Abstract
Acute ischemic stroke occurs when a blood clot obstructs cerebral blood flow, leading to ischemia and potentially irreversible brain damage. While large vessel occlusions are known for their catastrophic effects, medium vessel occlusions (MeVOs) also contribute significantly to stroke-related disability. These occlusions, which occur in smaller, mid-sized vessels, can result in substantial neurological deficits depending on their location and the availability of collateral circulation. The detection of MeVOs poses unique diagnostic challenges, as their subtle presentations are often overlooked in standard imaging. Timely and accurate identification is critical for initiating appropriate therapies, including intravenous thrombolysis, endovascular thrombectomy, and secondary prevention measures. This editorial takes you on a visual journey through the world of MeVOs, exploring their locations, challenging cases, and the diverse techniques used to identify them. With detailed illustrations, it demonstrates how to recognize these occlusions on both advanced and conventional imaging, including guidance on spotting them on digital subtraction angiography. Finally, it delves into how these strokes are treated, offering a comprehensive and engaging look at the unique challenges and solutions in MeVO management.
Introduction
Acute ischemic stroke (AIS) usually strikes when a blood clot blocks an artery in the brain, cutting off blood flow and triggering a chain reaction from ischemia to irreversible brain tissue damage. Time is of the essence: in a typical large vessel occlusion (LVO) stroke, approximately 2 million neurons are lost each minute that blood flow is interrupted. 1 While the neuronal loss in medium vessel occlusion (MeVO) strokes is generally less, the extent of damage, and the resulting disability can vary significantly depending on the location of the occlusion, as these strokes still have the potential to cause disabling deficits. 2 The size of stroke depends on the location of blockage in the vascular tree: LVO strokes occur in major proximal arteries, MeVOs affect mid-sized arteries (1–3 mm diameter), 3 and small vessel occlusions involve the smaller branches (Figure 1). Each of these stroke subtypes are caused by different underlying conditions and treated in different ways, underscoring the critical need for swift and accurate diagnosis and intervention.4,5 MeVOs account for 25%–40% of all AIS cases and are characterized by blockages in the M2/M3 segments of the middle cerebral artery (MCA), A2/A3 segments of the anterior cerebral artery (ACA), or P2/P3 segments of the posterior cerebral artery (PCA), although the exact definitions vary (Figure 2). 6 Historically, MeVOs were thought to be highly responsive to intravenous thrombolysis (IVT). However, recent evidence reveals that up to 50% of MeVOs fail to reopen with this approach alone (Figure 3). 2 Additionally, a pooled analysis of two prospective cohort studies found that outcomes for MeVO stroke patients managed medically without endovascular thrombectomy (EVT) are generally unfavorable: One-third are not functionally independent at 90 days, half do not achieve excellent outcomes, and around 10% do not survive the initial three months after their stroke. 2 While current guidelines endorse EVT primarily for LVOs, many clinicians are now extending EVT indicationsto treat MeVOs as well. 6 Several randomized controlled trials are currently underway to demonstrate the potential benefits of EVT for MeVO strokes, and their results have recently been published or are expected soon.7–9 However, before EVT can be considered for MeVOs, detecting the occluded vessel and making an accurate diagnosis of a MeVO stroke are essential steps. In this pictorial editorial, we dive into the stroke subtype that is caused by MeVOs, exploring their unique challenges, diagnostic considerations, and treatment approaches.
Acute ischemic strokes caused by vessel occlusions of different sizes. (a) Shows an LVO stroke due to an M1 occlusion. (b) Shows a MeVO stroke in one of the M3 branches. (c) Shows acute scatter thromboembolic strokes in far distal small vessels, too small to be visible on CTA. LVO: large vessel occlusion; MeVO: medium vessel occlusion.
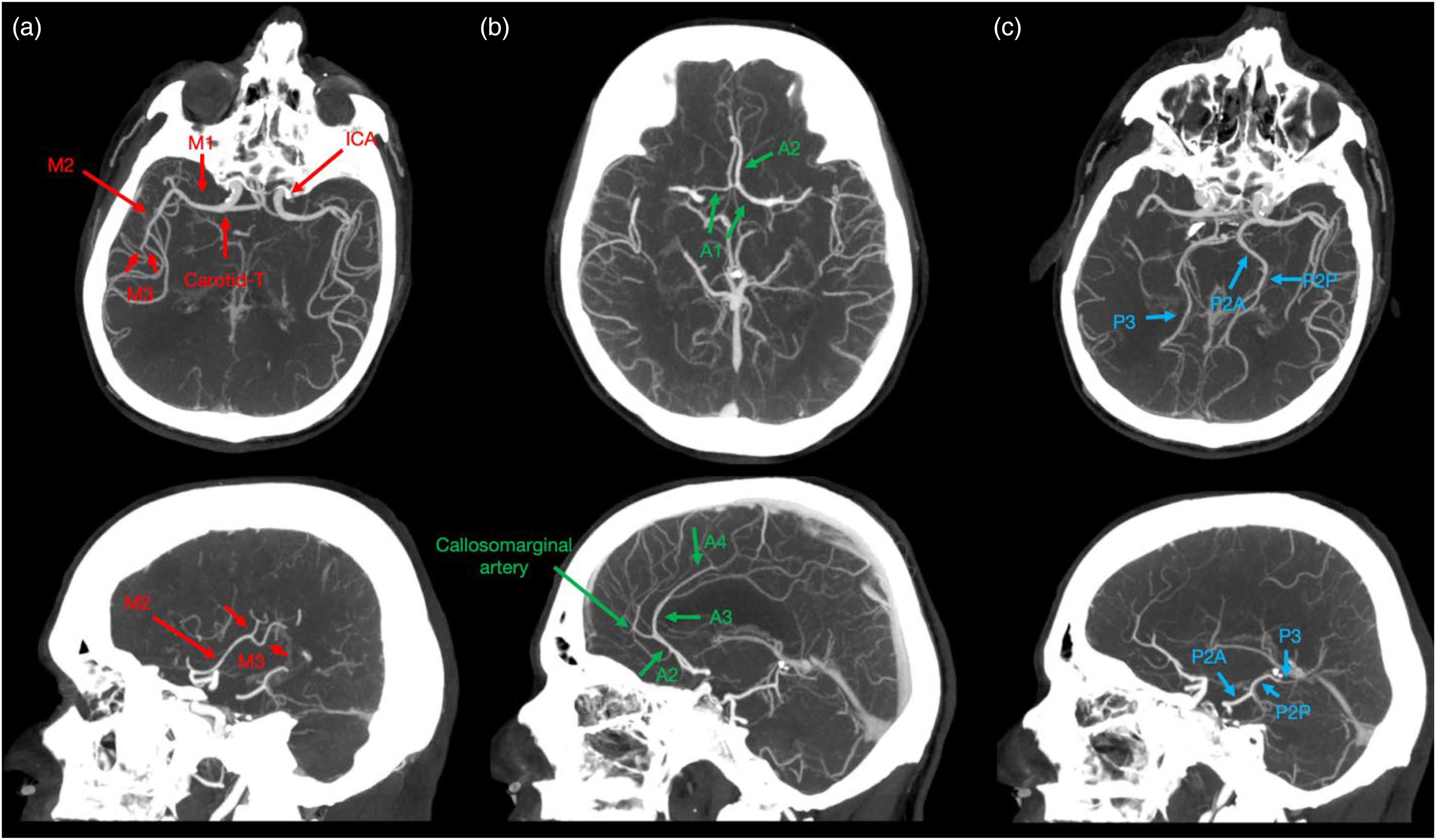
Segments of the intracranial arteries on CTA. (a) Shows the intracranial segment of the ICA, the “Carotid-T”, where the ICA divides into the M1 branch of the MCA and the A1 segment of the ACA. The M2 segment, or insular segment, originates at the main bifurcation, courses posterio-superiorly within the insular cleft, and terminates at the peri-insular sulcus. Here, the M3 segment, known as the opercular segment, begins, traveling laterally along the frontoparietal operculum and continuing as the M4 segment at the external surface of the Sylvian fissure. While in some cases, the MCA bifurcation pattern consists of a simple bifurcation, branching patterns can sometimes also be more complex, with three or even more branches, leading to multiple M2 segments of the MCA rather than a single defined segment. There are usually multiple M2 branches, and numerous M3 and M4 branches. (b) Shows the A1 segment originating from the Carotid-T and terminating at the anterior communicating artery. The A2 segment starts here, runs anteriorly along the rostrum of the corpus callosum, and continues as the A3 segment at the origin of the callosomarginal artery. The A3 segment terminates posteriorly above the body of the corpus callosum and continues as the supracallosal A4 segment. (c) Shows the P2 segment, which originates after the posterior communicating artery. The P2 segment consists of the P2A (anterior) part and the P2P (posterior) part, which course through the cisterna ambiens. It then transitions into the P3 segment, which passes through the quadrigeminal cistern and becomes the P4 segment within the sulci of the occipital lobe. MCA: middle cerebral artery; ICA: internal carotid artery.
