Abstract
Introduction
It is estimated that approximately 100,000 individuals, all ages included, have sickle cell disease (SCD) in the United States. 1 SCD is an umbrella term that encompasses a group of genetically inherited hemoglobinopathies and includes variants such as sickle cell anemia (SCA/HbSS), hemoglobin SC disease (HbSC) and hemoglobin S/ß thalassemia (HbSß). SCA is the homozygosity of Hemoglobin S (HbSS), making it the most severe and frequently occurring hemoglobinopathy, affecting approximately 65% of individuals with SCD.2,3 When oxygen levels are low, hemoglobin S (HbS) can form long polymers that are inflexible, leading to severe vaso-occlusion and chronic anemia. 2
In contrast, SC hemoglobinopathy is a milder presentation of SCD because unlike HbS, HbC molecules do not readily polymerize under low oxygen tension. 4 Individuals with HbSC disease still have aggregates of hemoglobin molecules within red blood cells which can lead to sickling, resulting in symptoms similar to SCA. 2 HbS-β thalassemia is a compound heterozygous condition that results from the inheritance of a β thalassemia gene from one parent and a HbS gene from the other parent. The heterogeneity of the β-Thal mutations leads to differing amounts of β-globin synthesis and consequently different amounts of Hb A. HbS-β thalassemia is usually classified in 2 types: HbS-β+ thalassemia and HbS-β0 thalassemia. In HbS-β0 thalassemia, the production of HbA is absent making it clinically indistinct from sickle cell anemia while in HbS-β+ thalassemia, the amount of HbA produced is variable. 5
Regardless of genotype, individuals with SCD are at an increased risk for multiple comorbidities, including chronic pain, organ dysfunction, and cardiovascular complications.6-8 Beyond these comorbidities, individuals with SCD also experience a host of physical impairments that affect multiple systems. These include impairments to the neuromuscular system (eg, pain or balance), the musculoskeletal system (eg, strength or range of motion), and the cardiopulmonary system (eg, endurance or energy expenditure). 9 In children and adolescents specifically, research has shown that SCD is associated with changes in muscle and bone composition, impaired muscle strength, impaired cardiopulmonary function at rest, and deficits in motor performance and physical function. 9 Importantly, children and adolescents with SCD are reported to have an increased number of physical impairments and limitations compared to their healthy peers. 9 In an effort to better understand the possible implications of these physical impairments, Silva et al, 2023 found that children with SCD walk a significantly shorter distance during the 6-minute walk test (6MWT) than their healthy counterparts. 10 Having also found that the distance covered during the 6MWT was inversely associated with reticulocyte count and the IL-6 biomarker. Silva et al, 2023 hypothesized that the shorter distance may be attributable to increased levels of inflammation (ie, increased levels of IL-6) in children with SCD, thereby impacting their physical capacity. 10
Additional studies have found that time spent doing moderate and high intensity physical activity (PA) was lower in children with SCD than in their healthy peers.11,12 Specifically, children and adolescents with SCD engage in a level and amount of PA that is 24%–58% lower than their peers, despite preliminary evidence supporting the benefits of PA in alleviating certain symptoms associated with the disease, such as pain. 13 The potential benefits of PA in this population also extend to improvements in the cardiorespiratory system and muscle function when engaging in regular endurance exercises that include resistance training. 13 Nevertheless, some studies have shown that acute and intense exercise can increase the risk of sickling due to dehydration and oxidative stress in individuals with SCD. 14 As such, there is little consensus on the safety of high-intensity exercise for children with SCD,15,16 which highlights the need for additional research to confirm whether specific types of PA should be encouraged to confer benefits in this population.
While research within this field remains relatively new, there is promise of potential therapeutic interventions with PA for the SCD population. While there are studies in the field of PA and SCD, 17 there is limited understanding of the implications of PA specifically for children and adolescents. Health complications often occur earlier in life for individuals diagnosed with SCD. These complications can include, for example, acute splenic sequestration (prevalence ∼30% in children under 6 years old), functional asplenia (prevalence ∼80% in children with SCA under 1 years old), and acute chest syndrome (∼30% of individuals with SCD have at least 1 episode throughout their life). 18 Due to the severity of these complications in childhood for individuals with SCD and the lack of understanding of the role of PA associated with acute splenic sequestration, functional asplenia and acute chest syndrome in this population, it is vital to better understand the impact PA can have on the symptomatology and management of SCD in childhood.
