Abstract
Purpose
The objective of this study is to report the initial experience with the PANTHER technique for ruptured intracranial aneurysms and to provide a descriptive comparison with patients treated using a single flow diverter (FD) under prasugrel single antiplatelet therapy (SAPT).
Methods
A retrospective analysis was conducted on patients with subarachnoid hemorrhage due to ruptured wide-neck aneurysms. The PANTHER group (n = 8) underwent reconstruction with non-identical telescoping hydrophilic polymer–coated (HPC) FDs under prasugrel SAPT. For the purpose of comparison, the outcomes of patients treated with a single HPC-coated FD (n = 21) were reviewed. The data included complications, angiographic occlusion, and clinical outcomes.
Results
The PANTHER procedures were technically successful, without procedure-related hemorrhagic, ischemic, or thromboembolic complications. At early follow-up (median 5 months), complete occlusion was observed in all available PANTHER cases, whereas in the single FD group, 77.8% of assessable aneurysms showed complete occlusion. At long-term follow-up, complete occlusion was documented in both groups.
Conclusion
In this single-center series, the PANTHER technique was technically feasible and associated with high early occlusion rates and favorable clinical outcomes under prasugrel SAPT. A descriptive comparison with single FD treatment suggests a potential hemodynamic benefit of non-identical telescoping constructs. Larger prospective studies are warranted to validate these preliminary findings.
Keywords
Introduction
Aneurysmal subarachnoid hemorrhage (SAH) and its complications are associated with substantial morbidity and mortality.1,2 The primary objective of prompt therapeutic interventions after aneurysm rupture is to avert re-rupture. 3 Surgical clipping and endovascular coiling are the two main treatment modalities for acutely ruptured aneurysms.4–6 However, these treatments have limited efficacy in occluding all ruptured aneurysms. 7 The advent of endovascular devices has expanded the therapeutic options available for the management of ruptured aneurysms. Flow diversion has become an established treatment for non-ruptured wide-neck aneurysms unsuitable for coiling, because of its efficacy and safety.7,8 Flow diversion with a telescoping technique can be used to treat long aneurysms or to increase the flow diversion achieved at the base of treated aneurysms. Nevertheless, the use of flow diverter (FD) with a telescoping technique remains a contentious issue in the therapeutic management of aneurysms. A major disadvantage of FD treatment is the need for dual antiplatelet therapy, particularly in patients with acute ruptured aneurysm. However, in the past several years, experience with FD implantation under single antiplatelet therapy (SAPT) has been reported in both ruptured and non-ruptured aneurysms.9,10 Herein, we present our retrospective experience with the prasugrel-based aneurysm treatment with non-identical telescoping with flow diversion with hydrophilic polymer coating for endovascular reconstruction (PANTHER) technique for ruptured intracranial aneurysms unsuitable for coiling in the setting of acute SAH. The objective of PANTHER is to induce a more pronounced flow diversion effect concentrated at the level of the ruptured aneurysm, with the aim of reducing the risk of re-rupture and accelerating the occlusion of the aneurysm, in comparison to the conventional technique that uses only a single FD; both techniques under SAPT. In addition to the results of the main PANTHER cohort, the study provides a brief descriptive comparison of the results for patients treated with a single FD following an aneurysm rupture in a SAH setting.
Methods
Study population
A retrospective review of a prospectively maintained database was conducted to identify patients with ruptured aneurysms treated with HPC-coated FDs with prasugrel SAPT. Patients treated with uncoated FDs or other devices, such as coils, during the procedure, were excluded.
The primary cohort under consideration in this study comprised patients with ruptured aneurysms who underwent treatment using the PANTHER technique. Patients treated with a single FD only were included as a comparison group. Patients who had been pretreated or treated with uncoated FDs or other devices, such as coils, during the procedure, or who were treated more than three days after the aneurysm rupture were excluded from the study.
The demographic details, aneurysm morphology and location, FD type and size, platelet function test results, periprocedural and postprocedural complications, clinical outcomes, and angiographic follow-up (FU) data were recorded.
