Abstract
Cardiovascular diseases (CVDs) represent a leading cause of mortality worldwide, with a particularly high burden in low- and middle-income countries (LMICs). The lack of well-trained medical professionals in LMICs poses a fundamental challenge to the prevention of CVD. In response, a complex primary care-centered integrated care intervention (C-RISIMA) has been developed with a risk-stratified integrated care pathway and a team-based blended payment mechanism in primary care facilities in China. This paper presents a protocol for a mixed methods process evaluation of this complex intervention, conducted in a multi-center, two-arm, cluster randomized controlled trial. This process evaluation aims to explore how the C-RISIMA intervention is implemented within the context of China’s countywide health systems and incorporated into the existing practices, and how varied implementation shaped the outcome and sustainability of the intervention. The study incorporates quantitative data collection on intervention fidelity, dose, reach, and adaptation, as well as qualitative interviews and focus groups with involved stakeholders including healthcare professionals, patients, and administrators. The analysis will utilize a combination of realist evaluation and the Normalization Process Theory to distill insights about the intervention’s integration into routine clinical practice. The findings of this study will inform policymakers on the much-needed knowledge about implementing primary-care-centered integrated care interventions in similar settings, particularly in LMICs where the burden of CVDs is most acute.
Introduction
Background
Cardiovascular diseases (CVDs) are the leading cause of mortality worldwide, with 80% of cases from low- and middle-income countries (LMICs) (Joseph et al., 2017; Li et al., 2023; Liu et al., 2019; Wang et al., 2023). Currently, an estimated 330 million people in China are living with CVDs, which accounts for over 45% of total deaths (Hu & Writing Committee of the Report on Cardiovascular Health and Diseases in China, 2024; National Health Commission, 2024; Zhang et al., 2023). The total healthcare expenditures caused by CVDs is over 600 billion RMB in China along with substantial indirect costs due to loss of productivity and long-term care. The increasing economic burden makes it urgent for China to implement effective primary prevention strategies (Hu & Writing Committee of the Report on Cardiovascular Health and Diseases in China, 2023).
Strengthening primary care with an emphasis on risk factor management has significant potential in preventing cardiovascular diseases (CVDs), as most events are avoidable through effective risk factor control (Smith et al., 2004). However, it is challenging to provide individualized and effective primary care due to the shortage of skilled professionals in China (Qin et al., 2022). Additionally, the fragmentation of preventive and curative services further exacerbates the situation (Li et al., 2020; Wu et al., 2016). It is estimated that nearly half of all ambulatory consultations in China take place in secondary and tertiary hospitals (National Health Commission, 2024). In contrast, primary care providers have limited capacity to effectively manage cardiovascular risk factors (Du et al., 2019). Stratifying individuals into homogeneous groups based on shared CVD risks has been acknowledged as a feasible and cost-effective approach to integrating care resources (Lewis et al., 2013), with its effectiveness being proven effective in small pilots in China (Hu et al., 2021).
Despite demonstrated effectiveness of integrating specialist and primary care based on risk stratification in CVD prevention, little is known about how risk-stratified integrated care with proactive participation of specialists in primary care can be incorporated into daily health services to achieve optimal impact (Schwalm et al., 2019; Wei et al., 2017), or the context needed for such interventions to deliver optimal impact as designed (Goodwin, 2019; May et al., 2022; Moore et al., 2015; Saarijärvi et al., 2020). In fact, the lack of risk stratification as a key function in primary care has been noted even for national reform pioneering pilots of people-centered integrated care in China (Wang et al., 2018). To address this gap of knowledge about how to introduce CVD risk-stratification in order to strengthen primary care as the center of and integrated health services in the Chinese context, a complex intervention, namely the Cardiovascular RIsk-Stratified Integrated MAnagement (C-RISIMA), has been developed and implemented as a cluster randomized controlled trial (RCT). This paper presents a protocol for a process evaluation of the C-RISIMA trial, aiming at understanding the mechanism underlying the process by which the intervention achieves (or does not achieve) its expected outcomes, and the contextual conditions required to ensure that the intervention could be effectively adopted into routine healthcare.
