Abstract
Background
Intracranial vessel perforation is a rare but life-threatening complication during endovascular neurointerventions. Despite several described bail-out strategies, there is no consensus on the most effective approach for rapid hemorrhage control. This study aimed to systematically compare common endovascular rescue techniques in a standardized experimental setting.
Methods
Patient-specific, 3D-printed vascular models of the anterior circulation were used to simulate standardized vessel perforations at two anatomical sites and three perforation sizes. The impact of the anterior communicating artery (AComA) crossflow was assessed. Proximal balloon guiding catheter inflation, local compliant balloon inflation, temporary coil deployment, and distal intermediate catheter insertion were evaluated against a non-intervention control. The primary outcome was the time it took for 50 ml of fluid extravasation to accumulate.
Results
As expected, extravasation time inversely correlated with perforation size (ρ = –0.95, p < 0.001). Local balloon inflation at the rupture site most effectively halted leakage. Proximal balloon occlusion was only effective in the absence of an AComA crossflow (p = 0.02). Temporary coil deployment modestly slowed bleeding, especially for small-to-moderate perforations. Distal intermediate catheter placement had no significant effect.
Conclusion
Managing iatrogenic intracranial vessel perforation is time-critical, as even a brief bleeding time can be fatal. The most effective hemostasis method is compliant balloon inflation at the rupture site. If unavailable, temporary parent artery coiling can reduce bleeding while maintaining perfusion, but it is less effective for larger perforations. Proximal balloon occlusion is only effective in the absence of collateral crossflow.
Introduction
Endovascular techniques have revolutionized the treatment of intracranial vascular diseases by offering a less invasive alternative to open surgery.1–3 Likewise, mechanical thrombectomy has become the standard of care for acute large-vessel ischemic strokes, greatly increasing the volume of neurointerventional procedures. 4 However, intracranial vessel perforation remains one of the most dreaded complications during such interventions. 5 Previous publications report intraprocedural perforation in 1% to 5% of endovascular treatments. However, these events carry up to a 50% risk of severe disability and even mortality.6–14 The clinical consequences of an iatrogenic vessel perforation range from minor contrast extravasation into the subarachnoid space to fulminant hemorrhage with intracranial hypertension, herniation, and even a fatal outcome. 15 Prompt, effective management of an intraprocedural perforation is critical, but decision-making must occur within seconds in a high-pressure environment. Initial general measures include immediate reversal of heparin anticoagulation to promote clotting and aggressive blood pressure reduction to mitigate ongoing bleeding. 15 Ultimately, achieving rapid mechanical hemostasis is the cornerstone of rescue therapy. 6 This typically requires endovascular “bail-out” maneuvers to physically stop the bleeding at its source while maintaining cerebral perfusion. A number of bail-out techniques have been described in the neurointerventional literature for vessel perforations. Balloon occlusion is a frequently employed strategy. Inflating a balloon catheter, either a proximal balloon-guiding catheter in a parent artery or a compliant microballoon across the perforation site, can temporarily halt arterial flow and facilitate hemostasis.6,16 Temporary deploying coils within the parent vessel have been used to create a subocclusive effect that slows flow and encourages clot formation. 17 In certain cases, sacrificing the affected vessel may be unavoidable in order to save the patient's life. 18 Each of these approaches has been documented in case series or reports, but no clear consensus exists on the optimal sequence or combination of techniques for a given scenario. Yet, current evidence is derived predominantly from retrospective observations, and there are no standardized guidelines as of yet to direct practitioners in this high-stakes emergency. To address this knowledge gap, we designed an experimental study to systematically compare the efficacy of commonly used bail-out procedures following a standardized intracranial vessel perforation, using a controlled bench-top model. Our aim is to evaluate and compare the relative performance of each technique to identify the most effective interventions for managing this critical complication in emergency settings.
Materials and methods
Concept
Four commonly used endovascular rescue techniques following vascular perforation were compared using patient-specific, 3D-printed vascular models. The primary outcome was the time required for 50 ml of extravasated fluid to accumulate, representing a critical threshold volume in the context of intracranial hemorrhage. 19
Perforations were induced in three different diameters: 0.35 mm, 0.57 mm, and 0.87 mm, mimicking the outer diameters of a standard 0.014″ microwire, 0.017″ microcatheter, and 0.027″ microcatheter, respectively. Two anatomical locations were selected as perforation sites: the apex of the internal carotid artery terminus (ICA-T) and the bifurcation of the middle cerebral artery (MCA). Furthermore, the impact of crossflow via the anterior communicating artery (AComA) on extravasation dynamics was assessed.