This scoping review aims to describe and synthesize previously reported data in the field of PA and pediatric SCD to map the current evidence in response to the following aims: (1) to provide a description of PA and physical fitness assessments, (2) to provide a description of PA interventions, (3) to document the impact of PA interventions on health-related outcomes, and (4) to document adverse events related to PA interventions.
Review Questions
The scoping review objectives translate to the following question: In children and adolescents (≤21 years of age) diagnosed with SCD, what are the assessment methodologies to measure PA levels and physical fitness and what PA interventions have been conducted in this population?
Methods
This review was conducted and is reported following the Preferred Reporting Items for Systematic Reviews and the Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) statement. 19
Search Strategy
A preliminary search was performed in PROSPERO and several other journal indices and databases to ensure no scoping reviews, systematic reviews, or meta-analyses on the same topic were underway. Search structures, subject headings, and keywords were tailored to each database by a medical research librarian (KJK) specializing in systematic reviews in consultation with the senior author (MC), based on current literature.12,20-22 Search terms included a combination of subject headings and keywords covering the population, concept, and context (Supplemental materials). Databases included: Ovid MEDLINE, Ovid Embase, Cochrane Central, Web of Science, and PEDro Physiotherapy Evidence database. No limits were applied to geographical locations or publication dates, but searches were restricted to human and English-language articles. The full search strings for all databases and other resources can be found in supplemental materials. The reference lists of included articles were manually scanned for additional articles. The last database search was performed on January 28th, 2025.
Study Eligibility Criteria
Eligibility criteria included observational studies, RCTs, NCTs and uncontrolled interventions (ie, pre- and post-tests without controls), regardless of the study objectives, published in a peer-reviewed journal. Only quantitative studies were eligible for inclusion. For inclusion, studies had to include samples comprising children, adolescents and young adults (21 years of age or younger) diagnosed with SCD. SCD is an umbrella term that represents a heterogeneous group of genetically inherited disorders, characterized by mutations in the gene encoding the hemoglobin subunit ß, including SCA, HbSC and HbSß-thalassemia. Sickle cell trait patients were not included in this review.
Furthermore, eligible studies had to include at least one assessment of PA and/or physical fitness and/or a PA intervention focusing on improving health outcomes as a primary or secondary outcome. PA assessments were defined as behavioral measures quantifying “any bodily movement produced by contracting skeletal muscles, with a concomitant increase in energy expenditure”. 23 Physical fitness assessments were defined as quantitative measures assessing 1 or more of the following indicators: cardiorespiratory fitness, muscle strength and endurance, flexibility, balance and agility.24,25 PA interventions were defined as interventions that caused “any bodily movement produced by contracting skeletal muscles, with a concomitant increase in energy expenditure”. 23 Exercise was considered as a component of PA to reach the end goal of the PA intervention.23,26 Examples include, but are not limited to, aerobic exercise interventions, resistance training, flexibility, balance, agility or stretching exercises.
Studies were excluded if they were: (1) in a language other than English, (2) conference abstracts, case reports, case studies, short communications, systematic reviews, meta-analyses, position papers, white papers/editorials, letters to the editor and protocol papers, (3) unpublished research (eg, theses/dissertations), and (4) studies with animals.