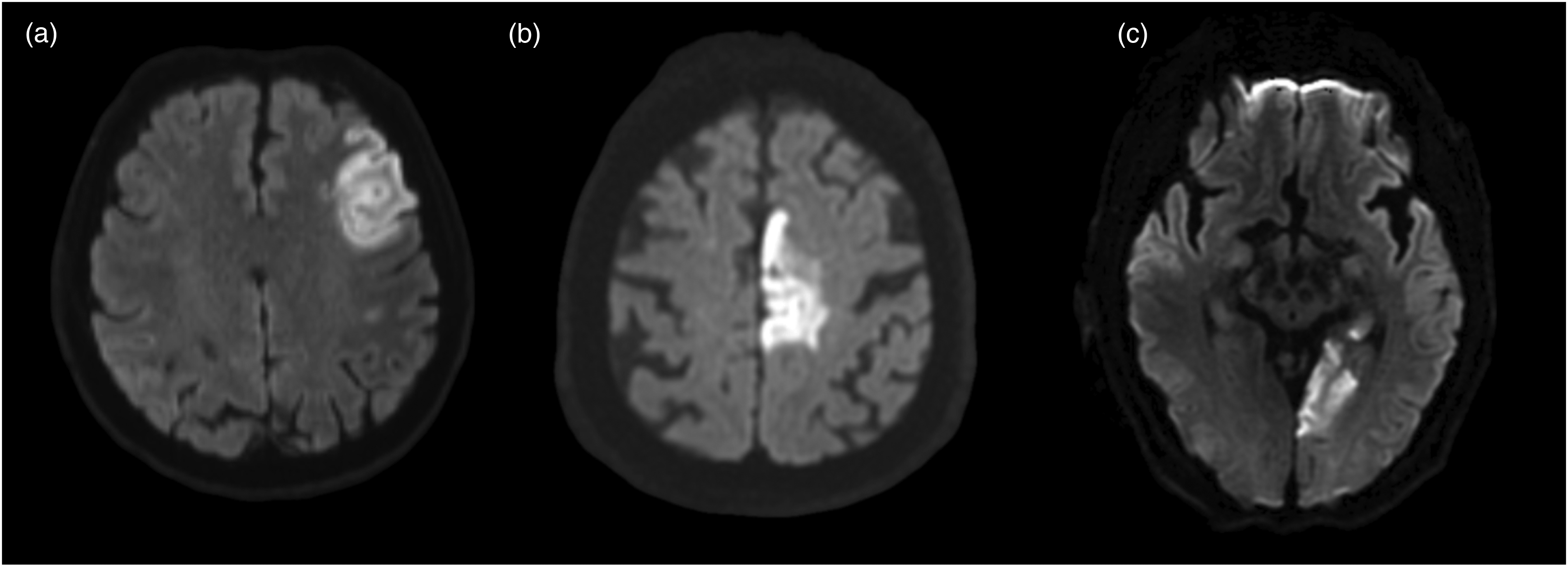
MeVO stroke patterns in different arterial vascular territories. (a) Shows a DWI-MRI of a M3 occlusion stroke. (b) Shows an A3 occlusion stroke and (c) shows a P3 occlusion stroke. MeVO: medium vessel occlusion; MRI: magnetic resonance imaging.
Challenges in MeVO diagnosis
MeVOs represent a complex frontier in stroke care. Unlike LVOs, which have clear diagnostic signs and established therapeutic pathways, MeVOs often manifest with subtle or atypical symptoms, challenging clinicians to make timely, accurate diagnoses, and rendering treatment decisions difficult. At the same time, they can result in significant neurological deficits if left untreated (Figure 4). 2 Unlike LVOs, which are often detected through standard imaging, MeVOs often require advanced imaging techniques like multiphase CT angiography (mCTA) 10 or magnetic resonance imaging (MRI) to identify subtle changes. Figure 5 shows a MeVO, easily missed without specific attention to detail and Figure 6 shows a missed M2 MeVO stroke, practically invisible by only looking at the axial imaging due to the straight vertical course of the vessel. Additional contributing factors for missed MeVOs, include fatigue from long call days, extended stays, lack of awareness, and night shifts, which further highlight the need to raise awareness and for specialized training in MeVO detection. On the other hand, we should also consider that false positive interpretations can be harmful, potentially leading to unnecessary inter-hospital transfers and futile, risky treatments.
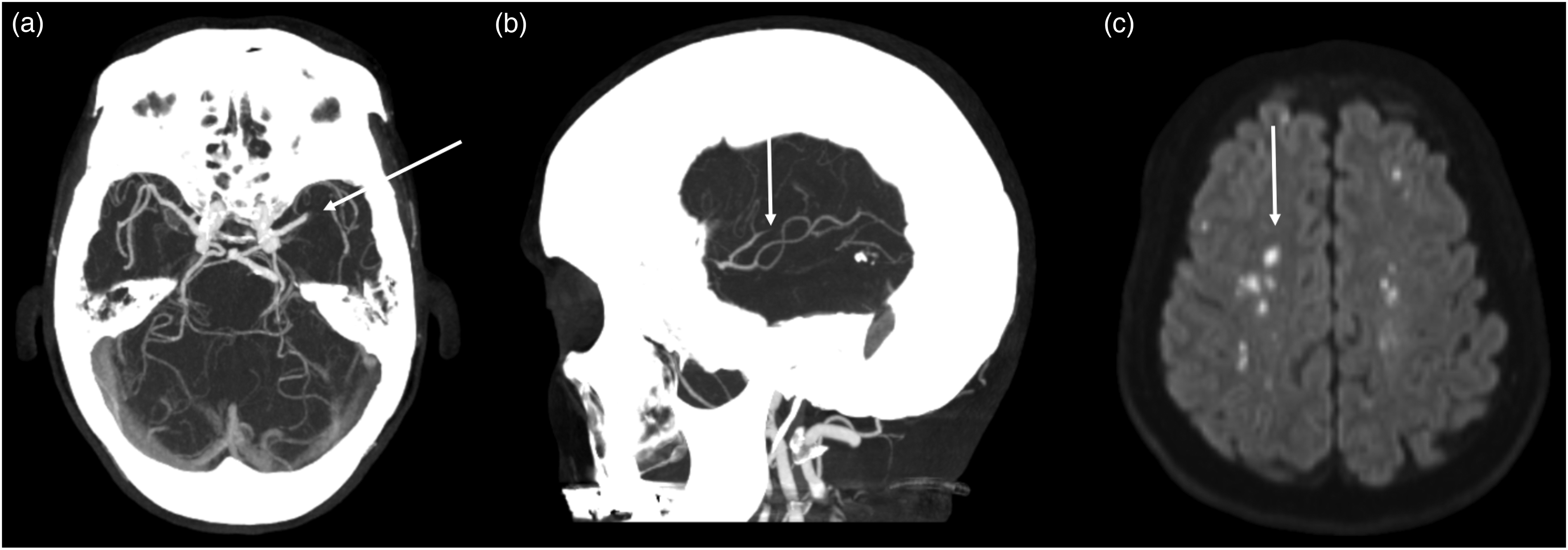
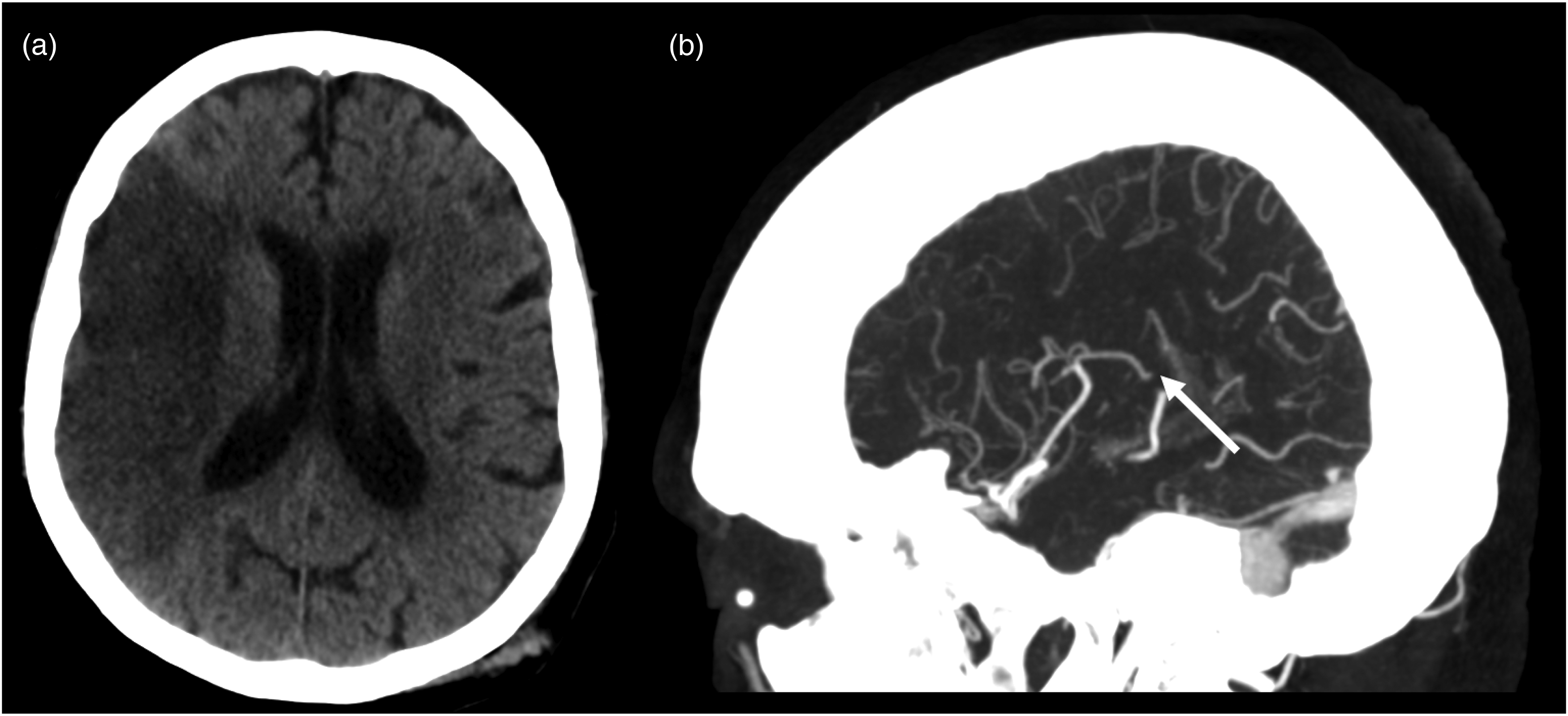
Large infarct after left P2 MeVO stroke. (a) Shows an unremarkable NCCT scan of the brain of a patient with visual disturbance. (b) Shows an acute P2 segment occlusion of the left PCA (white arrow). (c) Shows the MRI follow-up image with a large infarct in the PCA territory. NCCT: non-contrast CT; MeVO: medium vessel occlusion; PCA: posterior cerebral artery; MRI: magnetic resonance imaging.
