Each task will be administered using the RHDBank Discourse Protocol script and stimulus items.
Interview Conduct
Interviews will be conducted by Duke researchers with qualitative research experience. Interviews will be videorecorded and conducted via Zoom or phone, depending on the participants’ preference. Interviews are designed to take approximately 60 minutes to complete. Pilot interviews confirmed that interviews will take 60 minutes or fewer. All interviews will be conducted in English.
Analysis
Concept Elicitation Analysis
Final transcripts will be uploaded to NVivo 14 qualitative data analysis software for analyses (Lumivero, 2026). For the concept elicitation portion of the interview (not the discourse tasks), the flexible coding approach will be used. The team will review debriefing forms and transcripts to generate overarching concepts and themes present across interviews (Deterding & Waters, 2021). After transcripts have been developed, the team will categorize each transcript with participant attributes (e.g., demographic and clinical characteristics) for comparison and description. A code book for index codes will be drafted by the qualitative team and shared with the larger study team for their input. It will include index codes, ‘good quote’ codes, and analytic codes. Index (or structural codes) will be added to organize large pieces of text by general topics. This allows for subsequent rounds of transcript reading to be focused on each topic. For example, there will be a section of the interview that asks participants to describe a “good day” with their cognitive function. This entire section, including back-and-forth between participants and interviews will be coded as an index code for further comparison and analyses. Throughout index coding, analysts will keep track of potential themes that will then lay the groundwork for analytic codes. After index codes have been applied and output has been examined, analytic codes will be drafted by the team and refined after sharing with the larger team. Then, analysts will apply analytic codes to the transcripts. During analytic code application, exemplar quotes will be coded as “good quotes” to be considered as supporting documentation in results write-ups. After all codes have been applied, summary reports will be written to describe overarching themes by index code. The summary reports will also compare and contrast results by participant attributes.
Discourse Analysis
A TTP Corpus (https://talkbank.org/rhd/access/English/NonProtocol/TTP.html) has been developed in the RHDBank to facilitate discourse analytics and open-source data from this study. Each orthographically transcribed language sample will be coded using CLAN (MacWhinney, 2000; Minga et al., 2020, 2021, 2022). General discourse features (sample duration, words per sample, words per utterance, number of utterances, clauses per utterance) and discourse productivity score (utterance complexity and informativeness) will be calculated for each transcribed task. An analysis of questions is reserved for the unfamiliar object task. Utterances coded as questions will be further coded as polar (positive or negative -- e.g. Are you from NC? Aren’t you from NC?); content (may contain a question word where, what, who etc. -- e.g. Where are you from?), or alternative (A or B --e.g. Are you from NC or SC?). An analysis of narrative conceptualization will be completed using main concept analysis for the cat in the tree and cookie theft tasks (Marini, 2012; Richardson & Dalton, 2020). We will use the established main concepts to calculate a narrative conceptualization score. Discourse performance will be compared to other neurogenically compromised populations and controls in the RHDBank.
Ethics
The THINK-TTP protocol was reviewed and approved by the Duke University Health System Institutional Review Board (DUHS IRB) in 2024 (Pro00115540). UTSW and VUMC IRBs ceded oversight to the DUHS IRB, and DUHS served as the IRB of record for the study. Before beginning any interviews, all participants provided verbal or written informed consent.
Participant level documentation will be stored in REDCap and secure shared folders (Harris et al., 2009, 2019). Study team members from Duke will have access to all identified data; however, site investigators will only be able to view clinical data of participants from their respective site. All participants will be assigned a unique study identification number; interview recordings, debriefing forms, and transcripts will be labeled with the participant’s study ID. All data to be shared during meetings will be de-identified to maintain confidentiality.
Rigor
Interview Reflexivity
Using the debriefing forms to capture interviewer’s reflexivity, including bias, preconceptions, and a summary of what worked and did not work in the interview, allows interviewers to explore their positionality in relation to participants. These reflections help the study team identify areas for improvement, develop additional interviewer training tools, and make necessary modifications to interviewing procedures.