Study Selection
All studies identified from the search strategy were collated and uploaded into Covidence systematic review software. Duplicate citations were removed manually in Endnote. The first step consisted of 2 authors independently screening the titles and abstracts (MC and DPS) to identify potentially relevant studies. When the titles and abstracts were considered potentially relevant, the full text articles were obtained. The second step consisted of 2 authors independently reviewing the full text articles (MC and DPS) to confirm their eligibility. If an article did not meet the inclusion criteria, the reason for exclusion was recorded. The PRISMA flow diagram (Figure 1) shows the entire selection process from the original search results to the final selection of studies. PRISMA-ScR flowchart.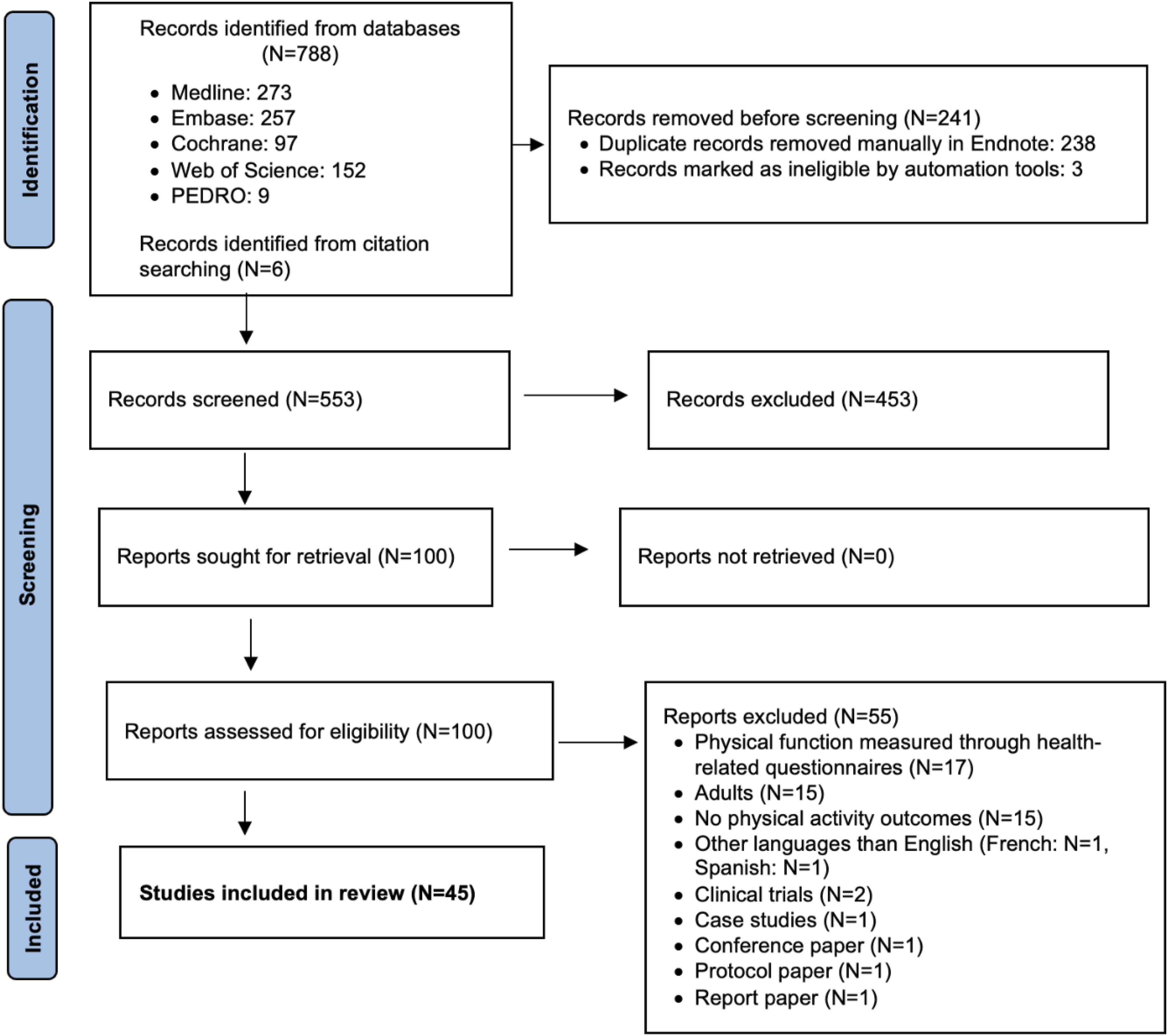
Data Extraction and Presentation
Data were extracted by the lead author (AL) and co-authors (DPS, LS, PN). To address review objectives, extracted data from eligible articles were as follows: authors, publication year, country, participant characteristics (ie, sample size(s), sex/gender [note: we did not distinguish between these terms because authors did not always make clear which one they assessed], age at enrollment, type of disease/genotypes, treatment), stated objectives, type and description of PA assessment(s), type and description of physical fitness assessment(s), type of PA intervention and description of PA intervention, reported adverse events, main results, and authors’ conclusion.
During the data extraction process, if data were incomplete or unclear in the full text, we contacted the corresponding authors by email (up to 2 times, 3 weeks apart) to obtain additional information.
Risk of Bias Assessment
In accordance with the methodological recommendations for scoping reviews, 27 no risk of bias assessment was performed.
