Abstract
Many groups (healthcare professionals, lawyers, philosophers, non-governmental organisations, bioethics committees, journalists, religious groups, etc.) participate in the bioethical debate about medical assistance in dying (MAiD). Bioethics literature on the topic involve different approaches (analytic, empirical, policy oriented, activist) and various normative perspectives (discourses based on deontological premises as the sanctity of life and human dignity, or on utilitarianism, libertarianism, etc.), some of which are at times irreconcilable. Regarding empirical studies, some voices (e.g., healthcare professionals) have been widely considered but the voice of people who request MAiD has been neglected. Understanding the personal and medical circumstances that lead to MAiD, which can only be achieved by listening to the phenomenological discourse of those involved, is key. This study aims to provide knowledge from the testimonies and experiences of patients who have initiated a MAiD request. We believe this research protocol can increase our understanding of a social and academic controversy that lacks important information to be complete. By doing so, this type of research could inform and improve end-of-life public policies, and particularly the health care of individuals who request a MAiD. We propose a qualitative phenomenological study using semi-structured interviews of people in the process of requesting MAiD, as outlined in the Ley Orgánica 3/2021, de regulación de la eutanasia (LORE), the Spanish Law on the Regulation of Euthanasia. The study is conducted nationally using a convenience sampling. The number of interviews is determined sequentially and cumulatively, depending on the richness of the narratives and the saturation of the information that has been collected. For the purposes of analysis, the interviews are transcribed verbatim and pseudonymised afterwards. Data analysis is conducted at the same time as data collection. The proposed study has received a favourable report from the Coordinating Committee on Biomedical Research Ethics of Andalusia (CCEIBA).
Keywords
Research context and background
The Spanish Ley Orgánica 3/2021, de regulación de la eutanasia (Law on the Regulation of Euthanasia), often abbreviated to LORE, allows people with a serious, chronic and incapacitating condition or with a serious and incurable disease to request Medical Assistance in Dying (MAiD): either euthanasia or assisted suicide. This new law was preceded by previous legislation on end-of-life decision-making allowing withholding and withdrawal of life-sustained treatments, terminal sedation, and the use of advance directives. Despite this regulation aimed at improving the conditions in which patients die, and involving their preferences as far as possible in the decisions made at the end of their lives (Jiménez García et al., 2014; Arimany-Manso & Martin-Fumadó, 2021), some high-profile cases in the media of suffering people requesting greater involvement of others in helping them to die suggested that the preceding regulation had limitations and gave rise to social debate in Spain in the 1990s and beyond. The LORE offers people with chronic or degenerative pathologies or with a prognosis of prolonged suffering greater control of the evolution of their illness and the timing and circumstances of their death, with the recognition of what has been called a right to exit (Aguilar et al., 2016). This new right also raises concerns among those who fear malpractice and abuse (Vermeer, 2021; Zurriaráin, 2019; Engelhart, 2021).
Theoretical background and current situation
Compared to other European legislation on MAiD, the Spanish LORE is considered particularly protective, as it defines the eligibility requirements restrictively and requires prior approval from a Guarantee and Evaluation Commission (a collegiate advisory body of a multidisciplinary, decision-making and supervisory nature, which shall act with functional autonomy and independence in the exercise of its functions: verify whether the necessary requirements are met, resolve complaints made by persons whose MAiD requests have been rejected by the responsible doctor, or resolve applications pending verification when there is a disparity of criteria between the members designated to grant or deny the MAiD, among others) before the provision can be carried out (not afterwards, as happens, e.g., in the Netherlands, Belgium and Luxembourg) (Velasco Sanz et al., 2021). Despite this, concerns have been raised that requests for MAiD may be motivated by inappropriate reasons or may result from personal circumstances and avoidable suffering, such as the lack of adequate care or a support environment (Downie & Schuklenk, 2021). According to some critical voices, a lack of assistance in situations of dependence and deficiencies in palliative care (Batiz Cantera, 2021; Dopchie, 2021; Raus et al., 2021) should invalidate any request for assistance in dying. The outlined concerns raise a number of empirical questions. Is treatable pain and suffering part of the motivation of those requesting MAiD? Are loneliness, depression or environmental pressures involved in the motivation leading to a MAiD request? Have those requesting the provision had access to palliative care? Does palliative care mitigate or eliminate the request for MAiD? What impact does end-of-life care have on requests for assistance in dying? What doubts, hopes and fears do people express in the process of a request for MAiD?