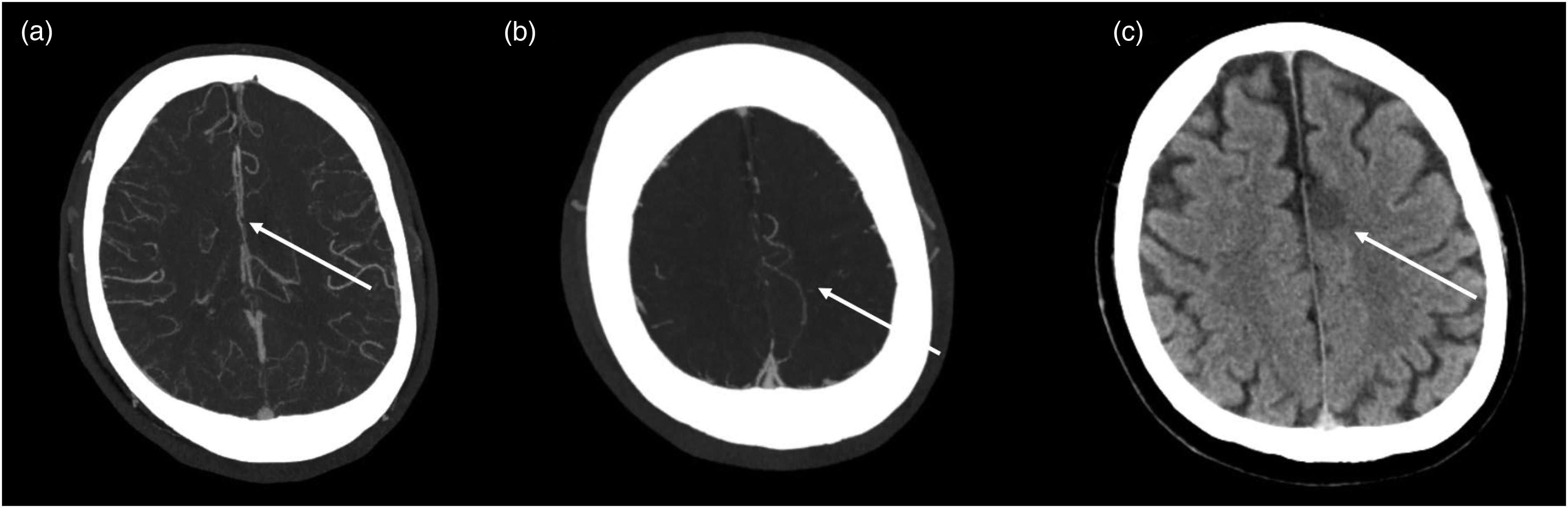
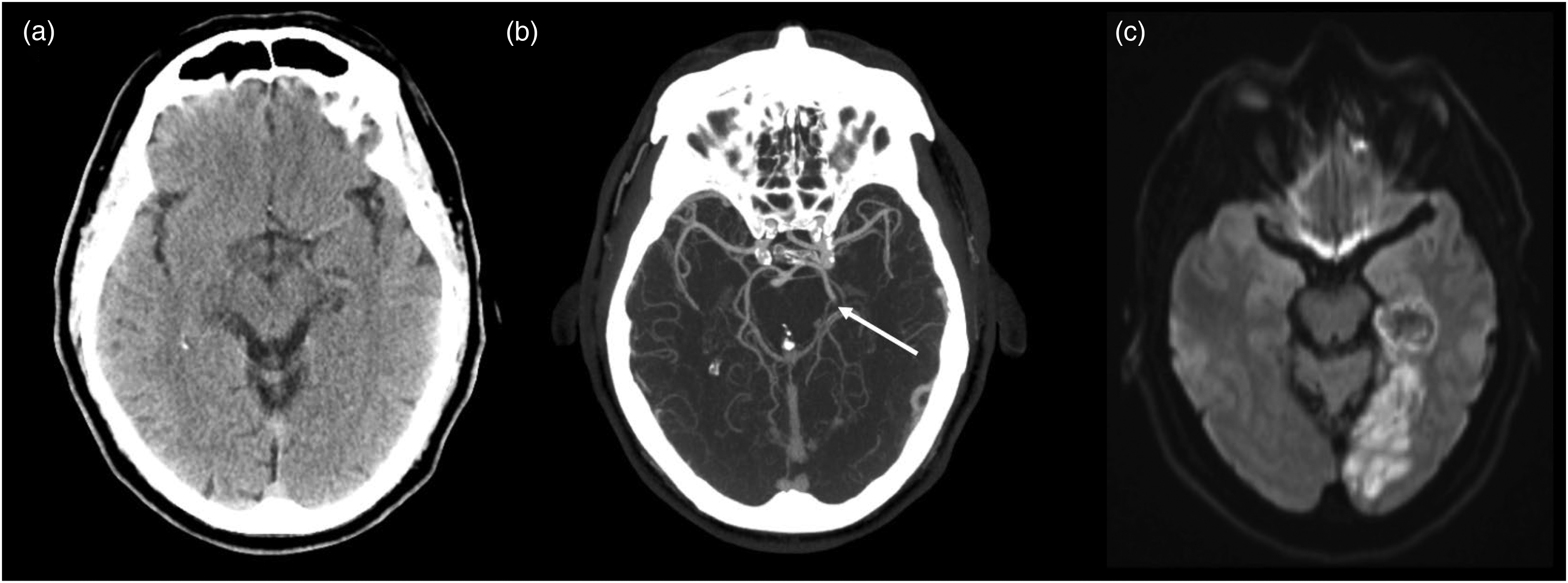
Challenging case of an A3 MeVO stroke. (a) CTA shows a very subtle interruption of opacification in the A3-Segment of the left ACA. (b) Shows a contrast hangup in the delayed phase of a mCTA and (c) shows the follow-up NCCT with a demarcated infarct in the cingulate cortex on the left. ACA: anterior cerebral artery; MeVO: medium vessel occlusion; NCCT: non-contrast CT.
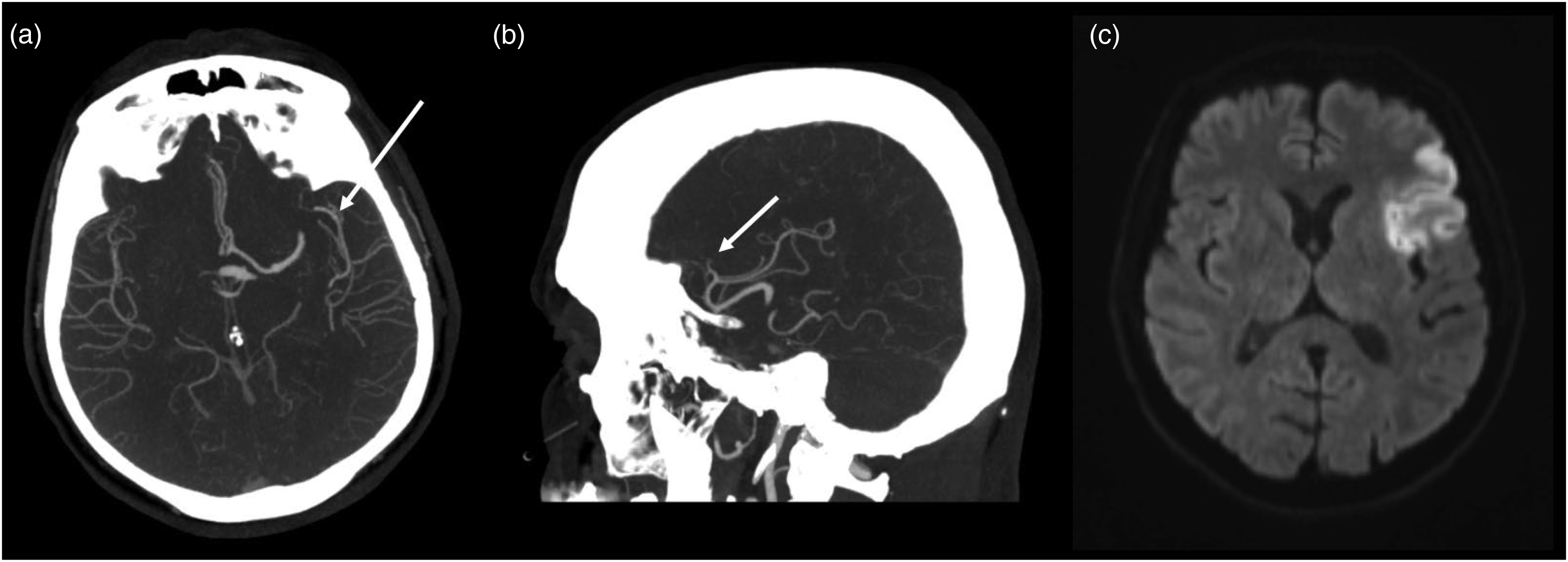
Missed M2 MeVO stroke that was challenging to detect on axial imaging. (a) Shows that this M2 segment occlusion of the left MCA is easy to miss by looking just at the axial imaging alone. (b) Shows that the occlusion is clearly visible in the sagittal plane. (c) Shows the follow-up MRI-DWI with stroke in the MCA territory. MCA: middle cerebral artery; MeVO: medium vessel occlusion; MRI: magnetic resonance imaging.