Vessel model fabrication
To construct the vascular models, a representative computed tomography angiography dataset of the anterior circulation was selected and segmented using MeVisLab 3.4.1 (MeVis Medical Solutions AG, Germany). Using Blender 4.1 (Blender Foundation, Netherlands), the model was reduced to include the internal carotid artery (ICA), MCA, and anterior cerebral artery up to the M2 and A2 segments, respectively. The inner diameter of the distal ICA measured 3.3 mm, while the proximal M1 segment measured 2.8 mm and the distal M1 segment 2.6 mm. The arterial branches were then merged into a single outflow per hemisphere. The model was mirrored and connected across the midline at the anatomical location of the AComA. Two configurations were printed. The first model type, “non-AComA” (non-crossflow variant), consisted of a solid bridge present at the AComA location without a patent lumen. Model type “AComA” (crossflow variant) consisted of a connecting lumen of 0.7 mm diameter, enabling flow communication between both hemispheres. For each combination of perforation site and diameter, one model was fabricated for each configuration (AComA and non-AComA), resulting in a total of 12 3D-printed vessel models. Models were printed using a Form 3+ stereolithography printer (Formlabs Inc., USA) with Formlabs Clear Resin V4.
Perforations were then drilled into the right side of each model at predefined, uniformly marked sites during the 3D printing process, according to the specified diameters. An exemplary vascular model, including perforation sites, is shown in Figure 1.
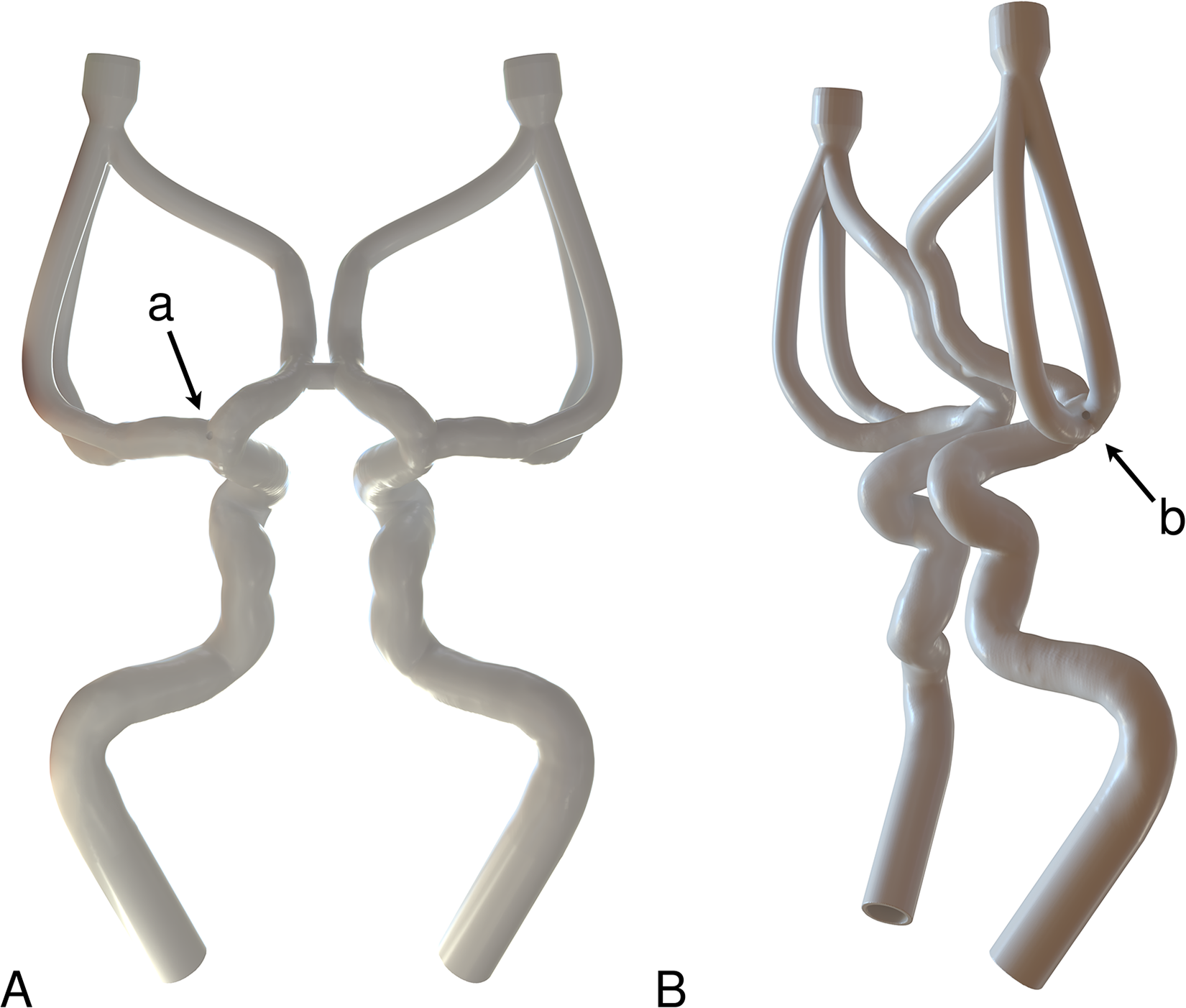
Rendered vascular model in frontal (A) and oblique-lateral (B) view, illustrating the perforation at the apex of the internal carotid terminus (a) and at the bifurcation of the middle cerebral artery (b).
