Abstract
Mixed-methods process mapping is a visualisation tool that identifies the steps, resources and personnel required to deliver a clinical practice, and has been previously used in an ad hoc manner to develop effective implementation strategies and solutions. To realise the potential of mixed-methods process mapping as an implementation tool, we aimed to develop and formalize the methodological steps and provide guidance for contemporary best practice approaches to using this approach for optimising implementation practice and research. Synthesising theory, evidence and expertise, we have identified 10 best practice recommendations and provide the first systematic framework for integrating mixed-methods process mapping into three core phases of health systems implementation, specifically: (1) engaging interest holders (and maintaining engagement), (2) identifying when, where, why, and to whom change is needed (and potential consequences), and, (3) identifying barriers and enablers, and co-designing implementation strategies. For each phase, we provide: (a) a rationale for using mixed-methods process mapping, (b) best practice guidance for combining mixed-methods process mapping with implementation practice and research, and (c) case studies exemplifying best practice. This article provides intelligence on mixed-methods process mapping to improve the consistency and quality of its use among implementation researchers and practitioners. We present a rationale, guidance, and practical tools for conducting mixed-methods process mapping to enhance the quality of implementation research and practice which can be used and adapted internationally. In doing so, it builds capacity and provides an opportunity for researchers and healthcare professionals to better understand and embed evidence-based innovations into health systems, improving service and client outcomes. Further research is needed to establish potential uses of mixed-methods process mapping to support other core components of implementation practice (e.g., adaptation), and to formally test the impact of this approach independently versus as part of a combination of implementation strategies.
Keywords
Background
Despite efforts to improve the efficiency of evidence translation into practice, there remains a stark difference between our knowledge of what works and the time it takes to get that knowledge routinely used in real-world settings (Smith et al., 2020). The field of implementation science focuses on the study of methods to accelerate the translation of evidence-based interventions, programs and policies into routine clinical practice to maximise client/patient outcomes and potential impact (Bauer et al., 2015). Implementation efforts can range from aiming to embed evidence into the system (implementation practice), undertaking a robust test of the effectiveness or cost-effectiveness of implementation methods, to an in-depth process evaluation (Metz et al., 2021). These efforts typically require healthcare staff to engage in information-gathering and idea-generation activities throughout an implementation process (e.g., specifying what behaviour needs to change, identifying barriers and enablers, co-design, monitoring, and evaluation).
Different members of the workforce are involved in implementation, including healthcare professionals (HCPs), managers, administrators, IT, etc. (also referred to as interest holders – i.e., those involved in or impacted by the existing or new process), and it is important to clarify their role in each step. Those tasked with an implementation challenge, from HCPs, through to implementation practitioners and researchers, must typically synthesise complex formal and informal data using - to varying extents - evidence-based theories, models, and frameworks to facilitate this journey whilst often simultaneously attempting to unpick the context-specific active ingredients of success (Taylor et al., 2023).
Whilst evidence-based implementation methods are necessary for enhancing replicability and building on existing knowledge to minimise the need to reinvent the implementation wheel, operationalisation can be long-winded, complicated, and overwhelming. This can lead to confusion amongst interest holders, as well as frustration around the time it takes to achieve impact on an individual site level and at scale (Taylor et al., 2019). This also poses a risk that implementation research in itself (the very thing that is supposed to optimise translation) can produce an evidence gap if there is a disconnect between the knowledge produced by implementation researchers, and the way in which HCPs and health services can apply it. One way to overcome this problem for both HCPs and implementation researchers is to explore new ways of how to extend the offerings of traditional improvement methods through an implementation science lens to speed up both the practical implementation process, and the rate at which implementation research is generated (Beidas et al., 2022). While there has been research on using theorised process maps to understand mechanisms of implementation success, the full potential and application of system and practice mixed-methods process mapping as a tool to support implementation and advance the science has yet to be thoroughly detailed (Kim et al., 2023; Klasnja et al.; Knapp et al., 2022; Lewis et al., 2022). In this article, we explain how the quality improvement method of ‘process mapping’ can be expanded to expediate the impact of both implementation practice and research.
