Abstract
Background
The Tigertriever stent retriever (Rapid Medical) can be actively expanded and contracted by the operator, which allows for several actuation-related parameters to be optimized to potentially improve device efficacy. These parameters have not yet been evaluated. We conducted a benchtop study to evaluate the effect of actuation frequency on clot integration within the stent.
Methods
A Tigertriever 17 device was deployed within a biological clot analog placed in a straight tube. The device was actuated between the maximally contracted and maximally expanded states with three different frequencies: passive (one-time opening,
Results
The clot integration factor was significantly higher (23% increase,
Conclusions
Faster actuation frequencies may result in improved clot integration with the Tigertriever device. This effect needs to be validated by clinical data.
Introduction
Nearly a decade ago, randomized clinical trials showed that mechanical thrombectomy was superior to medical management for the treatment of large vessel occlusion strokes. Extraction of thrombi in the fewest attempts is known to result in better clinical outcomes, but despite technical advances that have produced overall successful recanalization rates of 80% to 90%,1,2 first pass recanalization rates remain low at 30% to 45%.3–6 Good clinical outcomes (modified Rankin Scale mRS of 0–2 at 90 days) are also reported in less than 60% of cases.3,5–9 The primary potential device-related causes of poor outcomes include inconsistent success requiring multiple passes to achieve revascularization, vessel wall injury, and tractional load on the thrombus resulting in fragmentation and distal embolization.10–16
Tigertriever (Rapid Medical, Yokneam, Israel) is a relatively new clot retriever that allows for controlled actuation of the diameter via a core wire connected to the distal end of the device. The core-wire is proximally connected to an actuator handle that operators can use to adjust device diameter under fluoroscopic visualization17,18 depending on clot characteristics and vessel diameter/location. The ability to radially actuate the device and “relax” it during clot retrieval may reduce distal embolization by facilitating better clot encapsulation19,20 and may reduce vessel wall injury. 21 In contrast to other stent-retrievers that open passively, the Tigertriever provides a unique opportunity to optimize several parameters such as amplitude and frequency of the actuation cycle or rate of expansion and contraction in order to improve device efficacy. These parameters have not been evaluated previously. Here, we conduct an in vitro study to evaluate the effect of the actuation frequency on clot integration with the Tigertriever 17.
Methods
A jig was manufactured to hold three silicone tubes of 2.4 mm (3/32 in.) inner diameter. The tubes were submerged in 37 °C water. Radiopaque thrombi in 2.4 mm diameter tubes were obtained from the University of Massachusetts. The clots are prepared from whole bovine blood with a solution of calcium chloride, thrombin, and fibrinogen and have physiological mechanical properties.
22
The clots were inserted into each of the tubes from the distal ends and positioned within the test section window (Figure 1); thrombus length within the tube was always 2.5 cm. A 017 microcatheter was inserted through the rotating hemostatic valve and positioned distal to the thrombus. A Tigertriever 17 was inserted through the microcatheter and the microcatheter was unsheathed to deploy the stent retriever within the clot. As the device has an active actuation mechanism, it does not self-expand when deployed; the diameter of the device is at its minimum in the deployed state. The Tigertreiver17 was then actuated with three different actuation frequencies:
Passive ( Slow ( Fast (

Image of the benchtop setup used to evaluate clot integration. Clots were placed within the segment marked as “Test Section.” The Tigertriever devices were inserted through the rotating hemostatic valves on the right side of the image.
The sample size per group was chosen based on the effect size reported in a previous in vitro study 23 and our estimation that the actuation of the Tigertriever may perform better than the pushing technique for conventional stent-retrievers. Fast, passive, and slow actuations were conducted in sequence for each trial and the trials repeated. In the passive group, there was a wait time of 2 min after opening the device. After actuation, each device was radially contracted to 3 clicks on the device handle. This brings the device to the retrieval state, which is used clinically to minimize device wall apposition and endothelial damage during clot retrieval. A flat-detector CT scan (Dyna CT, Siemens Healthineers, Erlangen, Germany) was then acquired of the device and clot. Each individual device unit was only used for a maximum of two trials, resulting in a total of 10 individual units being used for the 19 trials.
