Abstract
Objective: The Brain Care Score (BCS), previously validated to predict the risk of age-related brain diseases, incorporates 12 modifiable risk factors, including blood pressure and laboratory measurements. In the U.S., fewer than 50% of individuals recall these measurements, limiting the BCS’s accessibility. This study aims to evaluate the predictive validity and discriminatory power of a BCS derived from participant responses for incident stroke. Methods: We performed retrospective analyses using the prospectively collected COSMOS data. The BCS, ranging 0-19 points, was calculated from modifiable risk factors derived from participant responses only. A Cox proportional hazard model, adjusted for sex and age was used to estimate the association between a higher BCS, indicating better brain health, and incident stroke. Results: Among 17 150 participants (median age 70.8, 59.6% female) free of stroke and TIA at baseline and with complete BCS data, 187 (1.1%) experienced a stroke during a mean follow-up duration of 3.6 (SD:0.7) years. A 5-point higher BCS was associated with a 36% lower stroke risk (HR:0.64 [95%CI:0.48-0.84], c-statistic:0.68). Discussion: A BCS derived from participant responses showed similar predictive performance and discriminatory ability compared to previous validation studies that use physical and laboratory measurements. Future studies could consider incorporating the BCS derived from participant responses when physical and laboratory measurements are not readily available.
“Our findings demonstrate that a BCS derived from participant responses is associated with incident stroke in a U.S. cohort.”
Introduction
Stroke is one of the leading causes of mortality and disability-adjusted life-years worldwide. 1 In 2021, stroke was the fourth leading cause of disability-adjusted life-years (DALYs), accounting for 161 million DALYs globally, with projections indicating this will double by 2050. 2 It is estimated that at least 60% of stroke cases are attributable to modifiable risk factors and could, therefore, be prevented.3,4 Despite accumulating knowledge on the importance of risk factor management and clear prevention messages from the American Heart Association 5 and American Academy of Neurology, 6 the prevalence of stroke continues to rise.5-7
The Brain Care Score (BCS) was developed as a comprehensive and quantitative score to empower people with actionable steps to take care of their brain and reduce the incidence of stroke, dementia, and late-life depression. 8 The BCS ranges from 0 to 21 points, with a higher BCS indicating better brain health. The BCS has previously been validated in the UK Biobank, showing that a higher baseline BCS was associated with a lower incidence of stroke. 9
Current models and tools, including the BCS, that are designed to aid people in prevention and risk factor management often require both a medical examination for blood pressure assessment and laboratory measurements to obtain blood sugar or cholesterol levels,7,9-11 which limits the out-of-clinic utility and accessibility of these models. Previous studies showed that fewer than 50% of people in the U.S. recall their specific blood pressure, HbA1c level, or cholesterol levels,12-18 and trends in awareness of these risk factors among the U.S. population have not improved over recent decades. 19 As such, asking users to remember specific measurements makes the scores less accessible than their intended broader use.
Previous literature has shown that self-reporting of common health questions, such as “Has a doctor ever told you have hypertension?” or “Has a doctor ever told you have diabetes?” can effectively identify the population at higher risk without the requirement of a recent medical examination.20,21 Substituting recent medical examination measurements with health information derived from participant responses in scores could make these scores more accessible in case physical or laboratory measurements are not available. Therefore, we aim to assess the predictive validity of a BCS that utilizes only modifiable risk factors derived from participant responses in its association with incident stroke in the COSMOS cohort.
Methods
Study Design
The COcoa Supplement and Multivitamin Outcomes Study (COSMOS) study is a large, pragmatic, placebo-controlled, randomized clinical trial in the United States testing a cocoa extract supplement and a multivitamin. The objectives of COSMOS were to examine whether daily cocoa extract or multivitamin supplementation reduced the risk of cardiovascular disease, cancer, and other important health outcomes. The interventions consisted of a placebo-controlled, 2 × 2 factorial design, in which participants were randomized to receive daily cocoa extract, a multivitamin, both, or placebo. Results showed that cocoa extract supplementation was associated with a 26% risk reduction for the death due to a composite outcome of myocardial infarction, stroke, and CVD death. There was no significant association between either of the interventions and the incidence of stroke or other CVD events.22,23 Recruitment was conducted via the Women’s Health Initiative (WHI), 24 Brigham and Women’s Hospital survey’s respondents, 25 and by mass mailings throughout the US. 26 Participants were enrolled and randomized between 2016 and 2018. Exclusion criteria were previous stroke, renal failure, dialysis, cirrhosis, recent cancer, or other medical conditions that precluded participation, plus poor compliers (taking <75% of the study pills) during a ≥2-month placebo run-in. Baseline risk factors were collected through self-reported questionnaires, and cardiovascular outcomes were confirmed via medical record review. Follow-up occurred at 6 and 12 months, then semi-annually.26,27 Prior to enrollment in the trial, all participants gave written informed consent, and the study received approval and oversight from the Human Subjects Committee at Brigham and Women’s Hospital and Massachusetts General Hospital.