Process Mapping
Process mapping has been traditionally used as a business tool to understand existing work systems/processes, resource allocations and areas for performance improvement (Dani et al., 2019; Kalman, 2002). In recent years, process mapping has been applied to health systems to improve implementation efforts (Best et al., 2023; Kononowech et al., 2020; Morrow et al., 2023). In its simplest form, process mapping provides a visual representation of the steps involved in delivering a standard clinical practice, to identify and minimise variation. It is commonly used to clarify the sequence of steps required to provide care in different settings, identify who is involved, and the workflows between interest holders. Process mapping provides multiple interest holders working within and across organisations with a shared understanding of how different actors interact within a system towards the same goal (Holleran et al., 2019; Parker et al., 2007; Santana & Redondo, 2018; Schraagen et al., 2010). Clarifying who is responsible for what reduces risks of redundancies and improves efficiency. When used alongside practice audit data, it can also be a powerful tool to help HCPs to identify gaps between actual and ideal practice, an important first step in quality improvement. Process maps lay the foundation for developing care pathways outlining best practices. They also provide context and structure for decision support algorithms to generate data-driven recommendations for optimisation.
Implementation Science and Mixed-Methods Process Mapping
In recent years, process mapping has been used as part of a set of implementation steps (e.g., identifying modifiable behaviours for change) (Kononowech et al., 2020), and in combination with implementation frameworks (e.g., to identify barriers/enablers and co-design implementation strategies) (Morrow et al., 2023; Salvati et al., 2023) to achieve evidence translation (Best et al., 2023; Colligan et al., 2010; Kononowech et al., 2020; Morrow et al., 2023). Benefits from these approaches are also starting to extend towards the goals of systematically advancing the speed and science of implementation through, for example, consolidating implementation data collection activities (Morrow et al., 2023). Mixed-methods research uniquely supports the iterative and dynamic nature of implementation science by leveraging the strengths of both qualitative and quantitative approaches (Aarons et al., 2012; Creswell & Plano Clark, 2011). By integrating data through designs such as convergent parallel, explanatory sequential, exploratory sequential, and embedded, mixed-methods approaches comprehensively address complex, real-world implementation problems that cannot be thoroughly tackled by either approach alone (Curry et al., 2013). Mixed-methods process mapping is a valuable tool to support implementation practice and research, as it integrates qualitative insights (e.g., from interviews and focus groups) with quantitative metrics (e.g., audit data to illustrate time and frequency). This integration makes it a valuable tool to support implementation (Harrison, 2024).
To further realise this potential, the present study aimed to establish consensus on and guidance for contemporary best practice approaches to mixed-methods process mapping for optimising implementation practice and research.
The Development of Best Practice Recommendations
Drawing on existing evidence and professional expertise, we identified three core phases of implementation where the method can be applied: (1) engaging interest holders (and maintaining engagement), (2) identifying when, where, why, and to whom change is needed (and potential consequences), and (3) identifying barriers and enablers, and co-designing strategies (Taylor et al., 2013). Working through each core phase, we provide (a) a rationale for how mixed-methods process mapping can be applied to inform and contribute to each phase, (b) practical guidance for combining process mapping with implementation practice and research, and (c) existing examples from the literature and approaches with emerging potential. We also demonstrate which types of mixed-methods data integration approaches, as described by Creswell and Plano Clark (2011), could be considered in each phase.
Engaging Interest Holders (and Maintaining Engagement)
Rationale
Practical Guidance: How to Engage Interest Holders (and Maintain Engagement).


Example of Generic Process Map.
Example in Practice: Engaging Interest Holders (and Maintaining Engagement)
Medication errors, a leading cause of iatrogenic harm, (De Vries et al., 2008; Roughhead L et al., 2013; Saghafi & Zargarzadeh, 2014) occur in 5%–10% of in-hospital administrations (Patel & Balkrishnan, 2010). Efforts to reduce these errors (Patel & Balkrishnan, 2010) have led to the implementation of Electronic Medication Management Systems (EMMS) (Agrawal, 2009) and administration policies, which have improved safety guideline adherence (Eslami et al., 2008) and reduced documentation (Westbrook et al., 2010), prescribing and administration (Morriss et al., 2009) errors. However, EMMS usage and policy compliance by nurses remain inconsistent (Debono, 2014; Koppel et al., 2008; McLeod et al., 2015).