Endovascular treatment
All treatments were performed in patients under general anesthesia with a biplane angiography system. Intravenous heparin was not administered, because of the SAH. Arterial access was obtained with a 6–8 French (F) sheath, usually in the right groin, and a standard 6–8 F guiding catheter. In most cases, a 5 F intermediate catheter (e.g., Navien A Mourant 058, Medtronic, Dublin, Ireland) was used. All FDs were deployed over a 0.021-inch microcatheter. After navigation of the vessel distal to the aneurysm, an FD with a smaller diameter, undersized relative to the parent vessel, was first implanted at the level of the aneurysm neck. Thereafter, the microcatheter was inserted distally to the first FD over the movable wire. Subsequently, an oversized FD, which was usually longer than the first FD, was implanted. FD diameter and length were selected according to two- or three-dimensional measurements of parent artery diameter.
The subsequent schematic (Figure 1) illustrates the step-by-step application of the PANTHER technique in the treatment of a ruptured AcomA aneurysm. The same case is shown angiographically in Figure 2 as a representative procedural example.
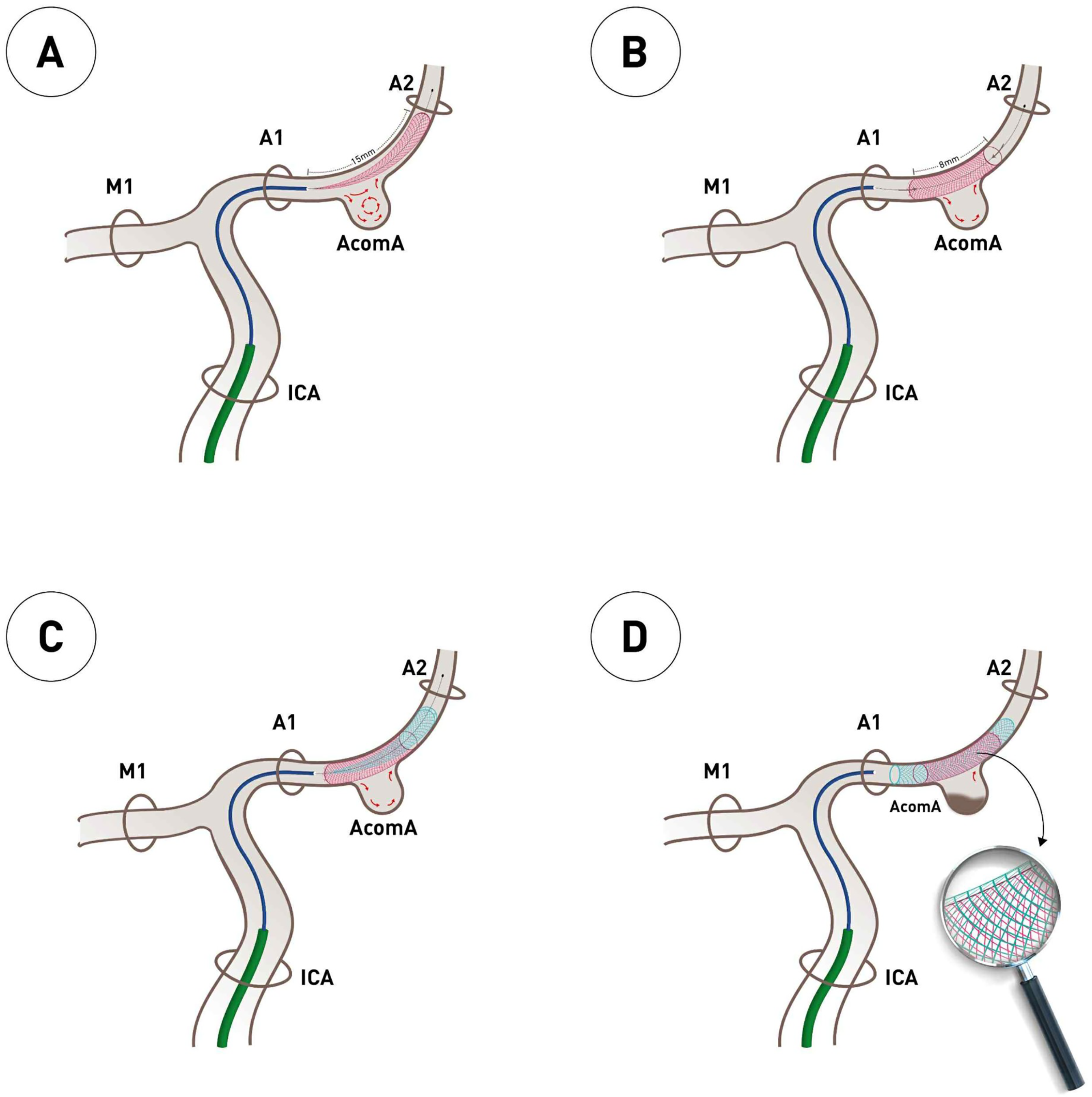
Stepwise schematic of flow diversion treatment of a ruptured AcomA aneurysm using the PANTHER technique with non-identical telescoping HPC-coated FDs under prasugrel SAPT. (A) A microcatheter is navigated from the ICA through the A1 segment into the distal A2 of the ACA. The first HPC-coated FD (2/15 mm), which is undersized relative to the parent vessel, is partially deployed across the aneurysm