C-RISIMA Study
Building on previous evidence from China and elsewhere, we designed a multifaceted intervention for CVD prevention focused on primary care in China. The components of C-RISIMA intervention include a risk-stratified integrated care pathway, along with a team-based blended payment mechanism that incorporates pay for coordination and pay for performance. The intervention was evaluated in the C-RISIMA study, a multi-center, two-arm, cluster RCT involving 64 clusters. The recruitment period will be completed before May 2025, and data collection is expected to be completed by the end of 2027. Details about the preceding work of C-RISIMA have been published in the main trial protocol, which is registered on https://www.clinicaltrials.gov.
In brief, the risk-stratified integrated care pathway proposes the incorporation of a specialist (cardiologist, neurologist, or endocrinologist from a local county-level hospital) into the family doctor team (FDT) to provide the stratified care pathway designed based on CVD risk and clinical guidelines. A typical FDT in rural China, including a village doctor from a village clinic and a general practitioner (GP) from a township health center, often lacks the capacity to adjust prescriptions for patients with uncontrolled hypertension and/or diabetes (National Health Commission, 2016). The specialist newly added to the FDT is expected to provide appropriate medical advice to the primary care team aimed at improving care for such patients, through monthly case discussions. With professional support from specialists, GPs and village doctors may have more confidence and skills in adjusting the medication plan for patients (Hu et al., 2021). In addition, enhanced frequency of patient follow-up by the primary care practitioners may increase adherence to treatment regimens. The integration of curative and preventive care could therefore benefit from the establishment of the new FDT.
Meanwhile, the new care pathways are also designed along with FDT enhancement (Details in Figure 1). C-RISIMA provides three different care pathways for individuals in three risk groups: low risk (10-year CVD risk: <10%), moderate risk (10-year CVD risk: 10%∼19%), and high risk (10-year CVD risk: >19%), based on the updated Laboratory-Based WHO Cardiovascular Risk Chart for East Asians considering their age, sex, blood pressure, smoking status, total cholesterol and presence or absence of diabetes (World Health Organization, 2019). The detailed content for the pathways is developed based on established guidelines and inputs from clinical experts. According