Abstract
Background
8F catheters serve as conduits to provide stable access and support for delivery of 6F aspiration catheters (ACs) in Acute Ischaemic Stroke treatment. Superbore catheters (SBCs) (internal diameter (ID) > 0.088″) are the latest innovation, designed for placement in the middle cerebral artery (MCA). SBCs may improve recanalization via enhanced control of blood pressure and flow, in comparison to balloon guide catheter (BGC) and regular guide catheter (GC) systems.
Aim
To objectively assess conduit catheter technologies in terms of deliverability, local impact on blood pressure and flow, and clot retrieval success in vitro.
Methods
Two in vitro models were used. Two SBCs, (CEREGLIDE92 and TracStar/Zoom88) and one BGC (EMBOGUARD) were used with 0.071″ ACs, and one GC (NeuronMAX) was evaluated with two 6F ACs (RED68 and SOFIA). M1-MCA occlusions were created with cohesive and friable thrombus analogues. Device tracking and clot retrieval were evaluated by two neurointerventionalists, under physiological haemodynamic conditions.
Results
SBC positioning in the MCA resulted in significant reductions of local blood pressure and flow in comparison to GC systems positioned in the Internal Carotid Artery (p = .000). However, when the SBC could not reach the MCA, they displayed similar haemodynamic control as the GC systems. SBC systems achieved high retrieval success for friable thrombi, likely due to flow reversal in the MCA during aspiration (p = .000). The TracStar/Zoom SBC system had lower rates of cohesive retrieval success due to difficulties in catheter tracking and poor clot alignment with the catheter bevelled tip.
Conclusion
Clot retrieval success is influenced by the location of the conduit for the 6F AC and the design of the AC tip.
Introduction
Aspiration thrombectomy is well established as an effective endovascular treatment of large vessel occlusion (LVO) acute ischaemic stroke (AIS).1,2 Traditionally, the aspiration procedure involves the use of a guide catheter (GC) or long introducer sheath (LS), placed in the internal carotid artery (ICA) for LVOs in the anterior circulation. These devices are used as conduits, to facilitate delivery of smaller, mostly 6F (internal diameters (IDs) = 0.068–0.072″) aspiration catheters (ACs), to the occlusion site. The design of these conduit devices has advanced, allowing more distal placement of GC's and LS's (80–90 cm), as well as larger and more flexible balloon GC (BGC) technologies. The most recent development is the generation of ‘superbore’ catheter (SBC) systems, that are designed to be placed in the middle cerebral artery (MCA).