neck. (B) Following the full deployment and adaptation to the wall of the first FD, it is shortened to approximately 8 mm. This intentional foreshortening increases the mesh density across the aneurysm neck due to the undersized FD, thereby enhancing the flow-diverting effect by decreasing porosity. (C) In order to improve wall apposition and expand the treated segment, a second HPC-coated FD with a larger diameter (3/18 mm) and a longer length is advanced over the first FD. (D) The final construct shows the complete deployment of both FDs. The overlapping zone across the aneurysm neck creates a denser heterogeneous mesh with lower porosity. The magnified view illustrates the interwoven struts and effective modulation of flow into the aneurysm, promoting the initiation of thrombosis.
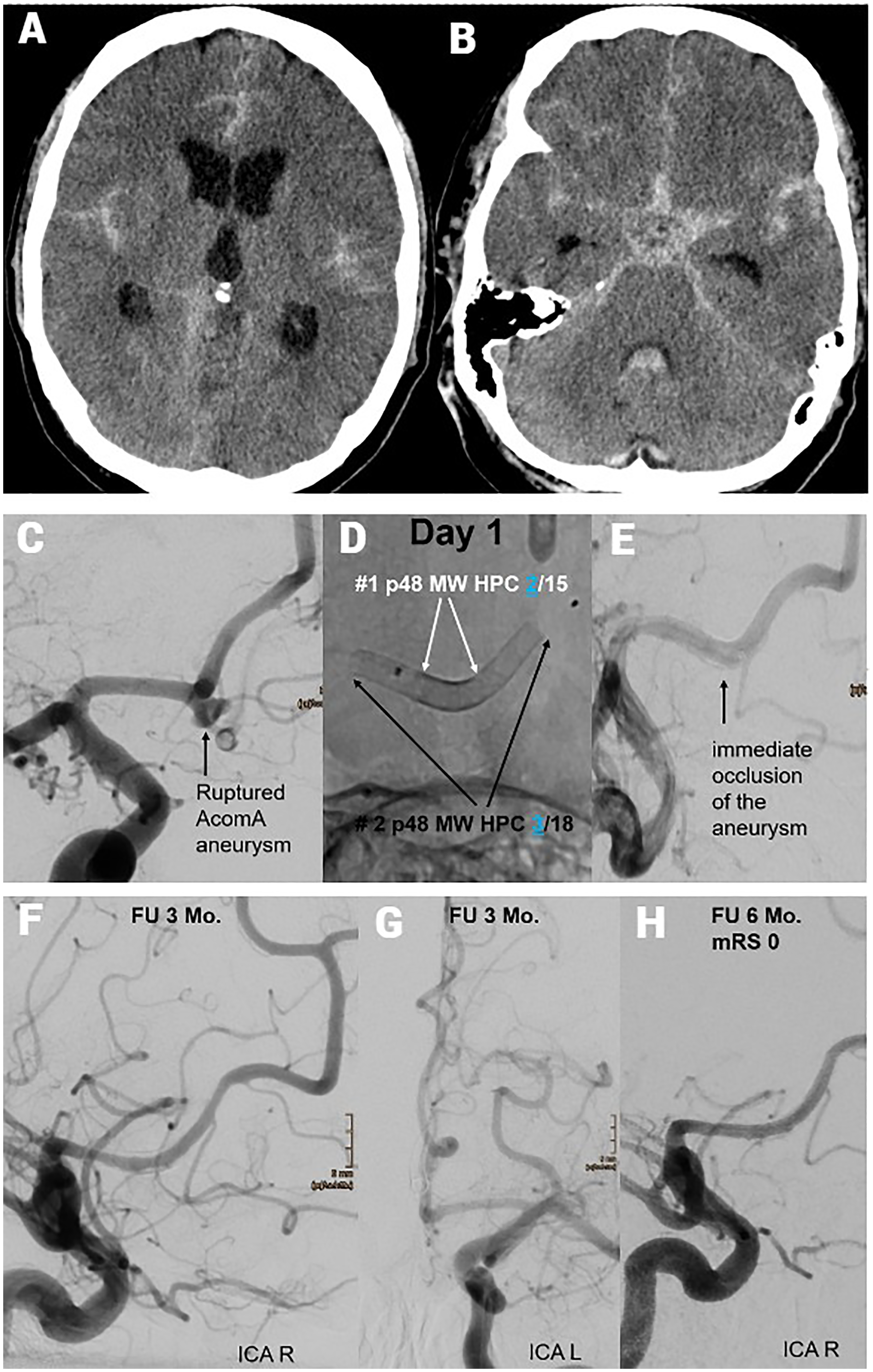
Endovascular treatment of a ruptured AcomA aneurysm using the PANTHER technique with non-identical telescoping FDs under prasugrel SAPT. (A) Initial axial non-contrast CT showing diffuse SAH, most prominently in the interhemispheric fissure. (B) Axial CT image at a lower level confirms SAH with additional haemorrhage in the basal cisterns. (C) Baseline DSA reveals a ruptured wide-necked saccular aneurysm at the AcomA. (D) Day 1: Deployment of two p48 MW HPC FDs in a non-identical telescoping configuration as part of the PANTHER technique. (E) Immediate post-procedural angiography demonstrates near-complete flow stagnation and early occlusion of the aneurysm. (F, G) Follow-up DSA at 3 months from right (F) and left (G) ICA injections shows complete aneurysm occlusion and preserved parent vessel patency. (H) Six-month follow-up confirms durable occlusion with no evidence of recurrence; the patient achieved an mRS score of 0.