Test chamber construction
An acrylic container with a total volume of 2100 cm³ was filled with 2050 cm³ of water, into which the vascular models were mounted. The inlet and outlet vessels were connected via silicone tubing to fixed valves mounted on the external walls of the container. The top of the container was removable for the replacement of the vascular models. Additionally, a vertical chimney with an open top and a base area of 10 cm² was mounted on the container lid. This way, 1 mm corresponded to 1 ml. A scale ranging from 1 to 50 ml in 1 ml increments was marked on the chimney. The initial vertical distance between the artificial perforation sites and the lower start marking was approximately 110 mm, and 160 mm to the upper end marking. On average, the 135 mm water column above the perforations generated a realistic intracranial pressure of approximately 10 mmHg. Perfusion of the vascular models was achieved via silicone tubing and a Y-connector connected to a Watson-Marlow 520S peristaltic pump (Watson-Marlow GmbH, Germany), delivering 400 ml/min of artificial blood, which consists of a 60 : 40 water–glycerol mixture. 20 The setup was calibrated to maintain an intraluminal pressure of 80 mmHg. Each of the two outlets was fitted with silicone tubing containing a valve to prevent backflow of the artificial blood into the vessels, thereby avoiding any additional, unintended leakage through the perforation site. An overview of the test setup is provided in Figure 2.

Experimental setup showing the test chamber, including the measurement chimney (a), the mounted vascular model (b), the corresponding inlet (c) and outlet (d), the Watson-Marlow 520S peristaltic pump (Watson-Marlow GmbH, Germany) (e), the fluid reservoir (f), and the access tubing with a hemostatic valve for device insertion (g).
Bailout procedures
Four bail-out procedures were compared with a non-intervention control group. For this purpose, device positions were uniformly marked on all vascular models, and each underwent the measurement process sequentially. A balloon guiding catheter (BGC) (8F FlowGate2, Stryker, USA) was fully deployed in the subpetrosal segment. Furthermore, a compliant occlusion balloon catheter (TransForm 4 × 15 mm, Stryker, USA) was deployed centrally at the level of the perforation site in the ICA-T and MCA. Balloon pressure was continuously monitored using a manometer and adjusted as necessary. In subsequent measurement series, a temporary coil was placed at the same location (Target 360 SOFT 3 mm × 10 cm; Target 360 SOFT 7 mm × 10 cm, Stryker, USA), and a 5F intermediate catheter (5F SOFIA, TerumoNeuro, USA) was introduced into the M2 segment of the MCA. An illustrative case of leakage in the model and its procedural effects is presented in Figure 3, while a clinical example of arterial perforation is shown in Figure 4.