BGC and SBC technologies aim to provide additional support for thrombectomy devices and enhanced control of local blood pressure and flow during the procedure. BGCs achieve proximal flow arrest via inflation of a balloon in the ICA, reducing antegrade flow to the thrombus and minimizing the risk of distal embolization3–5 (see Figure 1A). However, the effectiveness of flow arrest via the BGC is limited by its proximal position, making the degree of flow reduction achieved susceptible to the configuration of the Circle of Willis (CoW). Unlike BGCs, SBCs are designed to be placed intracranially in the MCA, distal to the CoW, enabling reduction of blood pressure and flow local to the thrombus.6,7 SBCs occupy a substantial proportion of the cross-sectional area of the vessel, due to their large IDs, reducing the available area for antegrade flow (see Figure 1A).6,8 A number of SBC devices are currently commercially available, including TracStarTM LDP/ZoomTM 88 (Imperative Care, Campbell, CA, USA),9,10 HiPointTM (Route 92 Medical, San Mateo, CA, USA), 11 SOFIATM 88 (Terumo Neuro, Aliso Viejo, CA, USA), 12 and CEREGLIDETM 92 (J&J MedTech Neurovascular, Irvine, CA, USA). 13 These devices are comparable in terms of their large IDs (≥0.088″), however, some have unique design features, such as the TracStar/Zoom catheters, which have a bevelled tip, designed to achieve greater surface area interaction with the thrombus, see Figure 2. 10 Great potential exists for application of SBCs in the treatment of AIS, however, apprehension surrounds the deliverability of these large catheters to distal locations, including the MCA. 7
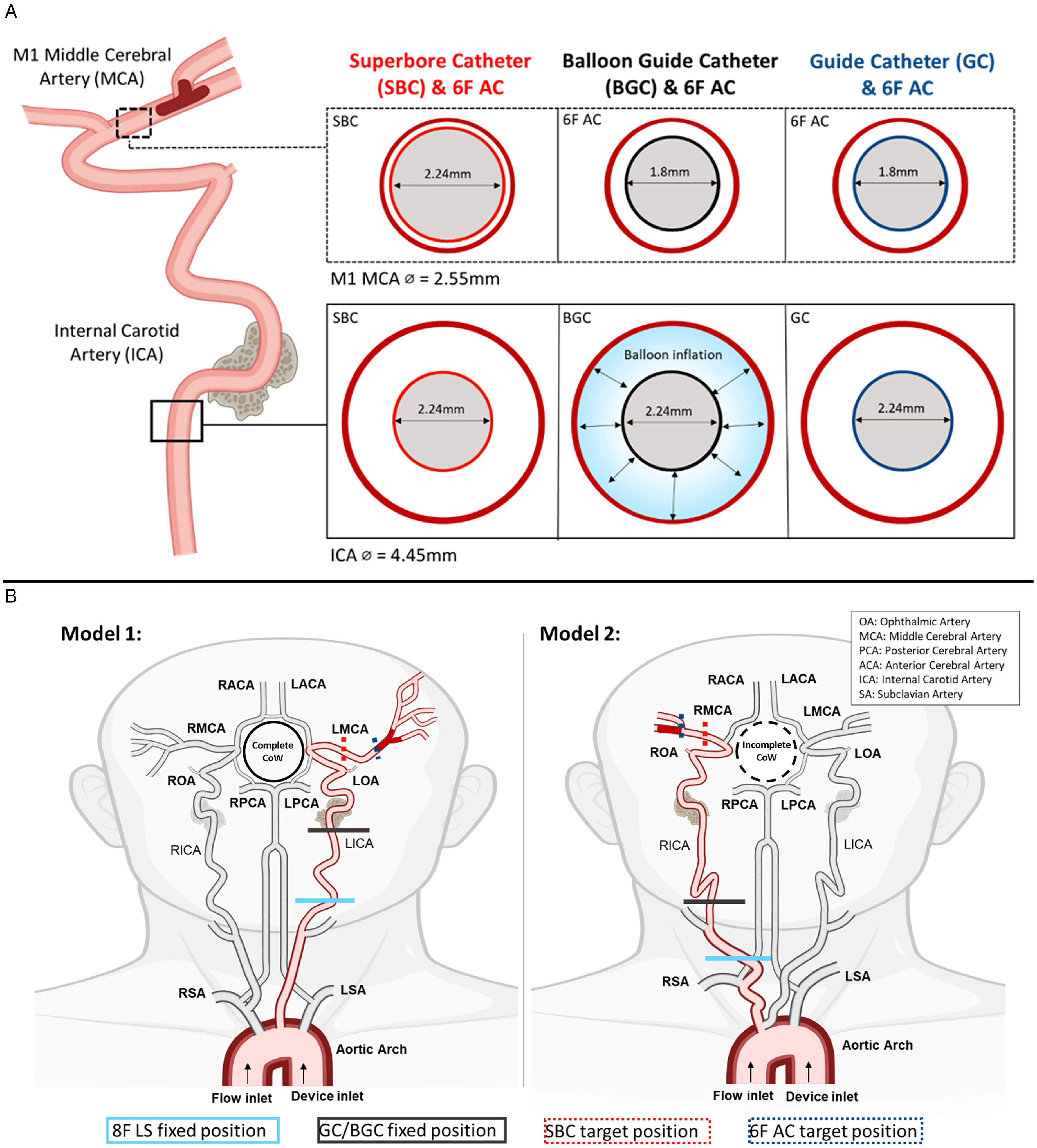
(A) Representation of the area occupied by each aspiration system in the M1 MCA and ICA, respectively, based on the measured IDs of the aspiration catheters (see Table 1) and the average diameter of the vessels. (B) In vitro anatomical models with the location of the occlusion, the target and the fixed positions of the catheters outlined. Model 1, intracranial model with a complete Circle of Willis (CoW) connected to a Type I Arch. Anatomical challenges in this model included a difficult ICA siphon and ophthalmic artery origin, and a dipping M1 segment. The diameter of the M1 was 3.14 mm. Cohesive and friable thrombus analogues were lodged to create an occlusion at the first bifurcation of the MCA. Model 2, intracranial model with an incomplete CoW connected to a Bovine Arch. This model possessed a tortuous common carotid artery and a loop in the cervical portion of the ICA. The diameter of the M1 was 2.88 mm. Cohesive thrombus analogues were lodged in the straight M1-segment, distal of the first MCA bifurcation.
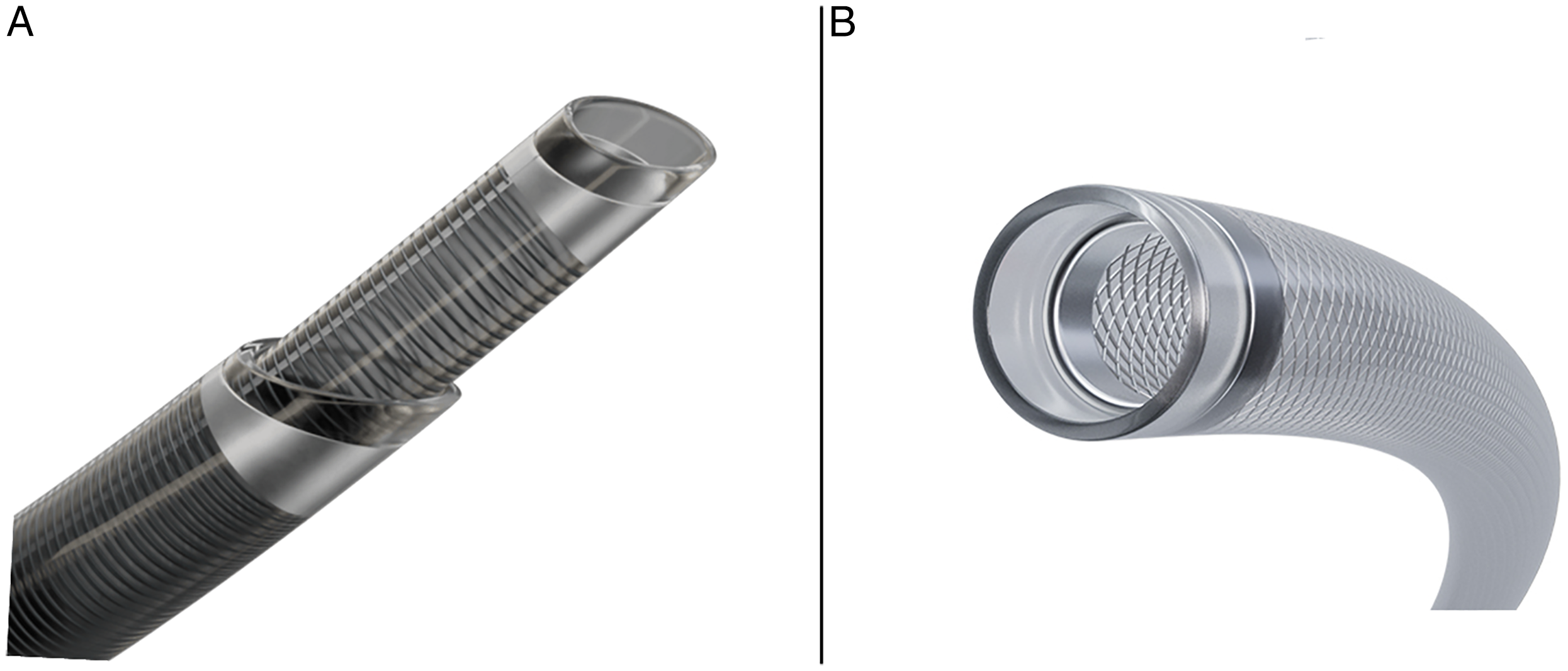
(A) Bevel-tip versus (B) straight-tip catheters.