Medication
In order to avoid potential rebleeding complications, particularly in cases where antiplatelet agents are being used, treatment was primarily conducted following the insertion of an EVD. Patients who did not have an EVD were therefore referred to the intensive care unit for its insertion, after which a loading dose of 30–60 mg of prasugrel was administered orally via a gastric tube 3 h before implantation. Treatment was initiated after evaluation of antiplatelet inhibition with a Multiplate analyzer (MP) or VerifyNow (VN).
In patients who had previously undergone EVD, a body-weight-adjusted bolus of Glykoprotein-IIb/IIIa-Rezeptorantagonist (Eptifibatide until the end of 2023, then switch to tirofiban due to discontinuation in the European Union) was administered intravenously during the procedure before FD implantation, and a loading dose of 30–60 mg prasugrel was administered post-implantation, with an overlapping, body-weight-matched continuous intravenous infusion of Glykoprotein-IIb/IIIa-rezeptorantagonist for 4 h. The administration of the glycoprotein IIb/IIIa receptor antagonist bolus and infusion served only to bridge the time until the P2Y12 receptor antagonist prasugrel (SAPT) takes effect after oral administration of the loading dose.
Post-procedurally, patients were administered 2 × 20 mg oral prasugrel daily, and platelet inhibition was evaluated with an MP or VN daily in the first week. The prasugrel dosage was optimized or maintained, as indicated by the test results.
