Abstract
Chronic kidney disease and its maintenance therapy (hemodialysis) lead to numerous physical, psychological, and social challenges for patients undergoing hemodialysis. Consequently, individuals receiving hemodialysis must adapt and cope with various aspects of their illness to effectively manage stressors related to the treatment. Identifying the level of patients’ adaptation to hemodialysis using an appropriate instrument provides valuable information for developing specific treatment plans, delivering effective medical and nursing care, and enhancing the performance of the healthcare team. Such measures ultimately contribute to achieving the highest possible level of functioning with minimal complications, reducing the risk of mortality, and improving patients’ quality of life. Existing instruments assessing adaptation among hemodialysis patients are generally non-specific, primarily based on psychological theories, and often influenced by the cultural and social contexts of Western societies. Moreover, these tools have not been developed from the direct experiences and perceptions of the patients themselves. Therefore, there is a need to develop a culturally appropriate, specific instrument for assessing adaptation in hemodialysis patients, grounded in their own experiences and perceptions. Accordingly, this study aims to design a protocol for the development and psychometric evaluation of a tool for measuring patients’ adaptation to hemodialysis.
Background
The growing prevalence of chronic diseases poses a major challenge to global health (Tayebi et al., 2023). Chronic kidney disease (CKD) is one of the most common chronic conditions in humans, currently affecting approximately 850 million individuals worldwide. It is projected that by 2040, CKD will rank as the fifth leading cause of years of life lost (YLL) globally (Francis et al., 2024).
End-stage renal disease (ESRD) represents the advanced stage of CKD, defined by a glomerular filtration rate (GFR) of less than 15 ml per minute per 1.73 m2, and typically requires renal replacement therapy (RRT) such as dialysis or kidney transplantation (Ebadi et al., 2021; Palleti et al., 2023). Approximately four million people worldwide are undergoing RRT, with hemodialysis remaining the most common modality—accounting for roughly 69% of all RRT and 89% of all dialysis treatments (Bello et al., 2022).
Patients receiving hemodialysis face a complex set of physical, psychological, and social challenges that significantly impact their survival and overall well-being. Physical stressors include restrictions in physical activity, alterations in sexual function, dietary and fluid intake limitations, polypharmacy, loss of appetite, and reduced energy levels—all of which directly disrupt their quality of daily life. Psychosocially, patients often experience chronic anxiety, depression arising from lifelong dependence on dialysis machines, reliance on healthcare teams and caregivers, changes in family roles, limitations in work life and social relationships, altered body image and self-perception, the lengthy treatment process, and ongoing concerns regarding disease prognosis (Kalantari et al., 2023; Mohammed et al., 2022). Given the chronic and progressive nature of CKD, successful management requires not only precise medical interventions to control physical complications but also the enhancement of patients’ adaptive capacity to achieve optimal functional and psychological outcomes.
Adaptation to hemodialysis can act as a protective factor against potential psychological complications by fostering positive lifestyle changes and improving patients’ ability to meet their personal needs (Barberis et al., 2017). Conversely, poor adaptation among hemodialysis patients may lead to treatment and dietary non-adherence, neglect of self-care behaviors, and a consequent decline in quality of life, ultimately affecting clinical outcomes and resulting in various physical, psychological, social, economic, and emotional consequences (Mafi et al., 2019; Nikpey et al., 2023).
Adaptation to chronic illness-related stressors has thus become an increasingly important area of research and intervention in medical sciences, as many individuals worldwide live with such long-term conditions (Dehghani et al., 2018). Adaptation is a complex and dynamic process that requires continuous evaluation (Biesecker et al., 2013).
