Abstract
Introduction:
Structured physical activity may improve physical, cognitive, and social functionality, as well as overall quality of life of people with high-grade glioma. Yet, engaging them in such activities remains highly challenging due to their unique disease burden and perceived “loss of control.” A greater understanding of patients’ interests, preferences, behavioral motivations, and perceived barriers around physical activity is needed to design tailored and patient-led physical activity interventions. This protocol describes the method and ethical considerations of a cross-sectional mixed-methods study that has been developed to collect such information from glioma patients and their carers.
Methods and analysis:
The International Physical Activity Questionnaire (IPAQ 7, short form) and semi-structured interviews will be used for data collection. IPAQ scoring protocol will be followed to examine objective data and Clarke and Braun’s thematic approach will be used to analyze the interview transcripts. Ethics: Ethical approval was obtained from Cardiff University’s Research Ethics Committee and from the regional NHS Research Ethics Committee. The main ethical concerns are to maintain patients’ safety and comfort and ensure that their consent remains informed and valid throughout the recruitment, data collection, management, and dissemination stage. Risks associated with their physical condition, emotional distress, and time commitment should be continuously assessed and necessary actions should be taken accordingly.
Conclusion:
The protocol will work as a step by step guide for future researchers for developing similar qualitative research and obtaining ethical approval when involving vulnerable individuals like high-grade glioma patients in studies.
Background
Primary brain tumors account for an estimated 2.3% of malignancies worldwide (Ferlay et al., 2019). More than 5,000 people are diagnosed with a primary malignant brain tumor each year in the UK (Cancer Research UK, 2015) and high-grade gliomas (HGG) are the most frequently diagnosed (Giammalva et al., 2018). Patients with these rapidly progressive tumors have a median survival of 12 to 15 months (Stupp et al., 2010) with a 5-year survival rate of only 10% (Tran & Rosenthal, 2010).
From an early stage in their illness, HGG patients tend to experience severe impairments in physical, neurocognitive (e.g. dysphasia, decrease in mental capacity), neurological (e.g. epileptic seizures), and social functioning (Long et al., 2016; Moore et al., 2013). Challenging treatment regimens often exacerbate these effects (Aziz & Rowland, 2003; Long et al., 2016). Moreover, along an unpredictable disease trajectory (Philip et al., 2013), patients can become increasingly dependent and isolated which, combined with their symptom burden, may significantly reduce their perceived quality of life (QoL) (Halkett et al., 2015; Lucas, 2010).
An important element of QoL is the ability to participate in daily social and self-care activities that generate feelings of inclusion, control, and empowerment. The World Health Organization’s (WHO) International Classification of Functioning, Disability, and Health (ICF) is a framework that defines how a person’s body structure, physical activity, and participation as a member of society—alongside relevant environmental factors—determine a person’s functionality and disability. This ICF framework highlights the participatory component when trying to estimate health and disability, an aspect which is often overlooked but which is likely to be of great personal importance to patients (Fox et al., 2015; World Health Organization (WHO), 2002). For example, just being able to move more independently, or to get up and walk for a short distance, may increase a patient’s chance of being able to join in with family and friends, which may, in turn, generate a feeling of inclusion along with an increased sense of self-efficacy. It is those elements of participation, inclusion, and a sense of control rather than simply the activity of walking that may matter most to patients.
Multidisciplinary rehabilitation, with its more holistic approach, indeed not only aims to maximize physical functioning per se but also to promote independence and to help people adapt to their condition by empowering patients through improved self-management (Bartolo et al., 2012; Dalzell et al., 2017; Dean, 2018). A recent systematic review led by Rick et al. (2017) showed that multidisciplinary oncological rehabilitation interventions with physical, vocational, and psycho-educational components led to a better quality of life and a higher rate of return to work than usual care. As a potential component of this, results from several randomized trials have provided evidence that structured physical activity has a positive effect on a broad range of debilitating symptoms associated with specific cancer diagnoses and subsequent treatments, culminating in clinically meaningful improvements in functional capacity and overall quality of life (Cormie et al., 2015; Dieli-Conwright & Orozco, 2015; Jones et al., 2007, 2010). Structured physical activity is defined as any kind of exercise that is planned and done regularly to improve health (Subramanian et al., 2015).