Results
Search Strategy and Study Selection
The literature search yielded a total of 788 studies and 6 additional studies were identified by reviewing the references of included studies. After duplicate records were removed, a total of 553 records were screened by title and abstract. Of these 553 records, 100 underwent a full-text screening and 55 studies did not meet the inclusion criteria. A total of 45 studies were included in this review as meeting the inclusion criteria. Figure 1 illustrates the flow diagram of the search strategy in accordance with the PRISMA-ScR guidelines.
Sample Characteristics of the Included Articles
Data Extraction, Objectives, and Physical Activity and Fitness Assessments of Included Studies (N = 45).

aWhen participants were referred to as gender, not sex in the included paper; Age at enrollment refers to the mean age of participants.
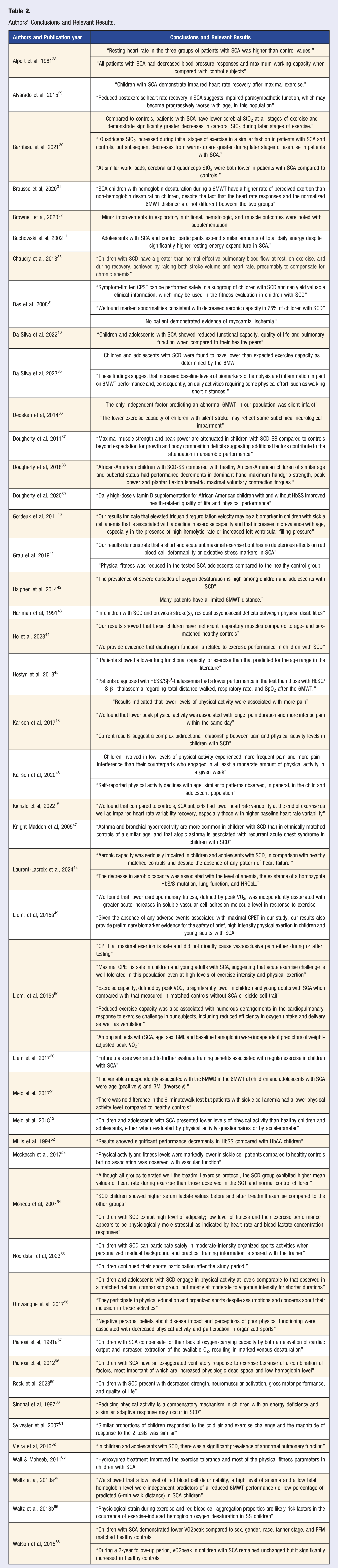
Authors’ Conclusions and Relevant Results.
PA Assessments
Few studies (13/45, 28.9%) reported assessing participants’ PA level, with 11/13 (84.6%) of these studies using more than 1 type of assessment. Laurent-Lacroix et al, 2024 and Liem et al., 2017 were the only studies that reported using only 1 type of PA assessment, namely they respectively used the Ricci and Gagnon questionnaire to assess self-reported PA levels 48 and a wearable heart rate monitor. 20 Seven other PA questionnaires were used to assess PA levels. One self-reported questionnaire for children was used to assess participants’ PA frequency. 46 The Brazilian version of the Physical Activity Questionnaire for Older Children and Adolescents was used in 2 studies.12,51 The International Physical Activity Questionnaire adapted for adolescents was used in 1 study, 53 the National Institute for Public Health and the Environment (RIVM) questionnaire was also used in 1 study, 55 and Omwanghe et al., 2017 used a combination of the National Health and Nutrition Examination Survey (NHANES), the Physical Activity Questionnaire (PAQ), and questions focused on a 7-day recall of physical functioning, adapted from the PROMIS Pediatric Item Bank on Physical Function Mobility. 56 Three studies assessed PA by using an Actigraph device, 55 with Karlson et al, 2016 using measurements from the 14-day study period 13 and Melo et al., 2018 using measurements from recordings over 7 consecutive days. 12 Two studies also used body movement recordings to measure total energy expenditure from PA over a 24-hour period.11,60 One study used a self-reported diary to assess PA 11 and 1 study used self-perceived moderate-to-vigorous physical activity. 55
Physical Fitness Assessments
Most studies (40/45, 88.9%) reported assessing participants’ physical fitness level. The most common type of assessment was the 6MWT, which was used as an assessment of physical fitness in 13 studies.31,35,36,40,42,44,45,51,53,59,62,64,65 These studies generally reported following the American Thoracic Society Guidelines and either provided participants with verbal encouragement or not. Eight studies assessed physical fitness with maximal cardiopulmonary exercise testing using a ramp cycle protocol20,33,48,49 and the modified Godfrey protocol.29,30,50,55 Seven studies used ad hoc treadmill protocols to assess physical fitness.38,39,47,54,61,63,66 Four studies used cycle ergometer exercise testing, with 2 studies using a protocol that required a progressive increase in workload,15,41 1 study using a modification of the Bruce and Hornsten format, 28 and 1 study using Jones stages I and III. 58 Maximal handgrip strength using a handgrip dynamometer,37-39 maximal vertical squat jump (3 warm-ups followed by 3 maximal squat jumps)37-39 were both used in 3 