Practical tips, training approaches, and strategies to enhance detection of MeVOs
The primary drivers of improved human performance in detecting MeVOs are training and experience. For instance, one study demonstrated that non-neuroradiologists without specialized neuroimaging training were considerably more likely to overlook vessel occlusions on CTA compared to neuroradiologists. 11 Radiologists with specialized imaging training also detect vessel occlusion with greater speed and confidence. 12 Although studies often compare radiologists at different training levels, having a neuroradiologist available to interpret acute stroke imaging in real-time is not feasible in all hospitals. Therefore, it is valuable to explore practical training approaches, tips, and strategies to improve MeVO detection.
Interpreting acute imaging in the clinical context
As previously mentioned, the detection of MeVOs can be tricky due to their relatively small caliber.3,6 Knowing where to look is thus of the essence to quickly find the occlusion. Clinical information of symptoms and their onset time are often key to direct the radiologist's eye to the affected area. For instance, leg-accentuated weakness points to the ACA territory. 13 Even superior and inferior M2 branches can often be clinically distinguished, as occlusion of the inferior M2 branch can result in contralateral sensory disturbances, neglect, Wernicke's aphasia (impaired speech comprehension but fluent articulation), or upper quadrant anopsia. Occlusion of the superior M2 branch on the other hand, rarely leads to neglect or visual field cuts but rather to hemiparesis, gaze deviation, and Broca aphasia (stuttering speech with good speech comprehension). Occlusion of the P2 segment of the PCA potentially leads to homonymous hemianopsia, memory impairment, sensory disturbance, impaired balance, or impaired consciousness,5,14,15 see also Table 1.
Intracranial vessel occlusions and their corresponding acute symptoms.

ACA: anterior cerebral artery; MCA: middle cerebral artery; PCA: posterior cerebral artery.
Knowledge of the time since symptom onset further gives a clue “how dark” the affected territory can be expected to be on non-contrast CT (NCCT). As a rule of thumb, decreasing tissue density is expected after 6 h after symptom onset likely representing irreversible tissue damage. 16 However, there is a substantial individual variability of how fast strokes progress. Strokes in so called “fast progressors” can be far developed after less than 6 h while the opposite can be true in slow progressors. 17
Advanced imaging techniques
Advanced imaging techniques can assist clinicians in detecting MeVOs with greater accuracy. 18
Multiphase CT angiography
mCTA captures multiple phases (Figure 7) of blood flow through the brain (early/arterial, middle/peak-venous, and late/late-venous). This dynamic imaging allows to observe how blood moves through vessels over time, visualizing delays in blood flow (Figure 7(b)). By capturing these changes across phases, mCTA makes it easier to spot regions with delayed filling, common indicators of MeVOs. 10
Multiphase CT angiography (mCTA) shows an A3 MeVO stroke. (a) Shows the first contrast phase with a thrombus in the A3 segment on the right that is very hard to spot. (b) Shows the first delayed phase with delated filling in the A3 segment, which is still visible in the second delayed phase shown in (c). mCTA: multiphase CT angiography; MeVO: medium vessel occlusion.
CT perfusion
CT perfusion (CTP) is another widely used advanced imaging technique that enhances the detection of MeVOs. During CTP, intravenous contrast is injected, and the brain is scanned multiple times. This series of images generates perfusion maps that show cerebral blood flow. The color-coding effect in CTP highlights MeVOs as wedge-shaped areas with reduced perfusion, making it easier to identify the occlusion site (Figure 8). 19
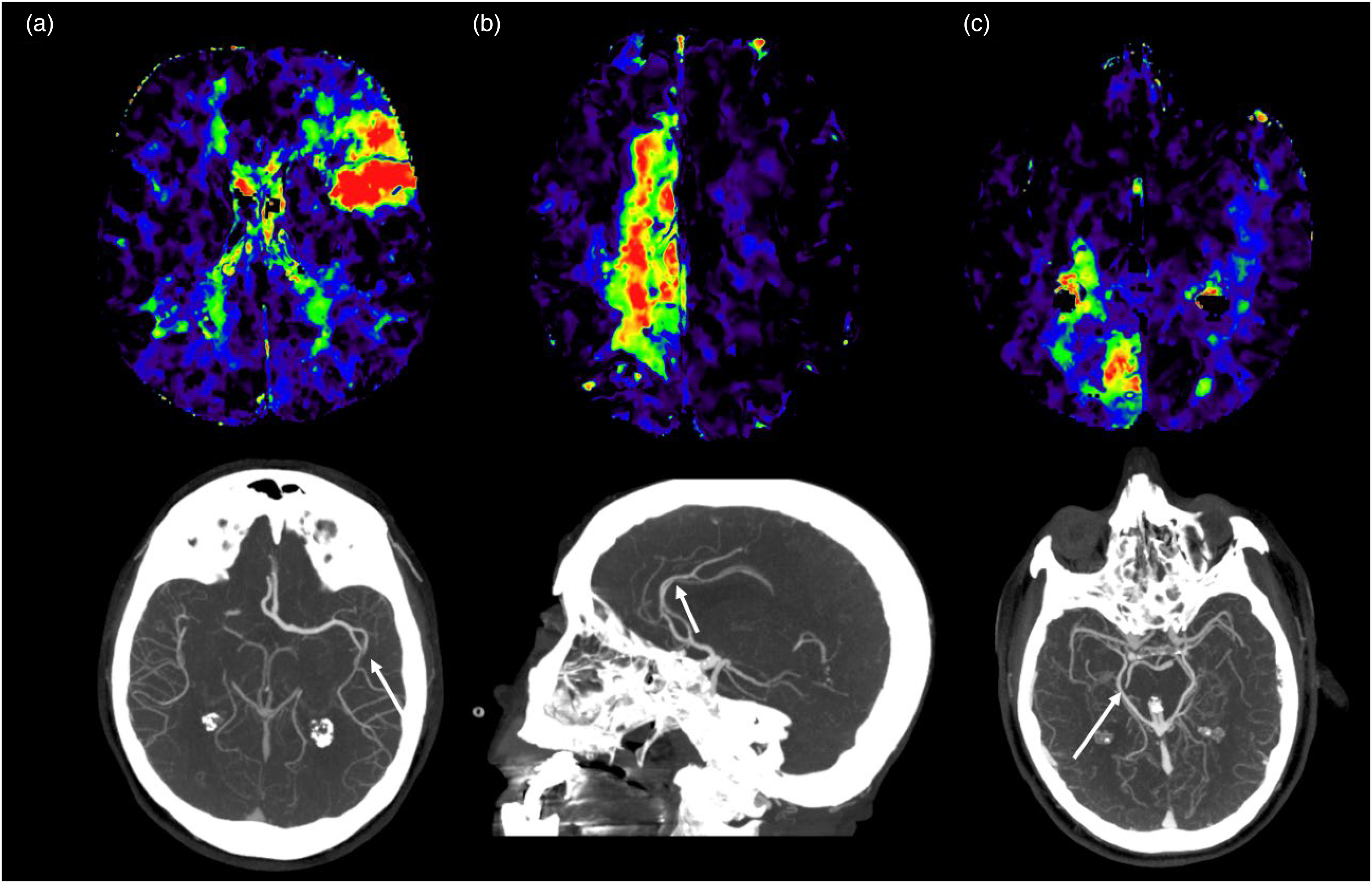
CTP in MeVO strokes. CTP with reduced perfusion in MCA (a), ACA (b) and PCA (c) MeVO territories. By going back to the initial single phase CTA imaging and after taking a closer look, it is possible to identify occluded vessels (white arrows) that were missed during the first imaging assessment of the CTA. ACA: anterior cerebral artery; MeVO: medium vessel occlusion; CTP: CT perfusion; MCA: middle cerebral artery; PCA: posterior cerebral artery.
Magnetic resonance imaging
MRI can provide additional information beyond CT in the diagnosis of MeVOs. DWI detects small ischemic cores caused by MeVOs more accurately than CT. 20 The hyperintense vessel sign (HVS) on FLAIR indicates the loss of a normal flow void and can assist in diagnosing MeVOs that are difficult to identify on MRA. Additionally, the susceptibility vessel sign on T2* reflects the characteristics of the thrombus, which can be useful in formulating treatment strategies.21,22 While MR perfusion requires contrast agents, non-contrast MRI can also predict salvageable tissue by assessing the difference between the areas indicated by DWI and HVS (Figure 9). On the other hand, MRI is susceptible to motion artifacts, which can hinder the detection of MeVOs in patients who cannot remain still during MRA imaging.