According to the European Association for Neuro-Oncology (EANO), strenuous exercise behavior is an independent prognostic factor for survival in malignant glioma patients in their guidelines for palliative care in adults with glioma (Pace et al., 2017; Ruden et al., 2011). Some recent studies have demonstrated that it is feasible to safely engage HGG patients in structured physical exercise programs, with beneficial effects on physical and emotional well-being, QoL, and cognitive function (Gehring et al., 2018; Heywood et al., 2017; Levin et al., 2015; Milbury et al., 2019; Sizoo et al., 2012). However, the evidence base is limited by small numbers of participants and there is a limited understanding of motivational factors for regular structured activity in this particular patient group.
Despite the promise of these recent findings, functional capacity and leisure-time physical activity levels of glioma patients tend to decline markedly over the disease and treatment trajectory (Culos-Reed et al., 2017; Piil et al., 2015). Due to their unique disease burden as well as their perceived “loss of control” and decreased sense of self-efficacy, engaging high-grade glioma patients in structured physical activity remains highly challenging (Acquaye et al., 2013; Cormie et al., 2015; Lowe et al., 2009; Wasley et al., 2018).
Therefore, greater understanding regarding patients’ interests, preferences, behavioral motivations, and perceived barriers around physical activity is needed to develop tailored, patient-led physical activity interventions for this patient group and encourage more physical activity in ways that patients and their families perceive as appropriate and do-able (Baima et al., 2017). This includes exploring the perceptions and beliefs patients hold with regard to the potential benefits of low-intensity physical activity and what they consider achievable with regard to increasing physical activity, not necessarily in the form of e.g. gym-type moderate to high-intensity physical exercising (Cormie et al., 2015) but rather in the form of regular, lower-intensity activities that can be incorporated as part of daily living at home. Since family carers play an important role as a potential source of motivation and encouragement (or discouragement) for cancer patients, it is also vital to explore their views on the potential benefits, barriers, and facilitators of physical activity for this patient group.
Few studies have explored the support needs (Langbecker & Yates, 2016) and exercise-related preferences of patients with primary brain tumors (Jones et al., 2007) as well as the potential barriers to, and motivators for, being physically active from the perspective of patients and carers (Fahrenholtz et al., 2019; Halkett et al., 2010). While describing support needs, Jones and Courneya (2002) advocated that without a structured intervention, most cancer patients would not get involved in exercise. Due to their complex and challenging symptom burden, this is particularly likely to be the case for HGG patients.
Important gaps exist in the understanding of key influences of exercise intent and behavior in HGG patients. Such knowledge is essential to be able to develop interventions that are doable and sustainable in the everyday life of these patients (Culos-Reed et al., 2017; Field et al., 2017; Jones et al., 2007; Lowe et al., 2010).
Incorporating such insights into the development of physical activity-based intervention studies is likely to improve recruitment, adherence, and outcomes for future studies that are of greatest relevance and importance to patients. Importantly, this will promote patient-centered care, a fundamental component of prudent approaches to value-based health care (Baumhauer & Bozic, 2016; Welsh Government, 2016). The main considerations when attempting to involve HGG patients in research on their personal experiences with remaining physically active are summarized and depicted in Figure 1.