Aspiration systems and device combinations.
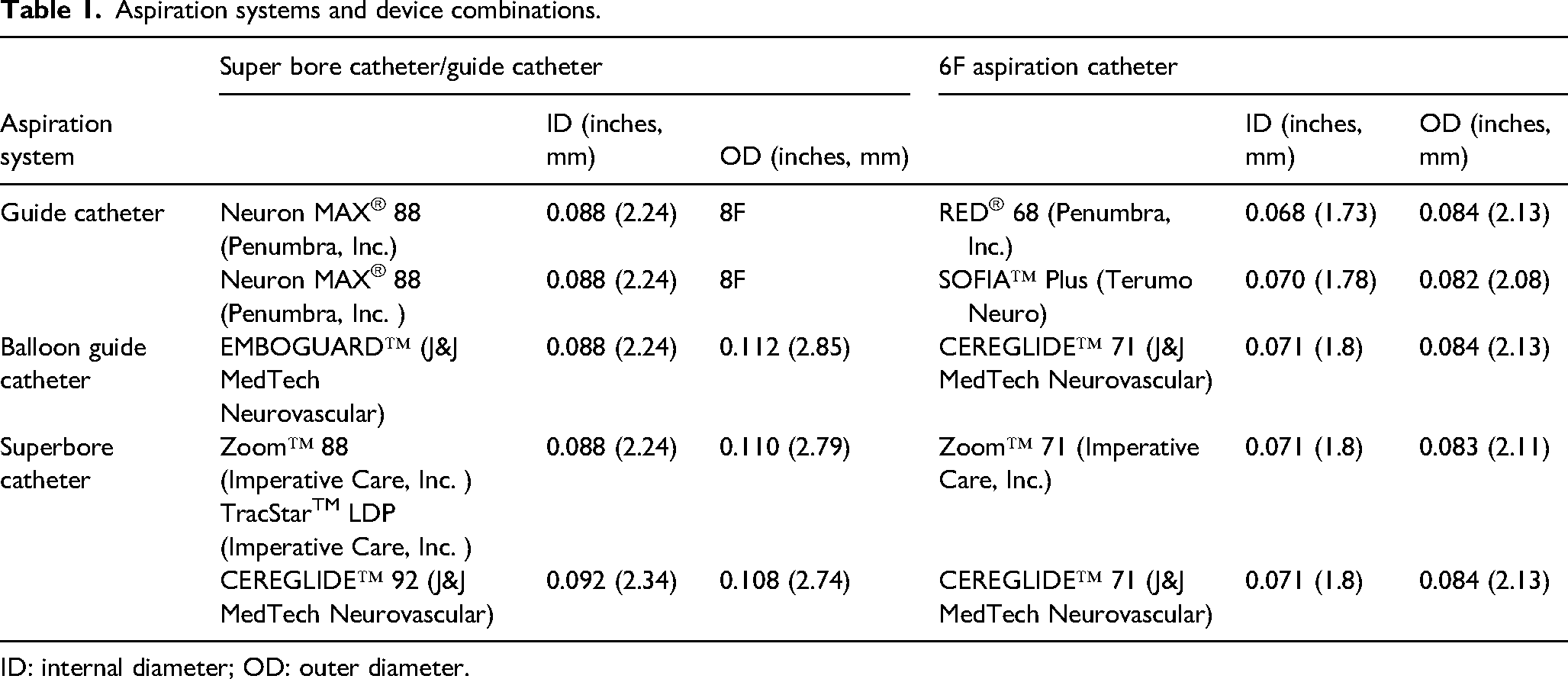
ID: internal diameter; OD: outer diameter.
The objective of this work was to conduct an in-vitro comparative analysis of aspiration thrombectomy with a 6F AC under three different procedural configurations, with an SBC, a BGC and a GC. The impact of catheter deliverability, catheter design, blood pressure and blood flow on clot retrieval success were assessed. We hypothesized that reduction of local blood pressure and flow in the MCA during clot removal enhances the likelihood of retrieval success.
Materials and methods
In vitro models
To replicate clinically challenging environments, two in vitro anatomical models with distinct ICA and Aortic Arch configurations were used: Model 1 and Model 2 (see Figure 1B). The models were prepared from silicone and 3D-printed from patient scans (MET centre, ATU, Ireland). A glycerol:water (40:60% v/v) blood mimic was circulated in the models at 37°C ± 2°C, to simulate in vivo conditions. In vitro recreation of physiological pressure conditions was achieved using a PD-1100 pulsatile pump (BDC Laboratories, Wheat Ridge, CO, USA) at 70 bpm ± 5bpm, to mimic the cardiac rhythm. Distal pressure was controlled by adjusting the height of a column of fluid at the outlet of the model. Low distal pressure was used in both test-setups to simulate the effect of poor leptomeningeal collaterals, creating challenging clot retrieval conditions. 14 The mean arterial pressure (MAP) was set to 120 mmHg. Blood flow through the MCA in Model 1 was set to 230 mL/min and to 150 mL/min in Model 2, to create varying flow conditions for retrieval.
