Abstract
The American Heart Association’s Life’s Essential 8 (LE8) is comprised of both biological and behavioral measures, and it predicts cardiovascular disease (CVD). Presently, however, its relation to measures of endothelial function implicated in CVD risk is unclear. In 318 midlife adults (198 female; 55 non-white, M = 42.3 years), endothelial activity was indexed by forearm blood flow (FBF) induced by reactive hyperemia, as well as circulating cellular adhesion molecules (VCAM-1, ICAM-1). LE8 measures included fasting glucose and non-HDL cholesterol, seated blood pressure, body mass index, diet quality, smoking and sleep duration (the latter assessed with self-report inventories). Multiple regression models revealed that lower LE8 scores were related to higher ICAM-1 concentrations (r = −0.41, P < 0.001), but not VCAM-1 (r = −0.06, P > 0.05). Higher LE8 scores were related to a higher FBF %-increase induced by reactive hyperemia (r = 0.29, P < 0.001). Findings suggest that LE8 scores relate to non-invasive and cellular markers of endothelial activity among otherwise healthy midlife adults. Longitudinal studies are needed to understand the divergent associations of LE8 with ICAM-1 and VCAM-1, and to determine if changes in LE8 predict future endothelial health or other contributors to atherosclerotic CVD.
Keywords
Introduction
Atherosclerotic cardiovascular disease (ASCVD) is the leading cause of morbidity and mortality in adults, and it is generally defined as the hardening and narrowing of blood vessels due to increased levels of inflammation, plaque formation, and reduced endothelium-dependent vasodilation. 1 In the United States, about 28.6 million adults present with some type of cardiovascular disease (CVD), and ASCVD specifically accounted for 4 of every 5 cardiovascular-related deaths in 2022. 2 The development of ASCVD is influenced by multi-faceted pathophysiological processes, many of which begin preclinically and prior to the onset of clinical symptoms and events. 3 Monitoring the preclinical risk factors associated with ASCVD development, such as endothelial function (EF), is important to its prevention and future cardiovascular health.
To define guidelines for improving cardiovascular health across the lifespan, the American Heart Association introduced the “Life’s Essential 8” score (LE8), a composite 100-point parameter comprised of four health behaviors (sleep duration, diet, nicotine use, and physical activity) and four health factors (body mass index [BMI], blood pressure [BP], blood glucose, and cholesterol), all of which have been associated with cardiovascular health outcomes and are impacted by behavioral, social and structural environmental factors. 4 To date, LE8 has been associated with many adverse health outcomes, such as CVD and cancer, as well as being predictive of future CVD risk and mortality.5-7 Being introduced recently in 2022, further research using the LE8 is still needed to understand its effectiveness in monitoring ASCVD risk, especially as it relates to preclinical disease markers.
Although an inverse relationship between the LE8 and ASCVD outcomes has been increasingly documented, there are still relatively few studies evaluating LE8 and early (preclinical) indicators of ASCVD risk. Endothelial dysfunction, defined as a pro-inflammatory and thrombotic state leading to blunted arterial vasodilation, plays a key role in the development of ASCVD and precedes the formation of structural alterations in vasculature. Its determinants include several physiological risk factors, such as hypercholesterolemia, blood pressure induced shear stress, and elevated inflammation. 8 Furthermore, certain health behaviors, such as smoking and physical inactivity, are associated with diminished EF.9,10
Flow-mediated dilation (FMD) induced by reactive hyperemia, a common method of assessing EF, assesses the dilation of an artery following an ischemic occlusion of blood flow to a part of the body. 11 The maximal vasodilatory response following an occlusion reflects the structural capacity of the arteries to dilate and the acute release of circulatory vasodilators, such as nitric oxide. 12 FMD may contribute to the assessment of ASCVD risk and is predictive of future ASCVD outcomes. 13 Venous occlusion plethysmography is a non-invasive, reliable method of monitoring strain-gauge-derived volumetric changes in blood flow post-occlusion (i.e., reactive hyperemia), with lower volumetric changes being associated with worse EF. 14
Assessing the concentration of soluble cellular adhesion molecules in peripheral circulation provides complementary and additional information about endothelial inflammation contributing to EF. Damage to the endothelium results in an inflammatory response and localized recruitment of leukocytes. The trans-endothelial migration and adherence of leukocytes to the surface of endothelial cells is mediated by cellular adhesion molecules. Intercellular cell adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1) are selectins, transmembrane proteins on the surface of endothelial cells and leukocytes, that mediate the adhesion of leukocytes to the endothelium and are upregulated by various inflammatory states. 15 Elevated soluble concentrations of ICAM-1 and VCAM-1 in fasting blood have been shown to be indicative of endothelial dysfunction, greater subclinical atherosclerosis, and predictive of future ASCVD.16-18
To date, little research has compared ASCVD risk determined by the LE8 with measurements of FMD or the concentration of cellular adhesion molecules, such as VCAM-1 or ICAM-1. Accordingly, the purpose of the present study was to assess the relationships of LE8 to multiple indicators of EF in otherwise healthy midlife adults. In this study, we specifically aimed to compare these functional (FMD) and circulating (VCAM-1 and ICAM-1) markers of EF with the LE8 score in 318 adults who were free of prior and current cardiovascular diagnoses and cardiometabolic health conditions. In line with current research, we hypothesized that lower LE8 scores would relate to greater blood CAM concentrations and lower FMD, both of which are indicative of worse EF.