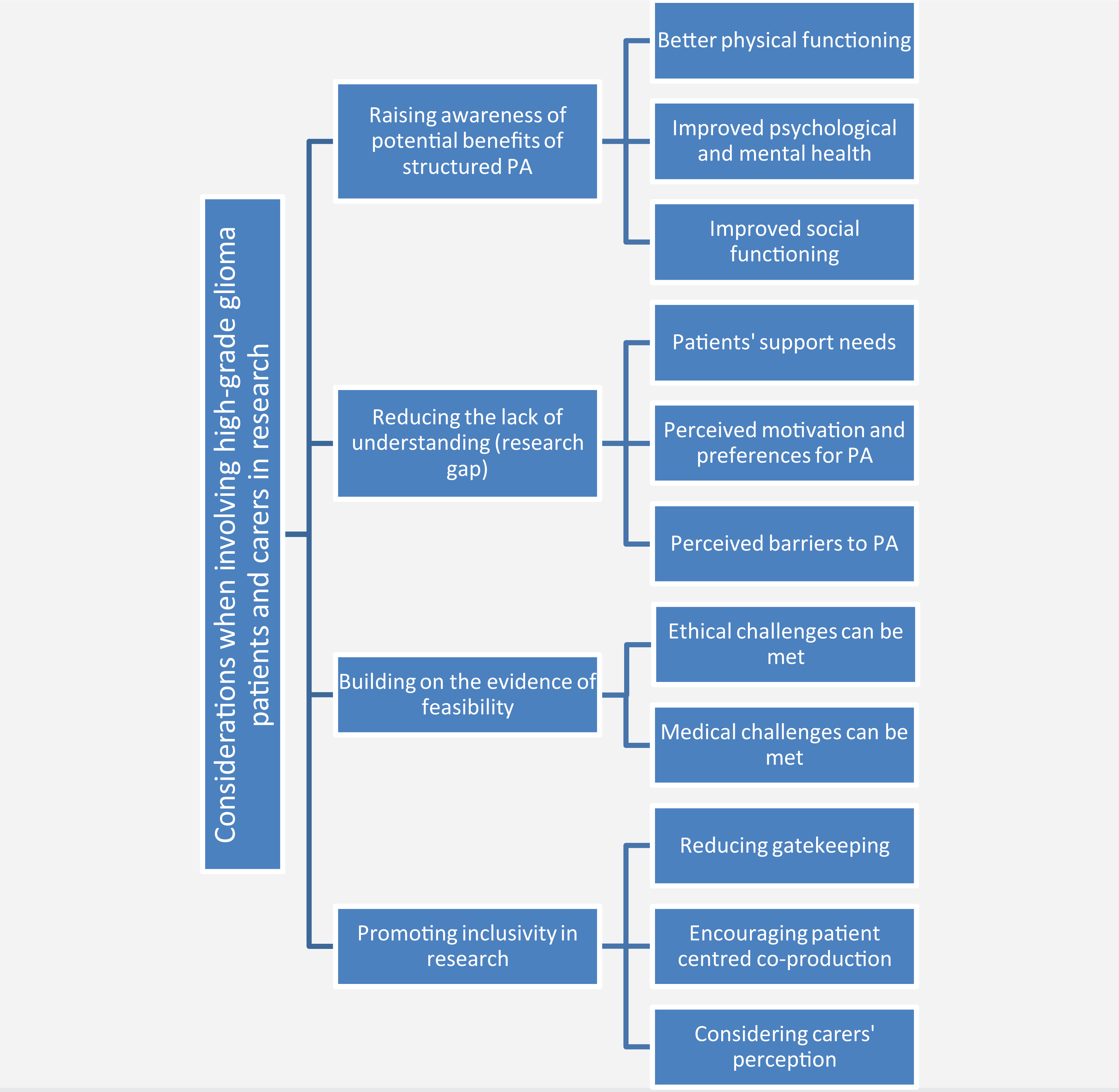
Main considerations when involving high-grade glioma patients in mixed-methods research on their physical activity (PA)-related personal experiences.
Study Objectives and Outcomes
The Structured Physical Activity in Brain Cancer (PhAB) study aims to identify HGG patients’ and carers’ preferences around physical activity as well as their perceived barriers to and facilitators of becoming more active in everyday life.
Objectives
Explore patients’ and carers’ perceptions around the benefits and risks of physical activity after a glioma diagnosis.
Identify what form and intensity of physical activity patients and carers think is appropriate to practice.
Investigate the perceived barriers to engaging in physical activities.