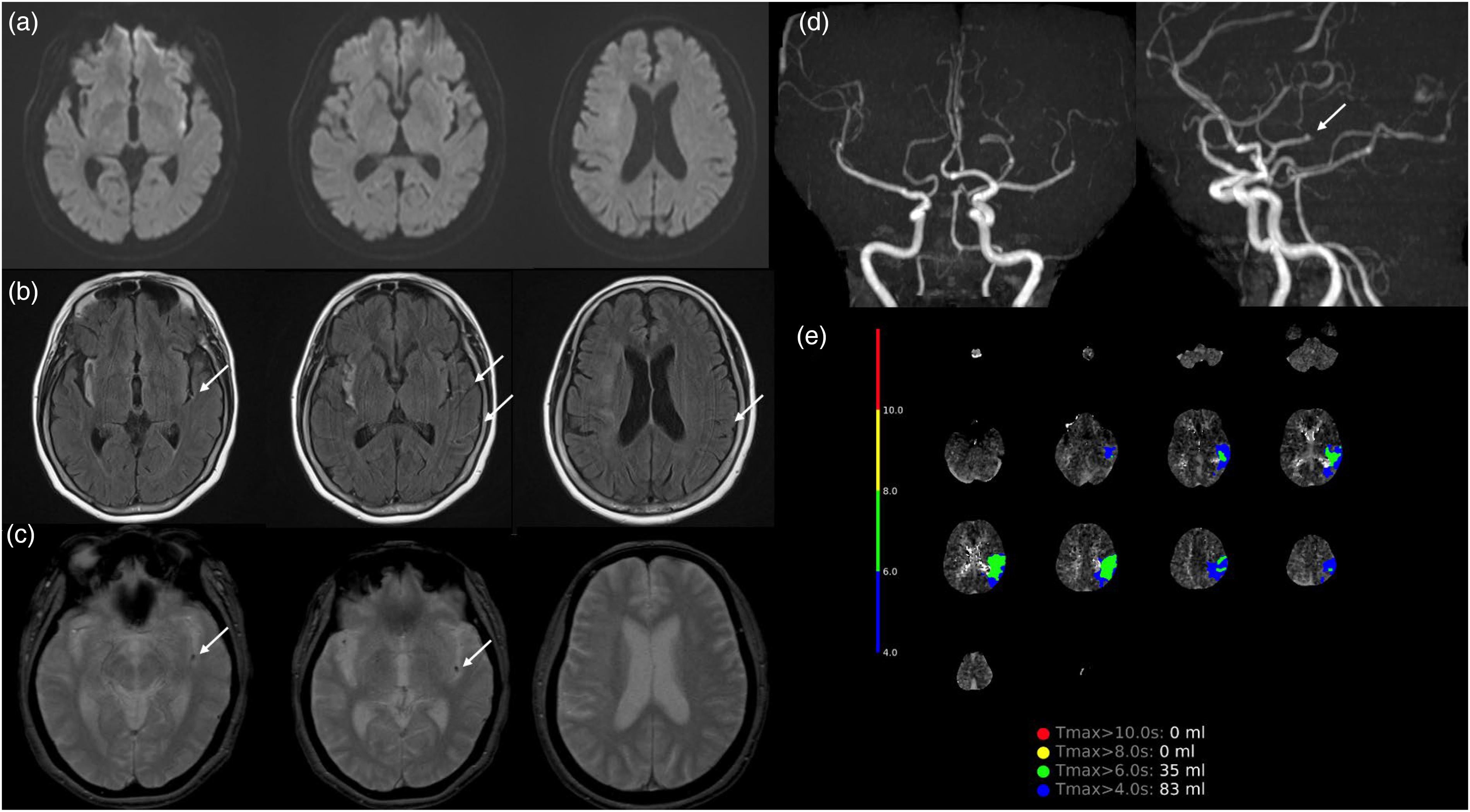
Mevo stroke detection on MRI. Baseline MRI of a patient with left M2 occlusion (a: DWI, b: FLAIR, c: T2*, d: MRA, e: MR-perfusion). (b) Shows a hyperintense vessel sign due to loss of normal flow void (white arrow) distal to the occlusion site. (c) Demonstrates a susceptibility vessel sign (white arrow) at the occlusion site. Although only a small infarct is visible on DWI (a), the area near the hyperintense vessel sign on FLAIR corresponds to delayed Tmax on perfusion imaging (e), suggesting the presence of a relatively large salvageable tissue volume. MeVO: medium vessel occlusion; MRI: magnetic resonance imaging.
Mevo detection without advanced imaging
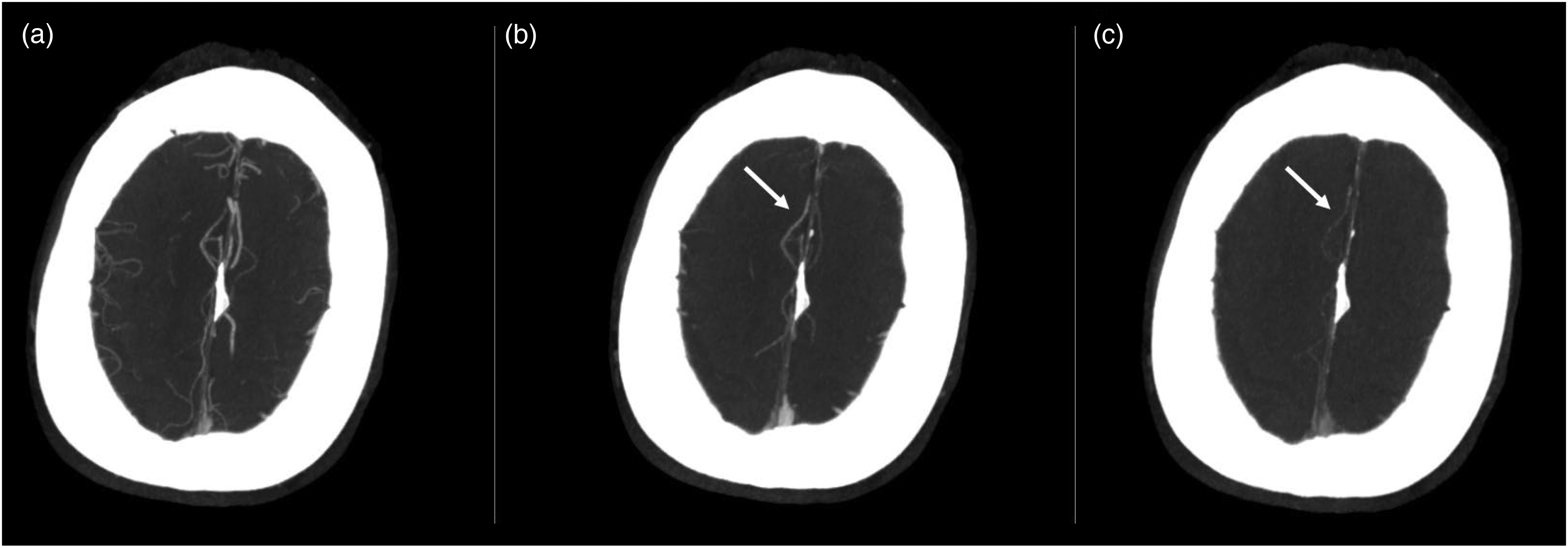
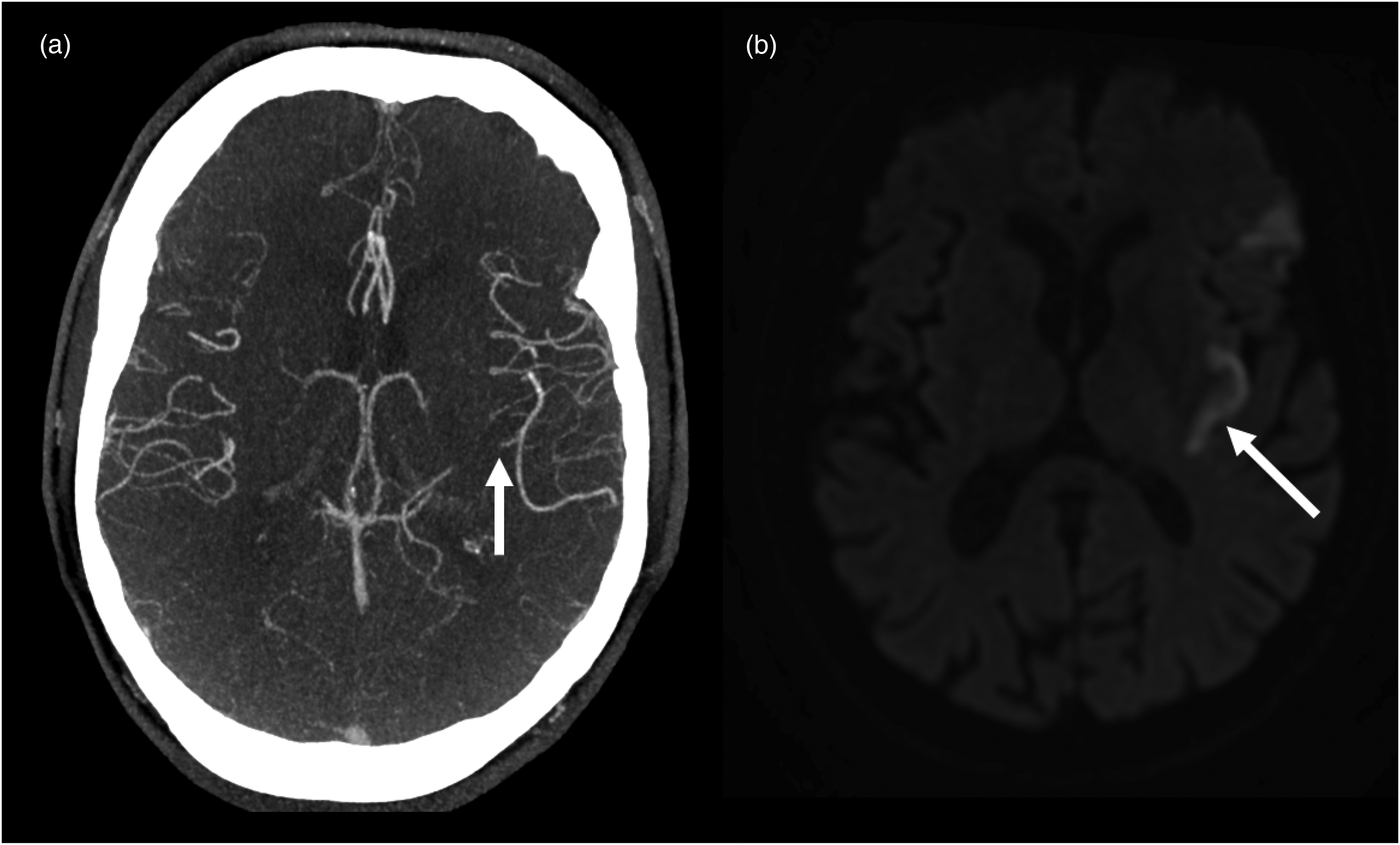
However, advanced imaging is often unavailable in smaller hospitals and low-/middle-income countries. In such cases, a helpful tip is to examine maximum intensity projection reconstructions in all planes, as already demonstrated in Figure 6, or as seen in Figure 10, where a subtle right M2 clot becomes clearly visible in the coronal view. Areas with fewer vessels, reduced visible contrast within a vessel, or smaller vessel calibers can also indicate the presence of an occlusion on routine CTA if advanced imaging is unavailable. These observations are somewhat similar to analyzing perfusion imaging, as these areas appear to receive less blood flow compared to the opposite side of the brain (Figure 11). It is easier to identify these signs in LVOs, but they can also be observed in MeVOs, particularly when the occlusions are more proximal.