Although it can be suggested that the mere existence of a recognized right to MAiD might be reassuring as an option even if it is never used, some others believe that it might constitute a form of pressure for some dependent people, who may find in the provision a way to stop being a burden on their family or caregivers (Downie & Schuklenk, 2021; Ortega Lozano et al., 2022; Zurriaráin, 2019). What impact has the recent legalisation of euthanasia had on the well-being of seriously ill patients and on their relationships with their families and/or caregivers? Does the discourse of those applying for MAiD suggest the presence of subtle, or not so subtle, pressures in the application processes?
If the exact circumstances of those who request assistance in dying, as well as the profound reasons that motivate those requests, are ignored, then the reliability of many of the statements and prognoses that are frequently expressed in debates on the drafting and the application of the LORE, and on MAiD in general, must be called into question.
Qualitative methods, particularly interviews and focus groups, and narrative approaches are increasingly seen as valid for, and having an impact on, bioethics research. Their use is becoming more widespread. Narrative medicine reveals that qualitative research in bioethics not only involves working at the intersection of different disciplines (ethics/philosophy, social sciences, medicine), but also between different poles of a spectrum of scientific paradigms, namely positivist versus interpretivist traditions (Roest, 2021; Roest et al., 2021). Moreover, as emphasised by feminist bioethical theories, the comprehension of the experiences can only be achieved by listening to the phenomenological discourse of those involved (Tong, 1997; Mills & McConville, 2023; Scully, 2010; Sherwin, 1992) and policies can only be evaluated and adapted if all actors are part of the discussion (Tronto, 2013).
Justification for the research and how it can be applied
The purpose of this project is to identify and understand the circumstances and motives of MAiD requests and thus contribute to providing qualitative data on some of the main ongoing bioethical controversies related to the regulation of MAiD in Spain. The aim is to provide those responsible for public policies in this area, healthcare professionals, social workers, ethic committees, and so on, with rigorous and professional information offered by empirical bioethics, avoiding normative positions that are ideologically charged or merely speculative. Research on phenomena like MAiD, which involve health issues and life processes, requires an intersectional approach that incorporates a dialogue between contextual elements and the experience of individuals in end-of-life situations.
There are no previous similar studies either in Spain or internationally, making this study an innovative scientific proposal. The results, by including a central perspective that has been absent from end-of-life discussions for a long time, will enrich bioethical debates about MAiD and contribute to significantly improving public policies in this field of clinical practice. This study can also be considered a first test of the processes that have been helping people to die in Spain since the approval of the LORE.
General objective
To explore and analyse the circumstances of, and motivation for, requests for MAiD.
Specific objectives
• To describe the psychosocial context of people who request MAiD. • To explore the experiences, values and emotions present in requests for MAiD and in MAiD processes. • To identify and understand the contexts in which MAiD is requested. • To detect possible biopsychosocial profiles of those who request MAiD. • To make proposals for the improvement of MAiD processes.
No hypothesis is formulated as this is a qualitative phenomenological study that is not based on prior assumptions or judgments about the possible narratives of people who request MAiD.
Methodology
Design
A qualitative phenomenological study with semi-structured interviews of people in the process of requesting a MAiD.
Scope
The study is conducted nationally from November 2022 to November 2024 in Spain.
Participants in the study
Apart from keeping to the characteristics of people who can ask for MAiD established in the LORE, participants need to fit in the following inclusion/exclusion criteria: A. Inclusion • People that have begun the process of requesting MAiD (regardless of whether they decide to continue until the end of the process or not). • People of legal age who have the capacity to give informed consent. B. Exclusion • People whose physical, psychological, or social situation prevents them from participating in an interview. • People whose vulnerability may be worsened by participating in the study.
Sample and recruitment strategies
Convenience sampling is used, determined by the number of individuals requesting MAiD who agree to participate in the study. The number of interviews is determined sequentially and cumulatively, with an estimated minimum of six to ten participants and no pre-established maximum. The final total is determined by the richness of the narratives and the saturation of the information that has been collected. Individuals interested in participating in the research receive detailed information about the study. After this initial information phase, those who choose to participate can give their consent orally or in writing.