Explore the potential facilitators of being more active in daily life.
Identify patients’ preferred environment and mode of receiving physical activity interventions.
Outcomes
The primary outcome will be an improved understanding of patients’ and caregivers’ perceptions of the barriers and facilitators of physical activity.
The secondary outcomes will be i) updated knowledge on the current physical activity level (minutes spent in low or moderate activity/day) of glioma patients, and ii) gained insight on their preferences for individualized physical activity interventions.
Methodology and Methods
This is an exploratory study with a cross-sectional design, conducted by the Marie Curie Palliative Care Research Centre (MCPCRC) at Cardiff University, Cardiff, UK. A mixed-methods approach will be undertaken, combining semi-structured interviews with open-ended questions with a short physical activity questionnaire. Qualitative interviews are a widely used technique when the primary research aim is to explore the thoughts of the respondents on a topic in their own words. A validated assessment tool (International Physical Activity Questionnaire, short form; IPAQ 7; see Supplemental Appendix 1) will be used to assess patients’ self-reported current physical activity levels. In addition, a research diary for observational, methodological, theoretical, and analytical notes (Hughes, 2011) will be kept to further contextualize the interviews and support reflexivity as crucial part of the qualitative research process.
Participants will be recruited in collaboration with Velindre Cancer Centre (VCC) in Cardiff, one of the largest specialist cancer treatment centers in the UK. All researchers involved have attended Good Clinical Practice (GCP) training and those researchers conducting interviews have obtained an Enhanced Certificate from the Disclosure and Barring Service (DBS).
Sampling and Participants
Sampling will be purposive and participants will represent the following criteria to be included in the study: An adult (aged 18 years or older). A patient (or carer of a patient) with a histologically confirmed or radiologically and clinically suspected HGG (WHO grade III or IV glioma). Able to understand the purpose of the study and provide informed consent. Conversant in English.
Patients receiving treatment at a site other than the collaborating cancer center and occasional carers (carers who are not taking care of the patient regularly) will not be able to participate. Nominated carers will require the patient’s consent to take part in the study.
The target sample size for this explorative study will be 6–10 patients and 6–10 carers. Qualitative data plays an increasingly important role in healthcare research, reflecting the experience of patients and the vital role of carers. Importantly, it aims to give a perspective derived from the cohort rather than population level data. The target sample size is also in keeping with recommendations for thematic qualitative analysis (Pope & Mays, 2006; Vasileiou et al., 2018) and reflects sample sizes from other studies involving HGG patients (Catt et al., 2011; Collins et al., 2014; Fahrenholtz et al., 2019; Halkett et al., 2010; Lobb et al., 2011; Piil et al., 2018; Sterckx et al., 2015).
Recruitment
A consultant oncologist who leads a multidisciplinary clinic for glioma patients at the VCC and a research nurse will support recruitment at the VCC. The research nurse will assess the emotional, physical, and psychological suitability of patients to be approached about the study during outpatient appointments. Remote recruitment will be done for those who are receiving remote and virtual consultations during the COVID-19 pandemic.
Patients deemed physically, emotionally, and cognitively well enough by the clinical team will be approached about participating in the study. If they express an interest in taking part, the research nurse will provide patients, as well as their accompanying carers, with a Participant Information Sheet (PIS), containing detailed information about the study objectives and procedures, as well as the risks and benefits of taking part. They will also be given an informed consent form (ICF) and asked to consider the provided study information for a minimum of 24 hours before deciding on whether they would like to participate in an interview. The research nurse will then obtain verbal consent for the research team to contact the patient and/or carer within a week of their clinic appointment to enquire if they are indeed interested in participating and to answer any questions that might have arisen after reading the study information material. With those patients/carers who choose to take part in the study, arrangements for an interview will be made for a date and time that is convenient for them. Due to the ongoing Covid-19 pandemic, all interviews will be conducted remotely over the phone. Participants will be asked to email a scanned document or photo of their signed consent form back to the research team who will then password-protect this digital copy and store it separately from the anonymized interview and questionnaire data. Alternatively, participants will be able to request a prepaid self-addressed envelope from the research team to be sent to them to return their signed consent form in the post. If both options to obtain documented consent remotely are impractical for a participant, the interviewer will ask their permission to record their verbal consent over the phone before starting the interview. The interviewer will go through the consent form point by point and ask the participant to agree to each statement individually. In place of a signature, the interviewer will state that they are signing an electronic copy of the consent form on behalf of the participant. The consent-related audio recordings will be conducted and stored separately from the audio-recordings of the subsequent interviews. A Standard Operating Procedure (SOP) for Remote Data Collection and Consent, developed by the MCPCRC, will be followed throughout (see Supplemental Appendix 2 for a synopsis of this SOP).