studies. Maximum symptom-limited cardiopulmonary exercise testing (Bruce protocol conducted according to the guidelines of the American Heart Association 34 and incremental cycle ergometer protocol 15 ), muscle torque (plantar flexion isometric MVC torques),38,39 neuromuscular motor skills (Bruininks-Oseretsky Test of Motor Proficiency),39,59 and physical fitness indices (ie, vertical jump apparatus, grip strength dynamometer, back strength dynamometer, leg strength dynamometer, flexibility, whole body reaction time)54,63 were each used as a measure of physical fitness in 2 studies. The following measures of physical fitness were each used in 1 study: 24-hour protocol (measurement of standard daily activities), 11 functional capacity (modified shuttle test), 10 motoric functions questionnaires (assessed using the modified Motor Assessment Scale), 43 activity of daily living questionnaire (assessed using the Barthel Index), 43 physical performance (100-yard potato race and 20- and 40-yard freestyle swimming test), 52 Timed Up and Down Stairs assessment, 59 Timed Up and Go assessment, 59 muscle function and strength, 32 and Canadian Agility and Movement Skill Assessment (CAMSA). 55
Physical Activity Interventions
Two studies (2/45, 4.4%) piloted a physical activity intervention in children and adolescents with SCD.
Liem et al, 2017 20 prescribed a progressive 12-week individualized home exercise training program comprised of 3 sessions per week of cycling on stationary bicycles. Heart rate and total exercise time were used to monitor participants’ progress. During weeks 1 to 6, prescribed exercise duration ranged from 10 to 30 minutes and targeted heart rate ranged from 50 to 100% of the heart rate at ventilatory threshold. During weeks 7 to 12, prescribed exercise duration was maintained at 30 minutes and targeted heart rate was maintained at 100% of the heart rate at ventilatory threshold. No adverse events were reported. 20 Liem et al reported that mean peakVO2 (baseline: 28.5 ± 5.6 mL/kg/min and 6-week: 30.8 ± 5.6 mL/kg/min, P = 0.01) and peak workload (baseline: 103 ± 20 watts and 6-week: 111 ± 26 watts, P = 0.03) significantly improved after 6 weeks of individualized home exercise training. 20 No significant improvement in exercise parameters, including VO2 peak and peak workload were reported at the end of the program. There was also no significant improvements in total white blood cell count, absolute neutrophil count, absolute monocyte count, Hemoglobin, platelet count, and D-dimer. 20
Noordstar et al, 2023 55 conducted a 10-week moderate to vigorous intensity organized sports program. Participants’ medical background and practical training information were shared with the certified trainer before the start of the first training session. Training sessions were soccer-related activities (dribbling, shooting, and passing), practiced individually, in pairs, or with the whole group for a total duration of 90 minutes. Children participated in a soccer match at the end of each training. Noordstar et al. reported that no intensity restrictions were imposed. All sessions were supervised by the medical team. One adverse event (vaso-occlusive episode) that required hospital admission and treatment with opiates was reported by the study team. 55 At baseline, Noordstar et al reported that children spent between 4.2% and 13.2% of their time engaging in moderate-to-vigorous physical activity and they were physically active for at least 60 minutes per day for 2 to 7 days. At the end of the program, children spent between 4.5% and 9.7% of their time engaging in moderate-to-vigorous physical activity and they were physically active for at least 60 minutes per day for 3 to 7 days.