To understand this inconsistency, an ethnographic study was conducted in two hospitals (A and B), examining nurses’ use of EMMS, workarounds instances, and their perceptions (Debono et al., 2017). The study involved developing a ‘generic process map’ based on medication administration practice and policies, which was refined through training, document analysis, and discussions with the EMMS Implementation Teams at each hospital. This iterative process resulted in multiple versions of the process map for each hospital (see Additional file 1, Figure A1.1) and (Additional file 1, Figure A1.2), created using Microsoft Visio software (Debono, 2014).
The process maps, reflecting the steps and perspective of medication administration, were formed with local experts. Hospital A’s map was clinically oriented, with components based on medication administration routes, while Hospital B’s map was regulatory structured, with components based on medication administration requirements (Debono, 2014).
Nurses were shadowed across 91 shifts (or part thereof), and the process maps served as visual prompts. These maps facilitated interest holder network establishment, ongoing dialogue, and knowledge exchange between HCPs and implementation researchers. They provided a visual representation of medication administration processes, communicated existing practices and areas for improvement, and fostered a sense of ownership and commitment among interest holders.
Identifying When, Where, Why, and to Whom Change is Needed (and Potential Consequences)
Rationale
A common requirement in implementation projects is to identify target behaviours for change – that is, behaviours that could bring about the desired implementation outcome (Michie et al., 2011). Identification of these target behaviours can serve several purposes, including narrowing down the focus for relevant, targeted, and impactful implementation efforts, and identifying key behaviours that can be measured for monitoring and evaluation of implementation success (Bauer et al., 2015). The approach to specifying target behaviours for change will differ depending on the focus of the project – for example, optimising the use of evidence that already exists (e.g., hypertension management guidelines, or hand hygiene procedures) or the introduction of a new evidence-based intervention (e.g., use of precision medicine to guide cancer therapies, or introduction of a diabetes digital self-management intervention) into practice) (Bauer & Kirchner, 2020). Mixed-methods process mapping provides an opportunity to define clinical practices in behavioural terms – specifying the actors, steps, and decision points along the pathway (Morrow et al., 2023). It can be used to identify the behaviours causing existing/potential barriers and bottlenecks that need to be targeted by the implementation effort (Michie et al., 2011).
Drawing upon the Action-Actor-Context-Target-Time (AACTT) framework (Presseau et al., 2019) provides an approach for identifying and specifying each interlinked behaviour in the process map. Collaborative AACTT-specification with interest holders can serve not only to zone in on the sequences of actions and the actors responsible for their enactment, but also any variation in settings (contexts) and times (frequency or time-dependencies) across these actions and actors. AACTT-specification can therefore help to clarify which audit data to seek or additional data to collect (where possible), and more directly inform how subsequent process mapping steps are conducted. For instance, assessing barriers and enablers to particular AACTT-specified behaviours can help to uncover challenges that may be specific to a given context or time that may not otherwise be laid bare if only the action and actors are identified.
Practical Guidance: How to Identify Target Behaviours for Change.


Toolkit of the Resources Needed to Conduct In-Person and Online Mixed-Methods Process Mapping.
Example in Practice 1 – Upscaling the Use of Existing Telehealth Guidelines
Telehealth services, traditionally used in remote settings, were rapidly implemented during COVID-19 to maintain clinical care while avoiding infections. Despite high patient satisfaction, the impact on HCPs’ workload and job satisfaction is underexplored. A study was conducted to describe the activity of a rapidly upscaled telepharmacy service in an Australian metropolitan hospital, capturing patient use and clinician experience.
A retrospective audit compared service provision before and after COVID-lockdowns, supplemented with semi-structured interviews with all hospital pharmacists (n = 10) who worked in the outpatient pharmacy post-lockdowns (Carland et al., ASCEPT 2023, Adelaide, Australia). The interviews, informed by audit data, focused on telepharmacy experiences and workload impact. Pharmacists’ narratives helped draft two process maps for face-to-face and telepharmacy service models, refined with a pharmacist’s input.