Thrombus analogues
Platelet-contracted clot analogues were prepared following the procedure previously outlined by Johnson, McCarthy, 15 by combining defined ratios of platelet-rich plasma (PRP) and red blood cells (RBCs). To obtain these components, the blood was centrifuged (Centurion Scientific Limited E240) at 180 rcf for 10 min. The top layer of PRP was removed and stored in a separate container. The remaining blood was then centrifuged again at 1200 rcf for 10 min, to separate the RBCs from the other blood components. The platelet-poor plasma and buffy coat layers were removed as waste, leaving only the RBC-rich layer.
Two groups of clot analogues, cohesive and friable, were prepared to create an occlusion in the distal M1-segment of the MCA from blood mixtures with 5% and 25% haematocrit (%H), i.e. the volume percentage of RBCs in the blood mixture, respectively. These % H values were chosen to ensure the creation of two distinct clot models: (I) resistant to ingestion (5%H) and (II) prone to fragmentation (25%H). The cohesive clot analogues were used in both models to assess the catheter's ability to maintain grip of the thrombus during retraction from the vessel and into the conduit catheter in different anatomical environments. Friable clot analogues were used in Model 1 only, to evaluate fragmentation and distal embolization during retrieval. A minimum 5-min, maximum 15-min dwell time was enforced to ensure consistent deformation of thrombi in the system. The length of the occlusion following dwell time was required to be 18 mm ± 3 mm.
Experimental design
To assess a range of aspiration technologies, commercially available devices were selected, see Table 1. To simulate the clinical use of these devices, tracking and clot retrieval performance were evaluated by two experienced interventional neuroradiologists. Experimental testing was conducted in two blocks. In the first block, each physician performed retrievals with the SBC systems, always starting with the TracStar/Zoom SBC system and alternating between Model 1 and Model 2. In the second block, the GC and BGC systems were performed in a randomized order by each physician, maintaining the same alternation between models. Seventy-five clot retrievals were completed in total, 42 with Physician 1 and 33 with Physician 2.
Tracking
The position of the LSs were pre-defined to a fixed location according to Figure 1B. The Super Arrow-FlexTM (80 cm, Teleflex, Wayne, PA, USA) was used in combination with the SBC systems. 8F short introducer sheaths were used in combination with the GC and BGC systems that were positioned as distal as possible in the in vitro models. Physicians were instructed to track the catheters to the target positions, detailed in Figure 1B with reasonable effort and force, representative of clinical practice. Once placed, the LS/GC/BGC position was maintained for the duration of the experiment. The CEREGLIDE 92 SBC system was tracked using its custom delivery aid INNERGLIDE 9 (IG9) (165 cm, ID 0.030″, J&J MedTech Neurovascular, Irvine, CA, USA) and a guidewire (200 cm, Synchro 0.014″, Stryker Neurovascular, Cork, Ireland). All other systems were tracked using a 6F AC, a microcatheter (160 cm, Prowler Select Plus 0.021″, J&J MedTech Neurovascular, Irvine, CA, USA) and a guidewire (Synchro 0.014″).
The conduit (GC/BGC/SBC) and 6F catheters were initially tracked to their target locations without a thrombus present to create a baseline of their influence on blood pressure and flow in the MCA. Distal position of ancillary delivery devices during tracking was limited to the proximal part of the M1 to recreate the clinical scenario of avoiding crossing the occlusion. Subsequently, all catheters were withdrawn, and an M1 occlusion was created, after which the devices were reintroduced to complete clot retrieval. The final location reached by each catheter was recorded. Each catheter was used for multiple passes but never more than three passes. Before each pass, the catheters were visually inspected for any gross damage that could impact performance.
Local blood pressure and blood flow
Local blood pressure and flow were monitored throughout the experiment at pre-defined stages of the procedure, to understand the impact of the aspiration systems on local haemodynamics in the MCA. Ultrasonic flow meters (Sonotec GmbH, Halle, Germany) were used to measure the flow through the MCA at (1) baseline, (2) during device tracking (without a thrombus present), and (3) during and (4) after clot retrieval, to provide a quantitative measure of the flow through the MCA at each procedural stage. Flow reversal was defined as a change in the direction of antegrade flow in the MCA, which occurred when the proximal pressure exceeded the distal pressure during aspiration. A pressure wire (OpSens Medical, Quebec, Canada), placed locally in the M1, was used to make pressure measurements at baseline. A pressure transducer (BDC Laboratories, Wheat Ridge, CO, USA) attached to the hub of an AC with the tip positioned in the M1, was used to make pressure measurements during device tracking. Thereby, the influence of the aspiration systems on blood flow and pressure in the MCA was measured.
Clot retrieval
The 6F AC was advanced until it was in contact with the clot. Slack was retained in the catheter systems in line with the physician's current clinical practice. Aspiration was applied through the 6F AC via a 60cc VacLoc syringe for a duration of 2 min or until there was flow return in the syringe. The AC was then retrieved proximally into the conduit catheter. The system of the AC and conduit catheter were then retracted to the level of the introducer sheath and removed from the model. Outcomes of clot retrieval were recorded to assess the volume of flow through each vessel outlet of the MCA after the thrombectomy, to quantify the degree of flow restoration through the MCA territory. Retrieval success occurred when the clot was fully aspirated from the vessel resulting in complete flow restoration through the MCA. Distal embolization of the clot from the M1 resulting in a secondary occlusion in a distal MCA territory was also recorded. One thrombectomy pass was carried out for each experimental run.
Statistical analysis
One-way ANOVA was used to assess statistical differences between the aspiration systems and multiple comparison analysis was performed with Fisher's test (Minitab, version 19.1.1.) A significance level of .05 was used.