Common Instruments Used in Studies to Assess Adaptation in Hemodialysis Patients
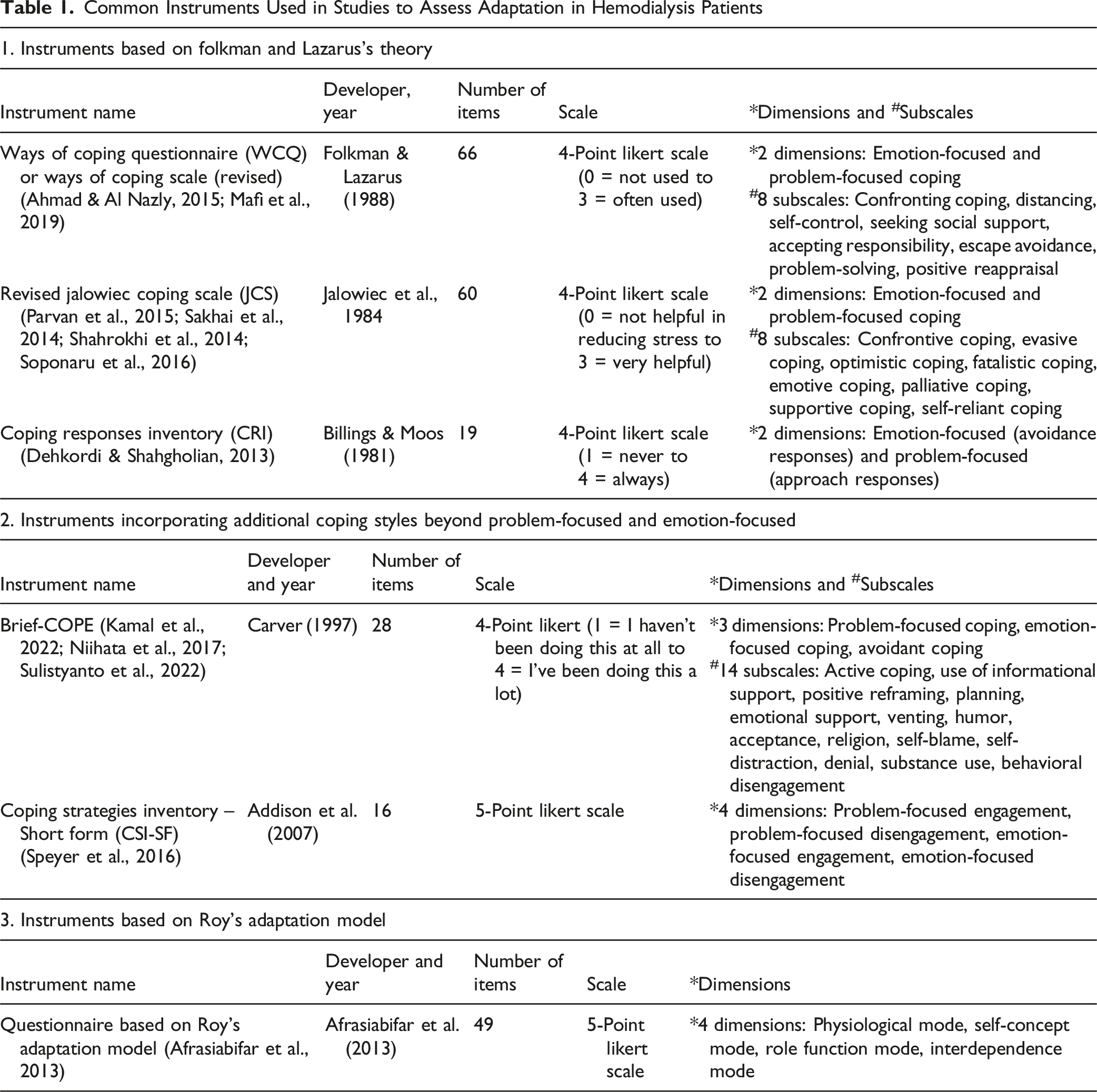
Carver and colleagues acknowledged that the two conventional coping categories were insufficient to capture the diversity of coping responses, leading them to develop the COPE inventory, which includes three categories of coping strategies (Carver et al., 1989). Zhao et al. (2022) later criticized both the Lazarus and Folkman and the Carver instruments, noting that these tools were developed almost exclusively using Western samples. Since cultural values significantly influence individuals’ coping strategies, tools designed within Western contexts may fail to capture coping mechanisms relevant to other cultural settings (Zhao et al., 2022).
Similarly, instruments developed under Roy’s Adaptation Model are often self-constructed, lacking rigorous design and psychometric evaluation, with only validity and reliability testing performed (Afrasiabifar et al., 2013). Other instruments, such as Pargament’s Spiritual Coping Scale (Pargament et al., 2000) and the Spiritual Coping Strategies Questionnaire by Baldacchino and Buhagiar (Baldacchino & Buhagiar, 2003), have also been used in some studies to assess religious coping among hemodialysis patients (Saffari et al., 2014; Taheri-Kharameh et al., 2016). However, these instruments solely focus on the spiritual dimension, whereas adaptation encompasses multiple interrelated domains.