The maps visualised workflow changes and task/role allocations (see Additional File 2, Figure A2.1 & Figure A2.2), revealing that telepharmacy involved complicated interactions and introduced repetitive administrative tasks. Inductive analysis of interviews and service delivery data showed that telepharmacy’s continued care provision resulted from a significant increase in pharmacists’ administrative workload and shifted medication management responsibility from patient to pharmacist.
Mapping telepharmacy revealed the hidden complexity of this substitution, visualised bottlenecks, and identified potential task re-allocation steps. Comparing face-to-face and telepharmacy maps highlighted the inherent efficiency and effectiveness of face-to-face service. It also raised questions about the value of telehealth services, particularly those driven by patient convenience rather than equitable care access.
Example in Practice 2 – Using Mixed-Methods Process Mapping to Inform the Design and Evaluation of a New Service-Level Intervention to Optimise the Use of Precision Medicine
Precision medicine trials often discover and validate new biomarkers for treatment response (Cahaney et al., 2022; Hadjadj et al., 2020; Lee et al., 2021), with tumour profiling aiding this process. However, treatments identified are often unapproved (‘off-label’) for paediatric use (Department of Health and Aged Care; Day, 2023), making access pathways opaque and time-consuming for HCPs. Paediatric oncologists, as the conduit between the patients and pharmaceutical companies, face challenges in obtaining off-label access, including regulatory navigation, negotiation with companies, and stringent documentation (Daly et al., 2023; Moerdler et al., 2019; Wong et al., 2020).
To address these challenges, ProCure was developed, a database and tool to streamline the off-label application process by providing drug profiles, off-label access literature, and contact information for relevant pharmaceutical companies (Mazariego et al., 2024; Tyedmers et al., 2025 (In preparation)). ProCure was designed for implementation, incorporating considerations of its practical use. This was guided by formal implementation process mapping interviews, and collaborative design sessions with potential end-users.
The first interview stage comprised of an implementation process mapping exercise; a visual representation of current practices associated with access to off-label therapeutics in a paediatric oncology precision medicine trial context (Antonacci et al., 2021; Kononowech et al., 2020; Morrow et al., 2023). A draft process map was used (see Additional file 2, Figure A2.3) to guide semi-structured interviews, asking participants to confirm or amend current practices. ProCure’s potential was discussed and steps were identified for its use or behavioural modification to encourage its use. With each interview, the process map was iterated to reflect amendments and new information.
The second interview phase included additional questions guided by the Consolidated Framework for Implementation Research (CFIR) (Damschroder et al., 2022), ensuring coverage of key potential barriers and enablers to ProCure use and any other factors affecting its implementation, which could be deductively coded.
Finally, collaborative design sessions were conducted using the final iterated process map (see Additional file 2, Figure A2.4) for additional feedback on ProCure’s best use in the off-label therapy application process. Three collaborative design sessions were organized, two with the project steering committee and one with a multidisciplinary team within a precision medicine molecular tumour board (MTB) meeting. These sessions facilitated the design process, allowing for immediate feedback, suggestions for improvements, and expert contributions to the resources’ development (Mazariego et al., 2024).
Example in Practice 3 – Using Mixed-Methods Process Mapping and Matched Audit Data to Co-design Outcomes for Hepatitis C Testing
Hepatitis C virus (HCV) infection affects approximately 124,000 people in Australia (The Kirby Institute, 2022). New treatments have cure rates of over 95%, however their uptake has been impeded by barriers to testing and treatment (Alavi et al., 2019; World Health Organization, 2017). Previous diagnostic pathways involved venepuncture testing and multiple visits, often leading to loss to follow-up (Conway et al., 2022). The GeneXpert point-of-care HCV RNA test, a finger prick test providing results in 1 hour, was developed to overcome these barriers (Grebely et al., 2017). It received Therapeutic Goods Administration approval in 2020 and is currently being rolled out as part of a national research program across Australia (Grebely et al., 2023). The study team are co-developing and testing an implementation intervention to enhance the uptake of HCV point-of-care testing. The first step uses process mapping to understand existing point-of-care testing practices, as well as barriers and enablers to optimisation.