Results
SBC tracking
Catheter tracking results for each system include tracking attempts during (1) baseline measurement of local blood pressure and flow (n = 5) and (2) clot retrieval (n = 5 for each clot type). This resulted in a sample size of n = 15 in Model 1 and n = 10 in Model 2, for each thrombectomy system. The CEREGLIDE 92 SBC and INNERGLIDE 9 system reached the target location in the M1 in 100% of cases in Model 1 (15/15) and Model 2 (10/10), with subsequent successful delivery of the CEREGLIDE 71 catheter to the target location. The TracStar/Zoom 88 SBC and Zoom 71 reached the target location in 80% (12/15) of cases in Model 1, in the failed attempts, neither the SBC nor the 6F AC could be tracked to the MCA. These failures were attributed to the orientation of the bevel tip of the catheter, which got ‘hung up’ on the ledge of the ophthalmic artery and could not be tracked further. In Model 2, the TracStar/Zoom 88 SBC reached the M1 MCA in 4/10 attempts (40%), however, the Zoom 71 AC was able to reach the MCA in all cases. In instances where the SBC did not reach the MCA, the SBC position was maintained in the ICA, just below the level of the carotid siphon. Inability of the TracStar/Zoom 88 SBC to reach the M1 in Model 2 was found to be associated with insufficient length of the device (4/6, 66.67%) and high tracking forces (2/6, 33.33%). See Supplemental Section 1 and Supplemental Figure S1 for additional information and results on SBC tracking. Tracking of the 6F AC to the M1 was successful in every case for the BGC and GC systems.
Local blood pressure and blood flow
When the SBC systems reached a proximal M1 position, they significantly reduced local blood pressure in the MCA, in comparison to a GC or SBC, positioned in the ICA, see Figure 3 (p = .000). The GC systems, NeuronMAX with RED68, or SOFIA, were not significantly different from each other and reduced the MAP in the M1 by an average of 18% and 26%, in Model 1 and 9% and 16% in Model 2, respectively. The SBC systems, CEREGLIDE 92 and 71, and TracStar/Zoom 88 and 71, had a large influence on the MAP when positioned in the M1, reducing it by 64% (5/5) and 67% (4/5) in Model 1, and 86% (5/5) and 89% (3/5) in Model 2, respectively. In one case in Model 1, for the TracStar/Zoom system, neither the 88 nor the 71 catheters could be tracked to the MCA, thus, pressure and flow measurements were not taken. In two cases during device tracking in Model 2, with the TracStar/Zoom system, the SBC catheter could not be tracked to the M1. In these cases, only the 6F AC reached the target, and as such, a reduction in MAP of similar magnitude to the GC systems was achieved, see Figure 3. In Model 1, with a complete CoW, the BGC system, EMBOGUARD and CEREGLIDE 71 reduced pressure in the M1 by 24%, similar to the GC systems. Model 2 had an incomplete CoW, missing a PCom and ACom, and as a result, inflation of the BGC significantly reduced the pressure in the MCA. A similar trend was observed on the average flowrate through the MCA relating to the final position achieved by the conduit catheter, see Supplemental Figure S2.
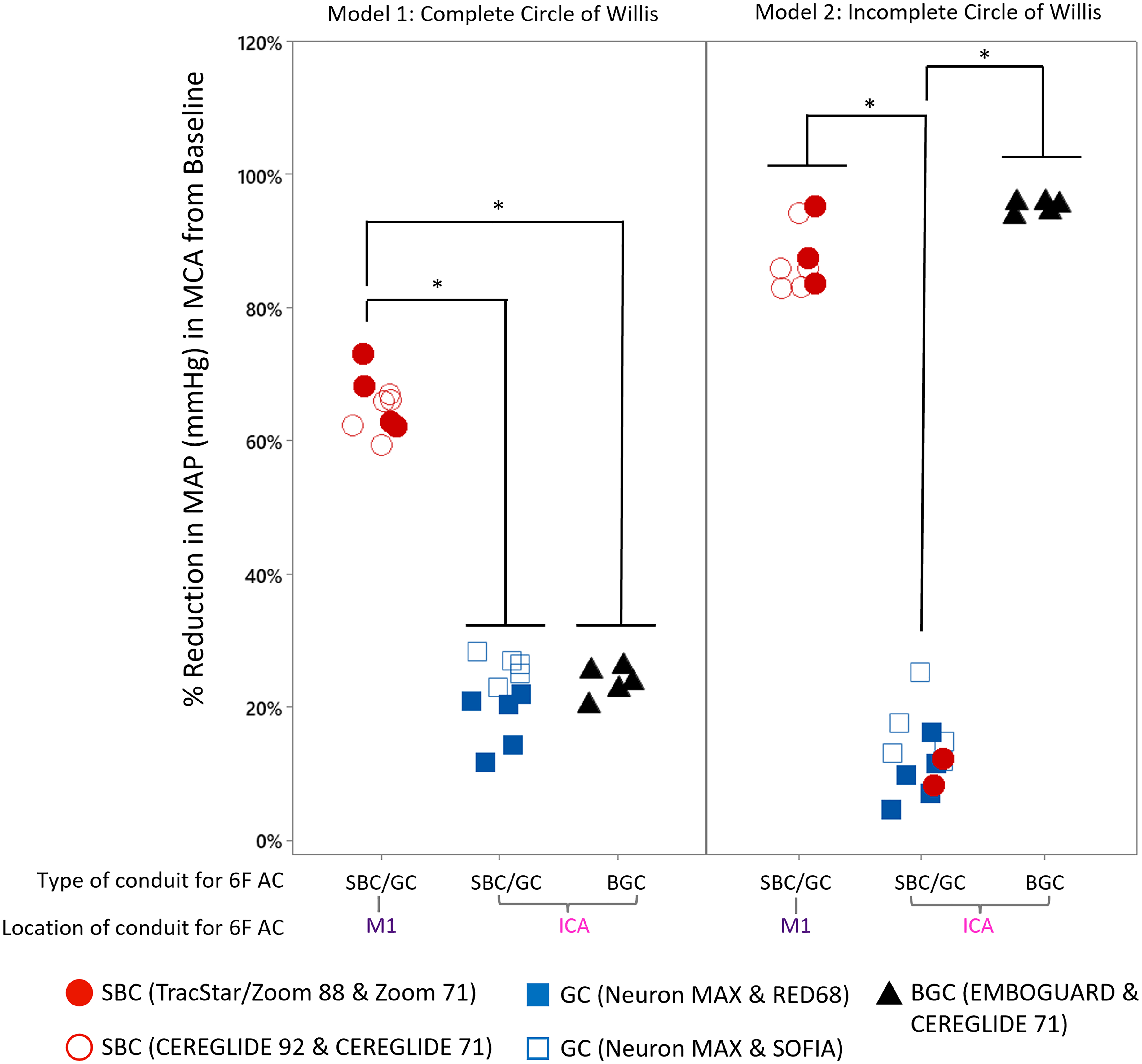
Percentage (%) reduction in MAP in M1 MCA compared to baseline for aspiration systems based on type and location of the conduit for the 6F AC in Model 1 and Model 2. In Model 1, n = 4 for the Zoom system as in one case neither the 88 nor the 71 device could be tracked to the M1 MCA, n = 5 for all other device combinations, *p < .001. Results were calculated using an analysis of variance with Fisher's test for pairwise comparison.