Brain Care Score
Development of the BCS
The BCS was created through a patient-informed, modified Delphi process 28 and includes all components of the Life’s Essential Eight (blood pressure, cholesterol, blood sugar, diet, physical activity, body mass index, smoking, and sleep),7,29 as well as alcohol use and social-emotional components. Specifically, 3 social-emotional components with strong epidemiological associations with the incidence of brain disease were included: (i) social engagement,30,31 (ii) stress,32,33 and (iii) purpose in life.34,35 The BCS is designed to have a motivational component (a higher score indicating better brain health) and offers a comprehensive, quantitative approach for longitudinal brain health monitoring5,9 (Figure S1).
BCS Derivation in COSMOS
The BCS was derived from the COSMOS questionnaires conducted at baseline (prior to randomization). To substitute the quantitative measurements of blood pressure, cholesterol level, and Hb1Ac level, we selected common health questions previously shown to identify populations at higher risk for hypertension (Has a physician/doctor ever told you have hypertension?) and diabetes (Have you ever been told you have diabetes?).20,21 While no data on a single common health question for cholesterol was shown to identify the population at higher risk, we combined 2 questions: (i) the presence of a family history of high cholesterol and (ii) current cholesterol-lowering medication use. People who had a family history of high cholesterol and did not use cholesterol-lowering medication were included in the higher-risk population. In contrast, those who do not have a family history of high cholesterol or those who are currently taking cholesterol-lowering medication were included in the lower-risk population. For the other components, if no direct translation of the BCS was available, the best available proxy was selected in a consensus meeting between the authors (JRS, RWPT, TNK, CDA, SDS, and NY). A complete overview of the questions used to derive the BCS from the COSMOS database and differences between the BCS and the COSMOS-derived BCS are described in Table S1. The COSMOS-derived BCS consisted of 11 of 12 BCS components, excluding social relationships due to a lack of an available proxy. The COSMOS-derived BCS ranged from 0-19 points. A complete overview of the used questionnaires can be found at https://cosmostrial.org.
Outcomes
Primary and Secondary Outcomes
Our primary outcome was the incidence of first stroke (ischemic or hemorrhagic) during follow-up. Secondary outcomes included first TIA and a composite outcome of stroke or TIA.
Endpoint Confirmation
Participants who reported stroke or TIA during follow-up signed a medical record release, and their records were adjudicated through detailed review by independent researchers to confirm or disconfirm events. Stroke was defined as the rapid onset of neurological symptoms lasting >24 h, and TIA was defined as a brief episode of a focal neurologic deficit lasting less than 24 h, with no permanent cerebral infarction. 36
Study Population
Participants with a history of stroke or TIA at baseline were excluded. Only those with complete COSMOS-derived BCS data were included.
Statistical Analysis
Statistical Analysis
In this study, we presented participant characteristics using means and standard deviations (SD) or medians and interquartile ranges (Q1 and Q3), as appropriate. We detailed the distribution of the BCS components and reported any missing data. For continuous variables, we applied the t test when the distribution was normal and the Mann-Whitney U test for non-normally distributed data. Categorical variables were analyzed using chi-square tests or Fisher’s exact test when appropriate. Normality was assessed using histograms, Q-Q plots, and the Anderson-Darling test. We performed a complete-case analysis, including only those with complete COSMOS-derived BCS data, aligning previous methodology.9,37,38 We assessed missingness by comparing baseline characteristics of included vs excluded participants.
Validation Analysis
To evaluate the accuracy of our proxy derived from participant responses in categorizing participants with high vs low blood cholesterol, we compared the mean cholesterol levels derived from participant responses between those identified as higher risk and those not at higher risk. This comparison was conducted among the subset of individuals who provided baseline cholesterol information. Cholesterol levels were reported categorically in increments of 20 mg/dL, starting at 140 mg/dL. We assigned participants the midpoint value of their reported category to calculate the mean cholesterol levels.