Four complementary methods are used to recruit participants: • Through the Spanish Right to Die with Dignity (DMD) organisation https://derechoamorir.org/. • Through Spanish associations of patients who may be eligible to request assistance in dying such as the Palliative Care Spanish Society (SECPAL), Cancer Care Foundation (CUDECA), Spanish Association of Amyotrophic Lateral Sclerosis (ADELA) and other organisations representing individuals with terminal diseases. • Via the INEDyTO website, with an open call for individuals who have initiated the request for MAiD to contact the research team, and for professionals who are in direct contact with MAiD cases and who want to pass the information on to their patients. In order to preserve the confidentiality of their personal data, the research team does not have access to the participant’s medical records, nor are they able to verify the existence of an MAiD request. In this case, screening is conducted through specific questions about the request process during the initial information meeting. • Through the Regional Guarantee and Evaluation Commissions, which are contacted individually through the Regional Health Councils. As mediators, they are asked to inform doctors in charge of MAiD processes about the existence of this research and about the opportunity for patients who have requested MAiD to participate in the study.
Contact procedure
All individuals who initiate the process of requesting MAiD are informed about the existence of the study by a designated person. In the case of the Guarantee and Evaluation Commissions, the responsible doctor (“médico responsable” in Spanish; defined by the law as the doctor who is in charge of coordinating all of the information and medical care for the patient during the MAiD request, and who is the main interlocutor in all matters relating to their attention and information, without prejudice to the duties of other professionals taking part in care) informs the patient. For collaborating associations, a designated person from the association is responsible for providing the information. In addition to a brief introduction to the study, the patient is given a form saying that they consent to being contacted. This consent form also provides the contact information of the research team (phone number and email) so that the patient can initiate contact if they wish. Whenever a patient signs the consent form, the designated person from the Guarantee and Evaluation Commission or the collaborating associations gets in touch with the research team. Within the research team, there is a person who is responsible for receiving the consent forms and contacting the patients.
Interview script and dimensions of the study
The in-depth interviews are based on a semi-structured script of questions that includes the following dimensions: (1) Reflections on one’s own death and desire to die; (2) Experiences of family and social support; (3) Experiences in healthcare; (4) Support needs in different areas, particularly in healthcare; (5) Motivations for the request; (6) Hopes and fears associated with the process; (7) Proposals for improvement.
Conducting the interviews
The interviews can be conducted in-person, by telephone or via video calls, using the format and in the location the interviewees prefer. Prior informed consent is requested for audio recording (see the Ethical Considerations section). If a participant asks for the interview not to be recorded, the benefits to the research of that participant’s testimony are considered to outweigh the potential bias or loss of literal information. In such cases, notes are taken during or after the interview. This limitation will be acknowledged in the results and discussion of the study. If a patient wishes to be accompanied during the interview, this is possible. The researchers who conduct the interviews have expertise in conducting semi-structured interviews on end-of-life topics and/or have clinical experience.
Transcript of the interviews
All the interviews are transcribed verbatim for analysis. The transcriptions undergo a process of pseudonymization to ensure that the interviewees cannot be identified.
Data analysis/information analysis
The analysis of the content is based on hermeneutic phenomenology (Mak & Elwyn, 2003; Pietkiewicz & Smith, 2014). The aim is to suspend preconceived judgments about what MAiD entails and learn from the main actors the reasons behind their request for it. To structure the analysis, the profiles of the participants are categorised using the following variables: “gender” (male, female, other); “age” (18 to 40, 41 to 64, 65 or older – following the literature on MAiD requests in other countries [Dees et al., 2013; Leboul et al., 2022]); “level of education” (a. primary education and first stage of secondary education; b. second stage of secondary education and non-higher post-secondary education; c. second cycle of higher education and doctoral degree); “euthanasia context”, according to the law (a. severe, chronic and debilitating condition; b. serious and incurable illness causing intolerable physical or psychological suffering); and “stage of the process” of the application. Additional socio-demographic information is considered to describe the heterogeneity of profiles and their influence on the narratives (e.g., cultural background, rural/urban environment, socioeconomic level).
The qualitative data is analysed and themes are defined. The data is coded into thematic trees. NVIVO software is used to support the analysis by coding and categorising the information. After the creation of categories, the main analysis ise carried out by one of the researchers, triangulating with the rest of the team to avoid potential biases, enrich the analysis, compare findings and work on the most significant thematic areas. Data analysis is conducted at the same time as the data collection. This makes identifying new criteria and emerging dimensions for exploration and analysis possible. The sample is built up until theoretical saturation is achieved. Based on similar studies (Leboul et al., 2022), it is estimated that this point could be reached with 15 interviews. After this review, the results will be prepared.
