Abstract
Background
Post-mechanical thrombectomy (MT) intracranial hemorrhage (ICH) is a major source of morbidity in treated acute ischemic stroke patients with large vessel occlusion. ICH expansion may further contribute to morbidity. We sought to identify factors associated with ICH expansion on imaging evaluation post-MT.
Methods
We performed a retrospective cohort study of patients undergoing MT at a single comprehensive stroke center. Per protocol, patients underwent dual-energy head CT (DEHCT) post-MT followed by a 24-h interval non-contrast enhanced MRI. ICH expansion was defined as any increase in blood volume between the two studies if identified on the DEHCT. Univariate and multivariable analyses were performed to identify risk factors for ICH expansion.
Results
ICH was identified on DEHCT in 13% of patients (n = 35/262), with 20% (7/35) demonstrating expansion on interval MRI. The average increase in blood volume was 11.4 ml (SD 6.9). Univariate analysis identified anticoagulant usage (57% vs 14%, p = 0.03), petechial hemorrhage inside the infarct margins or intraparenchymal hematoma on DEHCT (ECASS-II HI2/PH1/PH2) (71% vs 14%, p < 0.01), basal ganglia hemorrhage (71% vs 21%, p = 0.02), and basal ganglia infarction (86% vs 32%, p = 0.03) as factors associated with ICH expansion. Multivariate regression demonstrated that anticoagulant usage (OR 20.3, 95% C.I. 2.43–446, p < 0.05) and ECASS II scores of HI2/PH1/PH2 (OR 11.7, 95% C.I. 1.24–264, p < 0.05) were significantly predictive of ICH expansion.
Conclusion
Expansion of post-MT ICH on 24-h interval MRI relative to immediate post-thrombectomy DEHCT is significantly associated with baseline anticoagulant usage and petechial hemorrhage inside the infarct margins or presence of intraparenchymal hematoma (ECASS-II HI2/PH1/PH2).
Keywords
Introduction
Mechanical thrombectomy (MT) has become the standard of care in the treatment of large vessel occlusion (LVO) in the acute ischemic stroke (AIS) population.1–4 Hemorrhagic transformation (HT) of ischemic parenchyma resulting in intracranial hemorrhage (ICH) is thought to be a major driver of post-treatment morbidity in this patient population. 5 The incidence of post-MT ICH is variable but is reported to be as high as 15% with only a portion of these (4–14%) being symptomatic.6–9 The ICH found on post-MT scans range from subarachnoid hemorrhage related to the procedure, hemorrhagic conversion from ischemic territories, or new hemorrhage in an unexpected region.10–12 The proportion of post-thrombectomy ICH that demonstrate expansion in the acute phase (within 24–48 h) is not well documented. Delayed identification of ICH expansion can lead to poor neurological outcomes including significant disability or death. 13
Early detection of post-MT ICH expansion and risk stratification of such patients may allow for tailored management to improve outcomes. 14 The literature suggests several preprocedural and intraprocedural risk factors for post-MT ICH. Preprocedural risk factors include time to treatment, presence of atrial fibrillation (AF), elevated C-reactive protein (CRP), elevated mean arterial pressure (MAP), use of antiplatelet agents, thrombocytosis, number of thrombectomy passes, and a diagnosis of diabetes mellitus.10–12,15–20
In this study, we conducted an exploratory analysis to identify predictors of post-MT ICH expansion in immediate post thrombectomy dual-energy head CT (DEHCT) ICH-positive patients at a single comprehensive stroke center.
Methods
Study design
We performed a retrospective cohort study on MT patients at a single, comprehensive stroke center between January 2018 and December 2021. Per our institutional protocol, MT patients undergo immediate post-procedural DEHCT and an interval non-contrast enhanced MRI 24–48 h afterward if no contraindications to MRI exist. All MT patients who underwent both exams within the appropriate time frame and had evidence of ICH on the initial DEHCT were included. Patients who did not receive both examinations per protocol were excluded. The study was approved by the institutional review board (#221603), and data accession and storage were performed in accordance with the Health Insurance Portability and Accountability Act (HIPAA).
Definition of expansion events
Blood on the DEHCT was assessed on the virtual non-contrast (VNC). Blood on MRI was assessed in the T2-weighted fast field echo (FFE) sequence. Volume of ICH expansion was measured directly using the Sectra IDS7 volume calculation tool (Sectra AB, Linköping, Sweden) on either VNC or FFE (Figure 1). An expansion event was defined as any increase in the total blood volume between the post-MT DEHCT and the 24-h MRI scan (usually conducted 24–48 h post-MT). Expansion events included both increases of blood volume at one specific location or HT in a new location.