Cox Proportional Hazard Model
To determine the predictive validity of the COSMOS-derived BCS, we assessed the association between incident stroke and a 5-point higher baseline BCS through Cox proportional hazard regression models. A 5-point higher baseline was selected, based on prior research,9,37,39 to reflect a substantial yet achievable improvement in someone’s brain health. For example, a 5-point higher baseline BCS might be achieved by stopping smoking and reducing alcohol use (from ≥4 per week to 0-1 per week). All of the Cox proportional hazards models were initially adjusted for sex assigned at birth and age, aligning with the previously described methodology.9,37,39 We performed a similar analysis for our secondary outcomes of TIA and a composite outcome of stroke and/or TIA. For all models the proportional hazards assumption was assessed using Schoenfeld residuals. We found that the proportional hazard assumptions were violated for age in the secondary outcome models. To address this, we did not adjust the model for age directly. Instead, we accounted for age by categorizing it into tertiles, allowing us to include age as a stratification variable. This approach ensured that age-related effects were addressed while maintaining the validity of the Cox model. Results were presented as hazard ratios (HRs) with corresponding 95% confidence intervals (CIs). To assess the predictive accuracy of our models, we calculated and reported the concordance statistics (c-statistics).
We simulated and visualized the HRs and 95% CIs for our primary outcome as a dose–response curve across the BCS range of 0 to 19. Using previously described methods, 40 we used the mean BCS as the reference point and performed 10 000 simulations per model to generate these estimates. Furthermore, we evaluated the clinical utility of the model using decision curve analysis at the mean follow-up time.
Sensitivity Analysis
In a sensitivity analysis we adjusted for age, sex and randomized treatment of the COSMOS trial and tested for effect modification by the randomized treatment assignment. Furthermore, we stratified the primary outcome model by sex assigned at birth and age. Age was stratified at a cut-off of 75 years, aligning with the benchmark for prevention recommendations provided by the American Heart Association. 41 A Fine and Gray competing risk model was used to account for the competing risk of death.
A P-value of <0.05 (two-tailed) was considered statistically significant. Statistical analyses were performed using Rstudio Version 3.3.0. The manuscript follows STROBE guidelines.
Results
Cohort Characteristics
Demographic Characteristics.
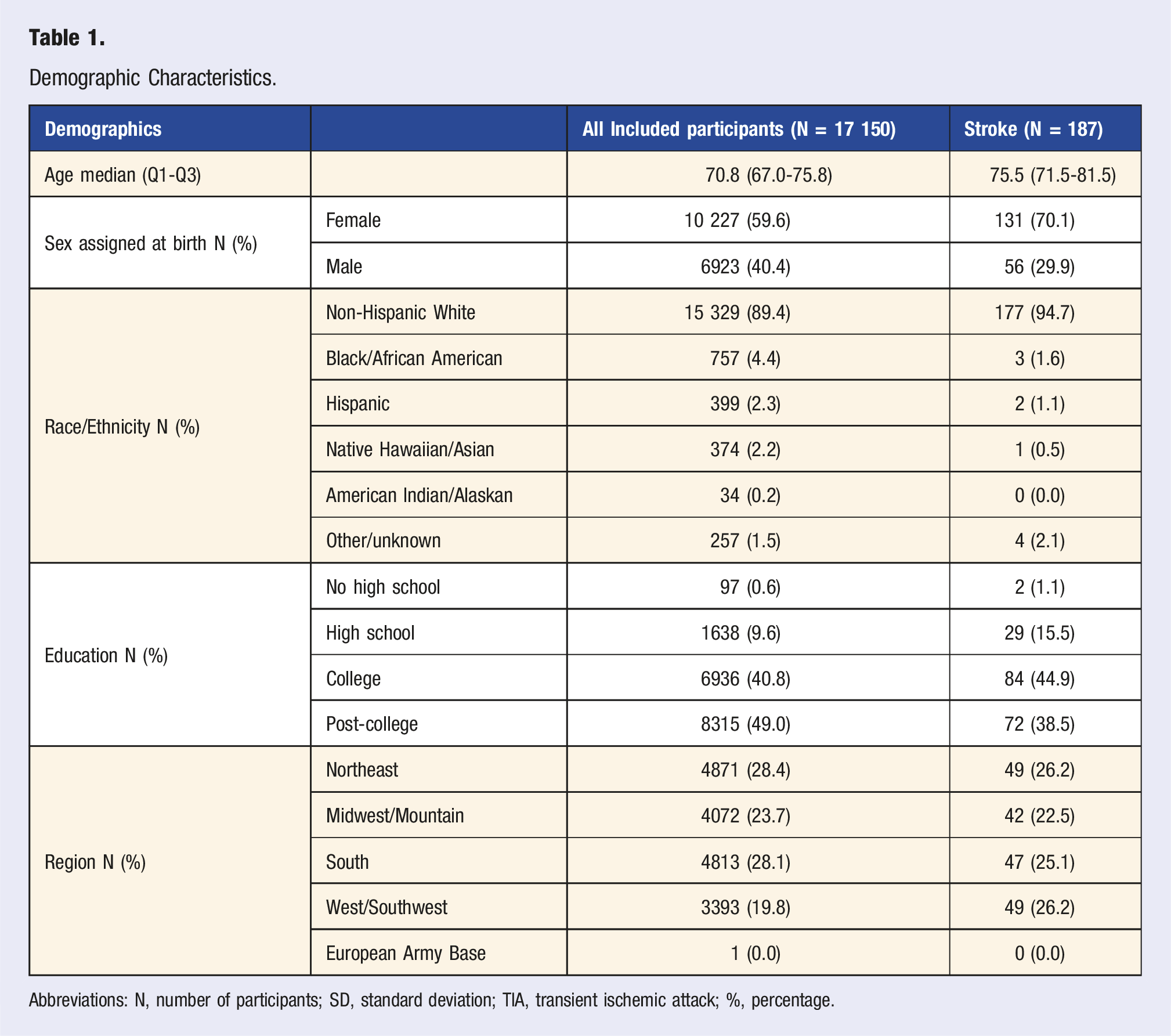
Abbreviations: N, number of participants; SD, standard deviation; TIA, transient ischemic attack; %, percentage.