to the pathway, participants in different risk tertiles receive tailored management plans, particularly regarding health education and follow-up frequency. Timely adjustments for management plans are also made based on dynamic risk monitoring for each participant. Design of the Cardiovascular RIsk-Stratified Integrated Management (C-RISIMA) Intervention. *The Follow-Ups Will Collect Individual Data for CVD Risk Evaluation, Conduct CVD Risk Communication, Measure Medication Adherence, and Provide Lifestyle Counseling and Personalized Health Advice to Participants.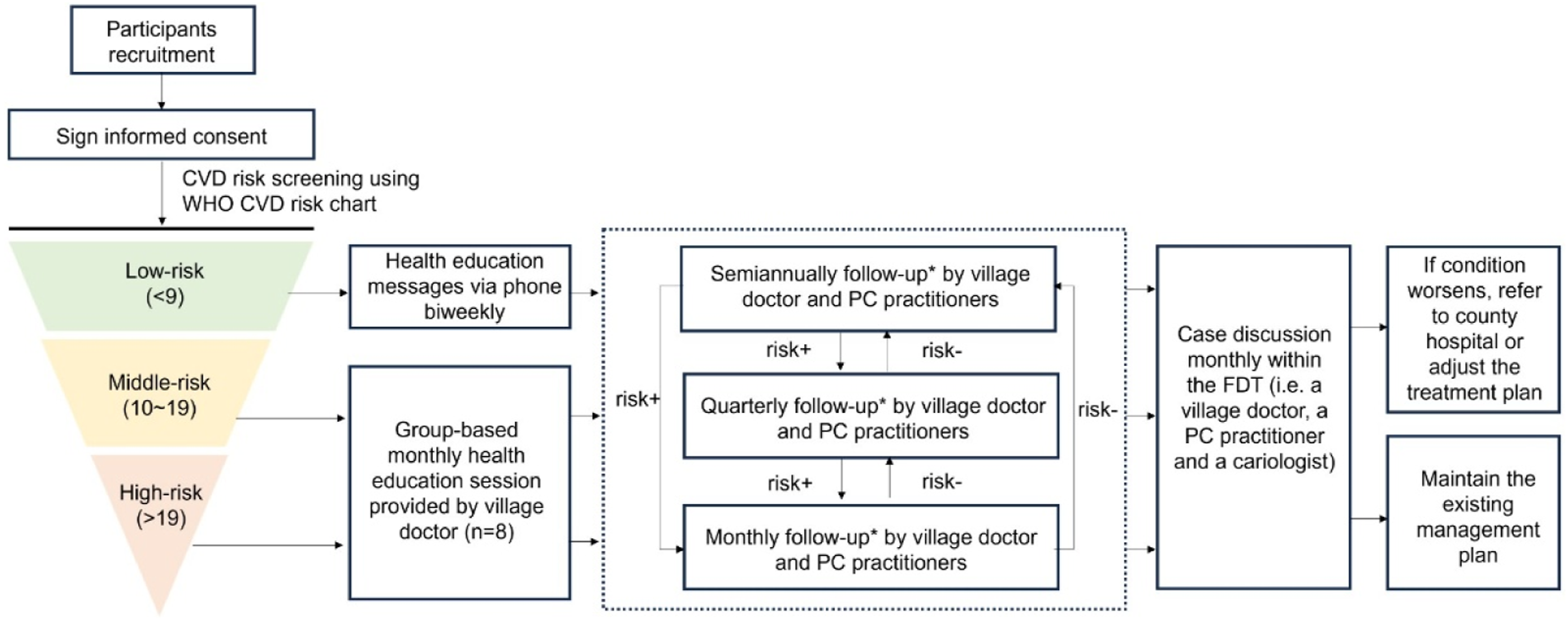
The team-based blended payment is a financial incentive provided to the healthcare professionals in the intervention groups. Coordinated services, including risk follow-up and case discussion, will be provided with payment to the corresponding providers. Besides, FDT will also receive financial incentives calculated based on service quality and CVD-related health outcomes at the population level over the study period. The detailed indicators for calculating performance-based payment include both specific process elements (i.e., compliance with the pathway) and targeted outcomes of the intervention (i.e., CVD events).
Considering that the C-RISIMA intervention involves the integration of personnel and services at both primary care facilities and county hospitals, its implementation may be highly sensitive to local health system characteristics. Since 2016, China has been implementing a model of county-wide tight medical alliances (CTMA) to establish primary-care-centered integrated services (World Bank Group, 2019; Yip et al., 2019). CTMAs are typically led by a county-level hospital and involve all township health centers and village clinics in their catchment areas (Details in Figure 2). In a CTMA, the leading hospital supports these facilities to improve the primary care services delivered by the FDTs. Since CTMAs vary in the aspects and depth of integration between county-level hospitals and primary care facilities, clusters will be randomized at the FDT level within each CTMA. Structure of a Family Doctor Team (FDT) and a County-Wide Tight Medical Alliance (CTMA).
Data will be collected from 2,560 participants diagnosed with diabetes and/or hypertension across 64 FDT clusters affiliated with 3 CTMAs in two counties in China. Each FDT will recruit 40 participants. The 64 FDTs will be individually randomized to participate in the C-RISIMA intervention for 12 months (see Figure 3). Design of the C-RISIMA Trial.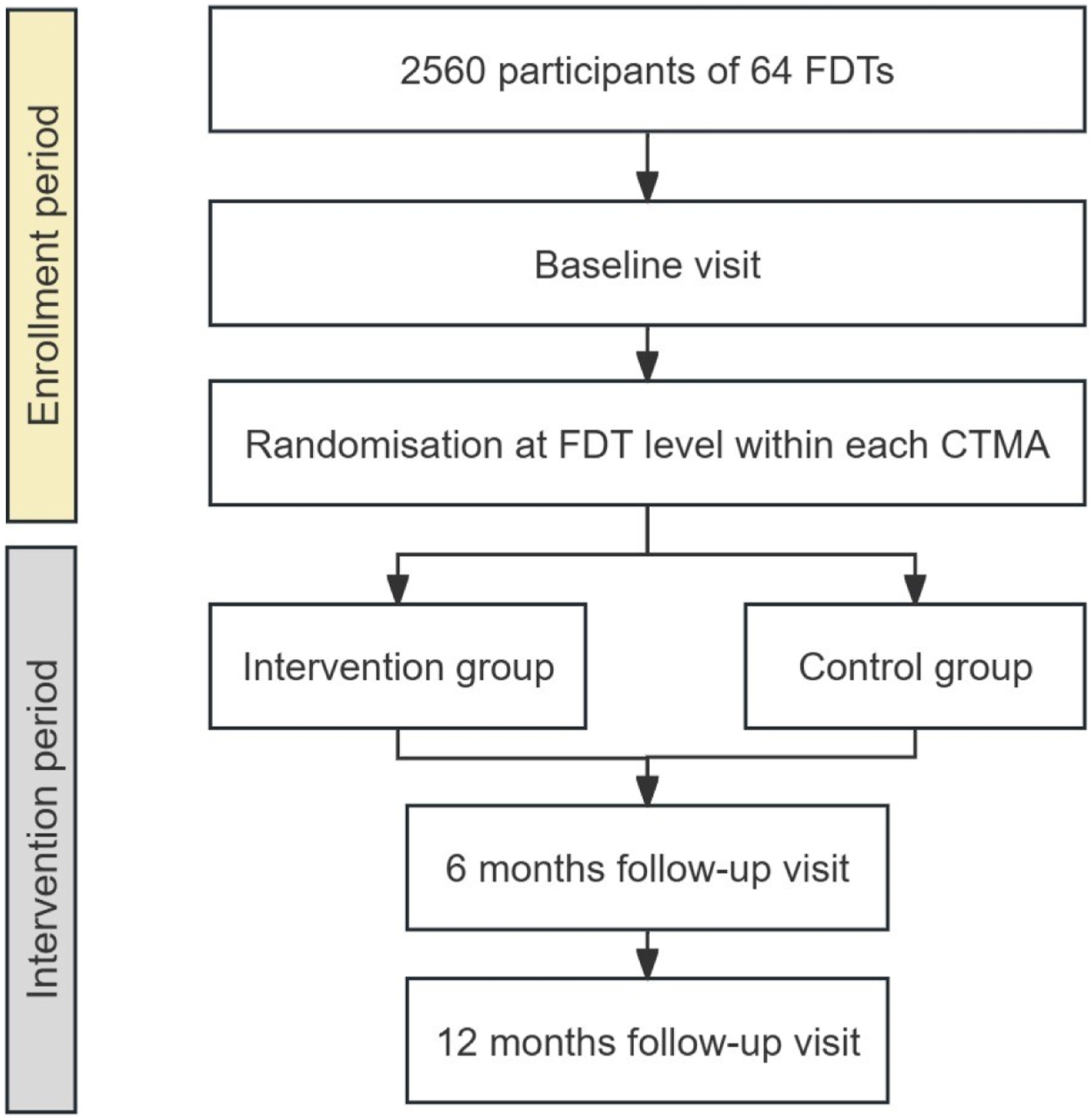
Process Evaluation