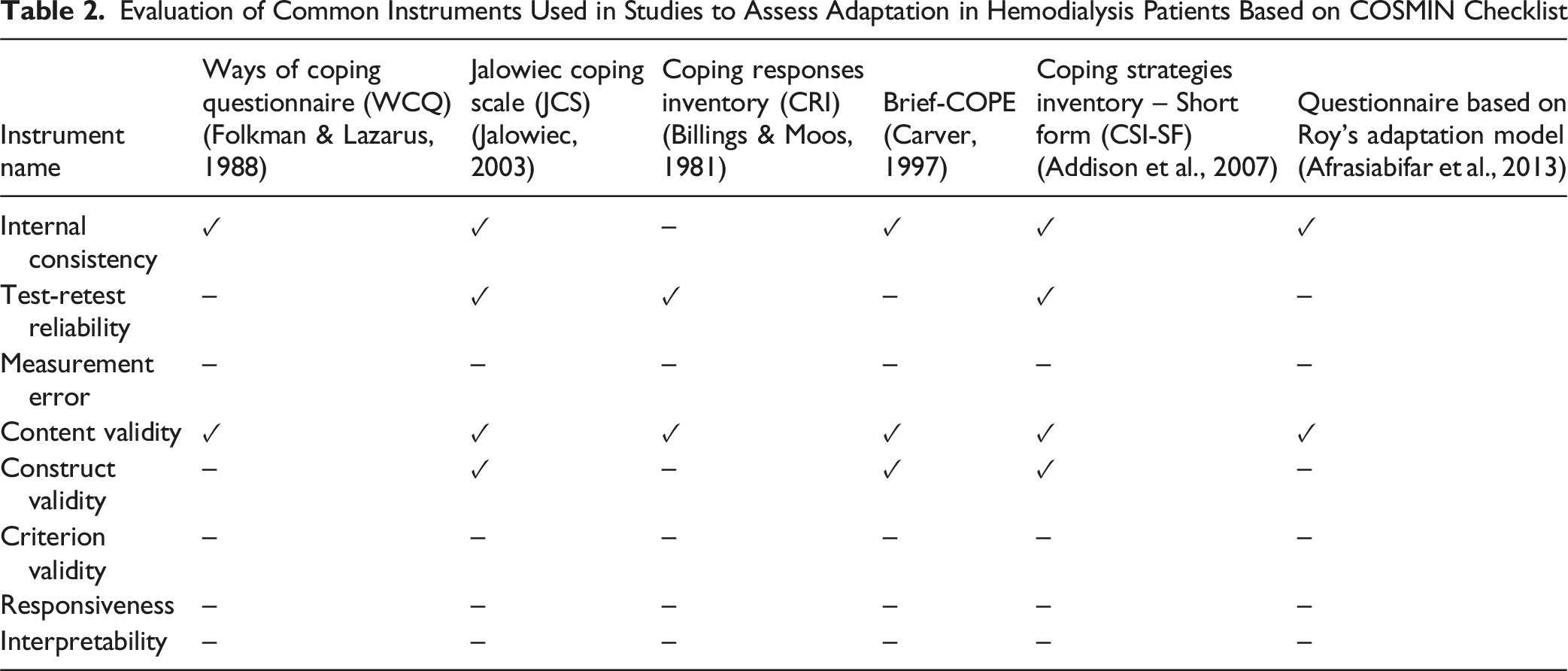
Evaluation of Common Instruments Used in Studies to Assess Adaptation in Hemodialysis Patients Based on COSMIN Checklist
Study Justification
Given the critical importance of adaptation in patients undergoing hemodialysis, the use of a standardized and appropriate instrument to assess their level of adaptation is essential. Such a tool enables a comprehensive understanding of the patient’s adaptive status, the influencing and associated factors, and the dimensions requiring support. This insight is fundamental for implementing effective interventions and offering practical recommendations aimed at enhancing the adaptation of patients to hemodialysis. Moreover, the instrument can provide valuable data to inform the development of personalized care plans, targeted therapeutic strategies, improvements in nursing practices, and optimization of healthcare team performance. By delivering actionable information regarding patients’ adaptive status, the tool may also serve as a decision-making benchmark for health system policymakers in planning and resource allocation to improve patient health outcomes.