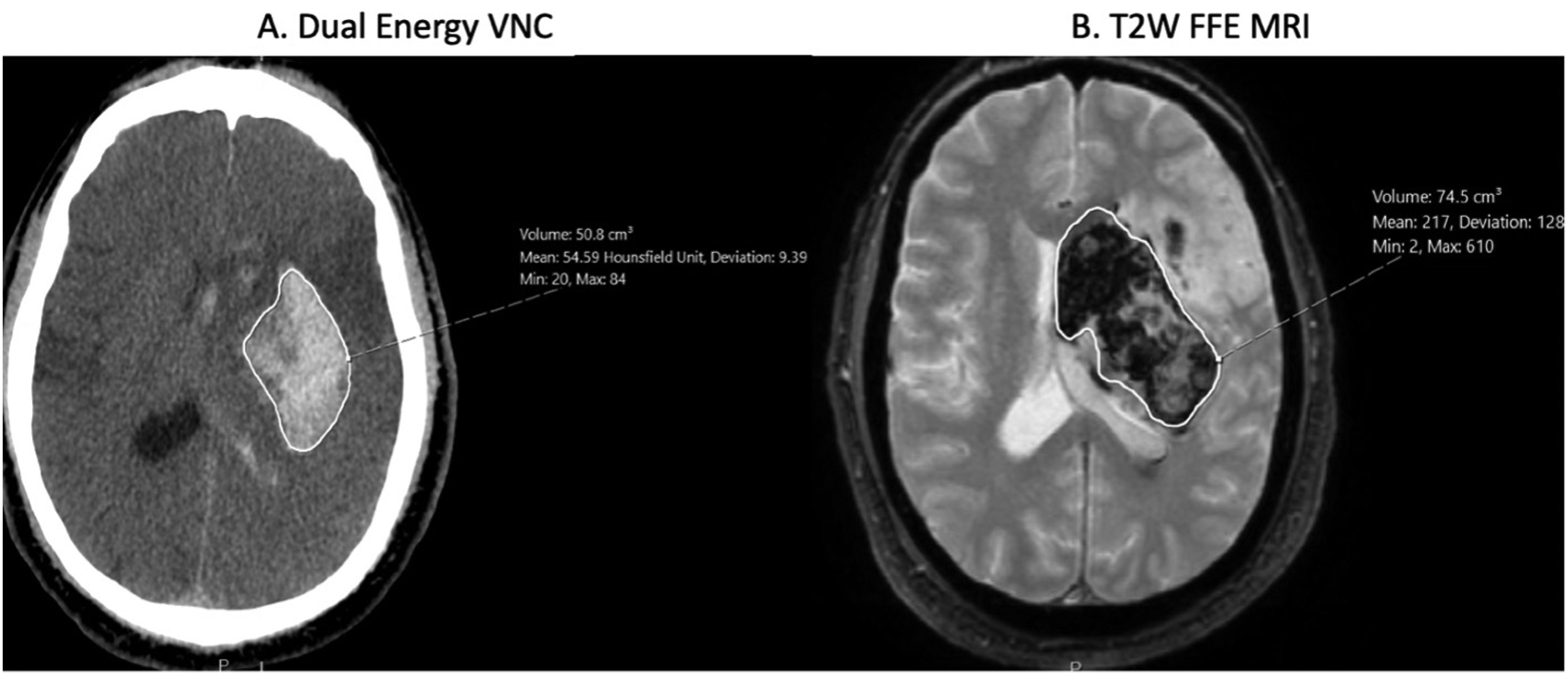
Hemorrhage expansion was calculated using Sectra's volume calculation tool instead of the traditional ABC/2 method. The software groups areas of similar densities (A. CT) or intensities (B. MRI) together, the borders of which can be manually adjusted.
Data collection
Data from patient records were manually extracted by the research team and stored securely using REDCap. 21
Statistical analysis
Multivariable logistic regression analyses were performed to identify independent predictors of ICH expansion. Variables used in our adjusted analysis were selected based on significance on univariate analysis (p < 0.05) and previously described post-MT ICH risk factors. Covariates included: patient age, systolic blood pressure, NIH Stroke Scale (NIHSS) on arrival, anticoagulant use, antiplatelet use, pre-thrombectomy HbA1C, European Cooperative Acute Stroke Study (ECASS) II score, ischemic territory, use of general anesthesia, and number of thrombectomy passes. Model selection was conducted by an exhaustive search using the listed variables applying a sequential replacement method. An array of models with different numbers and combinations of predictors was created in a sequential, stepwise fashion. Covariate selection was limited to between one and four covariates to reduce model overfitting. Model performance was assessed to maximized adjusted R² and minimized predictor error as measured by Mallow's Cp and Bayesian Information Criterion. With similar performing models, the likelihood ratio test was also used to compare goodness-of-fit between models. p-Value ≤ 0.05 was considered significant. All statistical analysis was performed using R version 4.2.2 (The R Foundation, Vienna, Austria).