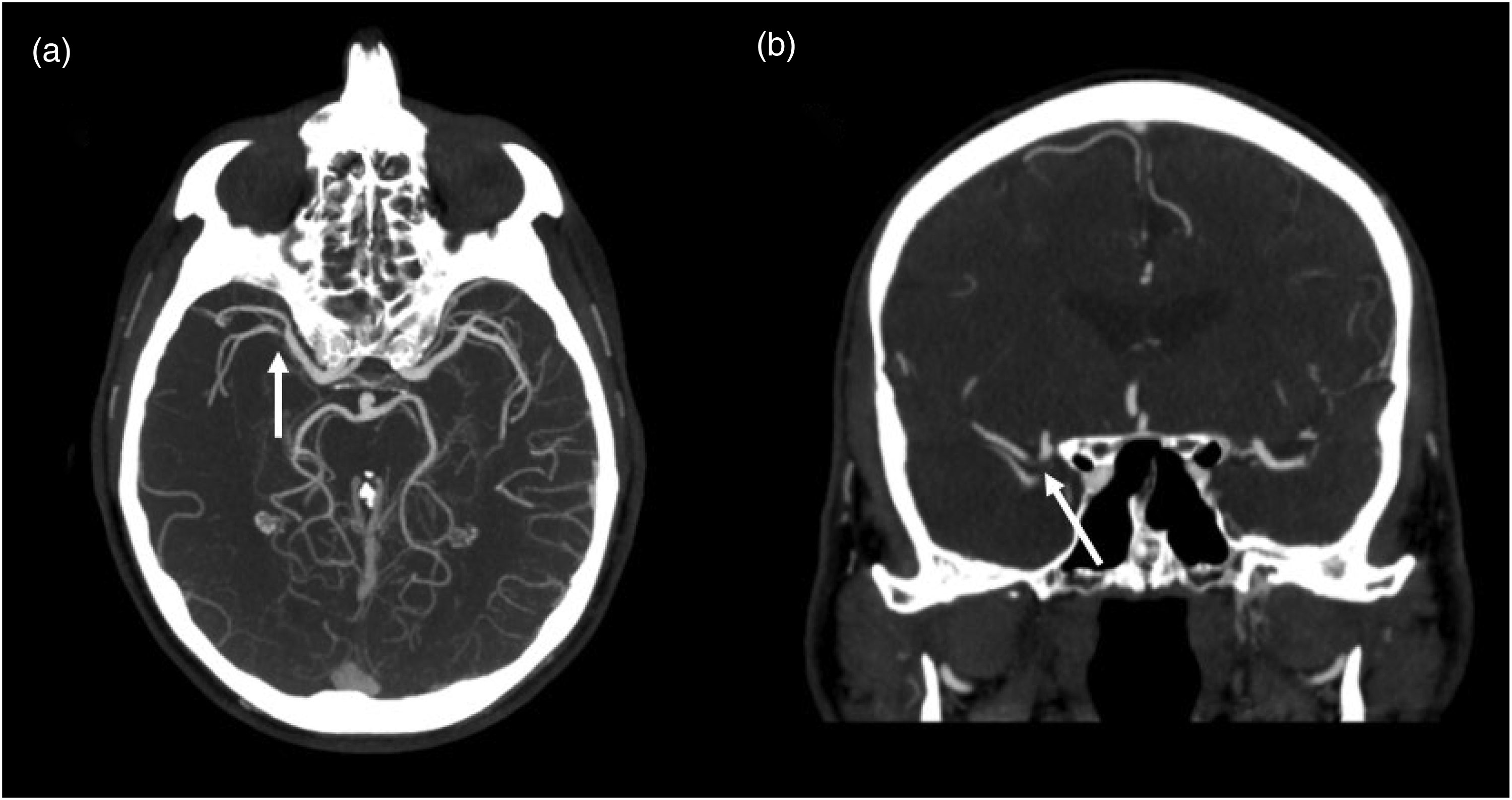
M2 thrombus on the right clearly visible on the coronal view. (a) Shows a proximal M2 occlusion on the axial plane that is easy to miss, whereas the big thrombus is clearly visible in the coronal view in (b).
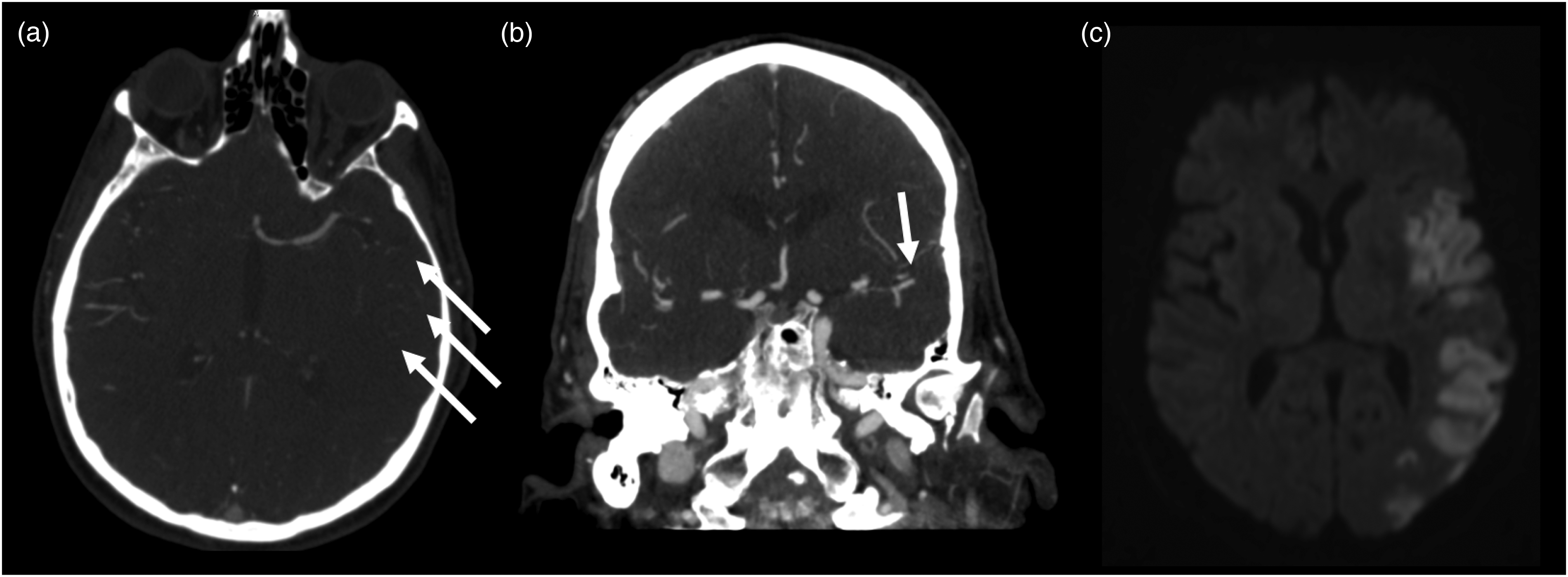
Occlusion detection based on asymmetric CTA vessel changes. (a) Shows fewer and narrowed vessels with reduced contrast enhancement on the left side compared to the right. (b) Illustrates that one of the two posterior-superior M2 segments of the MCA is occluded, leading to a large infarct, as visible in (c) on the 24-h DWI-MRI. MRI: magnetic resonance imaging; MCA: middle cerebral artery.