Methods
Participants
This cross-sectional study analyzed data collected from participants in the Neurobiology of Adult Health (NOAH) study. A total of n = 366 participants were initially recruited into the NOAH study (aged 28 – 56 years; 232 women, 133 men, and 1 other; 301 identifying as white; 34 identifying as Black or African American; 21 identifying as Asian or Asian American; 10 identifying as multiracial; 1 identifying as “other.”). 48 participants had incomplete data for one or more of the endothelial factors (ICAM-1, VCAM-1, and FBF %-increase) and were excluded from analyses, resulting in a sample of n = 318.
NOAH exclusion criteria were as follows: self-reported use of medications with central or peripheral autonomic effects on one or more occasions in the 14 days prior to testing (including antihypertensive or cardiac medications, antipsychotic medications, protease inhibitors or other anti-HIV medications, insulin, chemotherapy agents, immunosuppressants, prescription weight-loss medications and ephedrine); regular use (i.e., use on seven or more days in the 14 days prior to testing) of anti-anxiety medications, sleep medications, asthma medications and allergy inhalants, antidepressant medications, glucocorticoids, medical marijuana, more than two non-insulin medications for diabetes; consuming 35 or more alcoholic drinks in the last 7 days; consuming six or more alcoholic drinks on three or more occasions in the past seven days; use of illicit drugs on seven or more days in the past two weeks; major medical conditions, including CVD, severe hypertension (systolic BP >160 and/or diastolic BP >100 mmHg); cancer (treatment in last 12 months, except for non-melanoma skin cancer), hepatitis B/C, liver cirrhosis/failure, chronic kidney disease, and type I diabetes; self-reported history of a major neurological disorder or brain injury resulting in ongoing symptoms or cognitive impairment (e.g., multiple sclerosis, cerebral palsy, major head injury); history of schizophrenia or bipolar disorder; lung disease requiring regular or ongoing drug treatment; weight-loss surgery within the past 5 years; and for women, pregnancy.
Study recruitment methods involved mass electronic and print mailings to Allegheny County residents; radio, electronic (e.g., Craig’s List), and print advertisements in public places (e.g., Pittsburgh Port Authority Buses, local newspapers, community and park announcement boards); direct solicitation from the participant registries of the University of Pittsburgh’s Clinical and Translational Science Institute Pitt + Me Registry and University Center for Social and Urban Research Regional Research Registry; and distribution of study fliers at outdoor community events (e.g., farmers’ markets). All participants provided written informed consent after receiving a description of all study procedures prior to participation. Participants were compensated up to 250.00 USD for completing all baseline study visits and assessment protocols, and study approval was granted by The University of Pittsburgh Human Research Protection Office (Protocol number: 19030012). 19
Procedure
During the baseline period of assessment, NOAH participants attended 5 study visits. These included informed consent, medical history, demographic interviewing, and seated assessments of resting BP (visit 1); anthropometric assessments of height, weight, and body composition (visit 1 or 3); psychophysiological assessments of pulse wave velocity (PWV), cardiac autonomic activity, and forearm blood flow by VOP (visit 1 or 3); completion of questionnaires to assess health behaviors and psychosocial characteristics (visit 2) carotid artery ultrasound (not described here), and fasting phlebotomy (visit 4); and a magnetic resonance imaging (MRI) protocol (visit 5, not described here). Due to COVID-19 quarantine restrictions, some participants completed parts of the physiological protocol at visit 3 instead of visit 1. No differences were found between participants with visit 3 or visit 1 physiological data. The dates of data collection spanned 18 February 2019 to 16 December 2021.
Calculation of the LE8
The eight individual factors included in the LE8 were scored out of 100 points total as per the criteria recommended by Lloyd–Jones et al. 4 Daily sleep duration was determined using the Pittsburgh Sleep Quality Index, a self-report instrument used to assess sleep quality over a 1-month period. 20 Nicotine exposure and smoking status was determined through a self-report tobacco questionnaire (current smoker = 0, former smoker/quit <1 year or current inhaled = 25, former smoker/quit 1-<5 years = 50, former smoker/quit ≥ 5 years = 75, and never smoker = 100). Minutes of moderate-to-vigorous physical activity per week were collected using the Paffenbarger physical activity questionnaire, 21 administered during visit 4. Diet scores were determined using 2020 Healthy Eating Index (HEI) scores. The HEI is calculated based on 13 components related to diet (intake of fruits, whole grains, vegetables), with higher scores indicating heathier diet quality. 22 Physiological health factors were collected during scheduled laboratory visits. BP was measured using an electronic automated sphygmomanometer (Omron HEM-907XL Professional Blood Pressure Machine). For the assessment of BP, participants were seated with their dominant arm resting at the approximate height of the heart. After a 5-minute resting period, three BP readings were taken with 1-minute breaks between readings. An average of the last two BP readings was used for calculation of LE8. Height in inches (in.) was collected using a stadiometer. Weight and BMI were measured using a scale. Non-HDL cholesterol and blood glucose concentrations were measured from fasting blood taken at the beginning of the laboratory session. Composite LE8 scores were calculated as an average of the component scores ([sleep + nicotine exposure + physical activity + diet + blood pressure + non-HDL cholesterol + blood glucose + BMI]/8). Higher scores on this measure indicate lower risk for CVD.