Service providers from various sectors were engaged in interviews and focus groups to develop a preliminary process map (see Additional file 2, Figure A2.5). So far, we have conducted process mapping with 37 service providers from 15 community sites across four Australian states (New South Wales, Queensland, Canberra, and Western Australia).
Baseline audit data from provider surveys is being used to calculate the number of participants at each step of the point-of-care testing pathway for each site, to highlight gaps (loss to follow-up) where behaviour change and/or implementation support may be needed. The number of participants identified for testing during the initial consultation can be calculated from audit data on risk factors (such as current homelessness, history of incarceration and/or injecting drug use). This is compared to the number of HCV tests performed, and if there is a gap, highlights the need for better supports to engage at-risk participants in testing. Similarly, if the number of RNA tests performed does not match the number of participants who initiate treatment, this highlights the need for better treatment linkages. This audit data about risk factors, tests conducted, and treatment initiation can be used to design meaningful implementation outcomes to measure the effectiveness of the implementation strategy for the trial (Wolfenden et al., 2021). For example, a meaningful measure of reach is defined as the proportion of at-risk participants receiving point-of-care testing (Proctor et al., 2011).
Identifying Barriers and Enablers, and Co-designing Implementation Strategies
Rationale
Understanding how the decisions and actions of interest holders operate at multiple levels (provider-level, organisational-level, policy-level, consumer-level) and affect implementation in complex settings is key to designing effective implementation strategies. Conventional methods tend to focus on provider-level barriers to implementation, without considering how these are influenced by other actors in the broader system, process, and/or context (Morrow et al., 2023). This limits the understanding around what needs to be in place to optimise implementation, which subsequently can undermine desired outcomes. Mixed-methods process mapping can help to narrow down which specific actors influence the delivery of an existing or proposed intervention and therefore who should be interviewed to gain a holistic understanding of barriers and enablers of optimisation.
Conventional methods of barrier identification often take multiple interviews and/or focus group discussions with busy HCPs and considerable time to synthesise data and feedback. Iterative discussions typically occur sequentially: (1) identify target behaviours, (2) use an implementation framework to identify barriers and enablers, and then (3) use an implementation framework to co-design strategies to overcome barriers. Mixed-methods process mapping can reduce the time and number of interviews/focus groups needed to achieve these steps.
Practical Guidance: How to Leverage Mixed-Methods Process Mapping to Identify Barriers and Enablers and Co-design Implementation Strategies.

Example in Practice 1 – Optimising Lynch Syndrome Diagnosis
Lynch syndrome is a hereditary cancer predisposition, which requires a complex clinical diagnostic process involving multiple hospital departments. The Hide and Seek Project (HaSP) used a structured implementation approach to improve Lynch syndrome detection across seven Australian hospital networks (Morrow et al., 2019). Process mapping was used to understand the current clinical referral pathway, identify variation and gaps in practice, and define key behaviours for tailored intervention strategies (Morrow et al., 2023). Whilst a separate consultation activity was conducted to explicitly investigate barriers to identified target behaviours, the extent to which barriers could be inductively captured and theoretically coded through process mapping focus-group data was also explored.
Clinical interest holders attended two focus groups to develop site-specific process maps representing referral pathways. Focus Group 1 discussed current Lynch syndrome tumour testing and referral processes at their site. Transcripts were analysed to generate a computer-based process map, which (in line with Phase 2 practical guidance: how to identify target behaviours for change; recommendations 2.1–2.3) was presented alongside clinical audit data in Focus Group 2 to establish target behaviours for change. Six key areas of clinical practice variation across sites and site-specific target behaviours for change were identified.