Existing instruments used in previous studies are largely non-specific, predominantly grounded in psychological theories, and shaped by the cultural and social constructs of Western societies. The use of such tools—developed for particular cultural or social populations—may not provide adequate reliability or conceptual relevance when applied to groups with different cultural contexts. Consequently, the applicability and interpretability of their findings in other settings remain questionable. Furthermore, considering that adaptation is a dynamic and context-dependent process, its characteristics and dimensions may vary significantly based on factors such as disease type, lived experiences, personal beliefs, life history, personality traits, and cultural context. Therefore, there is a pressing need to design a dedicated questionnaire tailored to hemodialysis patients, grounded in their own experiences and perceptions.
Therefore, this study was conducted with the aim of designing a study protocol for the development and psychometric evaluation of an instrument to assess adaptation among patients undergoing hemodialysis.
Explanation and Justification of Method
The present study is a study protocol designed to conduct a sequential exploratory mixed-methods research with the aim of developing and validating an instrument for assessing adaptation among patients undergoing hemodialysis. Mixed-methods studies follow a pragmatic paradigm, in which the researcher collects and analyzes both qualitative and quantitative data and subsequently integrates them to achieve a more comprehensive understanding of the research phenomenon (Ivankova et al., 2006). The integration of qualitative and quantitative approaches guides the researcher toward a more in-depth understanding of the adaptation process in individuals receiving hemodialysis. The study will be conducted sequentially, consisting of a qualitative phase followed by a quantitative phase.
In the first phase, a conventional content analysis approach will be adopted to achieve a clear understanding of the concept of adaptation among hemodialysis patients and its underlying dimensions. In the second phase, the adaptation instrument will be developed, and its psychometric evaluation will be performed (Figure 1). The Protocol of Development and Psychometric Evaluation a Tool for Measuring Patient Adaptation to Hemodialysis
Phase 1: The Qualitative Phase
In this phase, since the aim is to explore, explain, and define the concept of adaptation among hemodialysis patients and its key dimensions, a qualitative approach based on conventional content analysis will be employed. The strength of this approach lies in its inductive nature—data are derived from participants’ direct experiences without imposing predetermined categories or theoretical frameworks. The researcher must become deeply immersed in the data to form a new understanding of the phenomenon under study. Participants will be encouraged to freely express their experiences without being constrained by preformulated questions or structures (Mehrdad et al., 2013).
Objectives
(1) To explore hemodialysis patients’ perceptions and experiences of adaptation. (2) To identify and define the main constructs and dimensions of the adaptation concept among hemodialysis patients.
Settings
The study will be conducted in hemodialysis units of public hospitals and private dialysis centers in the cities of Tehran and Khorasan, where the researcher has access to participants and can feasibly collect data.
Participants
Participants will be adult (≥18 years old) hemodialysis patients who meet the following inclusion criteria: • Having a confirmed medical diagnosis of chronic kidney disease (CKD). • Having undergone hemodialysis for at least six months (to ensure they have moved beyond the initial crisis phase and are able to articulate their lived experiences). • Being capable of verbal communication and willing to participate. • Absence of other diseases unrelated to renal function that could alter the characteristics of adaptation.
Sampling
Participants will be selected using purposive sampling, an appropriate strategy for qualitative research (Bolderston & sciences, 2012). The researcher will strive for maximum variation among participants in terms of age, gender, duration of hemodialysis treatment, socioeconomic status, education, occupation, and place of residence, among others. Sampling and data collection will continue until data saturation is reached.
Data Collection
Data will be gathered through in-depth, unstructured, face-to-face interviews conducted individually in a quiet, private setting approved by the participants.
At the beginning of each interview, after explaining the purpose of the study and obtaining verbal consent for audio recording, general questions will be posed to build rapport and familiarize the researcher with the participant’s background. Subsequently, the discussion will gradually move toward more specific and goal-oriented questions, such as: -“Can you please describe your experience with hemodialysis and life with it?”-
Follow-up probing questions will be used to elicit deeper insights, for example: • “Could you explain that a bit more?” • “What do you mean by that?” • “Would you please give an example to help me better understand?”
If a participant becomes fatigued, the interview will be paused and continued in subsequent sessions. Throughout the interview, nonverbal cues (such as gestures, tone, and pauses) will be noted. At the end, participants will be asked an open question such as:
“Is there anything else you would like to add that you think is important about adapting to hemodialysis?”