COSMOS-Derived Brain Care Score
A Brain Care Score Derived From the COSMOS Database.
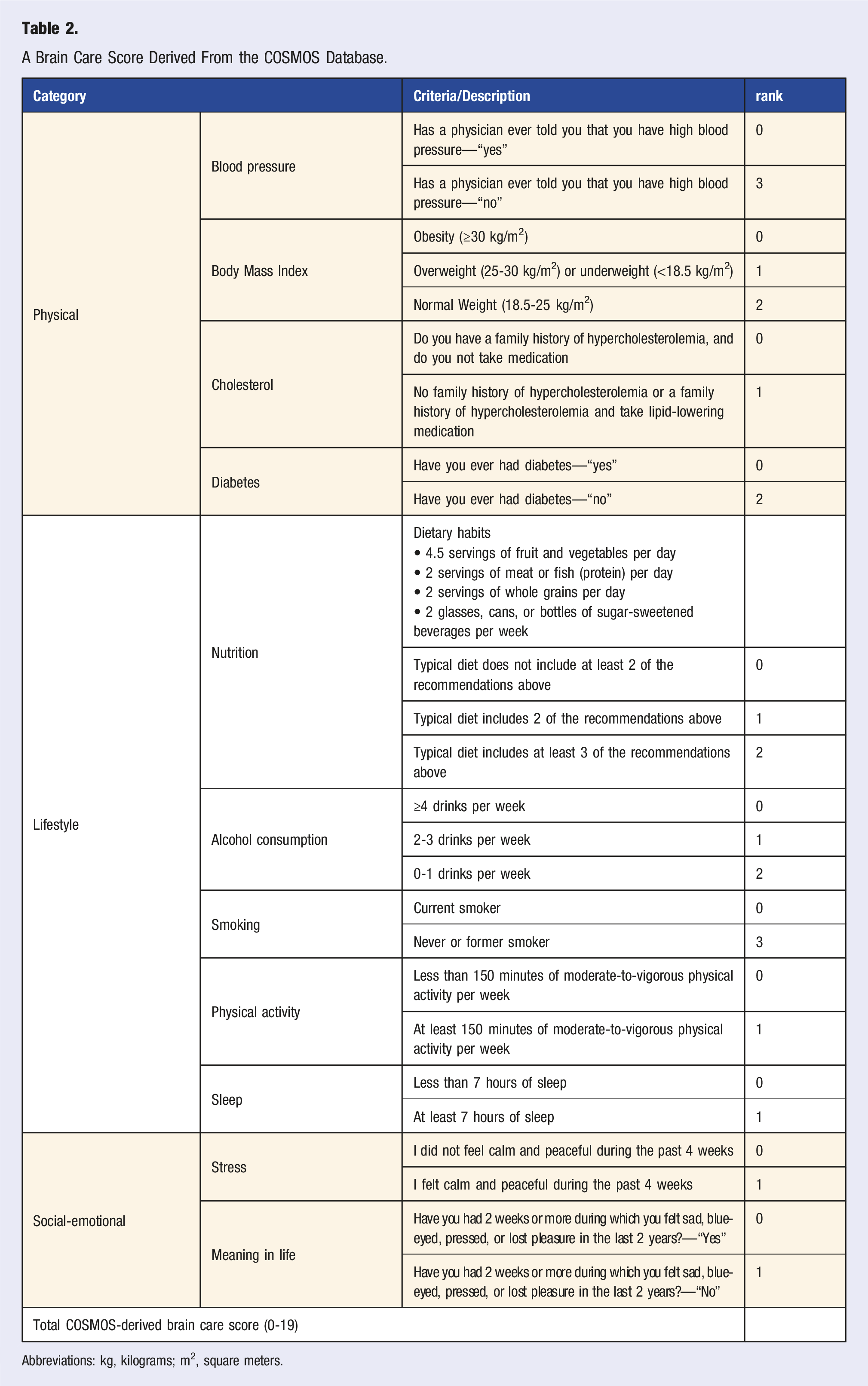
Abbreviations: kg, kilograms; m2, square meters.