Automated, artificial intelligence-guided detection of medium vessel occlusions
Artificial intelligence (AI) methods hold promise for automatic detection of vessel occlusions. Most automated vessel detection tools are primarily designed as triage aids for managing LVO strokes. These tools, often commercially available, exhibit high accuracy in identifying LVOs and flag neuroimaging scans for physician review. However, most algorithms are trained on LVO datasets, with limited studies examining their performance for MeVOs, particularly in the M2 and M3 segments.23,24 There are promising solutions for detecting MeVOs. For example, StrokeSENS is a tool developed by Circle Neurovascular Imaging, which uses mCTA and has shown potential for MeVO detection. Despite these advancements, automated MeVO detection remains still a relatively unexplored field. 25
Open science is playing an important role in recent advancements in the use of AI for stroke applications. Public imaging data with expert manual annotations are available, and open competitions to design new AI for stroke analysis tools are hosted regularly. 26 AI models for image classification and segmentation can be trained by leveraging data available from numerous sources. Classification models combined with explainable AI methods can tell whether a MeVO is present and importantly produce a saliency map that pinpoints the location of the occlusion (Figure 12). 27
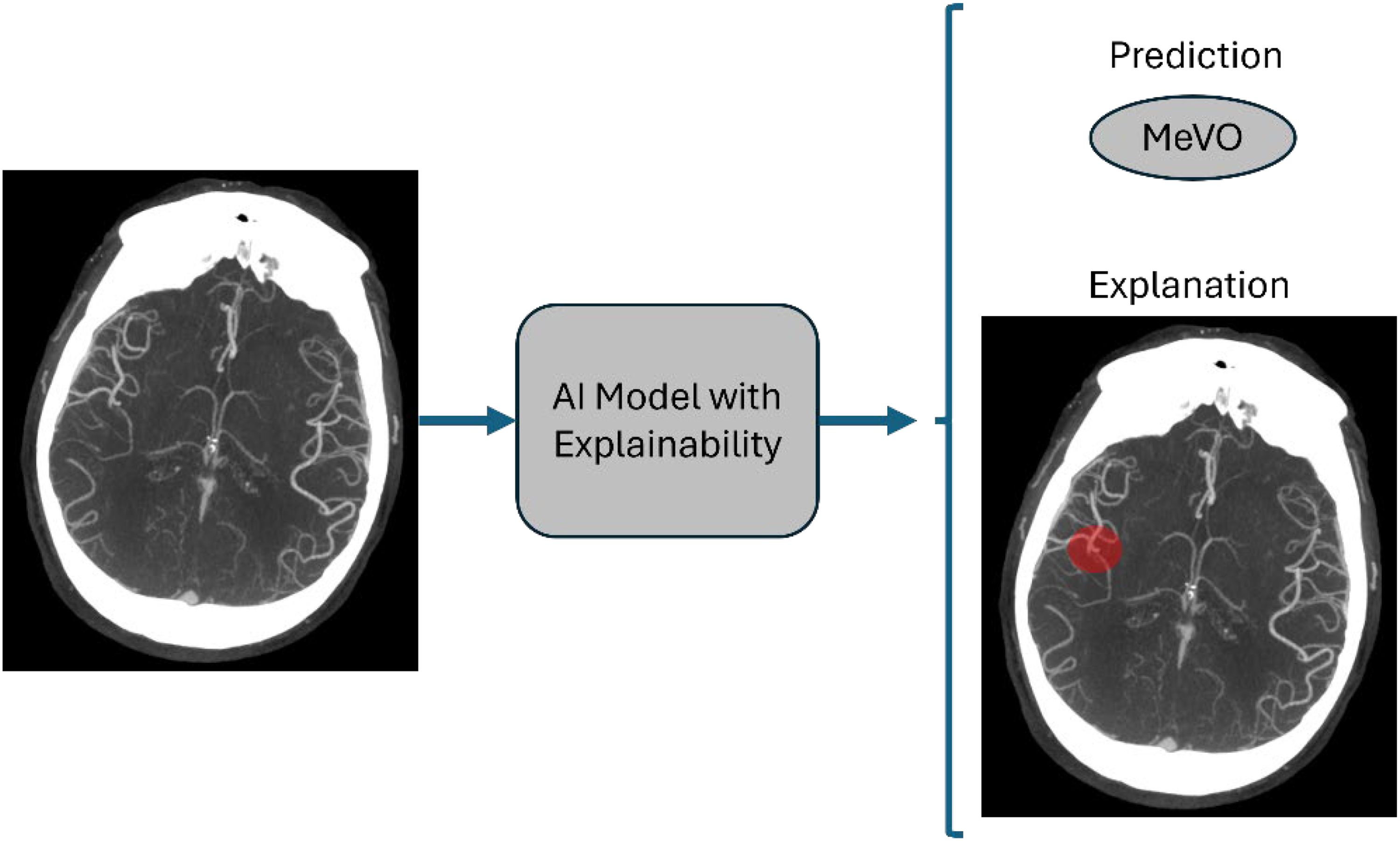
Illustration of an AI model with an explainability component to detect MeVOs. This figure shows how an AI model with an explainability component would work. The model would receive as input the patient's imaging information and the model produces to outputs: A binary prediction regarding the present or not of a MeVO and an explainability map, also known as saliency map, that highlights the region where the MeVO is present in the image (shown in red in the right image). AI: artificial intelligence; MeVO: medium vessel occlusion.
Primary vs. secondary MeVOs
Not all MeVOs develop independently. Some, referred to as secondary MeVOs, result from thrombus migration or fragmentation during treatments like IVT, EVT, or spontaneous. These secondary MeVOs are essentially former LVOs that have shifted to MeVOs. Compared to primary MeVOs, they often show more extensive ischemic damage, more severe baseline symptoms, limited response to thrombectomy and thrombolysis, and generally poorer outcomes. They can be recognized by an unusual infarct pattern, with infarcts appearing outside the typical downstream MeVO territory (Figure 13 and 14). 28 Multiple secondary MeVOs are often observed as a result of thrombus fragmentation, leading to perfusion deficits or early ischemic changes outside the primary MeVO territory. In such cases, it is highly recommended to carefully search for additional MeVOs.
Secondary MeVO. (a) Shows large early ischemic changes in the right MCA territory. The small M3 branch occlusion of the MCA shown in (b) (white arrow) is likely to be a small secondary MeVO due to a thrombus fragmentation from initial large proximal thrombus as the M3 branch occlusion cannot explainr this large infarct. MCA: middle cerebral artery; MeVO: medium vessel occlusion.
Secondary MeVOs with distal occlusions beyond the insular ischemic changes. (a) Shows a small M3 branch occlusion of the left MCA, which is unlikely to be the primary cause of the ischemic changes seen in (b) within the left insular region. This is due to the distal location of the occlusion relative to the affected territory. MeVO: medium vessel occlusion; MCA: middle cerebral artery.
Detecting MeVOs on digital subtraction angiography
Detecting the occluded vessel on digital subtraction angiography (DSA) is a critical step in successfully navigating wires and catheters to the site of the occlusion, but the detection and exact localization can be challenging for MeVOs due to their smaller size and the potentially smaller area of hypoperfusion distal to the occlusion, especially in cases of good collateralization. However, the same principles used for identifying occlusions on mCTA apply here. Look for signs such as delayed arterial filling and delayed washout to pinpoint the occluded vessel, as illustrated in Figures 15 and 16.
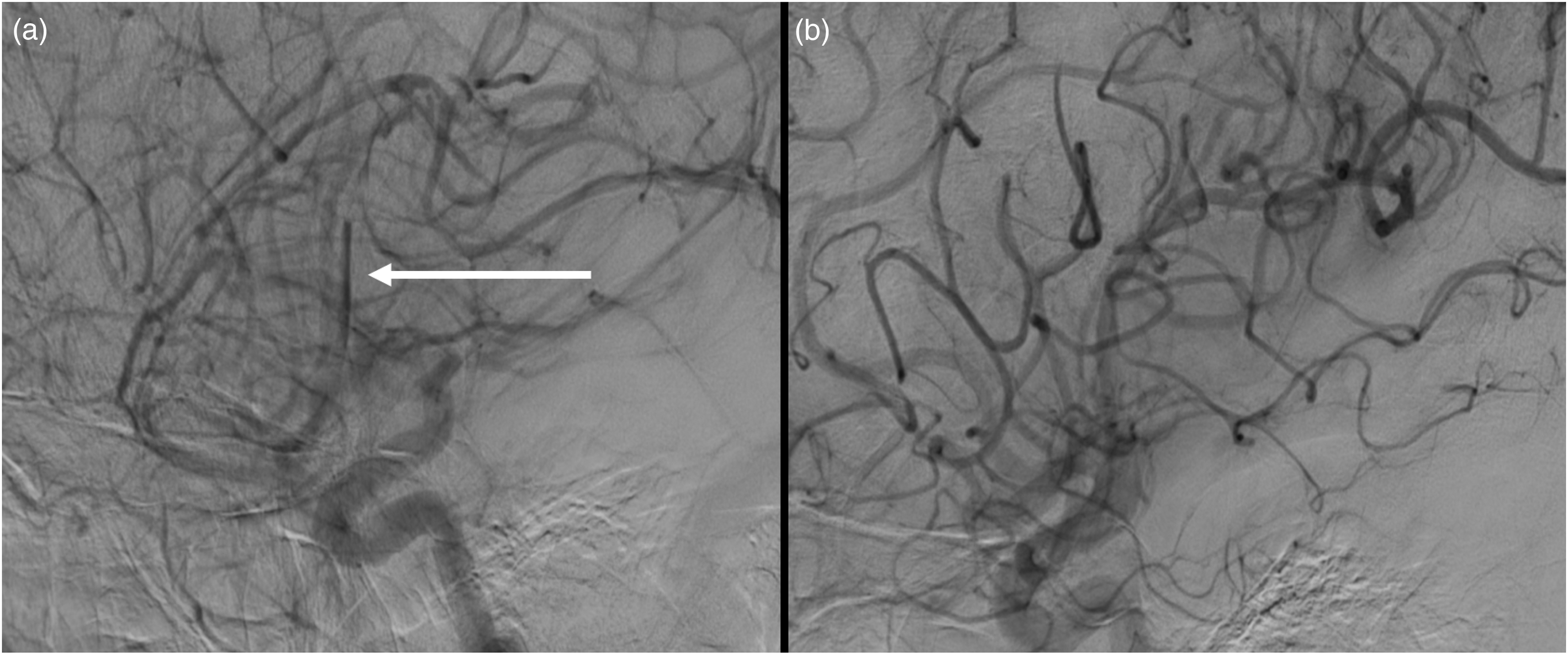
Example for recognizing and targeting occlusions on DSA. (a) Demonstrates delayed arterial filling and washout in the MeVO. Following successful stent-retriever thrombectomy, the previously clearly visible vessel occlusion is no longer apparent in (b), but there now appears to be a smaller, more distal occlusion. DSA: digital subtraction angiography; MeVO: medium vessel occlusion.