Cell Adhesion Molecules
Plasma ICAM-1 and VCAM-1 concentrations were quantified from fasting blood samples using the Ella Automated Immunoassay System Simple Plex Human VCAM-1/CD106 and Simple Plex Human ICAM-1/CD54 cartridges, respectively (ProteinSimple, Bio-Techne). Kits were run according to manufacturer’s directions, with samples run in duplicate. Intra-assay coefficients of variation for ICAM-1 and VCAM-1 were 3.55% and 2.04%, respectively.
Venous Occlusion Plethysmography
Venous occlusion plethysmography was performed using the Hokanson EC5R plethysmograph monitor (D. E. Hokanson Inc, Bellevue, WA) by trained research assistants. Measurements of forearm venous volume were collected using an indium-gallium strain gauge wrapped around the right forearm. To ensure a secure fit around the forearm, the strain gauge was wrapped around the point of greatest circumference (∼5 cm below the antecubital fossa). Supra-systolic pressures were determined from a prior blood pressure reading (40 mmHg over SBP). Participants were in a seated position with the right arm resting on foam cutouts designed to keep the forearm angled upward at the approximate height of the heart. The occlusion cuff was applied to the right bicep at the location of the brachial artery. An additional inflatable cuff was wrapped around the wrist to prevent confounding changes in blood volume from wrist and hand vasculature. 23
Baseline plethysmograph readings were collected from the strain gauge (%/min) with the occlusion cuff inflated to ∼50 mmHg for 7 s. The occlusion cuff pressure was released for 8 s in between inflations, and this process was repeated for a total of 12 readings. During readings, the wrist cuff was inflated to supra-systolic pressure. Baseline flow rates (FBFbase) were calculated as an average of the twelve readings (minimum of 6 valid readings required). To assess reactive-hyperemia induced vasodilation (FBFmax), participants underwent a 5-min occlusion after a 3-min resting period in which the occlusion cuff was inflated to supra-systolic pressure. After 5 min, the occlusion cuff pressure was released, and 8 readings were collected using the same protocol as baseline readings. The occlusion protocol was repeated, with a 5-min resting period in between occlusions. FBFmax was calculated as the average max flow rate from each post-occlusion protocol. The variable reflecting FMD is FBF %-increase post-occlusion: FBF %-increase = ((FBFmax – FBFbase)/FBFbase) x 100. FBF %-increase represented an indicator of EF, with lower values suggesting worse EF. 24
Statistical Methods
Analyses were performed using R Studio version 4.5.0 (4/11/2025). Continuous variables were visually inspected for normality; 3 outliers were removed for having values outside of 3SD above/below the mean. Only one participant identified as “other” for sex at birth and was excluded from analyses due to an insufficient sample size. Thus, the final sample size was n = 318. Missing data for study variables was addressed using the Full Information Maximum Likelihood (FIML) estimation. VCAM-1 and ICAM-1 were log-transformed to correct positively skewed distributions. To account for the possible influences of social disadvantage on cardiovascular health, the National Area Deprivation Index score (ADI) was incorporated into the present analyses. Age, sex at birth (male/female), and the ADI (national) were included in analyses as covariates related to the LE8 and the endothelial factors. Bivariate correlation coefficients were calculated between all study variables. Multiple linear regression models were used to assess the influence of the LE8 and its component factors on the endothelial health markers. Model A included covariates (age, sex, and ADI) and the composite LE8 score; Model B included covariates and the individual factors of the LE8. The models were tested independently, and no hierarchical entry of variables was used. To explore potential losses in precision when using the derived categorical LE8 factor components, Model B was re-run using continuous variables for the eight factors (BMI, LDL-cholesterol, SBP, fasting glucose, minutes of weekly moderate-to-vigorous physical activity, PSQI scores, HEI scores, and smoking [smoking years] alongside the relevant covariates.
Data and analysis code are available on Open Science Framework (https://osf.io/4wrcy/).
Results
Baseline Characteristics of the Study Population.