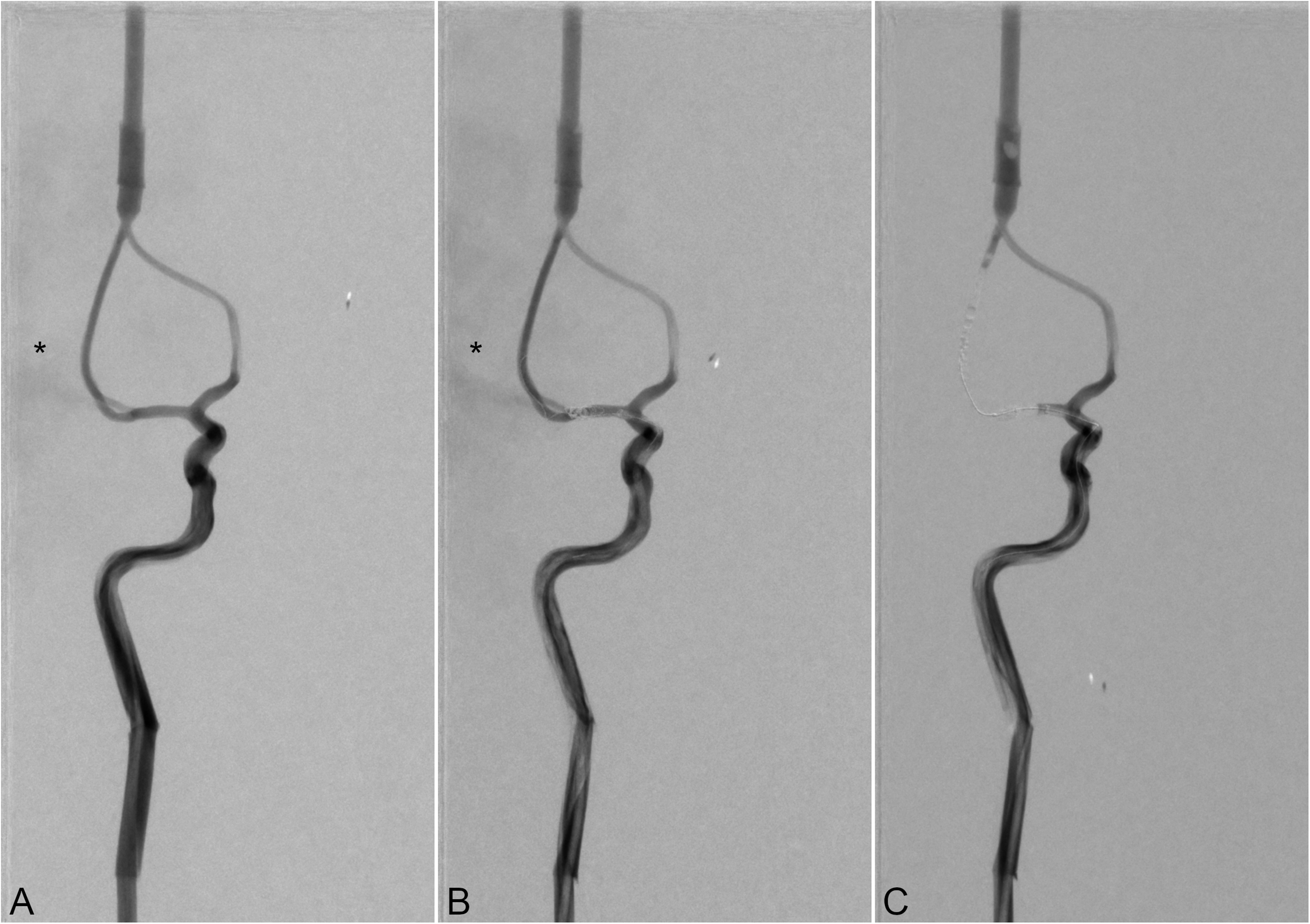
Digital subtraction angiography images in anteroposterior view of extravasation comparison in a non-AComA model with a 0.35 mm perforation at the middle cerebral artery bifurcation. Extravasation (asterisk) is observed without intervention (A) and after temporary coiling (B). No extravasation is seen following compliant balloon inflation at the perforation site (C).
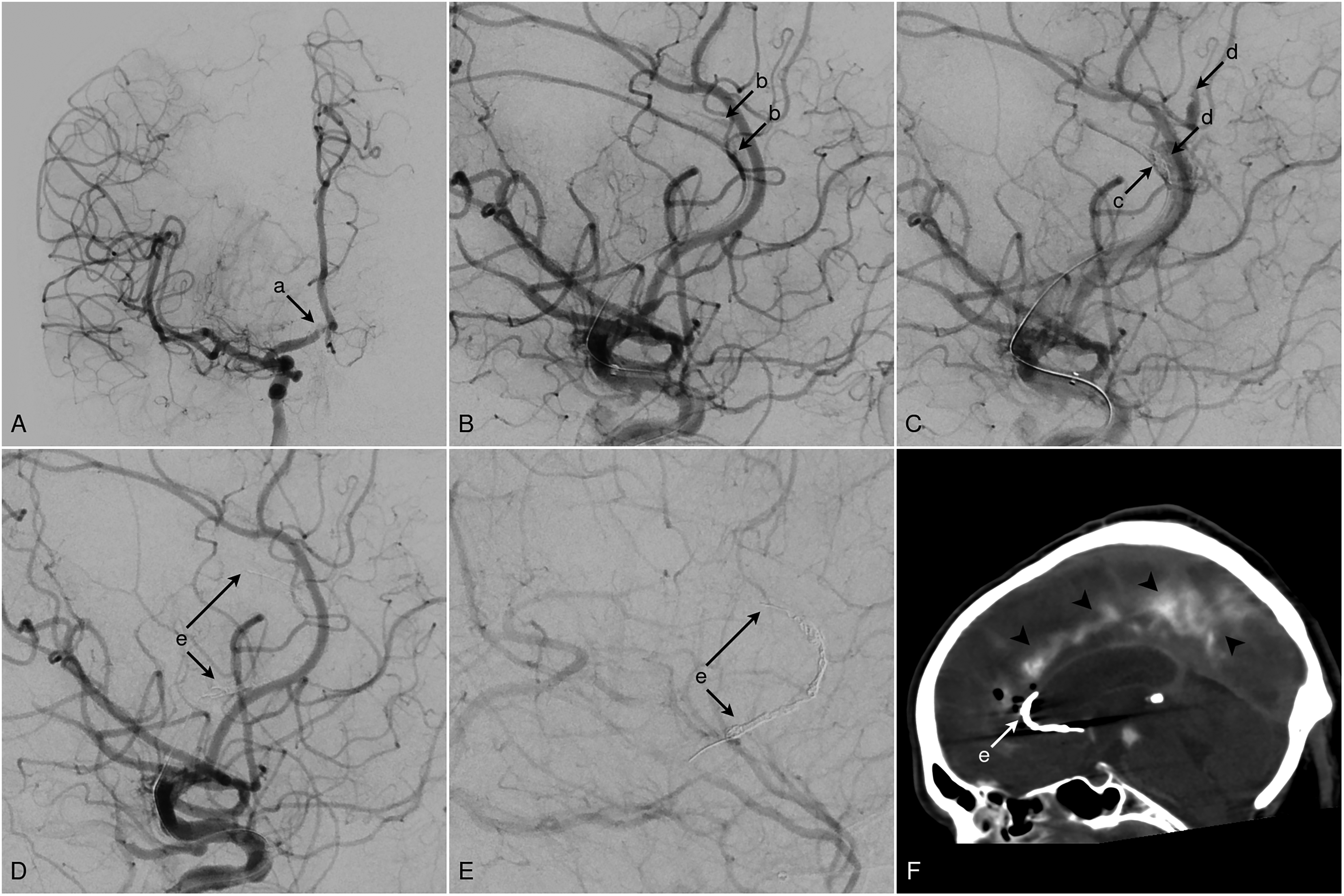
Patient in their 70s with an occlusion of the distal A1 segment of the right anterior cerebral artery. (A) Occlusion of the distal A1 segment (a). (B) During navigation beyond the thrombus into the A2 segment using a 0.014-inch microwire and a 0.017-inch microcatheter for mechanical thrombectomy, a wire perforation occurred, resulting in contrast extravasation (b). (C) Immediate temporary coiling was performed through the already positioned microcatheter (c). Control angiogram after 3 min still demonstrated persistent extravasation (d). Additional coils were deployed, resulting in the sacrifice of the A2 segment (e). The final control angiograms showed no further extravasation in either the arterial (D) or late venous phase (E). (F) Post-procedural NECT demonstrated a mild subarachnoid hemorrhage (arrowheads) and the placed coils in the A2 segment (e).
Leak measurement protocol
For each combination of model, perforation size, and location, the pump was calibrated and set to deliver a mean flow rate of 400 ml/min and an intraluminal pressure of 80 mmHg. As the water–glycerol mixture extravasated through the drilled defect, the fluid level in the tank's chimney column rose accordingly. Each measurement was recorded using a high-definition camera. The primary outcome measure was the time required for a leakage volume of 50 ml to accrue, as indicated by the chimney scale. The maximum duration per measurement was limited to 1800 s. Timing was determined by frame-by-frame video analysis. After each measurement, the water–artificial blood mixture was drained, and the container was refilled with water to the initial level marked on the chimney. All measurements were performed three times.
Statistical analysis