Clot retrieval
In Model 1, in every case where the SBC systems, CEREGLIDE 92 and 71 and TracStar/Zoom 88 and 71, could reach the MCA, the accompanying AC successfully retrieved the friable clots. In one case, neither the SBC nor the AC for the TracStar/Zoom system could be tracked to the MCA, and as such, clot retrieval could not be performed. The BGC system, EMBOGUARD and CEREGLIDE 71, also achieved high rates of complete clot retrieval success, with the entire clot removed in 80% (4/5) of cases (Supplemental Table S1). In comparison, the GC systems, NeuronMAX with RED68 or SOFIA, were unable to completely retrieve the friable clots and experienced high rates of distal embolization, 80% (4/5) and 40% (2/5), respectively.
Flow rate measured through the MCA after first-pass aspiration enabled quantitative analysis of the percentage of flow restored to the MCA from baseline, see Figure 4A. The SBC systems, once placed in the M1 successfully retrieved all friable clots, resulting in complete flow restoration. Analysis of flow rate through the MCA during aspiration revealed that the presence of the SBC in the M1 MCA facilitated flow reversal in the vessel, see Figure 4B. The BGC system, EMBOGUARD and CEREGLIDE 71, experienced one case of distal embolization that resulted in a partial flow restoration of 40%, due to the presence of a distal MCA branch occlusion. In comparison, the GC systems, NeuronMAX with RED68, or SOFIA experienced multiple cases of embolization resulting in significantly lower flow restoration through the MCA in comparison to the other systems (p = .000).
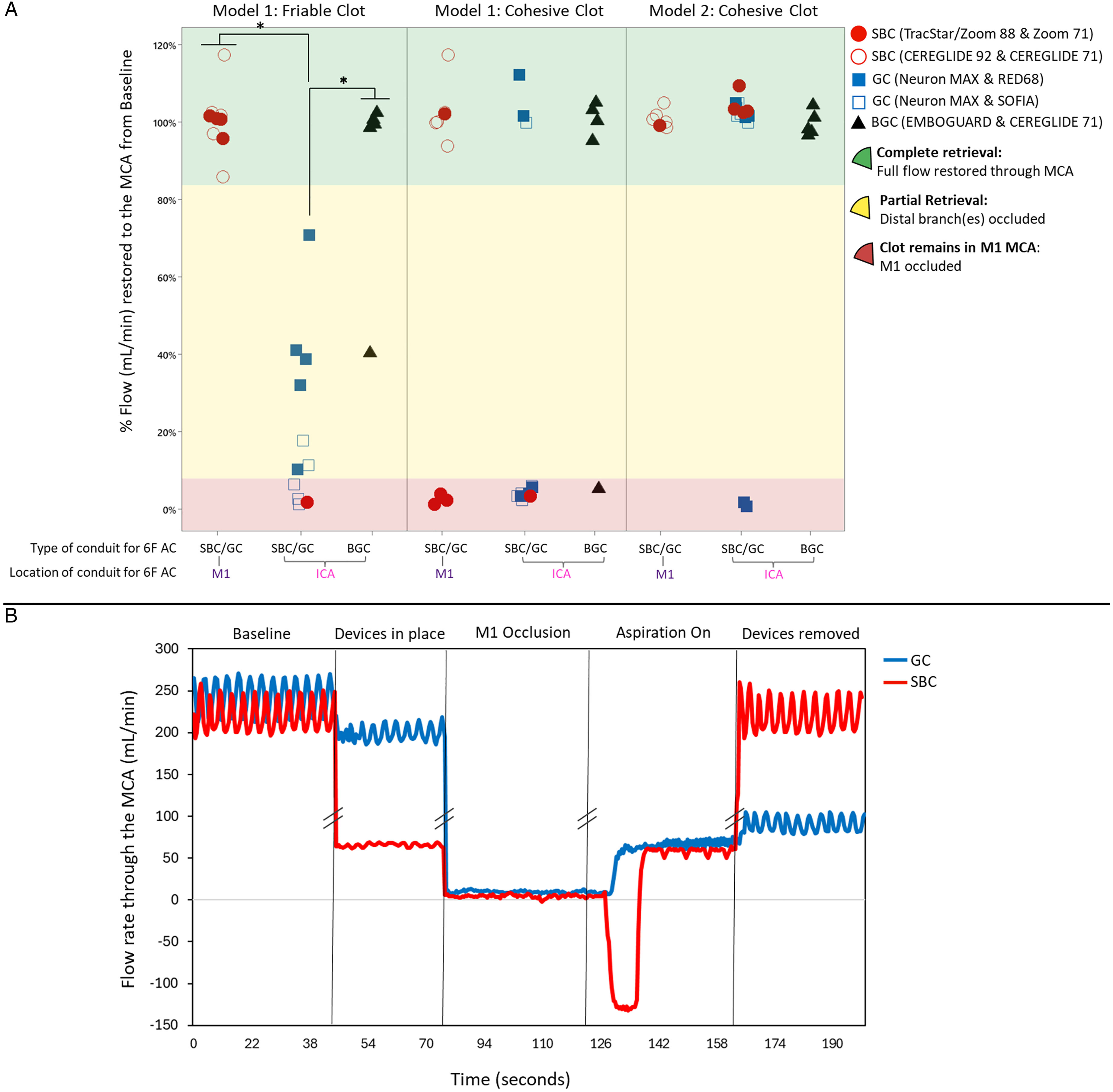
(A) Percentage (%) of flow restored in the MCA territory compared to baseline flow following first pass aspiration for Model 1; friable and cohesive clots, and Model 2; cohesive clots. n = 5 for each device combination. *p < .001. Results were calculated using an analysis of variance with Fisher's test for pairwise comparison, (B) Representative flow rate (mL/min) through the MCA throughout each procedural stage for GC and SBC systems. Flow reversal observed during aspiration with the SBC system as flow rate values through the MCA were recorded below 0 mL/min.
In contrast to its high rate of complete clot removal with the friable clots (4/5, 80%), TracStar/Zoom 88 and 71 had lower success removing the cohesive clots from Model 1 (2/5, 20%), compared to CEREGLIDE 92 and 71 (5/5, 100%) and the EMBOGUARD and CEREGLIDE 71 (4/5, 80%) systems, see Figure 4A and Supplemental Table S1. Failure of the TracStar/Zoom 88 and 71 to completely remove the clot resulted in a significantly lower average flow restoration in the MCA, 21% compared to 99% with CEREGLIDE 92 and 71 and 80% with EMBOGUARD and CEREGLIDE 71 (p = .035). RED68 and SOFIA lost grip of the clots during proximal retraction to the NeuronMAX in the ICA, in 2/5 (40%) and 3/5 (60%) of the cases, respectively.
In Model 2, NeuronMAX and RED68 was the only device combination which failed to restore complete flow through the MCA in 2/5 (40%) of the cases with the cohesive clots, see Supplemental Table S1. The AC was unable to maintain a grip of the thrombus during retraction to the GC, resulting in a mean flow restoration value of 62% (p = .027).