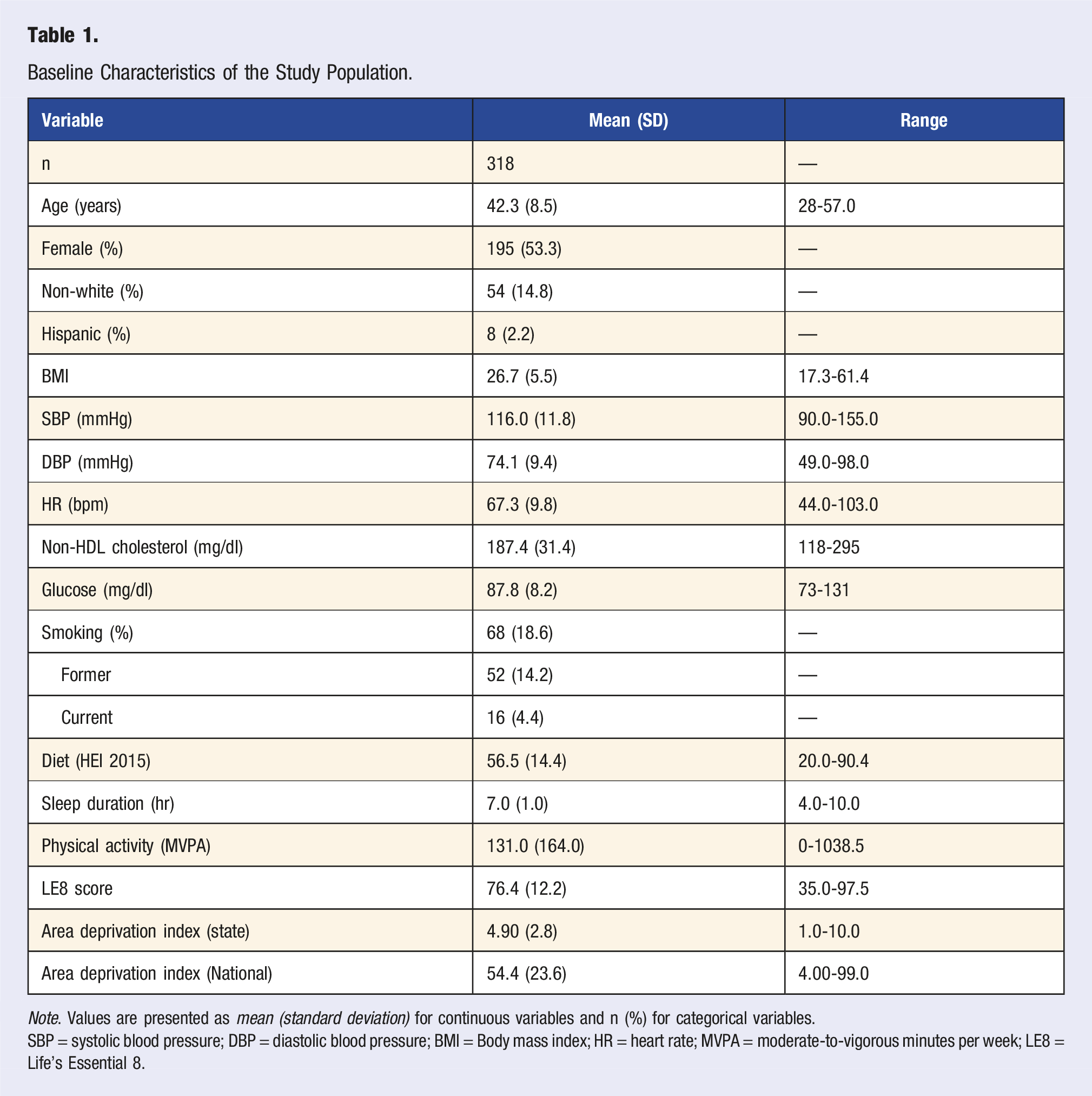
Note. Values are presented as mean (standard deviation) for continuous variables and n (%) for categorical variables.
SBP = systolic blood pressure; DBP = diastolic blood pressure; BMI = Body mass index; HR = heart rate; MVPA = moderate-to-vigorous minutes per week; LE8 = Life’s Essential 8.