The process map for recruitment and obtaining consent remotely is shown in Figure 2:

The process map for recruiting participants and obtaining consent remotely.
Data Collection
For patients, two data collection methods will be used: 1) a short questionnaire to gauge their current physical activity levels (IPAQ), followed by 2) a single in-depth interview. As the physical activity questionnaire is very short, the survey and the interview will be conducted over the phone on the same day to minimize the burden and time commitment for the patients. Administering the questionnaire before conducting the interview will reduce the self-reporting bias, potentially induced by the topics discussed during the interview. The physical activity examples included in the IPAQ questionnaire may also help patients to think about “physical activity” in broader terms rather than just gym-type exercising. For carers, only interviews will take place.
Physical activity questionnaire
Patients only will be asked to complete the short form of the International Physical Activity Questionnaire (IPAQ 7) that assesses their current physical activity level. The IPAQ 7 consists of 7 brief questions that ask participants on how many days in the past 7 days they have engaged in physical activity for at least 10 minutes as part of their everyday lives, categorized in the following way: vigorous activities (e.g. heavy lifting, digging, fast bicycling), moderate activities (e.g. carrying light loads, bicycling at a regular pace), walking (e.g. walking for recreation or exercise, walking to travel from place to place) and sitting. If appropriate, they will then be asked to estimate how many hours/minutes they spent on these activities.
Interview
All telephone interviews will be conducted using a semi-structured interview schedule. Two separate versions of this interview schedule have been developed for patients and carers, respectively (see Supplemental Appendix 3A and Appendix 3B). If possible, patients will be interviewed first and by themselves. However, where it is mutually agreed between the patient and their carer that the carer will remain during the patient interview, the patient will be interviewed in the carer’s presence (Morris, 2001). While it is likely to influence the patient in their responses and thus needs to be acknowledged during analysis, their presence might also be beneficial as carers might spontaneously cue, prompt, or fill in gaps during the phone interview, thus informing and enriching the collected interview data (Bjørnholt & Farstad, 2014; Halkett et al., 2010; Morris, 2001).
For all participants, some basic demographic information (age, when the patient’s glioma was diagnosed and, in the case of carers, their relationship to the patient) will be obtained at the beginning of each interview.
The domains covered in the interview schedules include patients’ and carers’ understanding of physical activity and what its benefits could be as well as their thoughts on what types of physical activity might be regularly achievable for them as part of their daily life. The interviews will also explore what participants perceive as barriers to becoming active, including any concerns they might have about making things worse, and what they think might encourage and help them to be active on a regular basis. Participants’ socioeconomic and environmental preferences and support needs for physical activity such as financial costs, who to exercise with, and where, will also be addressed.
Time requirement
Overall, participating in the study should take between 48–83 minutes in total for a patient (plus an additional 36–65 minutes for the interview with the carer), as detailed in Table 1 below:
Time Commitment Estimated for Participants.

Data analysis, reflexivity, and ensuring rigor
The IPAQ scoring protocol (IPAQ Research Committee, 2005) will be followed and MS Excel will be used to compile descriptive statistics for the quantitative data and to analyze the demographic characteristics of the participants (age, gender, duration of illness, current physical activity level).