Discussion
This study demonstrates the benefit of emerging SBC technologies to improve clot retrieval success. When positioned in the M1, the SBC systems achieved 100% success in removal of friable clots, with no cases of embolization, outperforming the GC and BGC systems. In the M1, the large IDs of SBCs (≥0.088″) occupy a substantial proportion of the vascular lumen, reducing the available cross-sectional area between the catheter and the vessel wall. Overall, this reduces the volume of antegrade flow in the vessel, due to increased resistance, see Figure 4B. This is in accordance with the Hagen–Poiseuille equation, see Equation 1. This effect resulted in reversal of antegrade flow (retrograde flow) during aspiration, as shown in Figure 4B, enhancing clot ingestion and minimizing distal embolization. A recent study by Glynn et al. (2025)
16
highlights that this effect is not simply due the size of the catheter, but the ratio of the catheter to the target vessel. While the in vitro system demonstrated the benefits of flow reversal, it is vital to consider the impact of dynamic vascular responses on its effects in vivo.
17
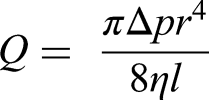
Equation 1. Hagen–Poiseuille equation.
SBCs have showed promise for direct aspiration, 6 however, this technique remains an off-label use of the device in the United States. This study is the first to demonstrate the benefit of SBCs on flow and pressure control to increase the likelihood of clot retrieval success under their approved indication as an access catheter. These haemodynamic advantages may translate clinically to higher first-pass success rates and improved patient outcomes when SBCs are used as conduit devices. 18 In addition, their large IDs provide a greater area for clot ingestion, further supporting their potential to improve clot retrieval by direct aspiration.
However, the benefit of SBCs is dependent on their ability to reach the MCA, and tracking of SBCs remains a concern for the clinical application of these devices. 7 In this study, TracStar/Zoom 88 failed to reach the MCA in 20% of the attempts (3/15) in Model 1, and in 60% of the attempts (6/10) in Model 2. Failure to reach the MCA limited their haemodynamic control to levels comparable to the GC systems. These failures were attributed to design features of the device, including its length, which became used up in tortuous vascular anatomies and the bevelled tip of the catheter, which got hung up on the ledge of the ophthalmic artery and could not be tracked further. However, it is important to note that two SBC technologies from Imperative Care, TracStar LDP and Zoom 88 were used interchangeably in this study. The differences in working length and coating length between these devices were not deemed to be significant for the purposes of this work. To overcome navigational issues, many next generation SBCs are now being designed with navigation aids, 19 such as CEREGLIDE 92 with INNERGLIDE 9 13 and HiPointTM with Tenzing 8. 11 Our findings align with emerging clinical data that report good deliverability rates for SBCs, 13 and demonstrate the value of navigation aids in overcoming anatomical barriers, see Supplemental Section 1 for further details.