For the statistical analysis, SPSS 29 (IBM, Redmond, USA) was used. Continuous variables are listed as mean ± standard deviation (SD), or median and interquartile range (IQR). Depending on the normal distribution, the unpaired t-test or the Mann–Whitney U-test was used for differences between two groups for continuous variables. The Kruskal–Wallis test, followed by Dunn's post hoc analysis, was used to compare the dependent variable across independent groups, as the data did not meet the assumptions of normality and homogeneity of variances. The correlation between time and perforation site was assessed using Spearman's rank correlation coefficient. Statistical significance was described as p-values < 0.05.
Results
Across all models in the non-intervention group, there was a strong negative correlation between perforation size and the time required to reach an extravasation volume of 50 ml. As the perforation size increased, the time needed to reach 50 ml of extravasation decreased significantly (ρ = –0.946, p < 0.001). The shortest time observed in the non-intervention group coincided with a perforation size of 0.87 mm at the ICA-T in the presence of an AComA. The most effective method to limit extravasation was balloon inflation of a BGC in the ICA in non-AComA models, as well as the placement of a compliant balloon directly above the perforation site, both in the ICA-T and MCA bifurcation, as well as in AComA and non-AComA models. In these cases, the experiment proceeded as predefined to the maximum observation time of 1800 s, during which extravasation remained below 1 ml.
In AComA models, inflation of a BGC in the ICA resulted in a mean time of 76.4 s (SD ± 61.9), which was significantly shorter compared to 1800 s (SD ± 0) in non-AComA models (p = 0.02). Across all models, perforation size was significantly associated with differences in the time needed to reach 50 ml of extravasation. With the smallest perforation size of 0.35 mm, the median time was 151.3 s (IQR 134–1402), compared to 60.0 s (IQR 46–1375) at 0.57 mm (p = 0.01), and 35.3 s (IQR 29–1361) at 0.87 mm (p < 0.001). The location of the perforation did not show a significant difference in extravasation time between the ICA-T (99.5 s; IQR 35–1388) and the MCA bifurcation (113.4 s; IQR 44.4–1402) (p = 0.26). The results are presented in Table 1. Each data point represents the mean of three repeated measurements. A detailed breakdown of individual measurements and corresponding standard deviations is provided in Supplemental Table 1.