Discussion
Our scoping review provides a description of the current methods used to assess PA and physical fitness in children and adolescents (≤21 years) diagnosed with SCD. Accurately measuring PA and physical fitness is essential to effectively evaluate associations with health-related outcomes, study PA intervention efficacy, and monitor changes over time. Our review highlights substantial heterogeneity in the assessment methods across the included studies, limiting generalizability. Moreover, we identified only 2 PA interventions for SCD patients that met our inclusion criteria, emphasizing a critical gap in the literature. These findings emphasize the need for the development of safe PA interventions tailored to this understudied population. This review serves as a foundation for future research aiming to improve PA outcomes in children and adolescents with SCD.
The potential benefits of PA for children diagnosed with SCD have been considered in previous studies. 13 Given that children and adolescents with SCD are prone to engage in lower levels of PA compared to their healthy peers,11,12 understanding the role that PA can play for this population is crucial. Buchowski et al. 11 have reported that adolescents with SCD exhibit PA levels up to 10.8% lower than their healthy counterparts (SCD: 1.32 ± 0.10 vs controls: 1.48 ± 0.12, P < 0.001). Karlson et al. 46 have observed that 23.1% of children with SCD engage in at least 30 minutes of PA only one to 2 days per week, 43.5% participate in 30 minutes of PA 3 to 5 days per week, and 22.4% maintain this level of PA 5 to 7 days per week. While no specific PA guidelines have been developed for children and adolescents with SCD, the general Physical Activity Guidelines for healthy children and adolescents recommend at least 60 minutes of moderate-to-vigorous intensity physical activity per day each week. 67 Omwanghe et al. 56 analyzed data from NHANES using self-reported questionnaires and found that children with SCD and their healthy peers engage in a similar proportion of PA. Specifically, 66% of children with SCD and 65% of healthy children reported engaging in at least 10 minutes of vigorous PA per week (P = 0.91). 56 The study also found that a significantly higher proportion of children with SCD (67%) reported engaging in at least 10 minutes of moderate-to-vigorous PA compared to 42% of healthy children (P < 0.01) 56
To assess PA levels, both objective and subjective methods were used in the included studies. Five studies used self-reported questionnaires,12,46,51,53,56 while 2 used accelerometers.12,13 A key limitation is that the majority of the included studies did not provide a comprehensive description of their PA assessment methods, likely because PA was not the primary outcome. This lack of detail reduces the reproducibility and comparability of findings across studies. To enhance transparency and facilitate reproducibility, we recommend that future studies provide thorough descriptions of their PA assessment methods. While self-reported questionnaires are widely used for their accessibility and cost effectiveness, they are often criticized due to their recall bias leading to over- or underestimation PA levels. 68 Incorporating objective measures can help mitigate these limitations by providing more precise and continuous data on PA. However, the higher cost and logistical challenges associated with accelerometers must be acknowledged, particularly in studies with limited resources. Future studies should be conducted to explore this topic further.
Selecting the appropriate method to assess PA is crucial given the lack of consensus on the optimal PA intensity for children and adolescents with SCD. Notably, only 2 studies included in this review focused on PA interventions in these patients. Liem et al, 2017 prescribed a progressive 12-week individualized home exercise training program, 20 while Noordstar et al., 2023 conducted a 10-week moderate to vigorous intensity organized sports program. 55 Both studies reported the feasibility and safety of their PA interventions.20,55 However, Liem et al. did not report significant training effects on exercise parameters (eg, peak VO2), total white blood cell count, platelet count, C-reactive protein and D-dimer. 20 The current literature therefore emphasizes the need for additional research to determine whether specific types or intensities of PA provide health benefits for children with SCD. Karlson et al., 2017 13 found that the majority of children with SCD (97.7%) engage primarily in light-intensity PA, while 43.4% engage in vigorous-intensity PA. Their findings reinforced that children with SCD are generally less physically active than their healthy peers. 13 However, there is little consensus regarding the safety of high-intensity PA for children and adolescents with SCD. 15 Some studies suggest that high-intensity PA can increase vaso-occlusive pain, while other studies suggest that acute and intense PA can increase the risk of sickling due to dehydration and oxidative stress. 14 Noordstar et al. reported 1 vaso-occlusive episode in 1 participant 6 days following the start of the moderate to vigorous training sessions. 55 However, the authors reported that this episode may not have been directly related to the PA intervention. 55 Further investigations are therefore necessary to establish evidence-based PA recommendations that find a balance between yielding potential benefits and taking into consideration safety concerns in children and adolescents with SCD.