Results
Patient demographics
A total of 394 mechanical thrombectomies were performed at our comprehensive stroke center between January 2018 and December 2021. A total of 262 (66%) patients met our initial screening criteria of having undergone both an immediate post-MT DEHCT and a 24-h interval MRI. Of these patients, 35 (13%) had hemorrhage detected on DEHCT. Of the 35 patients, seven (20%) were noted to have ICH expansion on subsequent 24-h MRI. These events included three instances of expansion of existing ICH and four instances of new HT in a discrete location (Table 1). A total of 132 patients did not receive the full protocol imaging and were thus excluded: 47 (12%) did not receive a DEHCT and 85 (22%) could not have an MRI or had an MRI > 48 h post-thrombectomy (Supplemental Figure S1).
Clinical presentation characteristics.
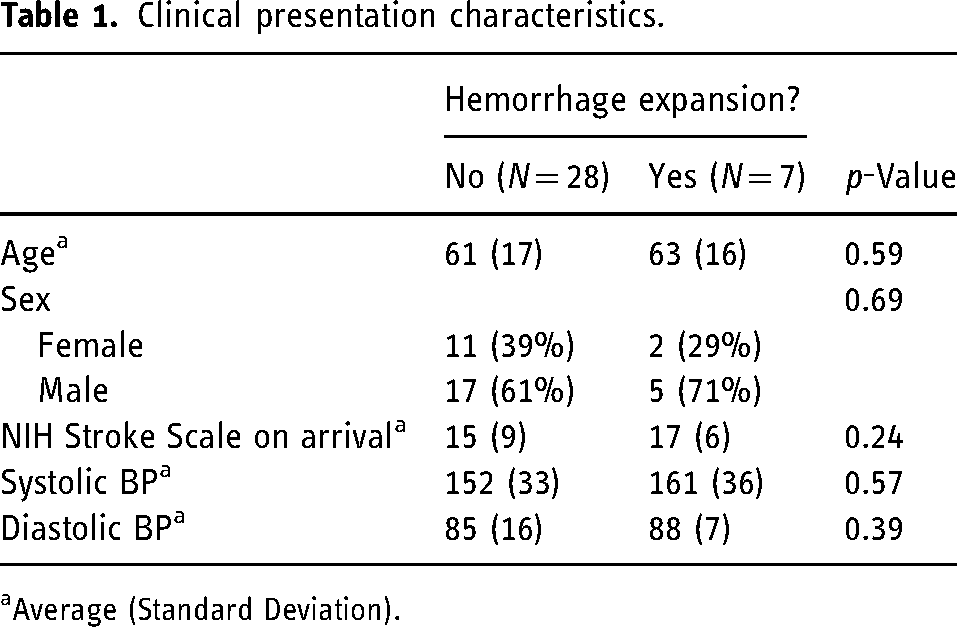
Average (Standard Deviation).
Pre-procedural risk factors
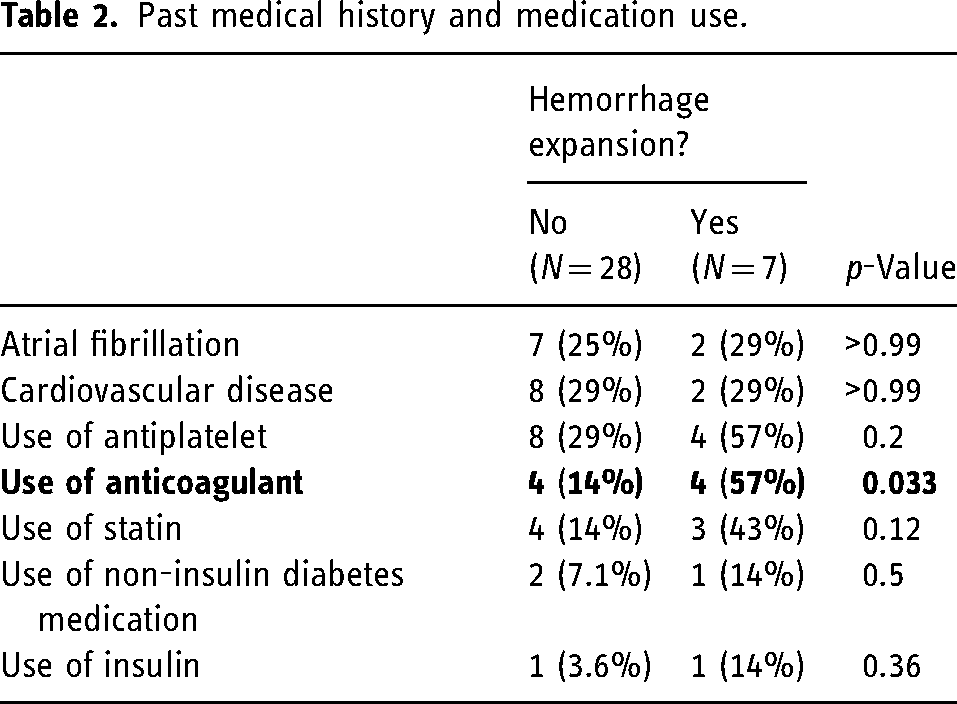
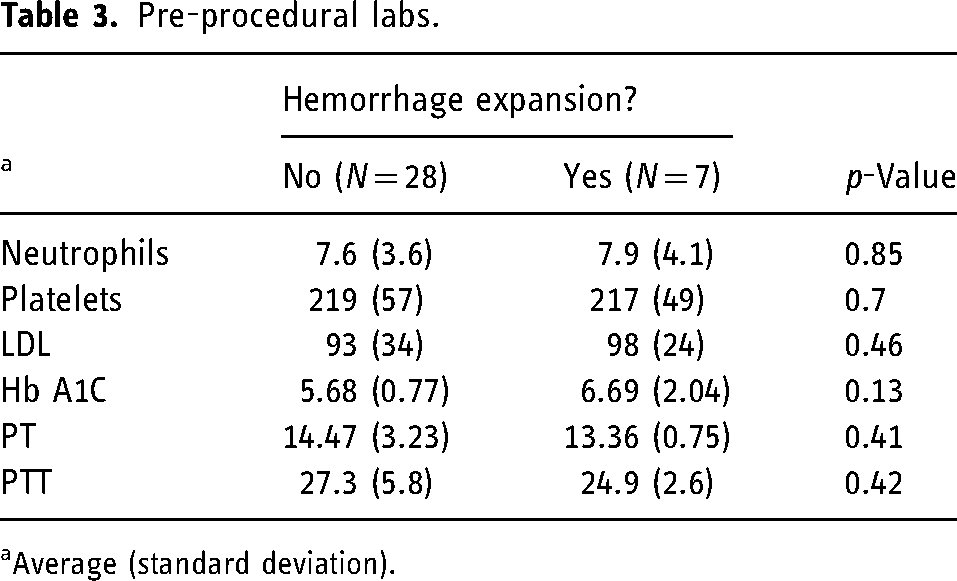
Past medical history, medication usage, and pre-procedural admission labs are compared between the ICH expansion group and the control group (Tables 2 and 3). Patients with ICH expansion had significantly higher rates of anticoagulant usage compared to the control group (57% vs 14%, p = 0.03), with no significant differences detected in the use of antiplatelets, statins, insulin, or non-insulin diabetes medications. There were also no significant differences detected in pre-procedural admission labs.
Past medical history and medication use.
Pre-procedural labs.
Average (standard deviation).
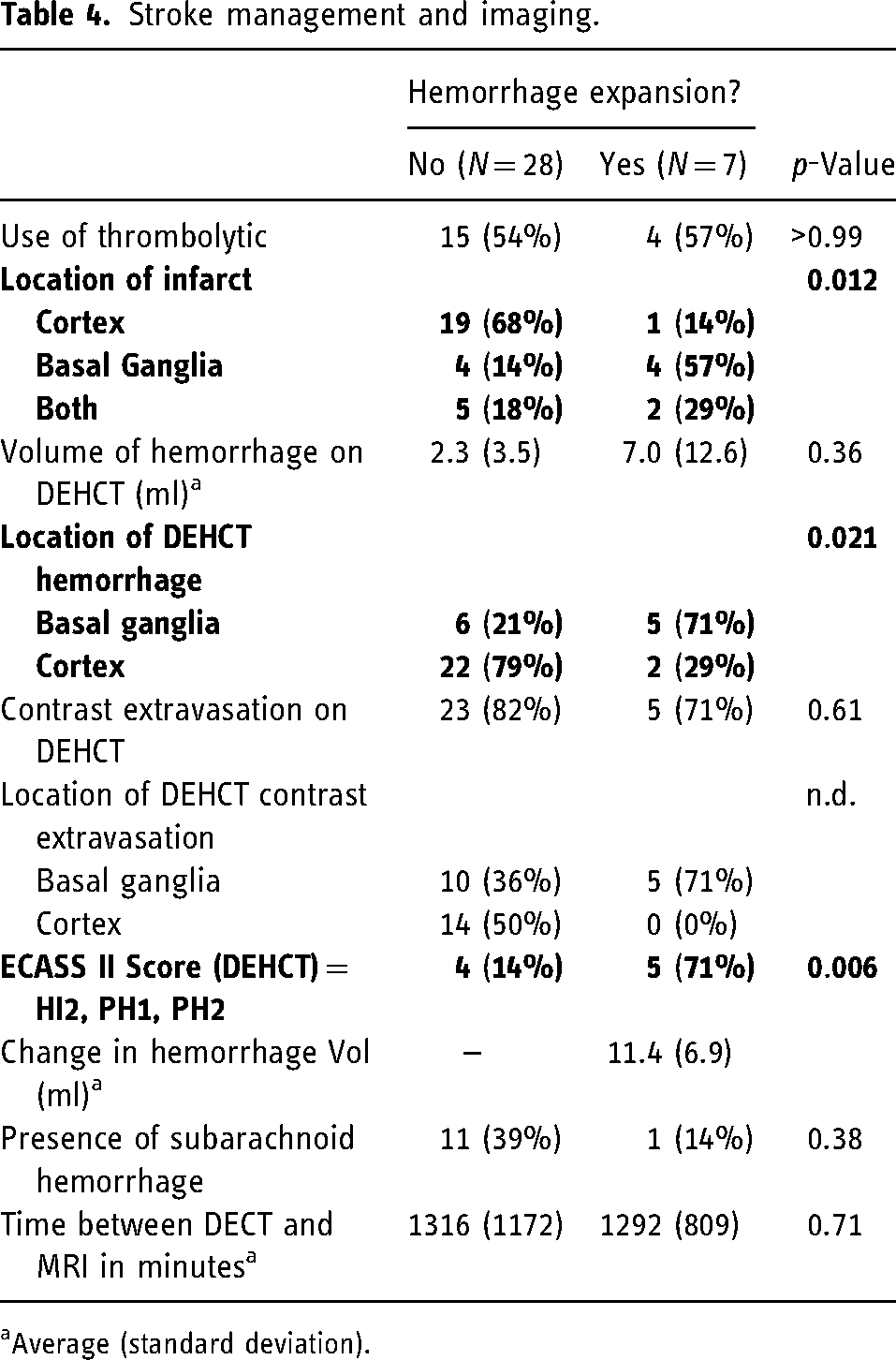