The semi-structured interview recordings will be transcribed, fully anonymized, and uploaded into NVivo v.12.0 (QRS International Pty Ltd, 2018), a qualitative data management software. Clarke and Braun’s (2013) thematic analysis (TA) will be adopted to analyze the qualitative data and will comprise six steps: 1) familiarization with data; 2) coding; 3) searching for themes; 4) reviewing themes; 5) defining and naming themes; and 6) writing up. To ensure the rigor and consistency of the analysis, two researchers will apply variety of perspectives and use multimodality forms of interaction with the data while coding the data independently.
The Study Management Team (SMT) at the MCPCRC includes two senior members with established track records of conducting high-quality research in the relevant field who will supervise and support the researchers throughout the project. This will include regular opportunities for discussion, reflection, and updating on the wider relevance of the research topic and the theoretical framework the researchers are operating within. These discussions will allow continuous assessment of integrity and objectivity during study conduct. Following the first interview, the transcribed interview will be reviewed as part of that supervision—along with field notes from the research diary—to ensure compliance with the protocol and methodological rigor. An expert team member will also co-code 10% of the qualitative data to ensure rigor and to support the researchers in their reflections on their interpretations. Insightful interaction with and creative interpretation of the data combined with digital data management offer a valid and tested analysis method for qualitative data (Maher et al., 2018). Consolidated criteria for reporting qualitative research (COREQ) guidelines (Tong et al., 2007) will be followed during the write-up of the study findings to ensure comprehensive and transparent reporting.
The anonymized data will be presented in a narrative format with a thematic structure. Anonymized quotations will be used—with participants’ consent—to illustrate and support identified themes. The obtained IPAQ data for patients’ current physical activity levels will be used to inform and contextualize the analysis and interpretation of patients’ interview responses. Due to the small sample size, it will not be possible to conduct statistical comparisons but when analyzing the interview data, it will be helpful to interpret interview responses in the context of current and recent activity levels which may provide additional perspective on topic areas including motivation, confidence, and self-efficacy. When similar data from a separate study conducted by the same research team in Bangladesh becomes available, cross-cultural comparisons will be attempted by re-analyzing the qualitative data with a thematic approach and exploring cultural differences.
Data management
All data will be stored by the research team following the requirements of the General Data Protection Regulation (GDPR, 2018; European Parliament and Council of European Union, 2016), Data Protection Act 2018 (DPA, 2018) and Cardiff University’s Records Management Policy (2020).
Digital copies of participants’ signed consent forms that have been returned via email will be password-protected and stored separately from the anonymized interview data. Consent-related audio recordings conducted prior to starting the interviews will be stored in the same way. Returned paper consent forms will be kept secure in a locked cabinet in a research office at Cardiff University. After each interview, the researcher will upload the digitally recorded audio data into an access-restricted folder on a secure University server. The obtained IPAQ questionnaire data will be inputted into Excel/NVivo and also stored on this secure server. All paper and digital files will be labeled with a study as well as a participant reference number. No identifiable data such as participants’ names and addresses will be included. Only anonymized data will be transferred via electronic media.
The transcription of the anonymized audio recordings will be conducted by a dedicated digital transcription company registered with Cardiff University. The company has high data security standards that will protect the security and integrity of the data they are entrusted with at all times. Uploading the audio files to the company’s platform and downloading the completed transcripts will be protected by military-grade 128-bit Transport Layer Security encryption, ensuring that data are not exposed during transfer. The research team will be provided with a dedicated password to protect file upload/ download links, thus ensuring that only authorized team members can download the completed transcripts. Finally, all encrypted data will only be stored on the transcription company’s platform for an agreed time and will then be completely and securely removed via a proprietary data deletion process.
All movement or deletion of voice data will be logged by name and date. Upon the permission from the funder, Tenovus Cancer Care (TIG 2019-09), all voice data will be deleted once the project is complete and the final report has been accepted by the funder. Direct quotations from the interview transcripts will only be used in anonymized form and only if participants have given their consent for the research team to do so.