Bivariate Correlations
As shown in Figure 1, ICAM-1 showed a significant negative association with the composite LE8 (r = −.41, P < .001). When assessing the LE8 components individually, BMI (r = −.34, P < 0.001), glucose (r = −.25, P < .001), non–HDL cholesterol (r = −.20, P < .001), BP (r = −.221 P < .001), diet (r = −.20, P < .001), and physical activity (r = −.20, P < .01) scores all showed significant inverse associations with ICAM-1. Translated to the raw variables, higher ICAM-1 concentration was related to greater BMI, blood glucose, non-HDL cholesterol, and BP, as well as less physical activity and worse diet quality. Additionally, higher ICAM-1 was associated with lower FBF %-increase (r = −.21, P < .001). Neither the LE8, its component factor scores, or FBF %-increase showed significant associations with VCAM-1. Note. N = 318. LE8 = Life’s Essential 8; ICAM-1 = intracellular cell adhesion molecule 1; VCAM-1 = vascular cell adhesion molecule 1; FBF = forearm blood Flow %-increase; BMI = body mass index; PA = physical activity; BP = blood pressure; Non-HDL = non-HDL cholesterol; Sex = sex (male/female); ADI = area deprivation index. Correlation coefficients displayed above use LE8 factor scores, and Not the raw values of factor.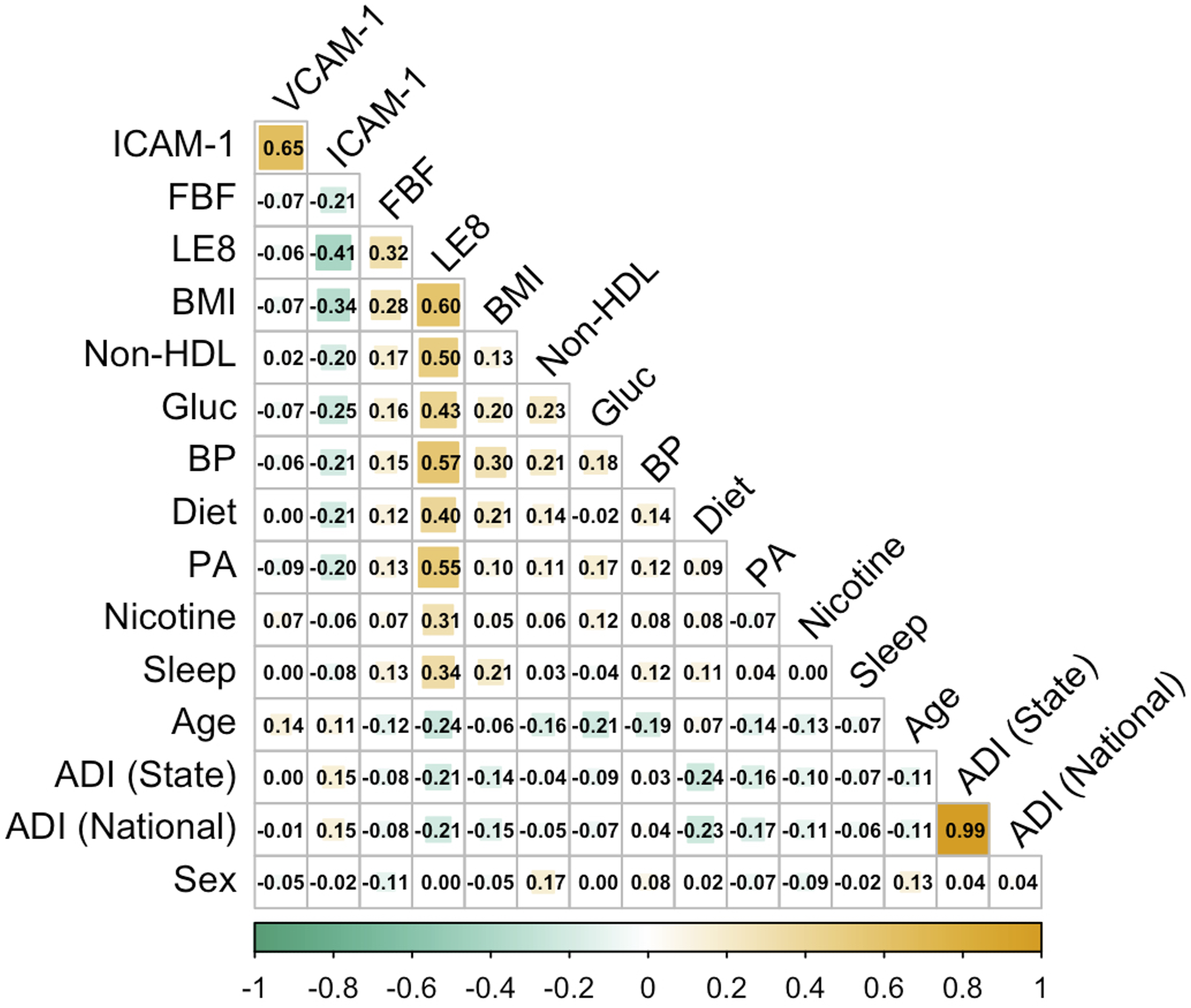
Greater FBF %-increase was associated with higher LE8 scores (r = .30, P < .001). Additionally, greater FBF %-increase related to LE8 summary scores for BMI (r = .28, P < .001), physical activity (r = .15, P < 0.01), non-HDL cholesterol (r = .17, P < 0.01), glucose (r = .13, P < 0.05), and blood pressure (r = .15, P < 0.05) scores. Translated to the raw variables, blunted reactive hyperemia (or lower FBF %-increase) related to higher BMI, non-HDL cholesterol, fasting glucose, and greater BP, as well as less physical activity.
Multiple Regression Models
Summary of Multiple Regression Results.