In light of the literature on realist evaluation, intervention outcomes of the C-RISIMA trial depend on the ‘implementation’ (e.g., how the intervention is delivered/received), the ‘mechanism’ (e.g., the extent to which health professionals change their practices and why), and the ‘context’ within which the intervention is delivered (e.g., CTMA) (Moore et al., 2015). Meanwhile, Normalization Process Theory (NPT), widely used to facilitate understanding the dynamics of the implementation process of complex interventions, is well-matched to our study in exploring how the new model is integrated within the existing practices (Murray et al., 2010). Therefore, the design of process evaluation is primarily informed by the key components (i.e., context, implementation, mechanism, and outcome) highlighted in the Medical Research Council (MRC) guidance for process evaluation of complex interventions (Skivington et al., 2021). The application of NPT will provide a deeper understanding of the contexts, mechanisms, and outcomes of the intervention, and how they interact (Dalkin et al., 2021). A grounded, translational framework was developed in our study, which combines the NPT and the Context-Mechanism-Outcome (CMO) framework of realist evaluation (May et al., 2016, 2022).
Aims and Objectives
This process evaluation aims to explore study how the C-RISIMA intervention is implemented and incorporated into the existing practices and how varied implementation shapes the outcome and sustainability of the intervention. Specific objectives are listed as follows: 1. To evaluate the implementation of the C-RISIMA intervention in terms of fidelity, dose, coverage, and adaptation. 2. To establish the local health system context for all clusters from the beginning to the end of the trial period to identify facilitators and barriers that may affect the implementation of the intervention. 3. To explore the mechanisms that motivate and shape the incorporation of the intervention into healthcare professionals’ daily work. 4. To explore how potentially varied implementation of the intervention influenced the patients’ behaviors and health outcomes of interest, as well as the sustainability of the intervention.
Methods
According to the MRC guidance on process evaluation, a clear description of the intervention and its causal assumptions, represented as a logic model, should be developed to identify appropriate areas for investigation. A logic model for C-RISIMA intervention was developed to assist in the evaluation of intervention components (details in Figure 4). The Logic Model of C-RISIMA.