Participants will then be thanked for their contribution and informed that they may be contacted later for member checking (to verify their statements) or follow-up interviews if necessary. The duration of each interview will depend on the participant’s endurance, availability of information, willingness, and mutual agreement.
Data Analysis
Qualitative data will be analyzed through conventional content analysis, using the continuous comparative method based on the steps proposed by Graneheim and Lundman (2004), as follows (Graneheim & Lundman, 2004): (1) Determination of the Unit of Analysis (2) Repeated Reading and Achieving a Sense of the Whole (3) Identification of Meaning Units (4) Condensation of Meaning Units (5) Coding (6) Creating Categories or Categorization (manifest content) (7) Extraction of Themes (latent content) (8) Interpretation and Final Reporting
Phase 2: the Quantitative Phase
Tool Development
To design the Adaptation Assessment Questionnaire for Hemodialysis Patients, we followed Waltz’s & Lenze (2017) four-stage norm-referenced approach (Waltz CF & Lenze, 2017).
Objectives
1. To develop relevant and appropriate items to adaptation constructs in hemodialysis patients
Method
As outlined by Waltz CF & Lenze. (2017), the essential steps in instrument development are (Waltz CF & Lenze, 2017): Step 1: Selection of a conceptual model – The presence of a sound theoretical foundation is essential for producing appropriate items that represent the full content domain of a new instrument. Conceptual models can be derived inductively, deductively, or through a combined approach. Given the inherently subjective nature of adaptation among hemodialysis patients, in this study the conceptual model will be developed inductively, based on participant experiences identified in the qualitative phase. Step 2: Explication of objectives for the measure – This step clarifies the measurement objectives. Step 3: Development of a blueprint – A blueprint will be drafted to determine and propose the necessary items to measure each construct according to the operational definitions. Step 4: Construction of the measure, including administration procedures, an item set, and scoring rules and procedures– Items will be formulated for each construct, guided by theoretical and operational definitions. Instructions for administration, scoring, and interpretation will also be developed.
Psychometric Evaluation
A methodological study will be conducted to examine the instrument’s psychometric properties.
Objectives
(1) To determine the validity (content validity, face validity, construct validity) of the tool (2) To determine the reliability (internal consistency, relative/absolute stability) of the tool (3) To determine the responsiveness/sensitivity of the tool. (4) To determine the interpretability of the tool. (5) To determine the feasibility of the tool. (6) To determine the scoring method of the tool.
Method
(1) Validity. Validity will be assessed in three domains: content validity, face validity, and construct validity (Sharif Nia et al., 2022).
1.1. Content Validity: Content validity will be examined through both qualitative and quantitative approaches (Sharif Nia et al., 2022).
Qualitative content validity: Ten experts (in instrument design, hemodialysis, and nursing) will review the questionnaire according to Haynes’ criteria—grammar, appropriate vocabulary, necessity, logical placement of items, and adequacy of scoring. Their feedback will be used for refinement.
Quantitative content validity: Content Validity Ratio (CVR) will assess the necessity of each item, based on expert ratings using a three-point scale (“not necessary” = 1, “useful but not necessary” = 2, “necessary” = 3). CVR will be calculated using below CVR formula and compared with Lawshe’s values. With ten experts, CVR values above 0.62 will be considered acceptable.
Content Validity Index (CVI) will assess item relevance on a four-point scale (“not relevant” = 1, “somewhat relevant” = 2, “quite relevant” = 3, “highly relevant” = 4). The Item-level CVI (I-CVI) will be calculated, with below I-CVI formula: • I-CVI <0.70 → item deleted • 0.70–0.79 → item revised • ≥ 0.79 → item accepted
To control for chance agreement inflation, the modified kappa statistic ( • K = 0.40–0.59 → weak agreement • K = 0.60–0.74 → good agreement • K ≥0.74 → excellent agreement (Sharif Nia et al., 2022).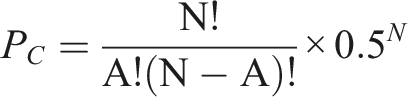

Finally, the Scale-CVI (S-CVI) will be computed, with ≥0.90 considered excellent and ≥0.80 the minimum acceptable threshold. At the end of each questionnaire domain, experts will also comment on domain adequacy.