Each Dyna CT scan was reconstructed with a small volume of interest around the device. The reconstructed axial slices were then imported into 3D Slicer (www.slicer.org). Three objects—the silicone tube, the clot, and the stent retriever—were periodically marked on the slices using the Paint, Draw, and Level Tracing functions in Slicer. The Grow from Seeds function was then used to segment the stent retriever and clot. The segmentations were then manually corrected as needed in the axial, coronal, and sagittal views. Two authors reviewed each segmentation to ensure consistency. Segmentation labels were exported in Nrrd format and then read 24 into Matlab (Mathworks, Natick, MA). In each axial slice, a spline was fit to the convex hull around the stent retriever wires and clot, thereby marking the boundaries enveloping the stent and the clot (Figure 2). The clot integrated within the stent was calculated as the intersection between the stent envelope and the clot envelope. All axial slices were processed to obtain the total clot volume and total volume of clot integrated within the stent. The ratio of integrated clot volume to total clot volume was recorded as the clot integration factor (CIF). 23 The CIF was used to statistically (ANOVA, Graphpad Instat, La Jolla, CA) compare the clot integration effectiveness of the three actuation frequencies. Tukey's post-test was used to compare pairs.
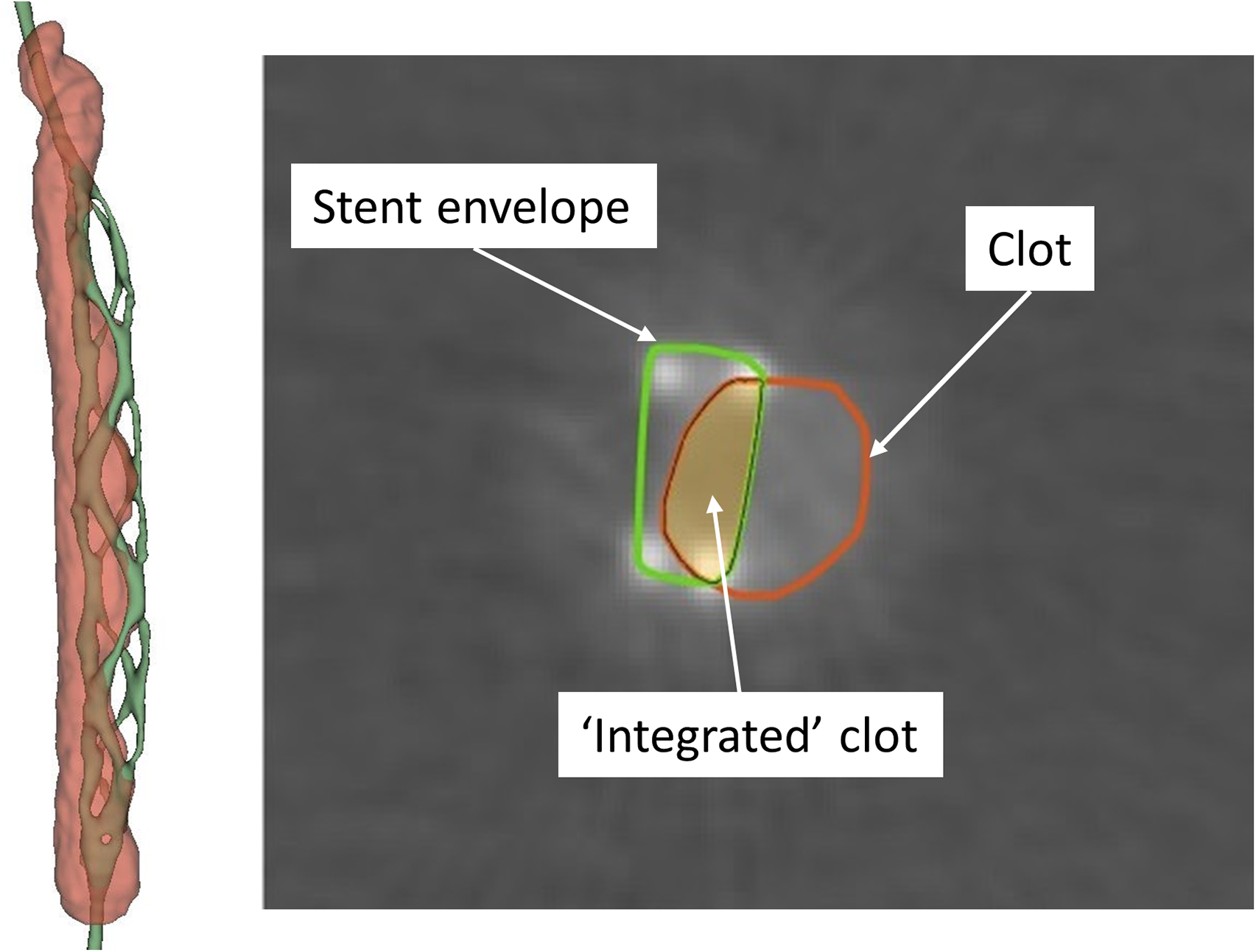
(Left) Image of clot and Tigertriever segmented in 3D Slicer. (Right) One axial cross-section showing the boundary (stent envelope) of the device and the boundary of the clot. The intersection of these two boundaries in each axial cross-section was used to calculate the volume of clot integrated within the stent.