The benefits that PA can confer for these patients can also extend to improvements in physical fitness outcomes. Most of the included studies assessed physical fitness, highlighting its relevance in this population. The 6MWT was the most commonly used assessment in 13 studies,31,35,36,40,42,44,45,51,53,59,62,64,65 while 10 studies evaluated physical fitness using a cardiopulmonary exercise test.15,20,29,30,33,34,48-50,55 Significant heterogeneity in assessment methods across studies limits direct comparisons and interpretations. Despite this variability, it is important to acknowledge the efforts made by researchers to measure physical fitness in children with SCD. Encouragingly, none of the included studies reported adverse events related to physical fitness assessments. Das et al, 2008 34 demonstrated the feasibility and safety of symptom-limited cardiopulmonary stress testing in children with SCD. Liem et al.20,49,50 reported that SCD patients were able to complete cardiopulmonary exercise tests without any major adverse events. These findings suggest that well-monitored physical fitness assessments can be conducted safely in children with SCD. Additionally, using physical fitness assessments that are close to patients’ daily activities may provide a more comprehensive understanding of their physical fitness status. Notably, 1 study introduced an innovative approach by incorporating an unconventional physical performance test, which included a 100-yard potato race and 20- and 40-yard freestyle swimming tests. 52
Children and adolescents living with SCD can have a variety of complications, and it is unclear if PA is associated with an improvement in those complications. SCD results in increased energy expenditure, which could be related to the lower levels of PA observed in SCD. 69 High intensity activity could lead to sickle cell-related complications, but light-to-moderate activity could confer benefits for individuals with SCD. It can be challenging to know that exact amount of PA that is both safe and has the potential to improve clinical outcomes in SCD. Merlet et al 70 demonstrated that moderate activity resulted in improvements in capillary networks, and improved indexes of PA. Clinically, it is still unclear if this results in reduced complications such as VOCs/painful events. Other clinically significant outcomes that may improve with PA in SCD include cardiovascular capacity and pulmonary function. Further investigation studying the impact of PA on SCD symptoms and complications continues to be needed.
To conclude, it is essential to recognize the impact of SCD on PA behavior and the substantial work that remains to be done to develop and implement safe and accessible PA interventions for this understudied population. This scoping review evidenced that there is significant heterogeneity in the methods used to assess PA and physical fitness in children with SCD and that evidence-based PA interventions in this population are limited. There is also a critical need to determine the optimal PA intensity for children and adolescents with SCD, as no guidelines currently exist to define what is both safe and effective. Conducting future research aiming to address these gaps is critical to allow for the formulation of evidence-based PA recommendations that are specific to the needs and challenges of children with SCD.
Supplemental Material
Supplemental Material - Physical Activity and Physical Fitness in Children and Adolescents Diagnosed With Sickle Cell Disease: A Scoping Review
Supplemental Material for Physical Activity and Physical Fitness in Children and Adolescents Diagnosed With Sickle Cell Disease: A Scoping Review by Ariane Levesque, Deepika Pugalenthi Saravanan, Peri Newman, Lauryn E. Six, Yevgeniya Bamme, Smita Dandekar, Kate J. Krause, Maria Chang Swartz, Gayle M. Smink, and Maxime Caru in American Journal of Lifestyle Medicine
Footnotes
Acknowledgments
We acknowledge Julia McAlexander for her valuable assistance during this review.
Author Contributions
AL, DPS, GS and MC conceived the study and participated in the design and the coordination. KJK and MC performed the literature search. AL, DPS, PN, LES, and MC performed the study selection. AL, DPS, PN and LES performed data extraction from included articles. AL, DPS, PN, LES, YB, SD, KJK, MCS, GMS and MC made substantial contributions to the interpretation of the data. AL, DPS, PN, LES, YB, SD, KJK, MCS, GMS and MC critically drafted or revised the manuscript for important intellectual content. AL, DPS, PN, LES, YB, SD, KJK, MCS, GMS and MC provided final approval of the version to be published and agreed to be held accountable for all aspects of the review in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Maxime Caru, PhD, PhD is supported by the Four Diamonds Research Funds, Department of Pediatrics, Division of Hematology and Oncology, Pennsylvania State Health Children’s Hospital, Hershey, PA, USA. Ariane Levesque, PhD is supported by the Four Diamonds Research Funds, Department of Pediatrics, Division of Hematology and Oncology, Pennsylvania State Health Children’s Hospital, Hershey, PA, USA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