Follow-up
Planned angiographic examinations were conducted at the following FU times: early (3–6 months), mid-term (9–18 months), and long-term (>19 months). Aneurysm occlusion was evaluated with the O'Kelly–Marotta (OKM) scale. 11 Neurological examinations were conducted by a neurologist or a certified stroke nurse periprocedurally, as well as during early and long-term FU (postprocedural period). The modified Rankin Scale (mRS) was used to evaluate outcomes. 12
Results
Patients and aneurysm characteristics
PANTHER group
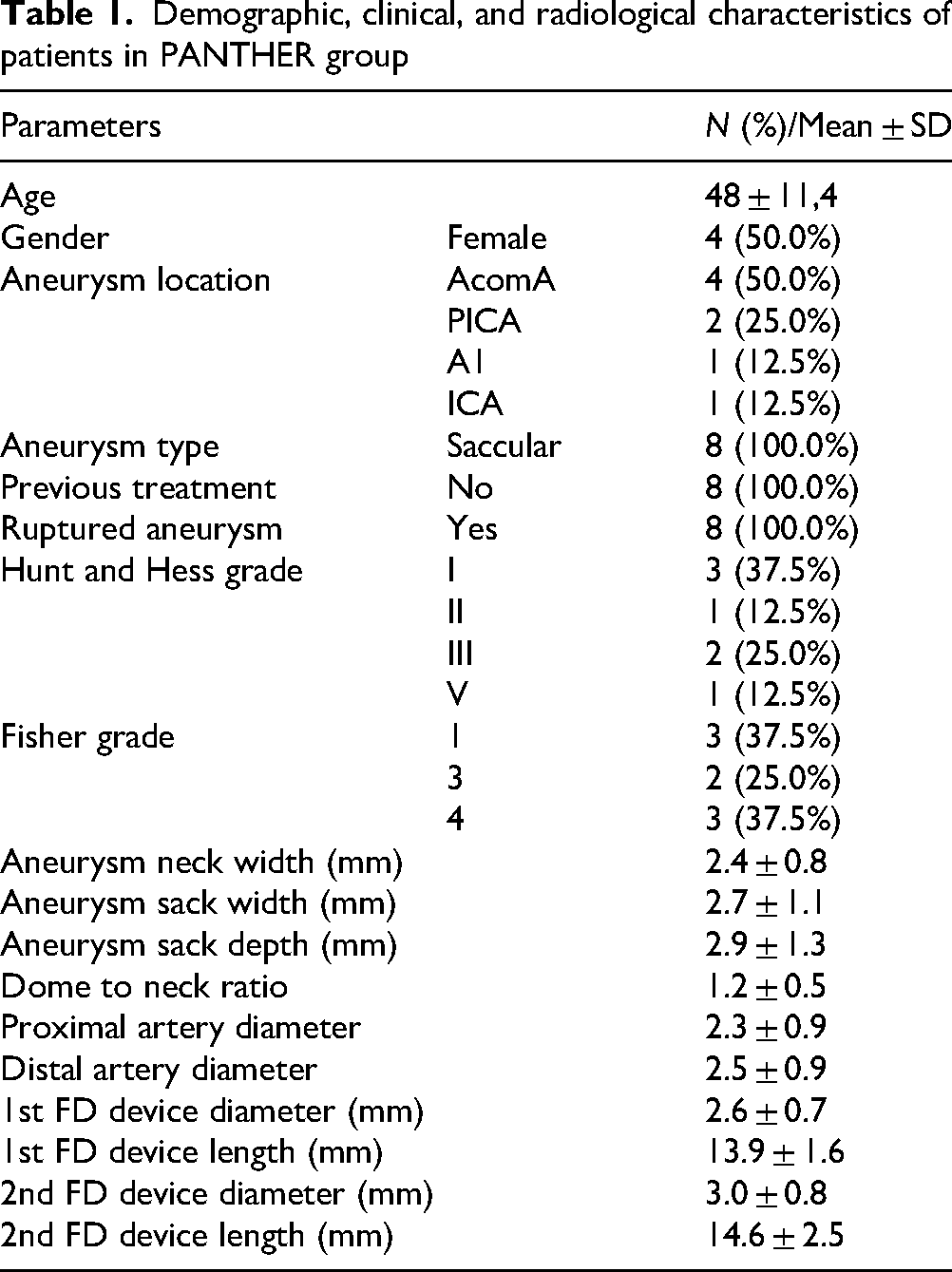
Between June 2021 and January 2024, eight patients underwent treatment of ruptured aneurysms through SAH with the PANTHERtechnique. The average patient age at presentation was 48 years. The aneurysms had a mean maximum diameter of 3.4 mm. The aneurysm locations were as follows: AcomA (n = 4), PICA (n = 2), A1 (n = 1), and ICA (n = 1). Six patients were treated with p48 MW HPC, and two patients were treated with p64 MW HPC. Demographic data, aneurysms, and FD characteristics of this group are summarized in Table 1.
Demographic, clinical, and radiological characteristics of patients in PANTHER group
Single FD group
In the period spanning June 2018 to January 2024, a total of twenty-one patients underwent treatment for ruptured aneurysms via a single FD. The mean age of patients at the time of presentation was 54.9 years. The mean maximum diameter of the aneurysms was found to be 3.1 mm. The locations of the aneurysms are outlined below: AcomA (n = 2), A1 (n = 2), A3 (n = 1), MCA (n = 1), ICA (n = 3), BA (n = 6), PCA (n = 2), PICA (n = 1) and V4 (n = 3). Twelve patients were treated with p48 MW HPC, and nine patients were treated using p64 MW HPC.