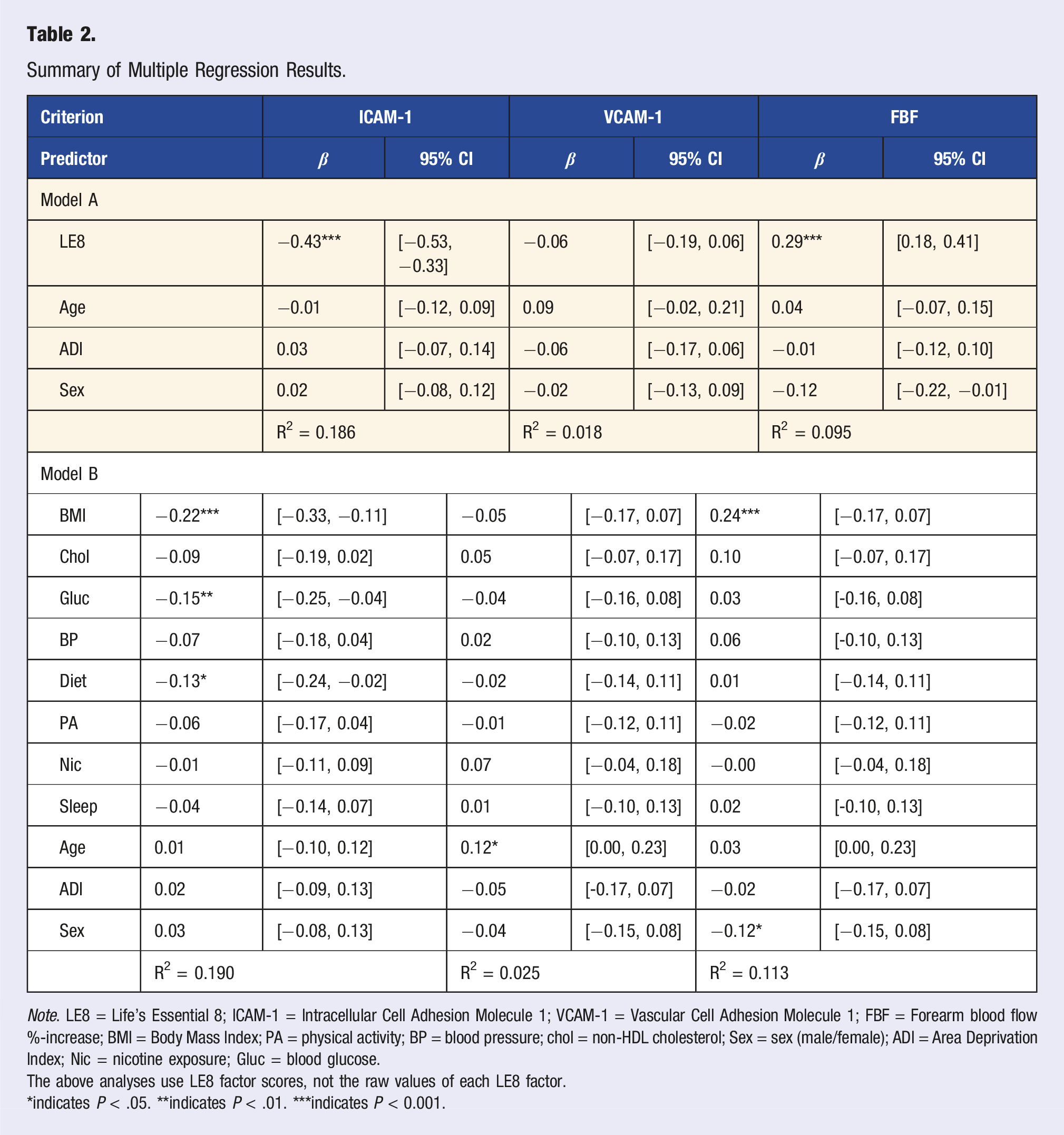
Note. LE8 = Life’s Essential 8; ICAM-1 = Intracellular Cell Adhesion Molecule 1; VCAM-1 = Vascular Cell Adhesion Molecule 1; FBF = Forearm blood flow %-increase; BMI = Body Mass Index; PA = physical activity; BP = blood pressure; chol = non-HDL cholesterol; Sex = sex (male/female); ADI = Area Deprivation Index; Nic = nicotine exposure; Gluc = blood glucose.
The above analyses use LE8 factor scores, not the raw values of each LE8 factor.
*indicates P < .05. **indicates P < .01. ***indicates P < 0.001.
VCAM-1 was not significant in Model A (R2 = 0.018, F = 5.608, P = 0.230) or Model B (R2 = 0.025, F = 8.447, P = 0.673). Age remained a significant predictor of VCAM-1 in Model B (b = 0.002, P = 0.05), with older age being associated with lower VCAM-1 concentration.
For FBF %-increase, Model A explained 9.5% of variation in FBF %-change (R2 = 0.095, F = 26.881, P < 0.001), such that the LE8 was significantly associated with FBF %-change (b = 0.29, P < 0.001). Model B significantly accounted for 11.3% of the variance in FBF %-increase (R2 = 0.113, F = 38.405, P < 0.001) with higher BMI scores (e.g., lower BMI [b = 0.24, P < 0.01]) being related to greater FBF %-change.
Utilizing continuous raw variables that comprise the LE8 factors instead of categorically derived values for Model B explained a comparable amount of variance in ICAM-1 (R2 = 0.189, F = 66.526, P < 0.001) and VCAM-1 (R2 = 0.024, F = 8.205, P = 0.695). When compared to the factor scores, the raw and continuous variables explained slightly more variation (∼3%) in FBF %-increase (R2 = 0.148, F = 51.391, P < 0.001).