Validation of Cholesterol Category Definition
A total of 13 847 participants had a cholesterol score on the BCS and self-reported their total cholesterol level in categories. Participants who had a family history of high cholesterol and did not take medication (N = 2928, 21.1%) (receiving 0 points for cholesterol) had a mean total cholesterol level of 195.8 mg/dL (95%CI: 194.5-197.0). Those who report a family history of hypercholesterolemia or reported a family history of hypercholesterolemia and take lipid-lowering medication (N = 10 919, 78.9%) (receiving 1 point for cholesterol) had a mean cholesterol level of 179.1 mg/dL (95%CI: 178.5-179.7). There was a statistically significant difference between the 2 groups (P < 0.01).
Survival Analysis
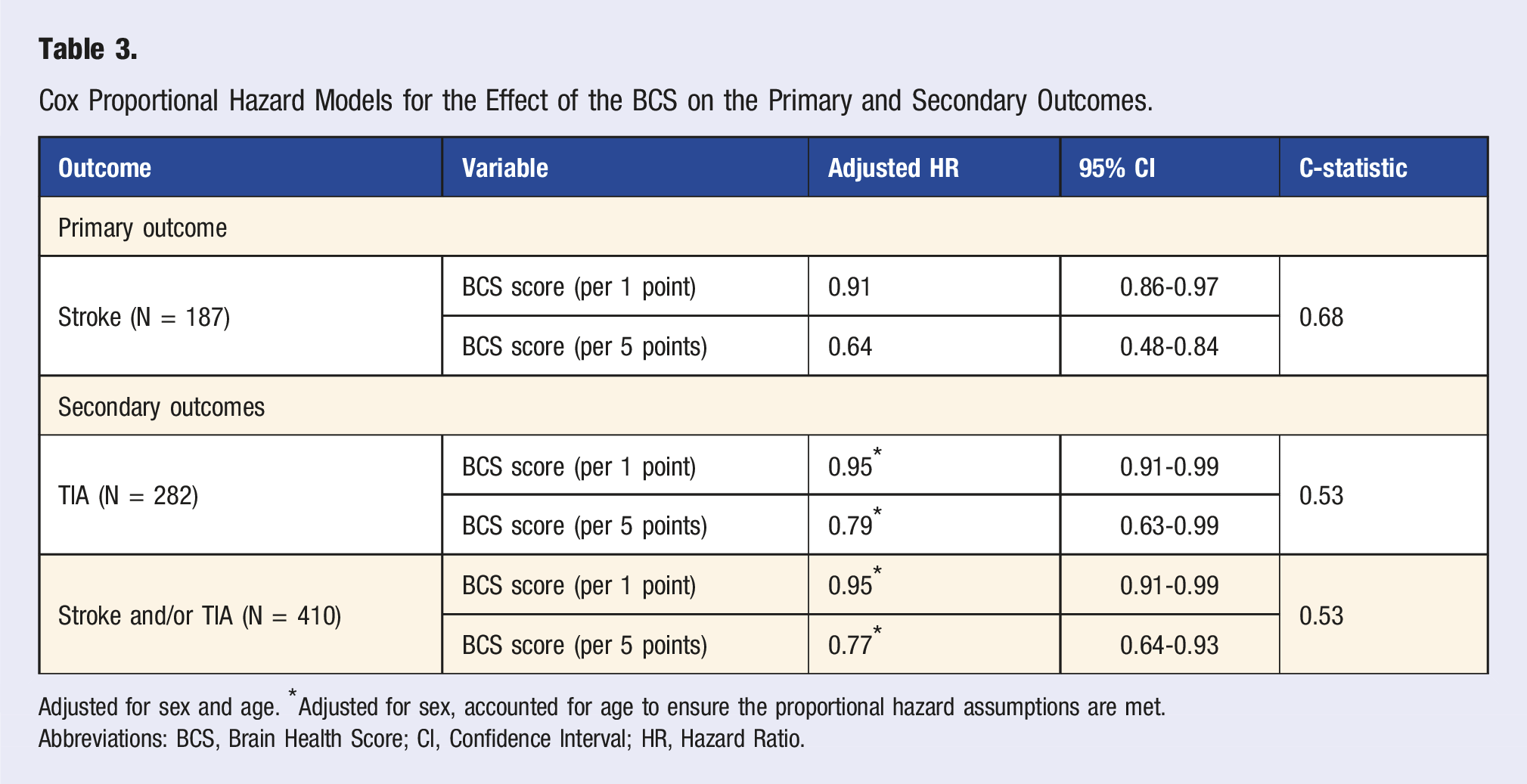
Primary Outcome
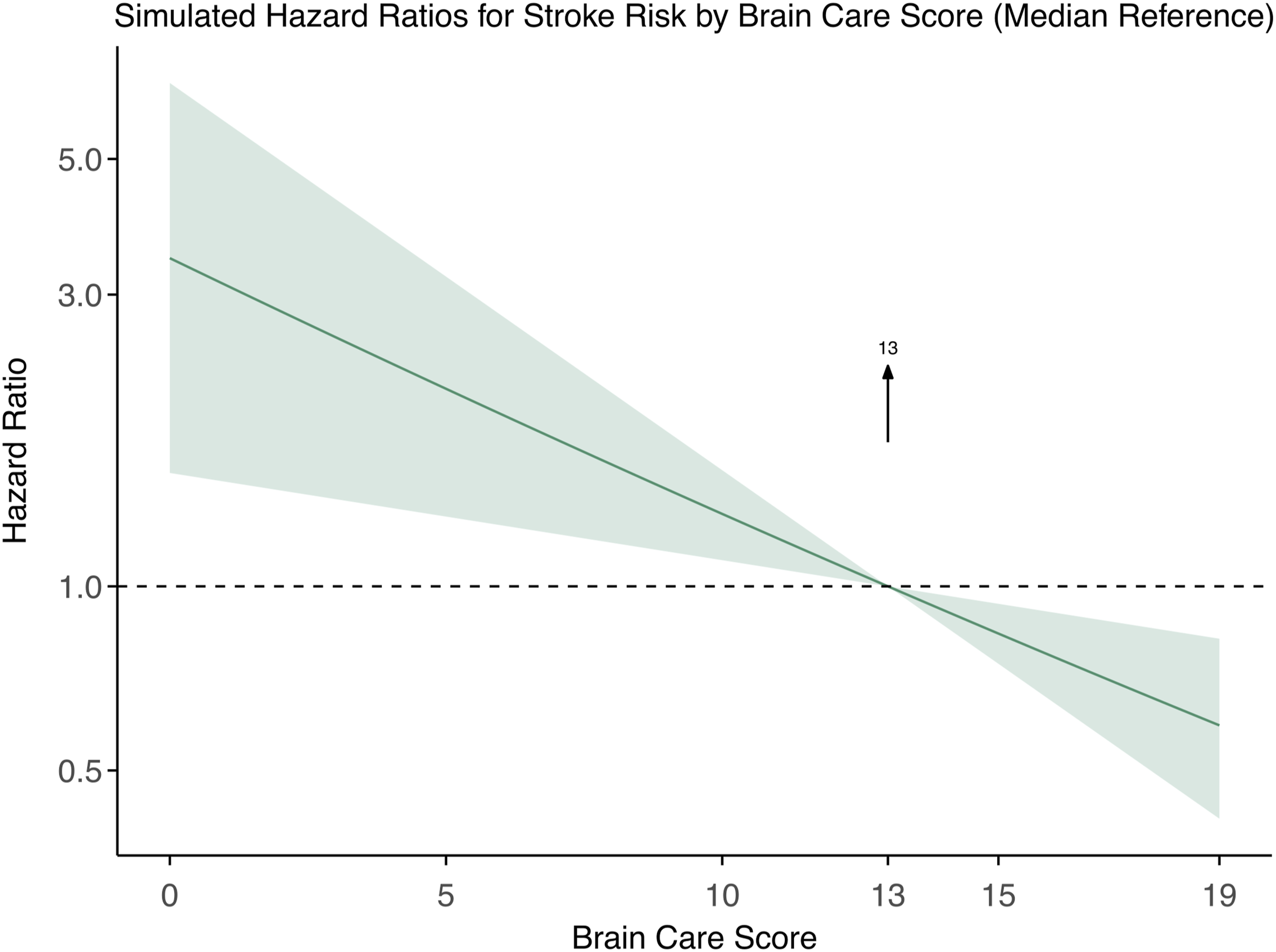
During a mean follow-up of 3.6 years (SD: 0.7 years), 187 (1.1%) people had a stroke (Table 1). In the lowest BCS quintile (BCS score ranging from 0-10; quintile distribution shown in Figure S4), 48 (1.4%) participants had a stroke, 117 (1.1%) in the 2nd to 4th quintile of BCS (BCS ranging 10-15), and 22 (0.6%) in the highest BCS quintile (BCS ranging 15-19). Figure 1 shows the cumulative incidence of stroke over time, stratified by BCS quintiles. In a Cox proportional hazards model, adjusted for sex assigned at birth and age, every 5-point higher baseline BCS was associated with a 36% lower risk of incident stroke (HR: 0.64 [95%CI: 0.48-0.84], c-statistic: 0.68 Table 3). A dose–response curve across the total BCS range is presented in Figure 2. Hazard ratios for