Figure 5 illustrates the various elements of the process evaluation and the incorporation of NPT constructs into the CMO framework. Process Evaluation Framework.
Sampling
Quantitative Study
All FDTs which were randomized to the intervention group were included in the study (n = 32/64). All 3 CTMAs and the 20 township health centers affiliated with the intervention FDTs will be included as the sample for establishing the local healthcare contexts.
Qualitative Study
Three intervention FDTs from each CTMA will be sampled as cases FDTs (n = 9) with the consideration of heterogeneity in the intervention implementation (details in Appendix 1). The judgment of case selection will be made on the information collected from the baseline informal interview (with senior staff from the local health bureau and CTMAs) and observations during the trial training process.
At least 2 administrators from each CTMA (6 respondents in total) and 9 administrators from township health centers affiliated with the case study FDTs will be recruited for qualitative research. The interviewees will be selected based on their familiarity with chronic disease management policies and healthcare services. The sample will be purposively chosen to ensure heterogeneity, considering factors such as age, gender, professional background, and seniority. Health professionals (n = 27) from these case study FDTs will also be purposively sampled to capture a diverse range of roles and experiences for semi-structured interviews. Additionally, one patient from each of the low-risk, middle-risk, and high-risk tertiles within each case study FDT will be purposively selected for interviews (n = 27), with attention to heterogeneity in terms of CVD risk levels. Furthermore, at least two additional patients from each risk tertile in the same FDTs will be purposively sampled for focus group discussions (n = 54).
When healthcare professionals are invited to participate in the main trial, they will be informed that a process evaluation will be conducted, with participation in this evaluation being optional. Administrators invited to take part in interviews will receive additional written information, and their informed consent will be secured. Patients who agree to participate in the trial will be informed that they may be invited to join an interview or focus group as part of the process evaluation. If selected, they will be provided with detailed information, and their consent will be obtained prior to their involvement in the evaluation.
Data Collection
Quantitative Data
The data of implementation will be collected from all 32 intervention FDTs to allow assessment of the fidelity, dose, reach, and adaptation of the intervention delivered in each FDT. Details of the quantitative data being collected on the delivery of the intervention can be found in Appendix 2. • Fidelity will be evaluated by assessing whether the C-RISIMA package is delivered as planned, in terms of frequency or content. • Dose will be measured by the total amount of intervention delivered, such as the total hours of health education provided. • Reach will be measured as the proportion of patients who are covered by the intervention. • Adaptation will be assessed by identifying any modifications made to the intervention during the trial period.
The collection of quantitative data for contextual factors will be conducted through questionnaires at the end of the trial and secondary data platforms including electronic health records and implementation forms during the trial process. The study will also collect the general characteristics of CTMAs related to integrated chronic care through an institutional survey. The questionnaire is informed by the World Health Organization’s six building blocks of the health system, which offer a comprehensive framework for assessing key areas that influence health system performance (World Health Organization, 2010). Data collection for health outcomes of interest (i.e., 10-year CVD risk scores) is illustrated in the main trial protocol, so the details will not be presented in this paper.
Qualitative Data
Context
Semi-structured interviews with administrators will be conducted at the baseline, midpoint, and endpoint of the trial to explain how contextual factors shape and affect the way intervention is implemented throughout the course of the investigation. The interview guide was developed with the experience within the research team and insights from the four NPT constructs for implementation contexts (May et al., 2016, 2022), which are strategic intentions (‘how do contexts shape the planning of intervention?’), adaptive execution (‘how do contexts affect the ways in which healthcare professionals identify workarounds to make an intervention and its components feasible in practice?’), negotiating capacity (‘how do contexts affect the integration of intervention into the existing working flow?’), and reframing organizational logics (‘how do social structural factors shape the implementation environment?’). It is possible to adjust this guide based on findings that emerged from previous interviews. The interviews will not be limited to the guide but can be flexibly adjusted based on the interviewee’s feedback and interactions. It may last for approximately 1 hour and will be recorded using an encrypted digital audio recorder. The audio recordings will be transcribed verbatim and anonymized before analysis.