Pre-procedural stroke management and imaging
The use of intravenous thrombolytics (57% vs 54%, p > 0.99) was not associated with ICH expansion (Table 4). Importantly, the interval time between DEHCT and MRI for patients who had ICH expansion and those without was not statistically significant (21.5 h vs 22 h, p = 0.71). The presence of subarachnoid hemorrhage was not associated with expansion. However, expansion was associated with infarction of the basal ganglia (86% vs 32%, p = 0.03), ICH located in the basal ganglia (71% vs 21%, p = 0.02), and non-HI1 ECASS II scores (71% vs 14%, p < 0.01) (Figure 2). The average increase in volume was 11.4 mL with a standard deviation of 6.9.
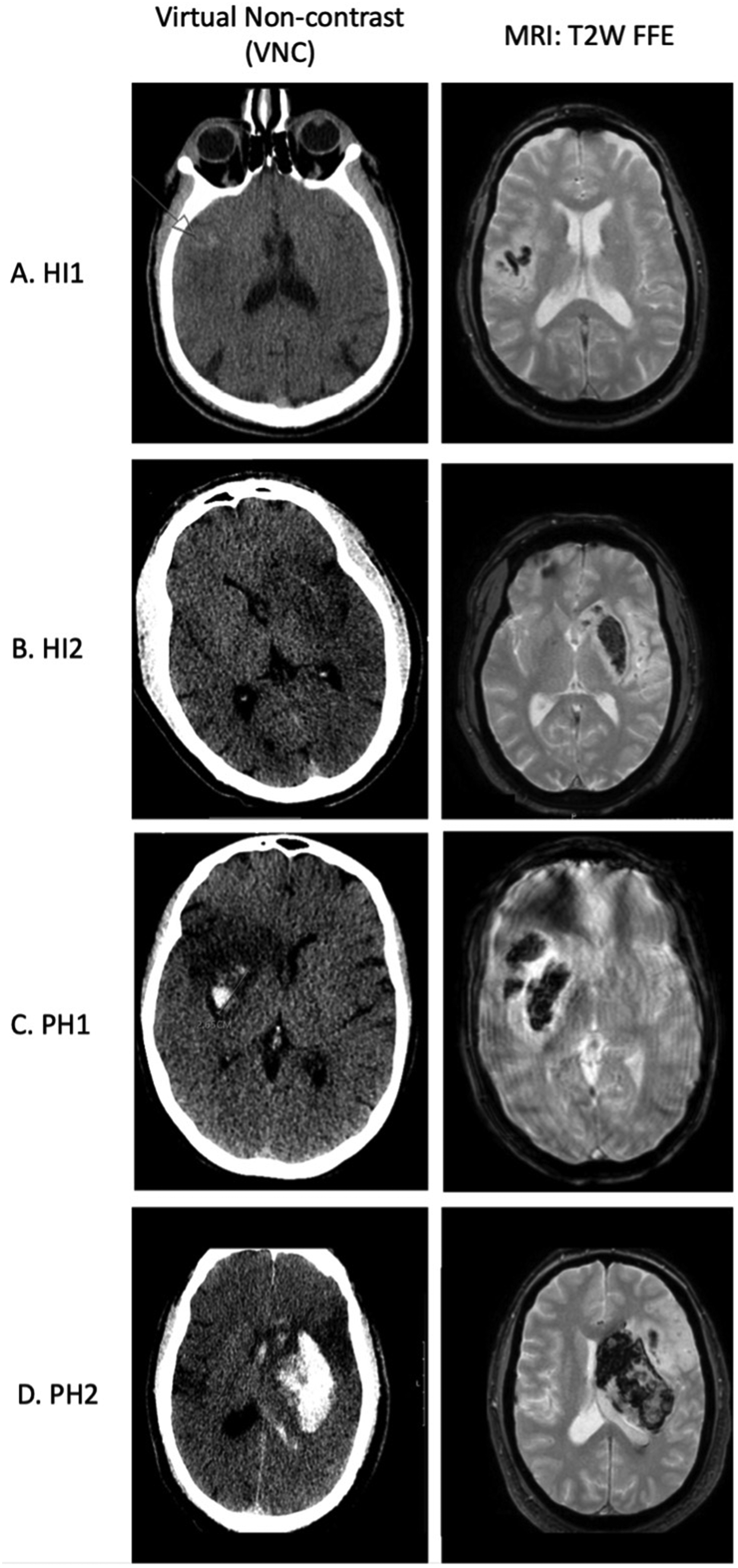