Discussion
Novel findings from the present study indicate that the LE8 score is associated cross-sectionally with multiple indicators of endothelial health among otherwise healthy midlife adults without clinical CVD. Specifically, higher LE8 scores were related to lower concentrations of ICAM-1 and larger reactive-hyperemia-induced increases in forearm blood flow (FBF %-change), likely reflecting better EF. Additionally, the LE8 score remained significantly related to ICAM-1 and FBF %-change, but not VCAM-1, after adjusting for potential confounding factors. Decomposing the LE8 into its component factors showed that BMI, diet, and fasting glucose contributed the most to the explained variance in ICAM-1, while only greater BMI independently associated with blunted reactive hyperemia (smaller FBF %-increase). There were no significant associations between VCAM-1 and individual components of the LE8. To our knowledge, this study is the first investigation of the relationship between the LE8 and multiple measures of endothelial health, including circulating CAM concentrations in midlife adults.
Since the LE8 was first introduced, several studies have shown that the score is related to conventional ASCVD risk factors and predictive of future CVD outcomes.27-29 Most research in this area, however, has focused on relatively older populations with diagnosed health outcomes, with less research observing the LE8 in relation to preclinical ASCVD risk in early to middle aged adults with no history or presentation of CVD or chronic illnesses. In turn, research using the LE8 score to assess subclinical atherosclerosis is limited, but several studies have associated greater LE8 scores with less carotid artery plaques and lower coronary arterial calcification (CAC) scores.30,31 Our findings add to this research by showing associations with multiple indicators of endothelial dysfunction, which is thought to be a precursor to atherosclerosis. Moreover, the indicators of EF discussed in the current study have been related to other measures of preclinical atherosclerosis. Previous studies have illustrated, for example, that both greater ICAM-1 concentration and lower FMD are related to carotid intima media thickness (cIMT) and CAC scores.32,33 Adding to the latter findings, the present evidence suggests that the LE8 score is also associated with indicators of EF in midlife populations. Of note, the internal consistency of the LE8 in the current sample was small-to-moderate (Cronbach’s alpha = 0.48). The factors comprising the LE8, though shown to be individually associated with cardiovascular health, may thus not have equal contributions to determinants of CVD or the development of clinical CVD. Components may also not show equally strong relationships with the other factor scores, suggesting that overall LE8 scores to not represent a single construct. Future studies utilizing the LE8 should continue to conduct ancillary analyses on its individual components and outcomes of interest to maximize interpretability and possible preventative or lifestyle interventions.
Despite being attenuated in later regression models, ICAM-1 and FBF %-increase showed bivariate relationships with BP, physical activity, and non-HDL cholesterol scores, but not with nicotine exposure or sleep. With respect to BP and cholesterol, worse EF has been shown in individuals with clinical hypertension and hypercholesterolemia.34-38 As discussed, aerobic physical activity has been shown to improve EF in healthy adults. 39 Low sleep quality has been related to the development of chronic diseases, including obesity and CVD.4,40 Even so, the link between sleep duration and EF in healthy adults remains unclear, with most studies utilizing samples of adults with medical conditions (e.g., Alzheimer’s), a small n, or lacking focus on long-term sleep behaviors. 41 Acute sleep disturbances have been related to greater ICAM-1 concentration and lower FMD in prior studies, but only in samples of young adults.42,43 To extend the findings of the current study, future research should assess EF and multiple indices of sleep quality in a large sample of healthy, midlife adults. Unlike sleep, the relationship between nicotine exposure and EF in healthy adults is well understood. Several studies have shown smoking to be related to worse EF and indicators of endothelial inflammation.44,45 In the present study, a very small percentage of participants were current smokers (4.4%), which could explain why no independent associations were found between nicotine exposure and the measures of EF.