Comparison of mean extravasation time between intervention groups and the non-intervention group, stratified by localization.
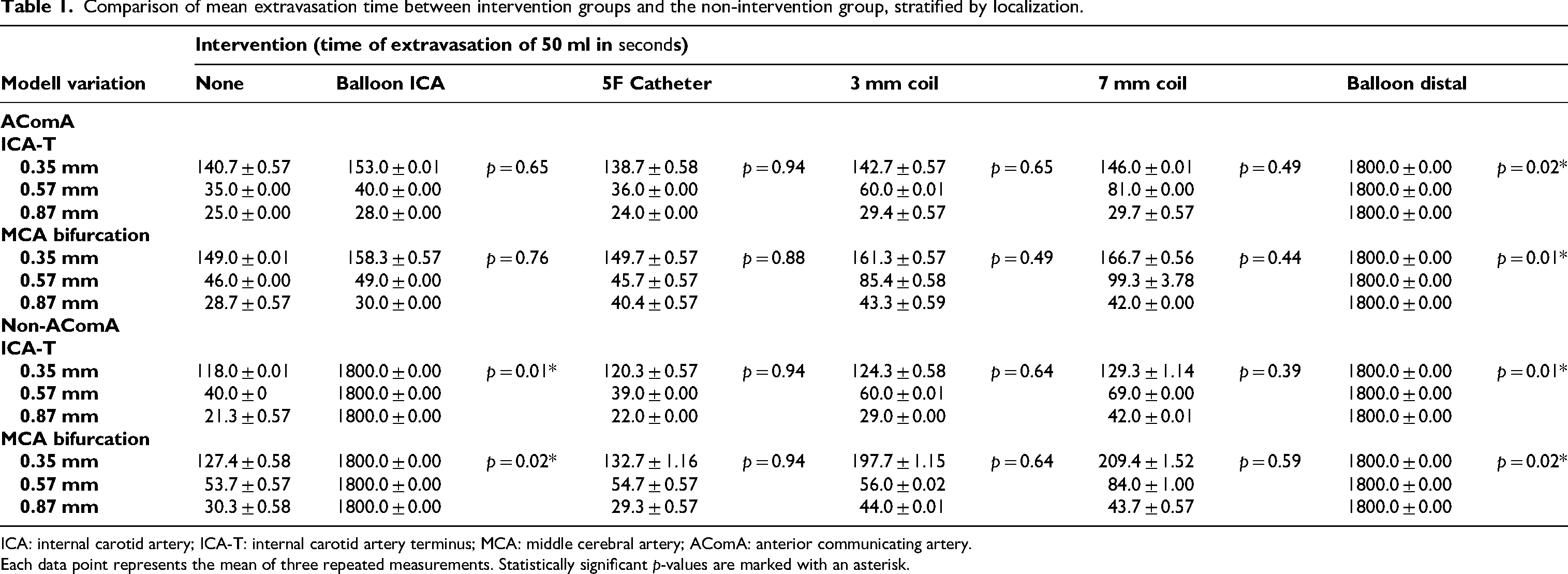
ICA: internal carotid artery; ICA-T: internal carotid artery terminus; MCA: middle cerebral artery; AComA: anterior communicating artery.
Each data point represents the mean of three repeated measurements. Statistically significant p-values are marked with an asterisk.