Endovascular treatment
All procedures in the PANTHER group were technically successful and performed using a short sheath (Terumo, Tokyo, Japan) with a 7F guiding catheter (Guider Softip, Stryker, Kalamazoo, MI, USA) in six cases (75%) or a 6F guiding catheter (Envoy MPC, Cerenovus, Irvine, CA, USA) in two cases (25%). An intermediate catheter (5F Navien A + 058, Medtronic, Dublin, Ireland) was used in 75% of cases. All FDs were deployed through a 0.021-inch microcatheter, in sevcn cases (87.5%) using Headway 21 (Terumo Neuro) and one case using Rapidtransit microcatheter (Envoy MPC, Cerenovus, Irvine, CA, USA).
In the PANTHER group, 37.5% of patients (3/8) and in the single FD group, 23.8% (5/21) received a glycoprotein IIb/IIIa receptor antagonist bolus and infusion for 4 h during treatment, in addition to the prasugrel bolus.
Angiographic follow-up
PANTHER group
Regular angiographic FU examinations were possible in all cases. Early FU (FU1, 3–6 months) was performed at a median of 5 months post-procedure. Complete aneurysm occlusion (OKM D) was observed in all cases (100%). Mid-term and long-term FU were performed in 75% and 25% of cases, at median time intervals of 12 and 22 months, respectively. In comparison, the complete occlusion rate immediately after the treatment session was 25% (2/8). In addition, 37.5% of patients presented with OKM A, and another 37.5% with OKM B.
Single FD group
Angiographic FU examinations were not possible in 14.2% of patients (3/21), due to complications after treatment in one patient and mortality before discharge and in the first six months in the other two. Early FU (FU1, 3–6 months) was performed at a median of 6 months post-procedure and showed a complete aneurysm occlusion rate (OKM D) of 77.8% (14/18). Mid-term and long-term FU were performed in 8.3% and 61.1% of cases, respectively (median 13 and months). In the early follow-up (FU), two of the four not complete occluded aneurysms demonstrated an OKM D. In the long-term FU, all of the aneurysms were found to be complete occluded (OKM D: 89.7% at mid-term FU and 100% at long-term FU). The complete occlusion rate immediately following the implantation of the FD was 14.2% (3/21). The distribution of patients according to their degree of angiographic filling (OKM) is as follows: 47.6% presented with OKM A, 28.6% OKM B, and 9.5% OKM C.
Clinical follow-up
PANTHER group
Treatment with FD was not the cause of clinical deterioration in any cases. The patients exhibited an average mRS score of 3.8 at admission, which decreased to 1.2 at discharge. In the post-procedural period, no patients showed clinical deterioration, and all patients exhibited an mRS score of 0 to 1 at the first FU and after the first 12 months.
Single FD group
Two patients perished due to poor conditions following the SAH. One patient was discharged from hospital care with severe neurological deficits, an mRS score of 4, due to ischaemic complications that occurred after FD implantation. The MP and VN tests revealed hyporesponsiveness to SAPT (10 mg prasugrel daily), despite confirmation of the prasugrel effect prior to the initiation of treatment. Absent other contributing factors, no other patients exhibited clinical deterioration as a consequence of FD treatment or following discharge.
Technical difficulties and complications
No technical difficulties in any treatments were encountered with FD implantation with the PANTHER technique.
Despite the treatment of patients with ruptured aneurysms and the use of antiplatelet agents, no hemorrhagic complications or aneurysm re-rupture was documented. In contrast, no instances of ischemic or thromboembolic complications were documented during or after the procedure. At the early FU, intimal hyperplasia was observed in two patients. However, this hyperplasia was no longer evident at the mid-term FU. In one patient, complete occlusion of the covered vessel was detected at the first FU, and in another patient, the covered vessel was found to have become thinner. Ischemic lesions were not observed in the supplied area of the covered vessel in both patients.
The aforementioned ischemic complication was documented in the single FD group. No further instances of thromboembolic or hemorrhagic complications were observed.