Process mapping naturally prompted discussion of intuitively-identified barriers and potential implementation strategies among HCPs. Barriers were retrospectively coded using the TDF (Cane et al., 2012), and proposed strategies were coded using the Behaviour Change Techniques Taxonomy (Michie et al., 2013). Where possible, the Theory and Techniques Tool (Johnston et al., 2021) was used to determine theoretical alignment between intuitively-identified TDF-mapped barriers and corresponding behaviour change techniques identified within strategies, as demonstrated in Figure 3. Process to Assess Theoretical Alignment of Intuitively Identified Strategies using the Theory and Techniques Tool. 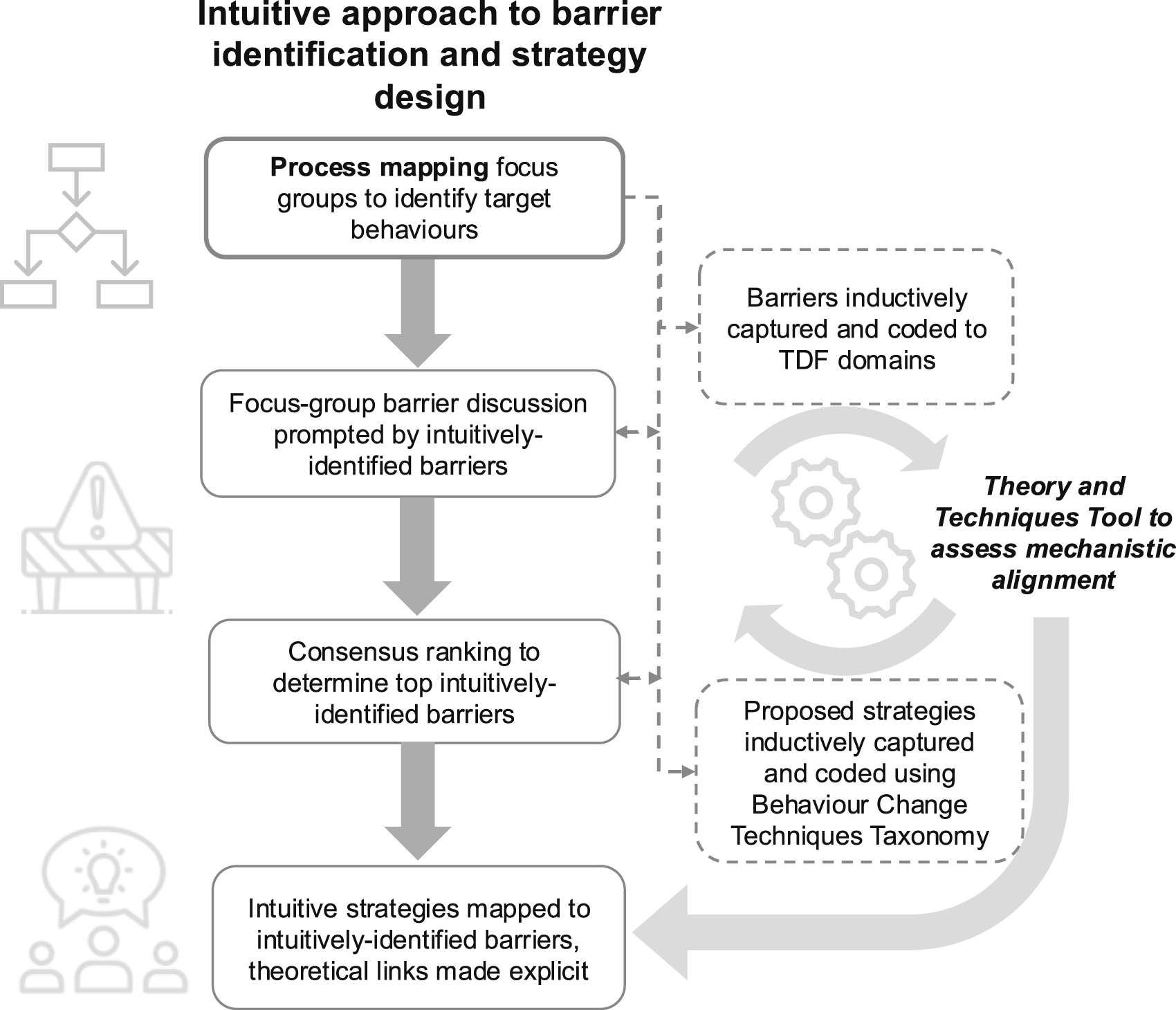
Ten barriers (across seven TDF domains) and 28 strategies were identified, with a significant proportion of intuitively generated strategies theoretically aligned (Morrow et al., 2024a). These were tabulated for consideration in later study phases, providing a pathway for targeted interventions. In this trial context, the activity served as part of a broader comparison between intuitive and theory-driven strategy development, rather than solely focusing on explicitly identifying barriers. However, in subsequent studies, there is potential to refine this approach by consolidating and systematically coding the barriers identified through the process mapping exercise, followed by subsequent consultation with interest holders to explore any additional barriers that might have been initially overlooked.