stroke, compared to the median BCS, decreased from 3.52 (95% CI: 1.65-6.5) at the lowest BCS to 0.56 (95% CI: 0.39-0.78) at the highest, demonstrating an inverse dose–response association. Decision curve analysis showed that the model provided greater net benefit than either treating all or none of the population at predicted stroke risk thresholds of 1%-2% (Figure S5). Cumulative Incidence of stroke over time stratified by Brain Care Score. Cumulative incidence (y-axis) over time (x-axis) in years, stratified by BCS. Red: Lowest quintile (BCS score 0-10), Yellow: Middle three quintiles (BCS Score 10-15), Green: Highest quintile (BCS score 15-19). Log Rank Test: P = 0.0075. Cox Proportional Hazard Models for the Effect of the BCS on the Primary and Secondary Outcomes. Adjusted for sex and age. *Adjusted for sex, accounted for age to ensure the proportional hazard assumptions are met. Abbreviations: BCS, Brain Health Score; CI, Confidence Interval; HR, Hazard Ratio. Simulated Hazard Ratios for Stroke Risk by Brain Care Score Using Median as Reference. Relationship between baseline Brain Care Score and stroke incidence adjusted for age and sex. The bold dark green line represents the relative hazard curve for stroke incidence across the Brain Care Score range, displayed on a logarithmic scale. The shaded light green areas denote the 95% confidence intervals. The risk curves are plotted relative to the median Brain Care Score, indicated by the arrow.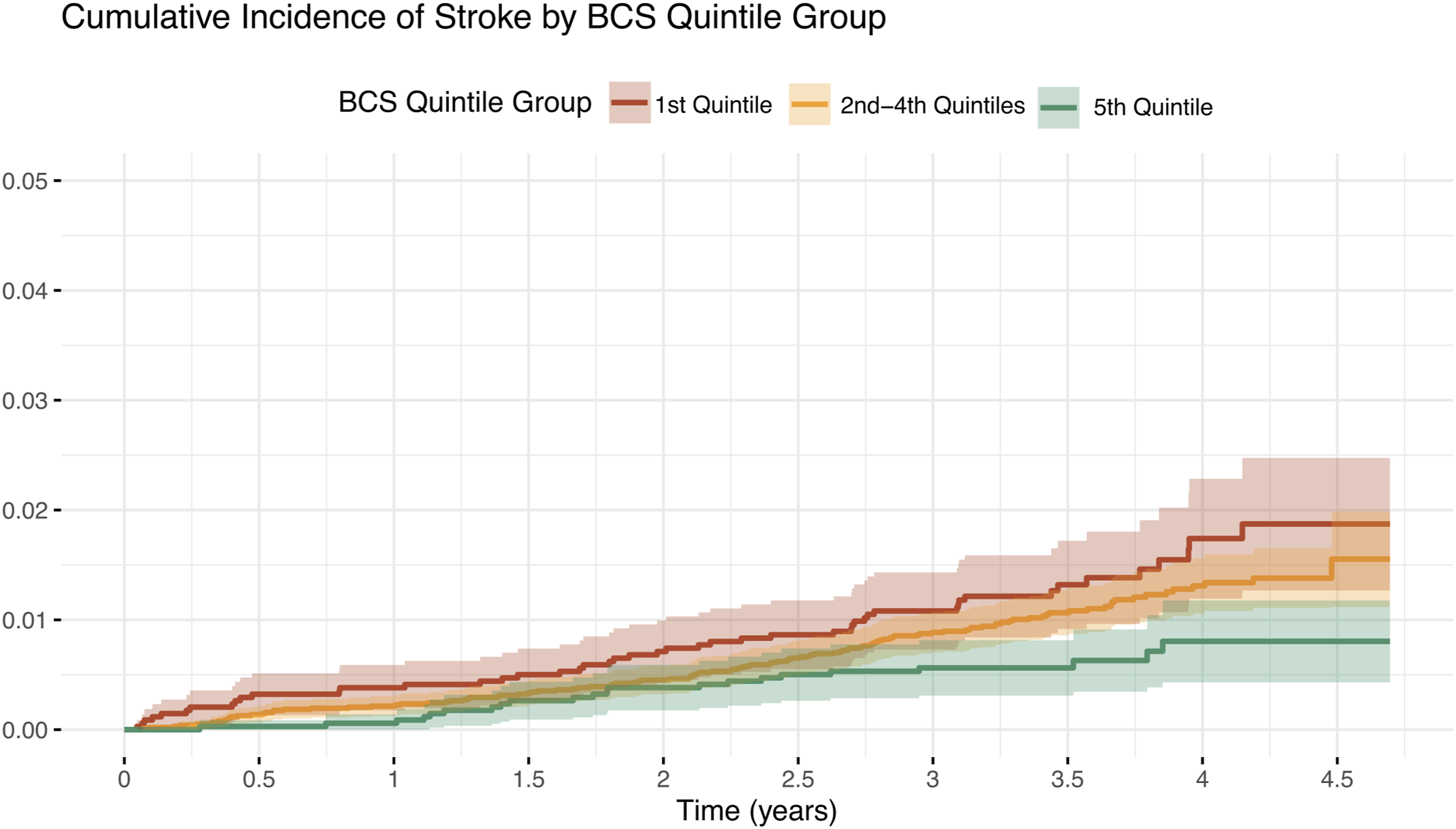
Secondary Outcome