Discussion
Vessel perforation during endovascular procedures remains one of the most feared complications due to its high morbidity and mortality.8,17,21,22 Even relatively small intracranial hemorrhages can lead to poor outcomes depending on their location. For intraparenchymal intracerebral hemorrhages, location-specific volume thresholds have been described. Volumes exceeding as little as 6 ml in strategic deep locations up to >48 ml in lobar regions are associated with significantly worse clinical outcomes. 19 In the context of subarachnoid hemorrhage (SAH), an accumulated SAH volume of ≥10 ml has been linked to a higher risk of unfavorable prognosis. 23 These findings emphasize that even modest bleeding, if in critical areas or in the subarachnoid space, can result in a dire outcome. Optimal management of an iatrogenic perforation, therefore, demands rapid hemostasis while preserving the parent artery patency in order to minimize hematoma expansion without causing ischemia in the territory supplied by that vessel. This principle is echoed in prior literature on intraprocedural ruptures, which emphasizes immediate control of bleeding in tandem with endovascular techniques that stop the hemorrhage, but ideally maintain distal perfusion.6,8,17 In our experimental study, we simulated arterial perforations and observed an inverse relationship between perforation size and the time to critical extravasation volume. Using the upper volume cutoff values from clinical studies as benchmarks, we found that in the nonintervention scenario, an extravasation volume of 50 ml was reached within 21.3 s for a large perforation (0.87 mm). By contrast, with a smaller perforation (0.35 mm), the same volume was reached in 149.0 s. In other words, larger vessel injury leads to fast extravasation, whereas very small perforations allow slightly more time before reaching volumes associated with poor outcomes. Nevertheless, even in the most favorable case, roughly 2.5 min was the maximum time before a 50 ml extravasation accumulated. This narrow window highlights the urgency with which immediate measures are required to control extravasation regardless of perforation size. Our findings mirror clinical data that prolonged active bleeding correlates with worse outcomes. In a recent retrospective multicenter study of 459 thrombectomy cases with vessel perforation, longer hemorrhage duration was strongly associated with mortality and disability. 21 In that series, median observed bleeding durations ranged roughly from 6 to 11 min before hemostatic control was achieved, and active bleeding beyond 20 min was linked to significantly poorer survival. 21 Direct comparison with our experimental study is limited. The cited study also did not differentiate between causes of bleeding, and smaller perforator ruptures were also included, which typically involve smaller lumina (<0.35 mm) and therefore exhibit lower extravasation volumes per time interval. These clinical observations, though not directly comparable to our controlled experiment, reinforce the notion that every minute of ongoing extravasation increases overall risk. Notably, that study did not find a significant difference in patient outcomes based on the location of the perforation. This is consistent with our experimental result that the extravasation time did not significantly differ through perforation location. The dominant determinants were instead perforation caliber, rather than the specific arterial segment affected. 21 We compared several commonly used endovascular rescue techniques side-by-side in our experimental model.6,8,17 The most effective method was inflating a compliant balloon over the perforation site, which achieved instantaneous suspension of extravasation. This was true regardless of whether an AComA was present or not. Clinically, this aligns with prior reports that when an intraprocedural rupture occurs, prompt balloon inflation across the tear can tamponade the bleeding and provide temporary hemostasis. 18 We observed that proximal flow arrest by balloon occlusion of the ICA was only reliably effective in stopping extravasation when the AComA was absent. If an AComA was present, proximal ICA occlusion alone failed to stop bleeding, because the contralateral ICA via the AComA immediately perfused the distal circulation and continued to drive fluid out of the perforation. This demonstrates the critical role of the AComA in providing collateral circulation during acute ICA occlusions, but in the scenario of an arterial perforation, a robust AComA becomes a liability. 24 The recruited cross-circulation will maintain blood flow to the injured site, thereby perpetuating the extravasation despite ipsilateral proximal occlusion. In our tests, ICA balloon occlusion in the presence of an AComA provided little benefit, highlighting that when collaterals are present, bleeding control requires a more distal solution at the site of rupture rather than proximal flow arrest alone. Interestingly, we found that the temporary placement of a coil at the perforation site, as described by Waqas et al., 17 showed in our study just a trend toward slowing the extravasation more in the MCA bifurcation rather than in the ICA-T, but with no significant difference from the non-intervention group. At the ICA-T, flow rates are higher compared to the MCA bifurcation.25,26 In our view, this hemodynamic characteristic may be one of the contributing factors to the observed trend regarding localization in the temporary coiling group. This technique involves deploying coils in the parent artery at the level of the rupture to create a partial obstruction without permanently sacrificing the vessel. Our results indicated that the effectiveness of this maneuver in delaying extravasation was more pronounced for medium and small perforation sizes. Using a coil sized approximately to the vessel diameter (3 mm) slowed the bleeding, and even an oversized coil (7 mm) provided a slight additional tamponade effect. There was a tendency that a larger coil delayed the extravasation slightly better than a smaller one. This aligns with the conceptual goal of this method. Unlike a fully occlusive balloon inflation, coil tamponade creates a subocclusive state, reducing flow but still allowing some antegrade perfusion to pass the coil mass. A complete flow arrest, such as that described by Waqas et al., 17 could not be achieved in our study. A likely explanation should be that our experimental setup, using fluid with blood-like viscosity, lacked the clotting mechanisms of living tissue. In vivo, merely reducing the blood flow can initiate primary hemostasis at the perforation site, sealing the vessel injury.17,27 Additionally, the surface of the platinum coils provides a pro-hemostatic milieu. 28 The absence of this physiologic clotting response in our setup could be the reason that the coil's effect was limited to a mechanical flow reduction, which could explain the relatively lower efficacy observed in our study. Prior reports have highlighted the advantages of this approach. Deploying a coil through an already-positioned microcatheter can be faster than preparation and advancing of a balloon catheter, and the residual trickle of blood flow maintained by the coil can lower the risk of ischemia in distal territories. 17 Our experimental data support the idea that coils can serve as an endovascular tamponade for vessel perforations, especially in scenarios where a balloon is not immediately available. However, the tradeoff is that coil deployment does not halt bleeding as thoroughly as a balloon. In our study, it only bought additional time by slowing the flow. The slightly better performance of a larger-diameter coil in our tests suggests that packing density in the lumen correlates with the tamponade effect. We also evaluated a technique reported anecdotally. It consists of advancing a 5F distal access catheter into the segment harboring the perforation to block the vessel. In our study, inserting a 5F SOFIA catheter (TerumoNeuro, USA) directly into the distal M2 Segment of the MCA did not produce a reduction in extravasation when compared to non-intervention. This is likely because the average native vessel diameter of the human MCA segment is larger than the catheter, leaving ample space around the catheter for blood to continue flowing. Specifically, the diameter of the MCA ranges between 2 and 5 mm. 29 A 1.7 mm outer-diameter catheter only partially occupies the lumen, leaving a substantial cross-sectional area through which blood can still pass. Thus, the occlusive effect of the catheter is negligible in a vessel of that size. Our experiment confirmed that positioning the 5F catheter at the perforation site did not significantly slow the bleeding compared to the control scenario. The practical implication is that simply advancing an intermediate catheter in the artery is insufficient to tamponade a high-flow perforation, and more definitive measures such as balloon inflation or coil deployment are necessary. This observation is in line with the general principle that partial measures that do not substantially reduce flow will fail to stop a brisk arterial hemorrhage.