Post-implementation process evaluation interviews of clinician interest holders highlighted process mapping as a key determinant of implementation success, facilitating a shared understanding of roles and responsibilities within the multidisciplinary team, whilst also pinpointing critical gaps to address (Morrow et al., 2024b).
Example in Practice 2 – Enhancing Genomic Testing
Embedding genomics into clinical practice is complex, requiring a multidisciplinary team of genetic specialists, laboratory scientists and clinical context experts. Using a theory informed approach, the perceived and actual barriers and enablers to delivering clinical genomic testing in Australia were investigated.
Semi-structured, process map-guided interviews (n = 32) with those responsible for genomic testing were conducted (Taylor et al., 2019). The interview schedule was based on the TDF and used an outline process map shared with participants ahead of the interview (see Additional File 3, Figure A3.1) (Best et al., 2021a, 2021b). This map probed participants’ perceptions and experiences of barriers and enablers to genomic testing.
Examples of Standardisation by Form and Function Taken From the Process Map (Figure A3.2) and Applied to the Expert Recommendations for Implementing Change (ERIC) (19). Adapted From Hawe et al. (59)

The data were analysed to identify intuitive or experience-based implementation strategies (Taylor et al., 2023). Thirty-two barriers were identified, with 29 intuitive strategies addressing 23 barriers. An additional 20 theory-informed implementation strategies were generated to address remaining barriers. Comparing theory-informed and intuitive strategies, a 20% alignment was found. An algorithm was developed to specify strategy development and potential mechanisms of change, demonstrating how process map-driven data collection can contribute to implementation researchers’ efforts to make underlying theory transparent and explicit: The construct of A and distinct TDF domain B were selected because the HCPs were doing C behaviour. The strategy of D is derived from the BCT E because it will do F—this should reduce their G (distinct TDF domain) and change their H (overarching TDF theme) because of X (explanation) (Taylor et al., 2023).
Findings and Discussion
We have synthesised implementation science principles and expertise, and evidence from six case studies across areas of cancer, medication adherence, infectious disease control, and paediatric and adult genomics into a novel method for mixed-methods process mapping in health systems implementation. We have illustrated how mixed-methods process mapping can be systematically incorporated into three core implementation phases to (i) engage interest holders, (ii) understand what needs to change, and (iii) identify barriers and enablers to change and develop solutions. We have provided practical tools that can be adapted and used by implementation researchers internationally to standardise and streamline implementation research and practice. These include a process map template with pre-defined keys (Figure 1), and a toolkit of practical resources needed to perform mixed-methods process mapping (Figure 2) alongside detailed guidance (Tables 1–3), a summary of which is provided in Figure 4. Summary of Practical Guidance Across Core Implementation Phases.
We have established consensus on and guidance for contemporary best practice approaches to mixed-methods process mapping for optimising implementation practice and research using three key phases of implementation. First, interest holders need to be informally and formally consulted to identify target behaviours that have been verified with audit data. Second, researchers need to answer questions designed using implementation frameworks to carefully elicit information about specific behavioural and system determinants of change. Third, this learning contributes to the design and refinement of evidence based and feasible strategies to overcome barriers.
The standardisation of this evolving method aims to improve the quality and efficiency of mixed-methods process mapping in future implementation studies, enhancing the potential impact of the research both practically and scientifically. In doing so, it builds the capacity of researchers and HCPs to apply implementation research and practice to embed evidence-based innovations into health systems to improve service and client outcomes. While we encourage the adoption of these practical guidelines, it is important to consider the specific research scope in real-world applications, where not all practices presented here may be feasible. For example, an exploratory mixed-methods design approach may not be feasible for rapid quality improvement in implementation research due to its time-consuming and resource-intensive nature. To effectively operationalise mixed-methods approaches in process mapping, it is essential to consider a robust data integration plan before entering each core implementation phase. Alongside the example case studies we provided, the data integration plans suggest that across the implementation phases, qualitative data is likely to be predominant in the phases (i) engage interest holders and (iii) identify barriers and enablers to change and develop solutions, whereas quantitative and qualitative components may be given equal weight in phase (ii) understand what needs to change.