Data Verification
A subset of the debriefing forms (10%) will be checked against the audio recording to ensure their accuracy. Similarly, the matrix will be checked against debriefing forms by two members of the study team. Inconsistencies in data entry will be discussed among the study team and addressed accordingly.
Inter-rater Agreement to Assess Reliability
The analysis team will code one patient and one observer transcript together to establish coding rules and procedures. Then three analysts will independently code another transcript and assess inter-rater agreement (Guest et al., 2012; Patton, 2015). Any discrepancies in coding will be discussed and adjudicated by a fourth member of the team. Any changes to the codebook will be documented with a version date, and transcripts that were coded with old codes will be updated. To ensure consistency in coding, inter-rater agreement will be assessed for every 5th transcript or approximately 20% of all transcripts.
Conclusions
The THINK-TTP study is designed to address a critical gap in clinical trial readiness for the assessment of cognition in patients with TTP by identifying the specific facets of cognitive function affected in this population. Through dyadic interviews with patients and their close observers, this study will provide foundational qualitative evidence to inform the selection and development of appropriate clinical outcome assessments. Ultimately, these efforts aim to ensure that cognitive dysfunctions, the outcome identified by patients as a top priority, are meaningfully captured in the evaluation of existing and emerging therapies.
Supplemental Material
Supplemental Material - The THINK-TTP Study for Assessing Cognitive Function in Clinical Trials: A Qualitative Research Protocol
Supplemental Material for The THINK-TTP Study for Assessing Cognitive Function in Clinical Trials: A Qualitative Research Protocol by Theresa Coles, Laura Mkumba, Dana Thompson, Brian D. Adkins, Benjamin F. Tillman, Jamila Minga, Elizabeth Cieza, Jada Green, Fiorella Guerrero Calle, Kristin Byrne, Olivia Fernandez, Oluwatoyosi Onwuemene in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material - The THINK-TTP Study for Assessing Cognitive Function in Clinical Trials: A Qualitative Research Protocol
Supplemental Material for The THINK-TTP Study for Assessing Cognitive Function in Clinical Trials: A Qualitative Research Protocol by Theresa Coles, Laura Mkumba, Dana Thompson, Brian D. Adkins, Benjamin F. Tillman, Jamila Minga, Elizabeth Cieza, Jada Green, Fiorella Guerrero Calle, Kristin Byrne, Olivia Fernandez, Oluwatoyosi Onwuemene in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material - The THINK-TTP Study for Assessing Cognitive Function in Clinical Trials: A Qualitative Research Protocol
Supplemental Material for The THINK-TTP Study for Assessing Cognitive Function in Clinical Trials: A Qualitative Research Protocol by Theresa Coles, Laura Mkumba, Dana Thompson, Brian D. Adkins, Benjamin F. Tillman, Jamila Minga, Elizabeth Cieza, Jada Green, Fiorella Guerrero Calle, Kristin Byrne, Olivia Fernandez, Oluwatoyosi Onwuemene in International Journal of Qualitative Methods
Footnotes
Acknowledgments
The team would like to acknowledge Nicole Lucas at Duke University, Monika Ruscheinsky at UT Southwestern, and Niral Patel at Vanderbilt for their support during the initial IRB submission and the study start-up phase. The team also thanks the volunteers for their time and feedback on the pilot interviews, and the study participants for their important contributions.
ORCID iDs
Ethical Considerations
The study was reviewed and approved by the Duke University Health System Institutional Review Board (Pro00115540).
Consent for Publication
All participants provided either verbal or written informed consent.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was supported by a grant to OAO and TC from the National Institutes of Health/National Center for Advancing Translational Science 1R21TR004694-01A1.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: O.A.O has received honoraria from Sanofi, and T.C. consults for Regenxbio and owns Proof of Purpose Consulting, LLC. These relationships have no connection to the information reported in this protocol. All other authors declare no conflicts.
Data Availability Statement
Upon study completion, interview data will be available in an online database.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