Results
The CIF was significantly higher with the fast actuation (26.7 ± 3.1%) as compared to the passive (21.5 ± 3%) and slow (21.9 ± 2.8%) actuations (Figure 3,
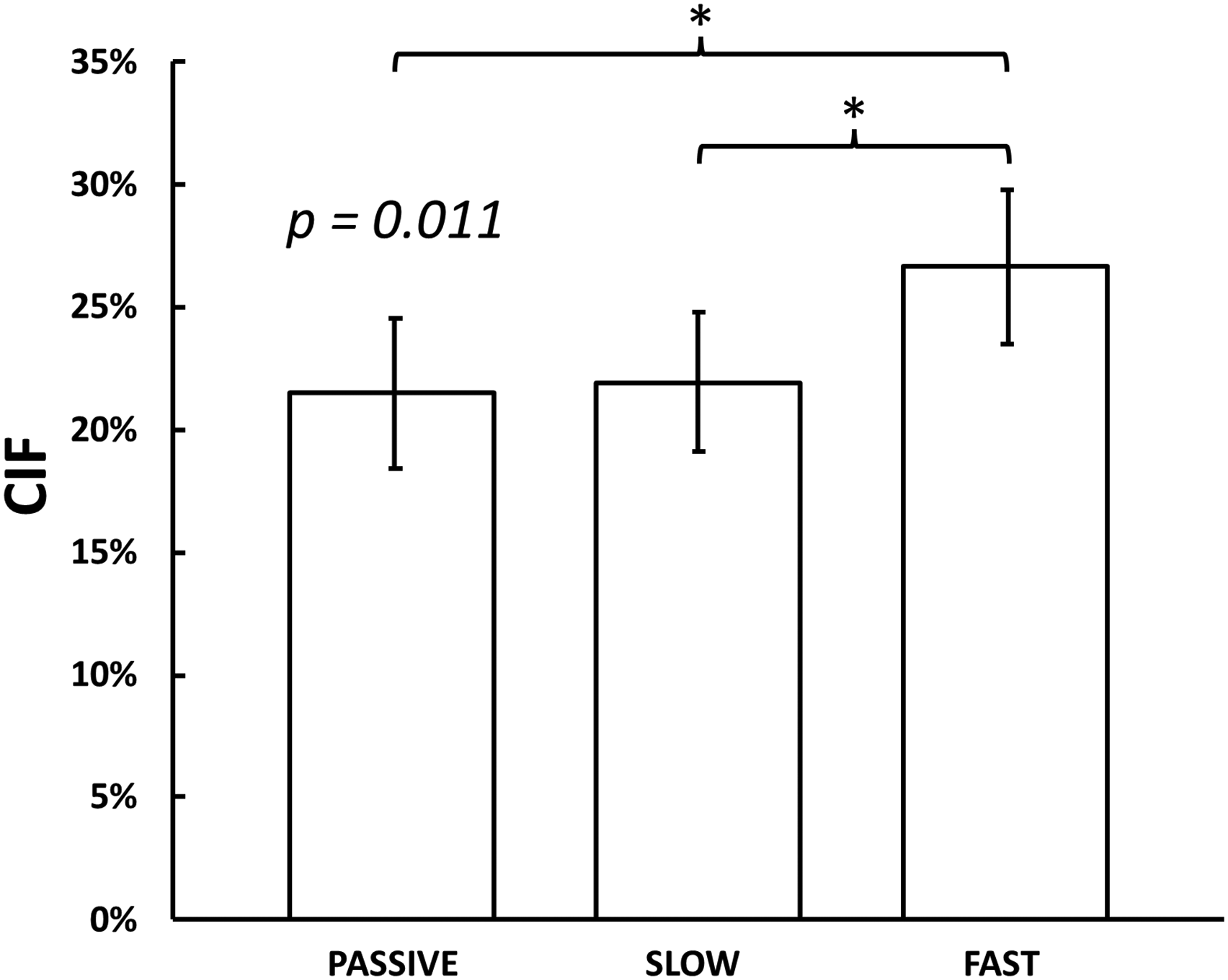
Clot integration factor (CIF, percentage ratio of volume of clot integrated within the stent to total volume of clot) for the three actuation frequencies—passive (single unsheathing), slow (20 s per actuation cycle), and fast (5 s per actuation cycle).