This methodology paper demonstrates a range of real-world benefits that can arise from using mixed-methods process mapping. Mixed-methods process mapping provides the ideal vehicle to incorporate implementation science theory, models, or frameworks. As a tool, it provides flexibility to respond to a wide range of research questions. This work highlights common uses of mixed-methods process mapping to date but is not exhaustive of the potential uses of this method to advance implementation science. Further research is needed to establish potential uses of mixed-methods process mapping to support other core components of implementation practice (e.g., how to effectively adapt processes and implementation approaches across settings), and to formally test the impact of mixed-methods process mapping, both independently and in combination with other implementation strategies, to achieve desired implementation outcomes.
Conclusion
We present a structured approach to accelerating the speed and quality of implementation in health systems through mixed-methods process mapping. By summarising 10 practical guidelines (Figure 4) across three core implementation phases, we provide a pathway for implementation researchers and practitioners, along with their clinical partners, to navigate complex implementation challenges. Together with the resource toolkit (Figure 2), these guidelines serve as a foundation to build capacity for targeted and efficient development and delivery of implementation solutions across contexts. Insights from our example case studies suggest that qualitative data tends to be predominant in the implementation phases of interest holders’ engagement and later in identification of implementation determinants and strategies, whereas a more balanced integration of both quantitative and qualitative data is likely required for identifying gaps for change, and understanding why these gaps exist. A proactive approach to data integration planning ensures mixed-methods process mapping is applied cohesively and effectively across implementation phases. Beyond offering a methodological framework, our work highlights the dynamic nature of mixed-methods process mapping and its potential for continuous refinement. We envisage that the proposed framework will be iteratively tested, adapted, and expanded based on learnings across various real-world healthcare settings.
Supplemental Material
Supplemental Material - Advancing the Speed and Science of Implementation Using Mixed-Methods Process Mapping – Best Practice Recommendations
Supplemental Material for Advancing the Speed and Science of Implementation Using Mixed-Methods Process Mapping – Best Practice Recommendations by Natalie Taylor, Carolyn Mazariego, Rachel Baffsky, Shuang Liang, Luke Wolfenden, Justin Presseau, Guillaume Fontaine, Jane E. Carland, Christine T. Shiner, Sarah Wise, Deborah Debono, Skye McKay, Stephanie Best, and April Morrow in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material - Advancing the Speed and Science of Implementation Using Mixed-Methods Process Mapping – Best Practice Recommendations
Supplemental Material for Advancing the Speed and Science of Implementation Using Mixed-Methods Process Mapping – Best Practice Recommendations by Natalie Taylor, Carolyn Mazariego, Rachel Baffsky, Shuang Liang, Luke Wolfenden, Justin Presseau, Guillaume Fontaine, Jane E. Carland, Christine T. Shiner, Sarah Wise, Deborah Debono, Skye McKay, Stephanie Best, and April Morrow in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material - Advancing the Speed and Science of Implementation Using Mixed-Methods Process Mapping – Best Practice Recommendations
Supplemental Material for Advancing the Speed and Science of Implementation Using Mixed-Methods Process Mapping – Best Practice Recommendations by Natalie Taylor, Carolyn Mazariego, Rachel Baffsky, Shuang Liang, Luke Wolfenden, Justin Presseau, Guillaume Fontaine, Jane E. Carland, Christine T. Shiner, Sarah Wise, Deborah Debono, Skye McKay, Stephanie Best, and April Morrow in International Journal of Qualitative Methods
Footnotes
Author Contributions
NT conceived the idea, drafted the manuscript, and led the writing process. DD, CM, JC, CS, SW, AM, and SB provided case studies. NT, AM, RB, and SL developed the recommendations and guidance. JP incorporated the AACTT framework. LW and GF helped to shape the introduction. All authors reviewed and refined the manuscript. All authors read the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