Mechanism
The interview guidance for the mechanism was prepared in advance drawing on insights from literature and normalization process theory (Hooker et al., 2015; Murray et al., 2010). The four NPT constructs of mechanism will serve as a scaffolding concept to guide the development of the interview guide, which are coherence (‘how is the intervention made coherent with stakeholders?’), cognitive participation (‘how are stakeholders engaged in the intervention?’), collective action (‘how is the intervention enacted in practice?’), and reflexive monitoring (‘how the intervention is understood and assessed by stakeholders?’). The guidance might need to be updated if findings from previous interviews reveal relevant information not covered in the original outline. The questions will not be limited to the guide but can be flexibly adjusted based on the interviewee’s feedback and interactions. During the interviews, the interviewer will use active listening and open-ended questions to encourage stakeholders to share their experiences, opinions, and reflections freely. Semi-structured interviews with healthcare professionals will be held after 6 months and 12 months from the start of the trial.
Outcome
Semi-structured interviews will be conducted to explain the practical performance of the intervention implemented, which makes how patients and providers change as implementation processes proceed become visible. Based on NPT, the development process of the topic guide will be partially informed by the following four constructs for implementation outcomes (May et al., 2022), which are intervention performance (‘what behaviors in patients have changed as the result of interventions?’), relational restructuring (‘how have interventions changed the way different providers are organized to each other and relate to within the FDTs?’), normative restructuring (‘how have interventions changed the resources or norms that govern providers’ action?’), and sustainment (‘how have intervention components been incorporated into providers’ behaviors?’). Serial interviews for the first 3 constructs will be conducted at 6 months and 12 months after the start of this trial. For ‘sustainment’ measurement, the interview will be conducted at 6 months after the end of the trial. To further understand patients’ perceptions of intervention, focus groups will be moderated at the end of the trial by experienced researchers to compare a sample of patients’ experiences of C-RISIMA care pathway with their experiences of usual care, and how the components of the care pathway have been operationalized from the perspectives of patients. The data collected from focus groups will be used as a supplement material for the evaluation of ‘intervention performance’, as focus groups are likely to generate richer data through diverse discussions. Researchers conducting interviews and focus groups will be blinded to the trial results. All the qualitative investigations will be recorded using an encrypted digital audio recorder, transcribed verbatim, and anonymized.
As part of the data collection process, the NPT constructs will be used as a conceptual inspiration when designing and developing the initial phase interview questions, rather than as a restricting framework to control the scope of the study. It is intended that the interview guide will be open-ended in order to capture emerging themes.
Analysis
Quantitative Analysis
The quantitative data collected will be used for descriptive statistical analysis to provide information about the overall extent of fidelity, dose, reach, and adaptation across intervention FDTs. All intervention FDTs will be further divided into two groups (i.e., “low delivery group” and “high delivery group”) based on the results of fidelity. Contextual factors collected through the questionnaires will be compared between these two groups using Mann-Whitney U tests for continuous data and chi-squared tests for categorical data, so as health outcomes of interest, including 10-year CVD risk, systolic and diastolic blood pressure, etc.
A multilevel logistic regression analysis will be conducted to assess the association between varied implementation and health outcomes. Stata version 18 will be used for the analysis.
Qualitative Analysis
The qualitative data collected from interviews will be read line by line and analyzed using thematic analysis after each collection time point. The data coding and analysis process will be conducted independently by three qualitative researchers, who are blind to the trial results, before discussion for a consensus on themes. Starting with an open coding stage, relevant meanings and phrases in raw data will be inductively identified as codes without applying any predefined frameworks. The relevant codes with similar patterns or concepts will then be categorized to generate themes and sub-themes. Before trial results are released, the themes will be discussed and refined within the qualitative research team to generate themes that reflect a more nuanced explanation. Once the themes are established, we will compare them with those in the pre-developed coding manual based on the NPT constructs for context, implementation, and outcome, to identify areas of alignment and divergence. NVivo V.10 software will be used.
Triangulation
A triangulation matrix will be used to integrate findings from both the quantitative and qualitative analyses. Summary statements will be developed for each key finding (i.e., findings about health outcome of interest, and acceptability of intervention from stakeholders) from both perspectives, and then the pairwise comparison will be conducted to assess whether the findings are in agreement, partially agreement (where they complement each other), dissonance (where they conflict), or silence (where only one data source contributes) (Tonkin-Crine et al., 2016). The triangulation will enhance the credibility and robustness of the findings by providing a more comprehensive understanding of the implementation process.