Two scans, dual-energy head CT (virtual non-contrast) immediately post-MT and MRI 24 h later. Example of ECASS II score grading in VNC. HI1 (A) is a petechial hemorrhage along the infarct margin and is not associated with expansion. HI2 (B) is petechial hemorrhage inside the infarct margin, PH1 (C) is hematoma <30% of the infarcted area with no/mild mass effect, and PH2 (D) is hematoma >30% of the infarcted area with mass effect. HI2, PH1, and PH2 are associated with ICH expansion.
Stroke management and imaging.
Average (standard deviation).
Procedural characteristics
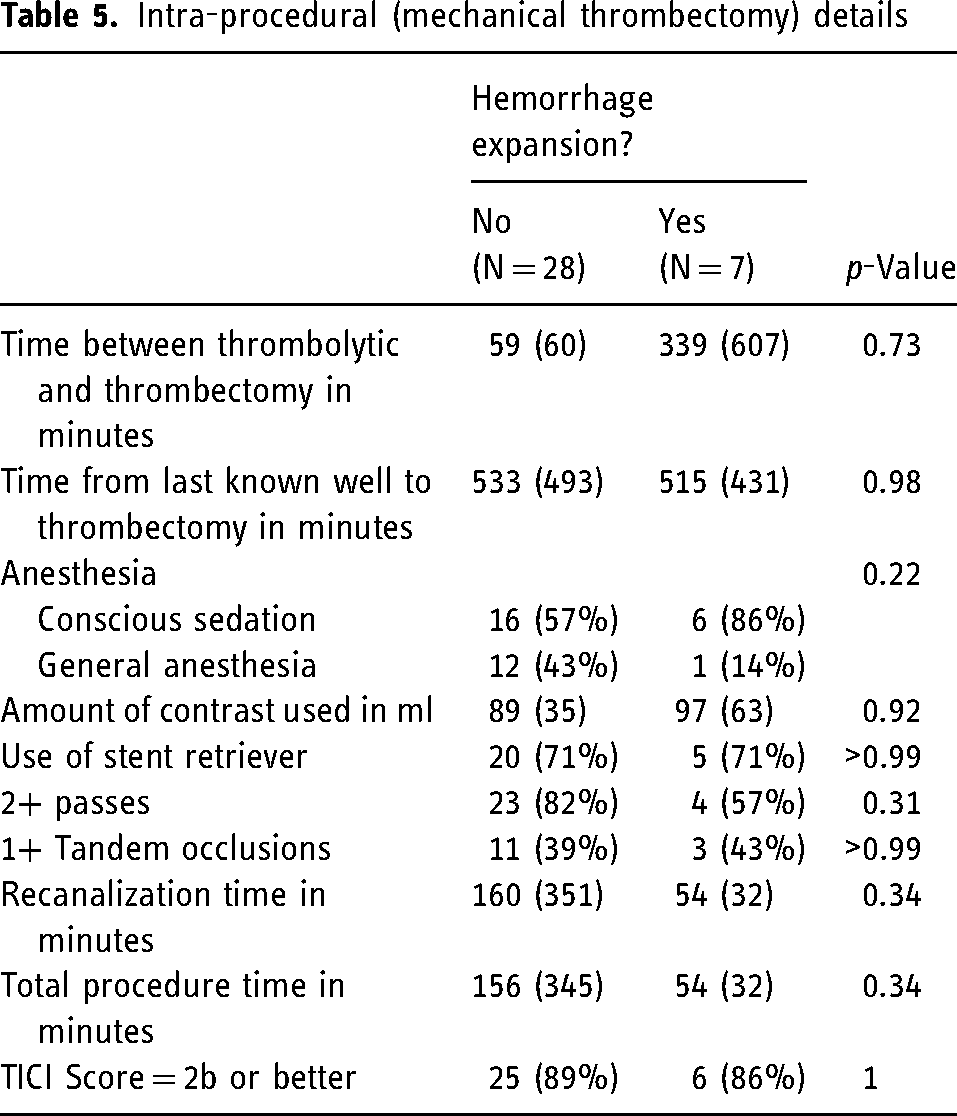
Intra-procedural factors, including time between last known well and thrombectomy, type of anesthesia, iodinated contrast volume, number of thrombectomy passes, presence of tandem occlusion, recanalization time, use of stent retrievers, and procedure time were not significantly associated with ICH expansion (Table 5).