Discussion
The Tigertriever stent retriever (Rapid Medical) allows active operator-controlled radial expansion. This creates an opportunity to study and optimize several actuation-related parameters to maximize clot capture. These actuation parameters have not been systematically studied previously. In this study, we evaluated the effect of actuation frequency on clot integration in a benchtop setup.
We found an approximately 25% increase in clot integration when the Tigertriever was actuated at 5 s/cycle (fast) as compared to 20 s/cycle (slow) or no cycling (passive). These results are concordant with a previous clinical study 18 that found repetitive expansion-contraction was more successful than single unsheathing (which corresponds to our passive group). The push-and-fluff technique with conventional closed-cell stent retrievers has also been shown to improve first-pass and overall recanalization rates as compared to single unsheathing. 25 The Tigertriever supplants the passive push-and-fluff effort with an actively controlled radial actuation, which may improve the ability to capture clots and recanalize vessels. Successful recanalization rates (including first pass effect) were greater with repetitive expansion-contraction as compared to single unsheathing in the previous clinical study, 18 but without statistical significance. The time-period of cycling was not evaluated in the previous study.
We hypothesize that the greater acceleration of the stent mesh wires in faster actuation cycles results in increased radial forces that improve clot integration. We also found no difference in clot integration between the single unsheathing passive actuation group and the slow 20 s/cycle actuation group. This may again point to a minimal level of acceleration required for better clot integration over passive opening. It should be noted that faster rates of expansion and contraction can potentially increase clot integration independent of the cycling frequency and the variables that dictate the temporal profile of the actuation cycle need to be evaluated in the future.
During clinical use, the Tigertriever could potentially be expanded slowly under fluoroscopic visualization during the initial expansion till roadmap or vessel displacement is observed 21 to determine a feasible “safe” amplitude/click-position on the actuator handle. The device could then be cycled quickly between the deployed state (completely contracted by the actuator handle) and this “safe” radial amplitude to minimize vessel injury. We observed no statistical difference in clot integration when the device was cycled 6 times versus 12 times in the fast group. The total actuation time may thus provide diminishing returns and the minimum number of cycles required to achieve maximal integration is another variable that needs to be evaluated in future studies.
Limitations
The study was conducted in a straight tube without flow. Tortuous/curved anatomies (with potential vessel displacement during actuation) and the presence of flowing fluid proximal to the clot would likely affect the results. Also, the CIF was calculated with the device in situ and clot retrieval was not attempted. We opted to use these simplifying conditions to control for these factors and assess the effect of actuation frequency during the first step of a Tigertriever thrombectomy procedure. We used a spline curve to represent the envelope of the stent. While different methods 23 can be used to represent the stent envelope, our comparisons are valid because the device is the same and stent segmentations were not different between the groups. Also, after removal of the device from the test setup, a larger proportion of clot was subjectively visualized to be enmeshed within the device after fast actuation as compared to the slow and passive groups in accordance with the quantitative CIF values (Supplementary Figure S1). We used the same biological clot (with approximate stiffness in the range of red thrombi extracted from plaques during carotid endarterectomy, Supplementary Figure S222,26) for all samples and the increase in clot integration observed with the fast actuation may change with varying clot stiffness and elasticity. While in vitro studies such as this can provide valuable information by parsing the different variables involved, the actual utility of optimizing these variables must be confirmed by clinical data.
Conclusion
The actuation mechanism of the Tigertriever device introduces the possibility of optimizing several parameters to improve clot capture efficacy. We evaluated one of these parameters—actuation frequency—and found that higher frequencies result in improved clot integration with the tested clot composition. This effect needs to be confirmed in a more physiologically representative in vitro setup, and most importantly, in the clinical setting.
Footnotes
Author contributions
CS is guarantor and contributed to development of experimental setup, analysis methods, data interpretation, and drafting of the manuscript. ND and MG contributed to data analysis and drafting of the manuscript. RS contributed to data analysis. DF contributed to critical revision of the manuscript.
Declaration of conflicting interests
DF has served as a consultant for Rapid Medical. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was partially funded by Rapid Medical.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