Public and Patient Involvement (PPI)
Two lay research partners have been supporting the study from the early stages. One was recruited from the existing research network and the other via Health and Care Research Wales’ (HCRW) Public and Patient Involvement Community (INVOLVE). The lay research partners were involved in developing the study protocol and took an active part in obtaining ethical approval.
Regular quarterly meetings will be held to receive continuous input from the lay research partners and provide them with study updates. Their suggestions, comments, and concerns will be addressed alongside the process of data collection, data analysis, report writing, publication, and dissemination of the findings.
Archiving and reporting
Study documents will be retained for 5 years and archived according to Cardiff University’s Record Management Policy (2020). Data will be managed in line with Cardiff University’s Data Governance Roles and Responsibility Policy (2019).
The results of this study will be reported to the funder. The research team also aims to publish the study results in peer-reviewed journals as well as present the findings at relevant national and international conferences. The lay research partners will be actively involved in the dissemination of the findings. The study team also has representations in the Supportive and Palliative Care Subgroup of the National Cancer Research Institute’s (NCRI) Brain Group and will use the group’s reach to further disseminate the findings. Participants will be notified about the availability of published data and if they wish, summary reports will be sent via email or post.
Ethical Approval and Research Governance Approval
Ethical approval has been obtained from Cardiff University’s Research Ethics Committee as well as from an NHS Research Ethics Committee. The VCC’s Research and Development Department has confirmed capacity and capability to support the delivery of the PhAB project. The study will be conducted according to the principles of Good Research Practice (including proper and appropriate conduct of research, professional integrity, honesty, statistical methods, use of data, interpretation of data, non-plagiarism) and the UK Policy Framework for Health and Social Care Research (NHS Health Research Authority, 2020; Medical Research Council, 2012). The study will comply with NHS Ethics Committee and Health and Safety regulations. It will also be conducted following the International Council of Medical Journal Editors’ recommendations for the protection of research participants (International Committee of Medical Journal Editors, 2019) and the World Medical Association’s Declaration of Helsinki 2013 (World Medical Association, 2013).
Discussion: Main Ethical Concerns and Risks vs Benefits
The overarching ethical concerns when involving patients in qualitative health research are to maintain their safety and comfort and ensure that their consent remains informed and valid throughout the study (Morse, 2007). The ethical concerns that will be carefully addressed at different stages of the study are depicted in Figure 3.

Main ethical concerns and how they will be addressed.
The main ethical issue associated with this study is the challenging health condition of the patients. Many of their carers tend to experience great distress in their new role as a carer and have to come to terms with, and adapt to, the rapidly changing support needs of the patient and their poor prognosis (Baillie et al., 2018; Long et al., 2016). As described in Figure 3, the research team will continuously check that participants’ consent is ongoing throughout the interview. The interviews will be conducted over the phone at a time convenient to the participants and entirely at their pace. The MCPCRC has a proven track record of conducting research with palliative care patients and highly experienced senior research team members will support and supervise this study throughout.
The risk associated with participating in this study is low as the participants will only be asked about physical activity but will not be required to actually engage in any kind of physical activity. The burden associated with study participation is also relatively low. Participating in an interview can, however, be cognitively, emotionally, and physically challenging for patients and their carers and comes with a time commitment. The research team will keep the time commitment to a minimum and monitor the participants’ well-being and comfort throughout the interview to encourage breaks (or terminate the interview) if needed. Even though these patients may be in the last months to a year of life, it is important to offer this patient group the opportunity to participate in research as a means of understanding their lived experience and improving services and interventions based on real-world findings (Terry et al., 2006; White & Hardy, 2010).
The risk of emotional distress is considered low given the nature of the questions to be asked. At times, having the opportunity to talk about their thoughts and experiences with an interviewer with the time to listen might result in participants sharing aspects of their experience that go beyond the explicit aim of the interview and might result in emotional upset. This can be an emotionally challenging situation for both the patient and the researcher, but it is important to respond with empathy and care and to leave it to the patient to decide if they would like to continue the interview or if they would like to take a break or re-schedule for another day (Sivell et al., 2019).