2.1. Face Validity: Face validity will be evaluated qualitatively and quantitatively with members of the target population (Sharif Nia et al., 2022).
Qualitative face validity: Ten hemodialysis patients will assess items for clarity, relevance, and difficulty. Feedback will guide revisions.
Quantitative face validity: An impact score will be calculated for each item using: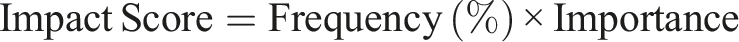
Here, frequency is the percentage of participants rating the item as 4 or 5 (“quite important” or “extremely important”), and importance is the mean score on the five-point Likert scale (5 = extremely important to 1 = not important at all). Items with impact scores >1.5 will be retained (Colton & Covert, 2007).
3.1. Item Analysis: Item analysis will serve as a preliminary evaluation of the questionnaire’s performance in the target population. The loop method will be applied. Initially, the overall reliability coefficient will be computed. Items whose deletion reduces reliability will be retained. Item-total correlations must be ≥0.30; items with correlation ≥0.70 with another item will be compared for redundancy—if semantically similar, one will be removed; if not, both will be kept. Items with no correlation ≥0.30 with any other item will be deleted (Polit & Yang, 2016).
4.1. Construct Validity: Construct validity will be assessed via exploratory factor analysis (EFA). EFA is useful for data reduction and clarifying the theoretical structure. Steps include: (1) Computing the correlation matrix for all variables. (2) Extracting initial factors. (3) Rotating the extracted factors (Grove et al., 2012; Polit & Yang, 2016).
Regarding sample size in factor analysis, recommendations vary: • 6–10 participants per item (Sharif Nia et al., 2022) • At least five participants per item, and no fewer than 100 participants overall, per Gorsuch (Gorsuch, 2014).
Data will be collected from hemodialysis patients who complete the instrument. Before conducting EFA, prerequisites will be checked: • Kaiser–Meyer–Olkin (KMO) measure for sampling adequacy (≥0.70 acceptable; 0.90 excellent) (Munro, 2005). • Bartlett’s test of sphericity for significance of correlations among items.
Factor extraction will be performed using principal axis factoring (Graneheim & Lundman, 2004). Factor naming will be based on the associated items and the theoretical framework established in the qualitative phase. Analysis will be conducted using SPSS version 23.
(2) Reliability. To assess the reliability of the questionnaire, two complementary approaches will be used: internal consistency and stability (Saffari et al., 2014).
1.2. Internal Consistency: Internal consistency will be determined by calculating Cronbach’s alpha coefficient in a sample of 30–50 participants. An alpha coefficient of 0.70 or higher will be considered acceptable for item retention within the scale.
2.2. Stability: Reliability in terms of stability includes both relative stability and absolute stability.
To examine relative stability, the Intraclass Correlation Coefficient (ICC) will be calculated. For this purpose, the instrument will be administered to a sample of 30 patients undergoing hemodialysis at two different time points, one week apart. The ICC will then be computed between the scores of the two administrations. Values of ICC closer to 1.00 indicate stronger test–retest reliability (Grove et al., 2012).
To examine absolute stability, the Standard Error of Measurement (SEM) will be calculated in the same 30-patient sample. SEM distinguishes real variation in participants’ status from variation due to measurement error (Ebadi et al., 2017). It will be computed using the following formula: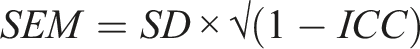
(SD: Standard Deviation, ICC: Intraclass Correlation Coefficient).
(3) Responsiveness (Sensitivity). Measurement instruments must be sensitive to real changes in the construct being measured. One method for assessing responsiveness is hypothesis testing, which examines whether the instrument can detect expected differences over time or between known groups (Ebadi et al., 2017).
To evaluate responsiveness in this study, the Pearson correlation test will be used to assess the relationship between patients’ adaptation scores and the duration of hemodialysis treatment. A statistically significant correlation would support the instrument’s responsiveness.
(4) Interpretability. Interpretability refers to the degree to which qualitative meaning can be assigned to changes in instrument scores (Shirinabadi Farahani et al., 2015). Based on the COSMIN checklist, interpretability of the developed questionnaire will be evaluated through the following indicators:
1.4. Minimal Important Change (MIC): The MIC will be calculated by multiplying the standard deviation of the change scores (test–retest) by a moderate effect size of 0.50. The MIC should exceed the Minimal Detectable Change (MDC) to indicate clinical relevance.