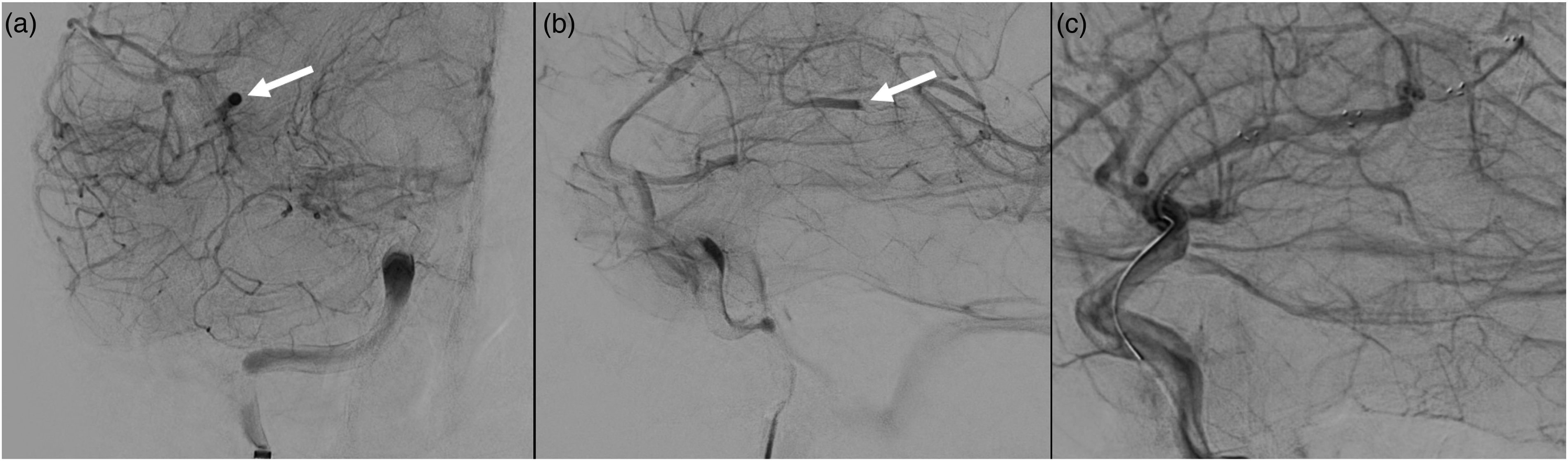
Another example for recognizing and targeting occlusions on digital subtraction angiography (DSA). (a) Displays delayed contrast washout in an occluded vessel in the coronal view, while (b) shows the same finding in the sagittal view, along with the visible proximal end of the thrombus. (c) Illustrates stent deployment, demonstrating the bypass effect of blood flow in the previously occluded vessel, with reduced visibility of the contrast hang-up.
Acute treatment of MeVO stroke
Intravenous thrombolysis (IVT) is a common approach to reopening occluded vessels, especially for smaller thrombi, where one might intuitively expect it to be the most effective treatment. 29 However, recent evidence shows that up to 50% of MeVOs fail to reopen with thrombolysis alone, highlighting the need for alternative or additional treatment strategies. 2 EVT is widely recognized as a highly effective intervention for LVO strokes, 30 making it an appealing option for managing MeVOs as well. However, implementing EVT for MeVO strokes presents unique challenges. One major limitation is the lack of high-level evidence, though several randomized controlled trials are currently underway or have been recently published to address this gap and determine the safety and efficacy of EVT for MeVO stroke.7–9 One major challenge neurointerventionalists face is that current endovascular devices are primarily designed for LVOs, making them less suitable for MeVOs due to their size and rigidity, but technology is steadily improving. 31 Despite these challenges, many interventional neuroradiologists have already incorporated EVT into their routine practice for certain subgroups of patients with MeVO stroke (Figure 17), underscoring the perceived benefits of this approach. 6
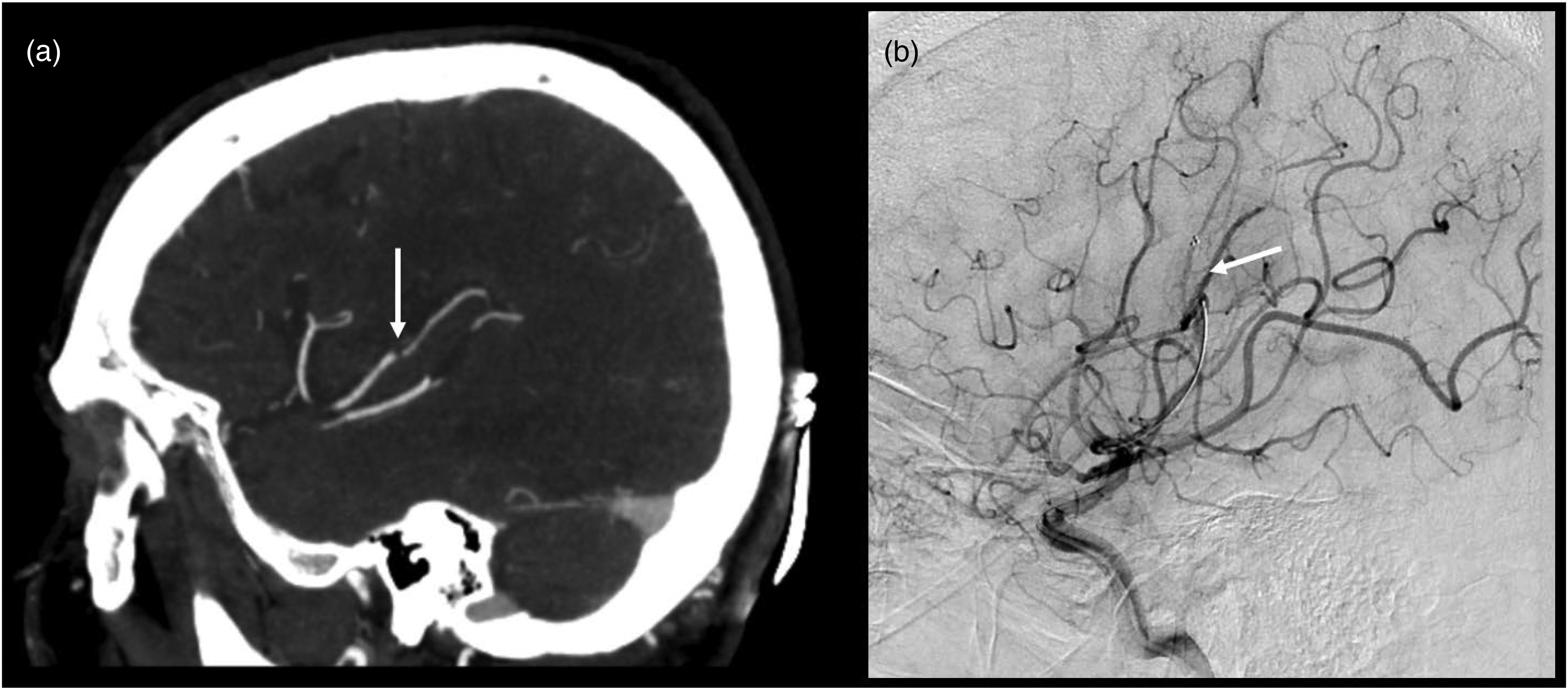
MCA M3 segment thrombectomy. (a) Shows a subtle M3-segment occlusion on the right in a patient with high NIHSS score. (b) Shows the first pass of a stent-retriever thrombectomy (white arrow). MCA: middle cerebral artery.
Conclusion
MeVOs represent a critical yet often challenging frontier in AIS care. Their subtle presentations are frequently missed on standard imaging, making accurate diagnosis particularly difficult. However, timely recognition is essential, as it secures the diagnosis, guides treatment with IVT, and some MeVOs could potentially even be effectively treated with EVT when detected early. Specialized training, along with practical tips and tricks such as those outlined here, can significantly improve diagnostic accuracy and enhance patient outcomes. Since the detailed outcomes of MeVO trials are not yet known and subgroup analyses are yet to be published, it is still crucial to emphasize the importance of recognizing a MeVO. Identifying these occlusions not only secures the diagnosis of AIS in general, but also enables appropriate acute stroke treatment, including IVT, stroke unit care, and secondary stroke prevention. Furthermore, it initiates a comprehensive stroke etiology workup.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Johanna Ospel is a consultant for Nicolab.
Data availability statement
Data generated or analyzed during the study are available from the corresponding author by request.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: Alexander Stebner received a stipend for his Research Fellowship in Calgary from the Swiss Society of Radiology.