Discussion
In recent decades, FDs have played an increasingly major role in a variety of neurointerventional treatments, particularly the treatment of intracranial aneurysms. 13 Furthermore, FD treatments are now used in a progressive manner for management of ruptured aneurysms, because of several factors, most notably advances in coatings and the ability to implant FD under monoplatelet inhibition, thereby minimizing the risk of hemorrhagic complications.9,10 Despite developments in neurointerventional techniques, coiling continues to be the preferred endovascular treatment for patients with ruptured aneurysms. 13 Nevertheless, the management of ruptured wide-neck intracranial aneurysms remains challenging in patients with SAH. For these treatments, the use of clipping and additional devices, such as remodeling balloons or self-expandable stents, may be considered. 14 The management of wide-neck intracranial aneurysms is complicated, and these aneurysms, particularly those that are small or flat, have high complication rates even when these devices are used. 15 Aneurysms of this type, as well as ruptured fusiform, blistering, or dissecting aneurysms, are reliable indications for FD treatment, even when patients have experienced SAH.9,16
Nonetheless, the use of FD for ruptured aneurysms remains debated. As with most potential devices, DAPT is required, despite the development of numerous coatings for implantation, to avoid thromboembolic complications, which can concomitantly increase the risk of hemorrhagic complications in patients with ruptured aneurysms. Historically, a dilemma has long existed in attempting to avoid ischemic complications and to prevent potential bleeding.17,18 HPC, a hydrophilic multilayer glycan-based polymer coating that can be applied to nitinol surfaces, is currently the only coating indicated for use with SAPT. Several recent studies have investigated the safety of this coating in various brain vessels, including the posterior and anterior circulations, and the proximal and distal cerebral vessels. Furthermore, the use of prasugrel SAPT has been examined in the context of ruptured aneurysms.9,10,19 The application of prasugrel in neurovascular cases, particularly in acute instances such as stroke or cerebral hemorrhage, is considered controversial by many neurointerventionalists, because of its association with elevated complication risk. However, our research team's publications have demonstrated the safety of this medication treatment strategy in both unruptured and ruptured aneurysms.9,10,19 Moreover, the frequency of hemorrhagic complications during surgical procedures under prasugrel-SAPT is low. 20
Both treatments, with a single FD and the PANTHER technique, may offer a potential treatment solution for ruptured aneurysms of various morphologies and sizes. These methods do not require navigation into the aneurysm fundus, thereby posing a lower risk than alternative methods requiring the implantation of coils or intrasaccular devices for the treatment of small and flat aneurysms. 21 The PANTHER technique creates an overlap of two FDs at the neck of the aneurysm to achieve a stronger flow modulation effect. In addition, the use of two FDs with different diameters results in heterogeneous coverage and flow through the devices, and thus decreases porosity and increases modulation efficacy.
Consequently, the PANTHER Technique has the potential to offer an additional advantage over the single FD technique in cases of ruptured aneurysms that cannot be adequately or completely covered with an FD due to the potential special anatomy and position of the aneurysm neck or the carrier vessel.
PANTHER offers several potential technical advantages. The implantation of an undersized FD, accompanied by targeted shortening of the device at the neck of the aneurysm, is a strategy employed to achieve a significant decrease in porosity and enhance efficacy. The subsequent implantation of the oversized FD has been shown to result in enhanced wall adaptation and a reduction in the risk of shortening or incomplete aneurysm coverage. These phenomena are particularly likely in patients with ruptured aneurysms because of the post-procedural regression of possible vasospasms and the corresponding widening of the vessel diameter. The overlap of the undersized and oversized FDs at the aneurysm's neck results in heterogeneous mesh coverage, thereby enhancing the flow diversion effect. It is important to note that the HPC coating has been demonstrated to reduce the risk of ischemia and thrombogenicity, thereby enabling implantation under SAPT (prasugrel).
Immediately following implantation of the FD, both groups exhibited a low rate of complete occlusion, with 25% in the PANTHER group and 14.2% in the single FD group. Despite the initial low occlusion rate, no instances of aneurysm re-rupture were documented.
In the early FU, 77.8% of ruptured aneurysms treated with a single FD demonstrated complete occlusion. However, it should be noted that all eight aneurysms in the PANTHER group were already completely occluded. The disparity in question diminished as the first year progressed. The ruptured aneurysms that were treated with a single FD demonstrated a complete occlusion rate of 88.9% by the end of the first year. In the long-term follow-up, complete occlusion was observed in all ruptured aneurysms.