2.4. Ceiling and Floor Effects: Total questionnaire scores will be standardized to a 0–100 range, and the percentage of respondents obtaining minimum (0) or maximum (100) scores will be calculated. The ceiling or floor effect should be less than 20%.
3.4. Score Distribution Across the Sample: The mean and standard deviation of responses are expected to vary across participant subgroups (e.g., gender, marital status, and education). Accordingly, mean adaptation scores will be compared among these groups to evaluate dispersion patterns.
4.4. Proportion of Missing Responses: A missing response rate between 10–20% (Grove et al., 2012) or up to 15–20% (Dong & Peng, 2013) will be considered acceptable.
(5) feasibility. feasibility refers to the simplicity and practicality of applying the instrument for measuring the intended construct. Short, easily understandable sentences, low percentage of missing response and a brief response time are key features that ensure the questionnaire is user-friendly and convenient for both patients and healthcare professionals (Sharif Nia et al., 2022).
(6) Scoring Method. The scoring procedure will depend on the response format of the items. For questionnaires employing a Likert-type format, scoring may be either: • Unipolar – with consecutive scoring from low to high values, or • Bipolar – with scoring from negative to positive values, possibly including a midpoint (Waltz CF & Lenze, 2017).
To transform raw scores into a 0–100 scale, a linear transformation formula will be applied for easier interpretation and comparison.
Conclusion
The measurement tool developed through this protocol can serve as a valid and objective instrument for assessing the adaptation level of patients undergoing hemodialysis. Its application will enable healthcare providers to plan and tailor care services, counseling, and referrals according to patients’ adaptive needs. Moreover, the results of this study may guide future research aimed at designing and implementing intervention programs to enhance the adaptation of this patient group.
A key strength of this study lies in the content development process, which will be based on interviews with the target population—hemodialysis patients themselves—ensuring that all items are derived from authentic patient experiences. This approach guarantees strong content relevance and contextual sensitivity.
A potential limitation is that, since the participants will be Iranian patients, the generalizability of the instrument might be constrained. However, we will seek maximum variation sampling to minimize this limitation and enhance the instrument’s representativeness.
Rigor
In this study, to enhance methodological rigor, the four trustworthiness criteria proposed by Guba and Lincoln (Elo et al., 2014) will be applied.
The credibility of the study will be strengthened through establishing effective communication and trust with participants, allocating sufficient time for data collection, repeatedly reading interview transcripts, engaging in prolonged interaction with the data, and achieving deep immersion in the analytic process. Member checking will also be conducted to verify the accuracy of the extracted meanings and interpretations.
To ensure transferability, purposeful sampling with maximum variation will be employed. In addition, the context, setting, sampling method, participant characteristics, and detailed procedures of data collection and analysis will be thoroughly described to allow readers to assess the applicability of the findings to other contexts.
Dependability will be enhanced by peer debriefing within the research team and external audit controls through six-monthly supervisory reviews of the research process and reports.
Finally, to establish confirmability, the research procedures and findings will be documented transparently and comprehensively to enable independent experts to audit and verify the consistency and neutrality of the interpretations.
Footnotes
Acknowledgments
This study is part of a doctoral dissertation in nursing, approved at Tarbiat Modares University under approval number 52/1/9156, dated December 23, 2023. The authors appreciate all the participants who will be participated in the present study. Also, we sincerely appreciate the cooperation of the Research Vice-Chancellor of the Faculty of Medical Sciences and Tarbiat Modares University.
Ethical Considerations
This study has been approved by the Ethics Committee of Tarbiat Modares University (Code: IR. MODARES.REC.1402.16, Date:24/12/2023). Informed consent will be obtained from all participants for study participation, answering interview questions, and recording interviews. Participation will be entirely voluntary, and patients were assured of their right to withdraw at any time. Confidentiality of collected data will be strictly maintained. The study will adhere to the Declaration of Helsinki and followed COPE (Committee on Publication Ethics) guidelines.
Author Contributions
Conception: Effat Afaghi, Eesa Mohammadi, Fazlollah Ahmadi, Design: Effat Afaghi, Eesa Mohammadi, Fazlollah Ahmadi Supervision: E
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Restrictions apply to the availability of data from this research because of the anonymity of the participants and confidentiality matters.