Ethical considerations
A reflective ethics approach is adopted, with ethical considerations being applied in all phases of the study in the following aspects: 1. Comprehensive information is provided to participants about the objectives and methodology of the study, including giving them the opportunity to ask questions, not to answer some of them if they do not feel comfortable with it, end its participation at any point and without any consequence on their MAiD request or their healthcare assistance. 2. Verbal and/or written informed consent process. 3. Data collection and processing of information is conducted within a strict framework of confidentiality and in accordance with the Ley Orgánica 3/2018, de 5 de diciembre, de Protección de Datos Personales y garantía de los derechos digitales (Organic Law on the Protection of Personal Data and Guarantee of Digital Rights). The personal information provided by each participant is pseudonymized and aggregated. A member of the research team is responsible for ensuring the pseudonymity and confidentiality of the data. The informed consent form provides information and contact details for the Data Protection Entity and the Data Protection Officer responsible. 4. Attention is given to the potential emotional impact of the interview on the person interviewed (when conducting the interview and by providing information on support resources). 5. The protocol has been reviewed by the accredited research ethics committee. 6. The study does not involve physical, social, or legal risks for identifiable participants. However, participation may have some emotional impact, particularly for individuals in potentially vulnerable situations. For this reason, it is essential that the recruitment process ensures participants’ freedom, and that they take the initiative to contact the research team or explicitly authorise the team to contact them. To facilitate free and non-coercive participation, the recruitment process includes two steps: authorization to be contacted and consent to participate. 7. Experience from previous studies (Jensen, 2011) shows that these interventions sometimes generate incidental positive or beneficial effects. However, the objective of the study is exclusively cognitive and not aimed at seeking beneficence; this information will be clearly stated in the consent document, and the participant’s understanding will be confirmed by the researcher to exclude any form of therapeutic misconception.
Some members of the research team have experience in bioethical analysis and some of them are members of ethics committees in institutions, healthcare centres and universities.
Impact of the expected results
• Re-evaluation of MAiD protocols and procedures, taking into consideration the testimonies of those who have requested MAiD. • Open debates with professionals aimed at transferring and disseminating the results of the patients’ experiences. • Transfer an understanding of the complexity of MAiD experiences to society, based on information from the testimonies of those requesting MAiD. • Establish future lines of research and generate hypotheses susceptible to qualitative and quantitative verification.
To provide feedback to the participating groups, a report on the motivations behind the euthanasia requests will be prepared and sent to the Guarantee and Evaluation Commissions and to Right to Die with Dignity (DMD) organisation and other participating associations.
Limitations of the study and contingency plans
Due to the limited time since the implementation of the LORE, as well as the characteristics of individuals in the process of requesting assistance in dying (vulnerability, physical/psychological suffering, lack of availability, lack of visibility, etc.), there may be difficulties in contacting individuals willing to participate in the qualitative interview process or in doing the interview before there is a declining in their health condition. However, alternative sampling strategies can help overcome these potential difficulties.
The first data on the number of assisted dying requests and procedures in some Spanish autonomous communities since the enactment of the law have recently been published. The records show that over 170 euthanasia procedures were performed in the first year, with significant disparity among the autonomous communities (Ministerio de Sanidad, 2021). This variability may also be due to information collection and registration systems. Despite efforts to systematise recruitment through the combined strategy, it is highly probable that a considerable number of potential participants are lost due to the perception of this study as an additional or unnecessary burden in patients’ already complex health situation. However, the minimum number of participants required to make this study viable is relatively low, and it is anticipated that the richness of the information obtained compensates for certain limitations in the quantity of data. As a methodologically and conceptually pioneering study, even with a small number of participants, the results provide necessary and important information. It should also be noted that some potential participants may express particular interest in participating in this study.