The present findings relating ICAM-1 to individual factors comprising the LE8 score replicate and add to prior literature. Several indexes of body size (e.g., BMI and waste circumference) have been shown to positively relate to ICAM-1 concentrations in healthy adults. 46 Several studies have found independent associations between indices of diet quality and ICAM-1 in healthy adults, with greater ICAM-1 relating to sub-optimal eating behaviors and lower dietary assessment scores (e.g., HEI).47,48 Because the prevalence of specific health conditions increases in midlife-to-older adulthood, such as metabolic syndrome or diabetes, 49 most studies looking at the independent associations between the factors of LE8 and ICAM-1 analyze samples with comorbid medical conditions, leaving a gap in research focused on otherwise healthy midlife populations. Along those lines, few studies have compared levels of fasting glucose with ICAM-1 in non-diabetic adults. Of note, one study found that serum-induced acute hyperglycemia elevated ICAM-1 levels, but not VCAM-1, in a small sample of healthy adults. 50 The small positive association found between ICAM-1 and fasting glucose in the present study may not be readily compared to existing research. Notwithstanding, previous literature has shown ICAM-1 concentration to be lower in healthy adults than those with conditions related metabolic health, such as obesity, Type-2 diabetes, and hyperglycemia.51-56 Low-grade adiposity-driven inflammation may serve as a potential explanation for ICAM-1 variation across levels of ASCVD risk. Adipose tissue is a source of inflammatory mediators, stimulating the release of pro-inflammatory cytokines and adipokines, which may foster increasing ICAM-1 expression. 57
In our regression models, we found that levels of VCAM-1 increased with age, though the effect size is relatively small. It is notable that no significant associations were found between VCAM-1, LE8, or its individual factors. Equally important, though ICAM-1 and VCAM-1 were moderately correlated, the negative association seen between ICAM-1 and FBF %-increase was not seen with VCAM-1. VCAM-1 and ICAM-1 display similar roles in the pathogenesis of atherosclerosis by initiating the adhesion of leukocytes to the media intima, a thin layer of endothelial cells coating the interior of arteries. 12 However, VCAM-1 displays a more pronounced presence in sites of lesion initiation when compared to ICAM-1, and has shown to be isolated to vasculature prone to damage from shear stress and turbulent blood flow.58,59 Though the role of VCAM-1 in atherosclerosis is well understood, literature tying the biomarker to preclinical ASCVD risk in healthy adults is limited. In a subsample from the Multi-Ethnic Study of Atherosclerosis, VCAM-1 concentration was greater in individuals with higher systolic blood pressure and LDL-cholesterol, though no significant association was seen with BMI. 60 Longitudinal studies of VCAM-1 in healthy adults have produced mixed results, with several studies finding that the biomarker does not relate to future ASCVD, improve risk assessment models, or predict CVD-related events (e.g., myocardial infarction).61,62 Overall, VCAM-1’s role in EF prior to the onset of ASCVD remains poorly understood. Even so, the current findings support the idea that VCAM-1 may not be sensitive to traditional indicators of ASCVD risk prior to the onset of clinical symptoms, especially in populations with good cardiovascular health. Thus, the lack of a relationship between VCAM-1 and ASCVD risk in the current study could be explained by a lower endothelial damage burden in the relatively healthy midlife sample.
The current study is the first to compare the LE8 score with FMD assessed with venous occlusion plethysmography. Brachial FMD is known to associate with traditional indices of ASCVD risk and holds predictive value for the development of ASCVD’s. 10 Though literature comparing the LE8 with brachial FMD is limited, one study using a sample of midlife to older adults with obesity found a positive association between FMD and the LE8. 63 FMD has been shown to be sensitive to varying degrees of ASCVD risk as defined by alternative scores other than the LE8; brachial FMD was related to lower Framingham Risk Scores in healthy adults. 64 Aligning with the findings of the current study, BMI and other indicators of body composition have been shown to be related to FMD in previous literature. Several studies have showed an independent association between BMI and FMD in healthy adults,65,66 as well as lower FMD and cIMT in non-obese adults when compared to those with obesity. 67 Adiposity-driven inflammation is known to decrease the bioavailability of nitric oxide through the upregulation of pro-inflammatory cytokines (e.g., TNF-a and IL-6), 68 which may serve as a potential explanation for the findings of the current study. Studies using nitroglycerine to elicit reactive hyperemia have found FMD to be lower in individuals with greater adiposity, adding credence to this theory. 69
Limitations
The present study is cross-sectional, precluding causal inferences. The findings of this study cannot be generalized to populations with established medical conditions due to the relatively healthy status of our sample. Venous occlusion plethysmography is a surrogate measure of EF, which indirectly assesses arterial diameter through volumetric changes in blood flow recorded at the surface of the skin. Additional methods of capturing FMD, such as brachial ultrasound tomography, may give a more accurate assessment of EF when used in conjunction with strain-gauge plethysmography. Information on second-hand smoke exposure could not be ascertained from the self-report inventories. Aspects of body composition cannot be addressed through BMI alone, such as fat percentage, muscle mass, or the distribution of fat mass in the body, highlighting the importance of using granular metrics to address body composition.70,71
Conclusion
The findings of this study revealed that better overall cardiovascular health (as determined by the LE8) related to lower ICAM-1 concentration and greater reactive hyperemia (FBF %-increase), which suggests the importance of the LE8 in reflecting subclinical endothelial health in midlife individuals. Factors related to adiposity and metabolism showed the strongest associations with ICAM-1 and FBF %-increase. Our observations suggest that the LE8 is related to multiple indicators of EF and has clinical and preventative utility in monitoring lifestyle factors related to ASCVD risk. Future longitudinal studies should examine if improvements in LE8 correspond to future changes in EF and vascular changes associated with ASCVD.
Footnotes
Acknowledgments
We thank Jaya King and Sara Boyko for their assistance in data collection and management. James R. Cirillo is now with the Department of Epidemiology, University of Pittsburgh School of Public Health, Pittsburgh PA. Kristen T. Stopfer is now with the Department of Human Development and Family Sciences, Texas Tech University, Lubbock TX.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health Grant P01 HL040962 (“Biobehavioral Studies of Cardiovascular Disease”).
Consent to Participate
Informed consent was obtained from all study participants, and study approval was granted by The University of Pittsburgh Human Research Protection Office (Protocol number: 19030012).