Intra-procedural (mechanical thrombectomy) details
Adjusted analysis
Multivariable logistic regression model controlling for confounders showed that anticoagulant usage (OR 20.3, 95% C.I. 2.43–446, p < 0.05) and ECASS II scores of HI2, PH1, or PH2 (OR 11.7, 95% C.I. 1.24–264, p < 0.05) had were significantly associated with ICH expansion (Table 6).
Adjusted analysis.

Covariates included: patient age, NIH Stroke Scale (NIHSS) on arrival, anticoagulant use, ECASS (European Cooperative Acute Stroke Study) II score.
Clinical outcomes
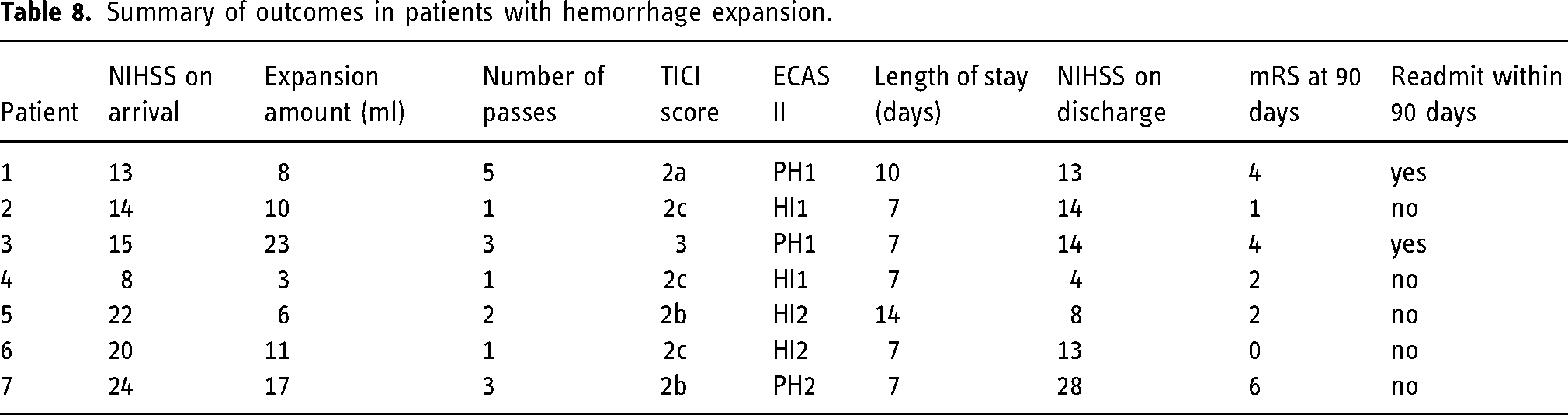
Outcomes were compared between the ICH expansion group vs. the control group (Table 7). No statistically significant differences were detected across all outcome variables. The outcomes of patients with expansion events are summarized in Table 8.
Outcomes.

Average (standard deviation).
Summary of outcomes in patients with hemorrhage expansion.
Discussion
It is estimated that about 15% of patients undergoing MT develop ICH. 6 While only symptomatic ICH has a well-documented association with worsened clinical outcomes (and the effect of asymptomatic ICH on outcomes remains mixed), not much is known about the evolution of post-MT ICH, that is, its transition into new ICHs or expansion of existing locations. 22 This study aimed to assess both of these events.