Importantly, when scheduling the interviews, it will be explained to patients and carers that if issues arise which are a cause of concern or distress, the researcher may encourage the patient or their carer to contact their clinical team at the cancer center (e.g. their specialist nurse) for support and advice. If needed, the researcher may also ask for the patient’s consent to do so for them. If a participant does not wish to carry on the interview at any point, the interview will be terminated immediately. As talking about one’s own or a loved one’s experience of illness and how it impacts daily life can bring up thoughts and emotions not only during an interview but also before and after, the PIS for both patients and carers include information signposting to free and confidential telephone support lines (run by Tenovus Cancer Care and Macmillan, respectively) which could provide support in such situations.
The ethical concern relating to the dissemination of study findings in the form of a summary report to participants is that there is a probability that the health condition of some patients may have deteriorated significantly or they may have died between the period of data collection and completion of the analysis. Such feedback to patients and families will thus have to be provided with great sensitivity and care.
Conclusion
This protocol is intended as a step-by-step guide for future researchers wanting to develop similar qualitative research and obtain ethical approval while involving vulnerable patients like high-grade glioma patients. The study will explore the thoughts and preferences of high-grade glioma patients and their carers about becoming physically active in their daily life. An analysis of this information aims to improve the understanding of patients’ support needs for physical activity and to enhance the development of more practical and useful interventions in the future. The long-term aim is to design and implement an intervention study based on the findings of this exploratory small-scale study.
Supplemental Material
Supplemental Material, sj-pdf-1-ijq-10.1177_1609406920982133 - Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol
Supplemental Material, sj-pdf-1-ijq-10.1177_1609406920982133 for Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol by Ishrat Islam, Silvia Goss, Annmarie Nelson and Anthony Byrne in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material, sj-pdf-2-ijq-10.1177_1609406920982133 - Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol
Supplemental Material, sj-pdf-2-ijq-10.1177_1609406920982133 for Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol by Ishrat Islam, Silvia Goss, Annmarie Nelson and Anthony Byrne in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material, sj-pdf-3-ijq-10.1177_1609406920982133 - Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol
Supplemental Material, sj-pdf-3-ijq-10.1177_1609406920982133 for Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol by Ishrat Islam, Silvia Goss, Annmarie Nelson and Anthony Byrne in International Journal of Qualitative Methods
Supplemental Material
Supplemental Material, sj-pdf-4-ijq-10.1177_1609406920982133 - Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol
Supplemental Material, sj-pdf-4-ijq-10.1177_1609406920982133 for Involving High-Grade Glioma Patients and Their Carers in Qualitative Research to Promote Structured Physical Activity: A Mixed-Methods Study Protocol by Ishrat Islam, Silvia Goss, Annmarie Nelson and Anthony Byrne in International Journal of Qualitative Methods
Footnotes
Acknowledgments
We thank the Wales Cancer Research Centre (WCRC), Cardiff and Vale University Health Board and End of Life Board of the Welsh Government for supporting Ishrat Islam; Marie Curie (MCCC-FCO-17-C.) for supporting Anthony Byrne and Annmarie Nelson, and Tenovus Cancer Care (TIG 2019-09) for supporting Silvia Goss. We are very much thankful to Prof Barry Bogin at Loughborough University for his contribution to developing the study design at the initial stage. Also, we are thankful to our lay research partners, Dr Kathy Evans and Ms Sarah Peddle, for their continuous support in reviewing the protocol and other documents relevant to obtaining ethical approval for the PhAB study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by Tenovus Cancer Care (Ref: TIG 2019-09).
Sponsorship and Ethical Approval
The study has been sponsored by Cardiff University. Ethical approval has been obtained from the Cardiff University Research Governance and the National Health Services (NHS) Research Ethics Committee in Wales (Health Research Authority and Health and Care Research Wales approval, Research Ethics Committee 3, Ref: 20/WA/0078; IRAS: 274453). This study has also been approved by the Velindre Cancer Centre authority.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