The ability of the AC to maintain grip of the clot during retrieval is determined by the aspiration grip force which is highly correlated with its ID, see equation (2).
20
In this study, the 6F ACs, RED68 and SOFIA, in combination with a regular GC, experienced high failure rates for cohesive clot retrieval. As shown in Supplemental Video S1, these ACs did not appear to have sufficient grip force to extract the clot from the M1 MCA. SBCs are advantageous in these scenarios as they provide greater surface area for contact with the clot, and their distal placement reduces the relay distance for ACs to travel during retrieval. This limits the potential loss of the thrombus or thrombus fragments and maintains distal access during multiple pass aspiration. In this study, placement of the SBC in the MCA resulted in 100% cohesive clot retrieval success for CEREGLIDE 92 and 71. CEREGLIDE 92 currently possesses the largest ID of any commercially available SBC, facilitating more effective clot ingestion and supporting the ability of the AC to maintain clot engagement during retraction. TracStar/Zoom 88 and 71, conversely experienced lower rates of cohesive clot retrieval success, attributed to the orientation of the catheter's bevel tip, which caused difficulty during catheter tracking, and catheter-to-clot interactions. Poor alignment between the clot and bevel tip resulted in the loss of the clot during retraction into the SBC in the M1 MCA (see Supplemental Video S2). Attempts to reorientate the bevel tip failed and no consistent technique could be achieved. These findings highlight the influence of catheter ID and tip design on aspiration grip force, with straight tip SBCs offering a clear advantage for complete clot ingestion and secure retrieval. However, they should be carefully considered, as operator blinding was not performed and this could have introduced potential user bias.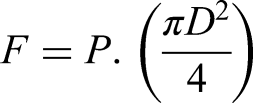
Equation 2. Equation governing the grip force at the tip of the AC.
The CoW is critical collateral network in the cerebral circulation 21 and variation in its configuration are common.22–25 In this study, two configurations of the CoW were used to assess the impact of aspiration technologies in varying haemodynamic environments, representing the extremes of an intact and a disrupted collateral network. Inclusion of additional anatomical variations may lead to intermediate haemodynamic responses, potentially reducing the effectiveness of the BGC systems, which were dependent on the configuration of the CoW to reduce local blood pressure and flow. In contrast, the ability of SBCs to reduce local blood pressure and flow, would not be affected, as their distal positioning in the M1-MCA, bypasses the CoW. In Model 1, collateral flow supplied via the anterior and posterior communicating arteries, limited the impact of inflation of the BGC on MCA pressure and flow. Conversely, a significant impact of the BGC was observed in Model 2 due to the absence of these primary collateral arteries. It is, however, vital to consider the potential risks of flow arrest in these scenarios, whether induced proximally by a BGC or distally by an SBC positioned in the MCA. In particular, in an anatomy with an incomplete CoW, as such patients may be dependent on pial collateral networks fed by the ipsilateral anterior cerebral artery, now being completely or partially blocked. Excessive or prolonged flow arrest in such scenarios may potentially convert salvageable territory into a complete infarct.8,26 While flow arrest may help to facilitate clot retrieval, its duration and extent should be minimized to balance procedural efficacy and patient safety.
In this study, despite varying degrees of haemodynamic control achieved by the BGC systems, higher rates of clot retrieval success were observed in comparison to the GC systems in both models. As three different ACs (RED68, CEREGLIDE 71 and SOFIA) were used across the GC and BGC systems, further testing was required to understand the mechanism for successful clot retrieval with the BGC system. Such testing was conducted with a friable clot in Model 1 (intact CoW) with RED68 and CEREGLIDE 71, in combination with EMBOGUARD and NeuronMAX, respectively, see Supplemental Figure S3. Application of proximal flow arrest via the BGC in this scenario did not significantly impact clot retrieval success for either AC.
Limitations
Two SBC technologies from Imperative Care, TracStar LDP and Zoom 88 were used interchangeably in this study. For the purposes of this work the differences between these devices, primarily differences in working length and coating length, were not deemed to be significant. Current aspiration thrombectomy practices often include aspiration through the conduit catheter. This was not performed in this study as aspiration through SBCs is currently not included as an indicated use in the United States. The operators were not blinded to device allocation, introducing potential for user bias. Development of sophisticated in vitro models has enabled repeatable recreation of AIS on the bench, facilitating critical insights into current thrombectomy strategies. However, these models do not replicate clinical dynamic vascular responses and furthermore, the insights from this study are limited to the dimensions of the in vitro models used, thus, the results from this testing should be confirmed in a clinical setting.
Conclusion
This research demonstrates the benefits of SBCs as conduit device in AIS. Once placed in the MCA, SBCs offer significant reductions in local blood pressure and flow, influencing clot retrieval success. In addition, the size, design and the construction of the AC tip were found to have a critical impact on catheter tracking and catheter–clot interactions. Together, these insights may inform device selection clinically and guide the innovation of next-generation aspiration thrombectomy devices.
Supplemental Material
Supplemental Material
Footnotes
Acknowledgements
All authors contributed to the drafting and revision of this manuscript. All authors reviewed and approved the version to be published.
Ethical approval
No patients or animals were used in this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been funded by Research Ireland and Johnson and Johnson MedTech Neurovascular (Grant No. EBPPG/2021/66).
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AG and RMcC are full-time employees of Johnson and Johnson MedTech Neurovascular; AG is also a full time PhD student at Trinity College Dublin. TA is a consultant for Anaconda, J&J MedTech Neurovascular-Neuravi, Ceretrieve, Cortirio, Optimize Neurovascular, Rapid Medical, and Spryte and is a co-founder/shareholder in Ceroflo.
Data availability statement
The data that supports the findings of this study is available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