To this end, we performed a single-institution, retrospective cohort study to identify predictors of ICH expansion following MT, as defined by interval increase in blood volume between post-MT DECT and 24-h interval MRI. By this definition, we included all events, including both true expansions of ICH in existing locations and new ICH in a different location. We found that prior anticoagulant usage and petechial hemorrhage inside the infarct margins (HI2) or the presence of intraparenchymal hematoma (PH1 or PH2) were significantly associated with ICH expansion. Notably, the use of intravenous thrombolytics was not predictive of HT, congruent with the existing literature. 7 In fact, ECASS II scores have already been independently shown to be predictive of both the development of symptomatic ICH and poor outcomes in patients with HT of ischemic brain tissue.7,23 Studies that have explored risk factors for initial post-MT ICH also find thrombolytic use to be independent of the development of ICH. 7 Additionally, studies exploring risk factors for ICH expansion also list anticoagulant usage as a potential risk factor. 24 Our data suggests that post-MT ICH expansion may behave in a similar manner to that of ICH seen in varying etiologies.7,25
In our unadjusted analysis, we found that basal ganglia ischemia or basal ganglia ICH were associated with ICH expansion. The pathophysiologic explanation between involvement of the deep brain structures and ICH expansion is unclear, however several studies have shown basal ganglia hemorrhages are associated with poor outcomes.26,27 Further research is needed to explore the link between clinically significant bleed locations (such as basal ganglia hemorrhage over cortical hemorrhage or procedural SAH) and its association with expansion. Additionally, a larger sample size is also needed to assess the link between ICH expansion and clinical outcomes as this study could not establish association between outcome measures like the length of stay, modified Rankin score at 90 days, or readmission rates with its low overall sample size.
In our retrospective cohort, we were able to assess factors associated with increased hemorrhage at 24 h on MRI compared to immediate DEHCT. These factors could be used clinically to assess which patients are most at risk for hemorrhage expansion. This would also help delineate what factors (changes to management protocols such as tightening blood pressure control, delaying initiation of antiplatelet/anticoagulant medications, reversal of antiplatelet/anticoagulant medications, or decreasing the interval between repeat imaging) may be modified to prevent this expansion.
There are several limitations that should be acknowledged. First, it is important to note that this is a single-center, retrospective cohort study, which may limit the generalizability of the results to other settings. Second, the small number of patients who developed post-thrombectomy ICH expansion may have limited the statistical power of our analyses. Over a 3-year period, only 2.7% of the included MT patients (7/262) had an expansion event. Thus, in conducting our adjusted analysis controlling for covariates, we were limited to one to four covariates to reduce model overfitting, limiting our ability to control for multiple confounding variables. Third, a significant portion of the patients who underwent MT (132/394) were excluded for failing to undergo either immediate post-MT DEHCT or 24-h MRI due to varied reasons (Supplementary Figure S1). The two scans at relatively specified time points allow us to track how hemorrhage in the initial scan evolves in an acute time setting as determined by subsequent 24-h imaging. If patients don’t have an initial scan or a subsequent scan in the acute period, we are unable to track the evolution of their bleeding post-MT. However, it is important to note that only nine of the 85 patients had ICH on DEHCT, which we believe is unlikely to have significantly biased our findings. Fourth, our study focused on imaging features of ICH expansion rather than symptomatic nature of the expansion. It was felt that due to the low number of hemorrhage expansions it would be difficult to clarify the significance of these expansions. We acknowledge that this could have prevented us from gaining a more detailed understanding of the clinical significance of post-thrombectomy ICH expansion and is recommended as the direction of future studies.
Conclusion
Our study found out that prior anticoagulant usage and presence of petechial hemorrhage or intraparenchymal hematoma independently predicts ICH expansion following MT for anterior circulation LVO. These findings suggest that we may be able to identify patients with immediate post-MT ICH expansion risk. Additionally, while there may be some clinically significant signals like basal ganglia hemorrhage (compared to procedural SAH or cortical hemorrhage) that may contribute to expansion, a larger scale analysis is needed to verify the veracity of such signals. Further studies with multicenter contribution and larger cohorts are needed to understand the natural history of post-MT hemorrhage, specifically the impact ICH expansion may have on the clinical outcomes of MT patients.
Supplemental Material
sj-docx-1-ine-10.1177_15910199241247884 - Supplemental material for Predictors of acute post-thrombectomy intracranial hemorrhage expansion in anterior circulation infarcts
Supplemental material, sj-docx-1-ine-10.1177_15910199241247884 for Predictors of acute post-thrombectomy intracranial hemorrhage expansion in anterior circulation infarcts by Akshay Bhamidipati, Nishit Mummareddy, Seoiyoung Ahn,c Gabriel Bendfeldt, Alexander T Lyons, Surya Gangavarapu, Jeffrey Chen, Jacob Jo, Naveed Kamal, Steven G Roth, Michael T Froehler, Rohan V Chitale and Matthew R Fusco in Interventional Neuroradiology
Footnotes
Authors contributions
AB, NM, SA, and MRF designed the study. AB, NM, SA, GB, JC, SG, and AL collected the data. AB, NM, JC, SGR, NK, RVC, MTF, and MRF analyzed the data. AB, GB, NM, JJ, SGR, NK, MTF, RVC, and MRF prepared the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