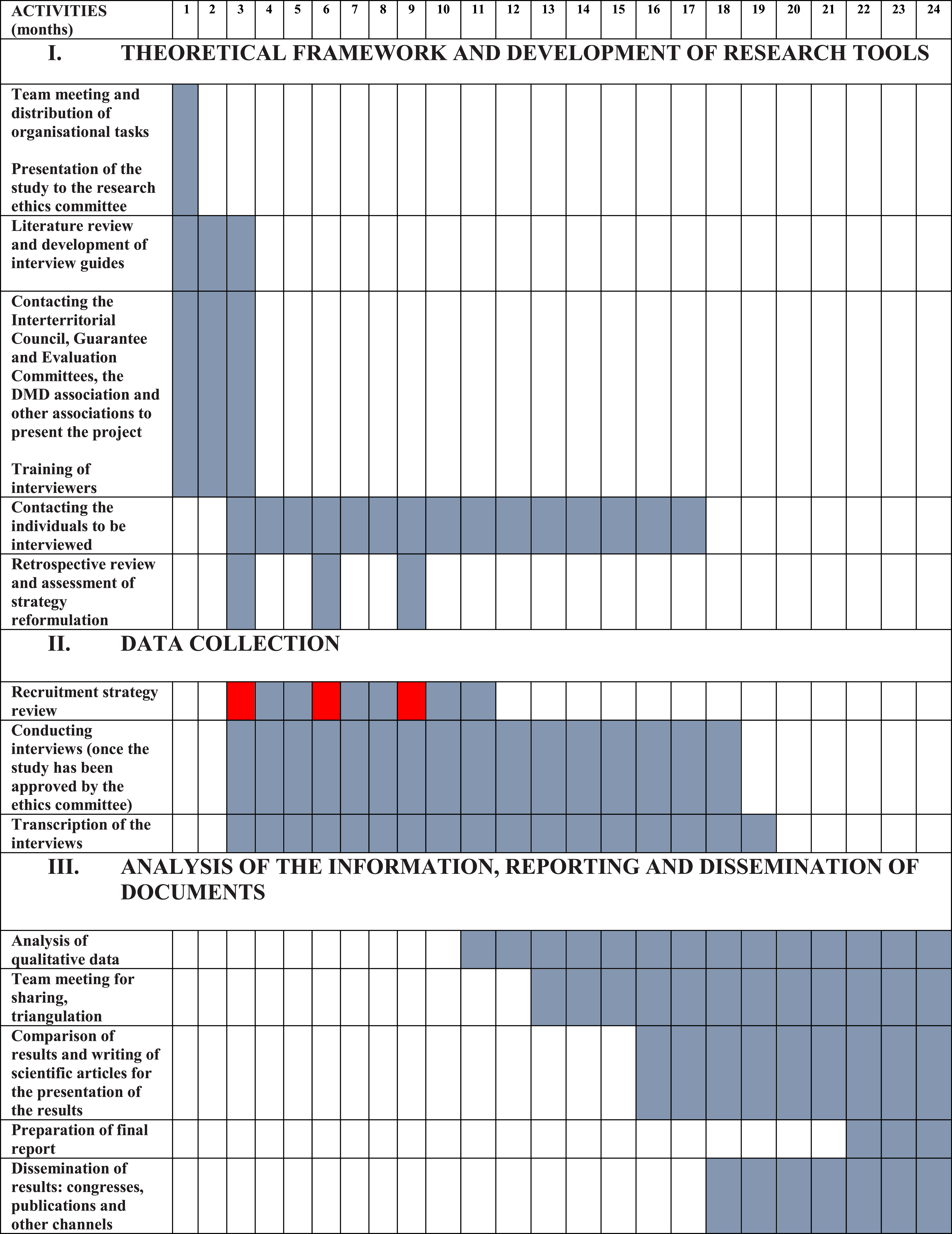
Three retrospective review points (at three, six and nine months) have been defined during the study period to assess the opportunity to reformulate the recruitment strategy (see Research schedule/work plan).
In the unlikely event of exhausting all recruitment efforts and not having any candidates by the ninth month of the project’s development, the recruitment strategy would be reformulated, attempting to gather the experiences of patients who have requested MAiD indirectly, from professionals (consulting doctors, doctors in charge of cases) who have been involved in providing such assistance.
Research schedule/work plan
Task Distribution Over the Study Duration.

The study received a favourable report from the Coordinating Committee on Biomedical Research Ethics of Andalusia (CCEIBA, Oct 2022).
Footnotes
Acknowledgements
We appreciate the assistance of Barnaby Griffiths with the language revision, as well as the support of the rest of the research team of INEDYTO Project
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been developed in the framework of the following research projects: INEDYTO (code PID2020-118729RB-I00), funded by the Spanish Ministry of Science and Innovation and «Narrativas de pacientes que solicitan la ayuda para morir» (code BEC-2022-019), funded by Fundació Grífols de Bioètica. Iris Parra Jounou thanks the funding of the Spanish Research Agency (code PID2019-105422GB-I00).