A total of 282 (1.6%) people had a TIA, and 410 (2.4%) had either a stroke and/or a TIA during follow-up (Table 3). In a Cox proportional hazards model, adjusted for sex assigned at birth and accounted for age, every 5-point higher baseline BCS was associated with a 21% lower risk of TIA (HR: 0.79 [95%CI:0.63-0.99], c-statistic: 0.53]) and a 23% lower risk of stroke and/or TIA (HR: 0.77 [95%CI:0.64-0.93], c-statistic: 0.53]).
Sensitivity Analysis
Sensitivity analysis also adjusting for randomized treatment yielded the same results, showing that every 5-point higher baseline BCS was associated with a 36% lower risk of incident stroke (HR:0.64 [95%CI:0.48-0.84], c-statistic 0.68). There was no evidence of effect modification by randomized treatment assignments (data not shown).
Stratification by Sex Assigned at Birth and Age
Of 10 227 females, 131 (1.3%) had a stroke. Of 6923 males in this cohort, 56 (0.8%) had a stroke. In a Cox proportional hazard model adjusted for age, every 5-point higher baseline BCS was associated with a 32% lower risk of stroke among females (HR: 0.68 [95%CI:0.48-0.95]) and a 44% lower risk among males (HR: 0.56 [95%CI: 0.34-0.92]). Table S4).
Among the 12 273 participants aged 75 or younger at baseline, 88 (0.7%) individuals had a stroke during follow-up. Among the 4877 participants aged 75 or older, 99 (2.0%) had strokes. In a Cox proportional hazard model adjusted for sex, every 5-point higher baseline BCS was associated with a 33% lower risk of stroke among those 75 or younger (HR:0.67 [95%CI:0.45-0.98]) and a 37% lower risk of stroke among those 75 or older at baseline (HR:0.63 [95%CI:0.43-0.94], Table S5).
Fine and Gray Model