Limitations
Our model presents several limitations. Firstly, it is a rigid vascular model that does not simulate autoregulatory mechanisms such as vasoconstriction or the recruitment of multiple distal collaterals. We used a glycerin-based solution with a viscosity comparable to that of blood. However, as previously noted, the model lacks an active coagulation system, which would influence hemostasis in the event of a perforation and induce a clotting response to bare metal coils. Our experimental setup provides only a simplified representation of the complex in vivo processes following vascular perforation and hemorrhage. In particular, the behavior of leaks in our setup does not fully reflect the physiological situation. In reality, bleeding occurs into a physically confined intracranial space, where the resulting increase in intracranial pressure is substantially higher than what was observed in our model. In our experiments, opening the chamber to atmospheric pressure likely underestimated this effect, leading to only a modest rise in pressure. Clinically, however, the same volume of blood would be expected to produce a much greater pressure increase. This discrepancy may have reduced the impact of transmural pressure on flow dynamics in our model. Consequently, the absolute values obtained in our experiments cannot be directly translated to the clinical situation. On the other hand, we developed a model that allows the relative comparison of different techniques for managing perforation-related extravasation under standardized conditions, while also incorporating anatomical variations such as the presence of an AComA. Future studies should aim to include in vivo factors. A prospective data collection focusing on the direct impact and comparison of the different techniques with regard to patient outcomes would be valuable.
Conclusion
In summary, managing an iatrogenic intracranial vessel perforation is a race against time. Our findings reinforce clinical data that even a few minutes of unabated hemorrhage can be fatal or disabling. The most reliable way to achieve hemostasis is inflation of a compliant balloon at the rupture site, which can completely stop extravasation. When a balloon cannot be used or is not available immediately, temporary parent artery coiling is an alternative to reduce bleeding while preserving distal perfusion. However, its efficacy diminishes with larger perforations. Final parent vessel occlusion should be considered a last resort.
Supplemental Material
sj-pdf-1-ine-10.1177_15910199251384352 - Supplemental material for Evaluation of bail-out techniques for managing cerebral vessel perforation: An experimental study
Supplemental material, sj-pdf-1-ine-10.1177_15910199251384352 for Evaluation of bail-out techniques for managing cerebral vessel perforation: An experimental study by Roland Schwab, Mai-Britt Wienecke, Stefanie Feierabend, Erelle Fuchs, Sebastian J Müller, Eya Khadhraoui, Maximilian Thormann, Stefan Klebingat and Daniel Behme in Interventional Neuroradiology
Footnotes
Acknowledgments
Not applicable.
Ethical considerations
Ethical approval was obtained as required according to the guidelines of the local ethics committees (Ethics Committee, Medical Faculty of Otto-von-Guericke-University Magdeburg, Magdeburg, Germany). This anonymous retrospective study, which was conducted in accordance with the Declaration of Helsinki, fulfills the guidelines of the federal state of Saxony-Anhalt. The data has been legally collected in accordance with section 15 paragraph 5 (University Hospital Magdeburg).
Consent to participate
The requirement for individual informed consent was waived by the responsible ethics committees due to the retrospective nature of the study and the use of anonymized data. Where applicable, participants or their legal representatives had previously provided general consent for the use of clinical data in research.
Consent for publication
Not applicable.
Author contributions
All listed authors provided a substantial contribution to the work. RS acquired, analyzed, and interpreted data for the work, designed the study, and drafted the manuscript. MW, SF, and SK acquired data for the work; EF provided linguistic revision/editing; SM, EK, MT, and DB provided critical revision of the manuscript. All authors approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed in this study are included in this article and its supplemental file.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