Rigor in the Study
This study employs a multifaceted strategy to ensure rigor in qualitative research, drawing on the established trustworthiness criteria, i.e., credibility, transferability, dependability, and confirmability (Guba & Lincoln, 1989; Maher et al., 2018). First, we seek to ensure credibility through prolonged engagement, whereby researchers actively interact with participants and observe the research setting through regular visits and conversation with the participants throughout the data collection and analysis. External experts will also be consulted as we collect and interpret data. We will also check within and between qualitative and quantitative data from all sources and stakeholder perspectives to cross-validate information collected. Moreover, careful attention will be devoted to achieving data saturation throughout the study to ensure that our research is grounded in sufficient evidence. Second, transferability is supported by the comprehensive collection of details about context to allow “thick description” that capture the research context, using the World Health Organization’s categorization of the six building blocks of the health system. This comprehensive documentation enables the readers to assess the relevance and applicability of the study’s findings in different settings. Third, we seek to maintain dependability by developing a comprehensive audit trail that records each stage of the research process—from data collection procedures and coding frameworks to analytic decisions and reflexive notes—thus ensuring transparency and facilitating replication. Finally, confirmability is strengthened through quarterly reflective meetings that involve both the research team and external experts, providing a structured forum for acknowledging predispositions of researchers, critically discussing interpretations and refining analytic approaches. Independent dual coding is also implemented to incorporate diverse perspectives and reduce individual bias.
Ethics
All procedures contributing to this work adhere to the ethical standards of the relevant national and institutional committees on human experimentation and conform to the Declaration of Helsinki, as revised in 2008. Data will be managed in compliance with the General Data Protection Regulation (GDPR). For interviews with various stakeholders and focus groups with patients, we will (i) maintain the confidentiality of both participants and locations, ensuring that no identifiable information appears in any reports or publications, and (ii) obtain written informed consent from all patients, healthcare professionals, and administrators that participate in this study.
Discussion
An adaptive implementation strategy is critical in rolling out complex CVD prevention by ensuring the uptake of promising evidence into practice, especially for complex interventions. This process evaluation will facilitate the understanding of the acceptability of a complex intervention for CVD primary prevention, how it was taken up, and the extent to which the findings from this trial can be transferred and applied in other settings.
A strength of our process evaluation lies in the combination of the realist evaluation and the NPT for the framework design. The utilization of NPT as a substantive theory within the CMO configuration enhances the explanation power of the potential mechanisms that facilitate or hinder the embedding of the complex intervention into routine practice, and effects produced by contexts. Further, this study provides multiple perspectives (provider, patients, healthcare system) for the understanding of implementation, which may be of interest to policymakers. Moreover, the credibility and robustness of study findings are further enhanced with the utilization of triangulation. However, since the study is limited to China, findings may be only applied partially to similar contexts.
Supplemental Material
Supplemental Material - A Primary-Care-Centered Integrated Care Intervention for Stratified Cardiovascular Risk Control in China: A Protocol for a Process Evaluation
Supplemental Material for A Primary-Care-Centered Integrated Care Intervention for Stratified Cardiovascular Risk Control in China: A Protocol for a Process Evaluation by Weijia Lu, Ziming Wang, Xinxin Xia, Yuan-Tao Huang, Yulin Kuang, Qihang Zhao, and Jin Xu in International Journal of Qualitative Methods
Footnotes
Acknowledgements
The authors would like to thank the Shaxian District General Hospital and Luzhai Health Bureau for hosting this research, in particular Xiaoying Wan, Kunxu Zhang, Xianjin Xie, Xiaohong Xie, Pengfei Liu and Ying Mei. The authors thank the Cross-Straits Medicine Exchange Association for their help and support, in particular Jian Jiang, and Xubing Jiang. They would also like to thank Shasha Yuan, Yuhui Shi, Gang Wu, Qiaoyan Xu, Xiaolin Wei, Junfeng Zhang, Yanchun Zhang and Xuefeng Zhong for their advice on the design of the trial, and Peter Bower for critical comments and edits on an earlier draft of the manuscript. Finally, they would like to thank the patients, and family doctor teams for their feedback in developing the intervention.
Ethical Statement
Author Contributions
Conceptualization: JX, WL; Literature review: WL, JX, ZW, XX, YH, YK; Methodology: WL, JX, YK, YH, QZ; Visualization: WL, ZW, XX, YH; Drafting of the manuscript: WL, JX, ZW, XX; Supervision: JX; Revision of the manuscript: JX, WL, ZW, XX, YH; Final review and approval: All authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Grant No. 71804004 sponsored by the National Natural Science Foundation of China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