The occlusion rate of 88.9% after 1 year in the group treated with a single FD appears comparable and, in some cases, slightly higher than the results of previous FD studies, such as the PUFS study, which recorded an occlusion rate of approximately 93.4% in the first year. Hanel et al. and Pierot et al. have reported marginally lower closure rates of 81.9% with the Pipeline device and 73.3% with the FRED device, respectively.14,22
A comparison of these results with the PANTHER group suggests that the enhanced flow diversion effect achieved with the PANTHER technique may expedite aneurysm occlusion and potentially enhance safety in ruptured aneurysms, particularly those with a wide neck and challenging anatomical configuration. However, the limited number of patients treated with the PANTHER technique does not permit the establishment of definitive statements from this comparison.
Furthermore, the ischemic or thromboembolic complication rates were found to be comparable in both groups. Despite the elevated material usage and the flow diversion effect, the PANTHER group exhibited no increase in the complication rates under the same medication strategy. Consequently, the PANTHER technique does not appear to increase the complication rates in the first experiences. The only ischemic complication documented in this cohort was due to a hyporesponse to the SAPT (Prasugrel). Consequently, the SAPT adaptation in patients with ruptured aneurysms remains a major aspect in these treatments.
The rate of intima hyperplasia was relatively low in the PANTHER group, at 25% at the early FU, and these cases demonstrated regression in subsequent FUs. None of these cases was considered to be hemodynamically or clinically relevant.
Nevertheless, the use of software and algorithms, which have been carefully engineered to enhance the precision of the device selection process, has enabled optimization of FD treatment outcomes and facilitated safer device implantation, even in small vessels.23,24 Furthermore, innovative corrective maneuvers are being developed for inadequately inserted FDs with simple-to-insert stents, such as the Stent Retriever or Comaneci device.25,26
Despite major advancements in treatment methods for ruptured aneurysms, including both micro-surgical and endovascular approaches, these interventions remain challenging and are associated with considerable complication rates in certain cases.
The objective of this study was to present the PANTHER technique as an additional treatment option, particularly for complicated aneurysm constellations. In our preliminary experience, the PANTHER technique appeared feasible and resulted in high early occlusion rates with favorable clinical outcomes. In addition, it is technically straightforward to use, in contrast to other methods, which can be more complicated and even dangerous in certain cases. Larger studies are needed to validate these findings.
Limitations
The study's limitations are attributed to its retrospective design, limited sample size, and incomplete long-term follow-up due to the relatively short time between some of the treatments and the study. Furthermore, the comparison with single-FD cases is purely descriptive and exploratory, without statistical testing, due to the limited number of cases and, consequently, the significance level. In addition, the results obtained are applicable only to HPC-coated FDs.
Conclusion
In our preliminary, single-center experience, the PANTHER technique appeared to be both feasible and promising for the treatment of ruptured intracranial aneurysms that were unsuitable for coiling in the setting of SAH. The technique achieved fast occlusion and favorable clinical outcomes under prasugrel SAPT, without hemorrhagic or ischemic complications observed. These findings, in conjunction with a descriptive comparison to single FD cases, suggest a potential hemodynamic benefit of the non-identical telescoping constructs. However, the need for larger prospective studies is evident to confirm these results and define the role of PANTHER in the management of ruptured aneurysms.
Footnotes
Abbreviations
Acknowledgments
None.
Author contributions
Conceptualization: Ali Khanafer; methodology: Ali Khanafer, Philipp von Gottberg, Abdallah Aburub; formal analysis and investigation: Ali Khanafer, Kamran Hajiyev, and Mohammad Almohammad; writing—original draft preparation: Ali Khanafer; writing—review and editing: Michael Forsting, Oliver Ganslandt, and Hans Henkes; resources: Ali Khanafer, Kamran Hajiyev, Philipp von Gottberg, and Abdallah Aburub; supervision: Hans Henkes.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
H. Henkes: consulting and proctoring for WallabyPhenox GmbH, co-owner of CONTARA GmbH. A. Khanafer: consulting and proctoring for WallabyPhenox GmbH. The authors declare that they have no further potential conflicts of interest.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Research involving human participants and/or animals
The study was conducted in accordance with the Declaration of Helsinki. Ethical approval was obtained from the institutional review boards of the Ethik-Kommission der Landesärztekammer Baden-Württemberg in Stuttgart (Reference No: F-2018-080).