In a sensitivity analysis utilizing a Fine and Gray hazard model for the competing risk of death, adjusted for sex assigned at birth and age, every 5-point increase in BCS at baseline was associated with a 35% lower risk of stroke (HR:0.65 [95%CI:0.49-0.85]), a 20% lower risk of TIA (HR:0.80 [95%CI:0.63-1.00]), and a 23% lower risk of stroke and/or TIA (HR:0.77 [95%CI:0.64-0.94]). Schoenfeld residuals were plotted for all Cox proportional hazard models (Figure S6(a)A-(e)E).
Discussion
In a cohort of over 17 000 US participants, we developed a modified BCS entirely based on health information derived from participant responses; this version closely aligns with the original BCS and previous validation studies in other databases.9,37,39 We found a statistically significant association between a higher baseline BCS and a reduced risk of incident stroke. Our results demonstrate the predictive validity of this BCS derived from participant responses to help patients better understand their personalized stroke risk even when physical and laboratory measurements are not readily available.
Our findings demonstrate that using a health screening tool with risk factors derived from participant responses has a similar predictive validity compared to a tool that requires laboratory values or a medical examination. A previous study that assessed the association between a 5-point higher baseline BCS that utilized blood pressure assessment and laboratory measurements and stroke incidence among 400 000 UK Biobank participants found a 5-point higher BCS associated with a 40% reduced risk of stroke with a c-statistic of 0.71, 9 similar to our present findings with a 36% reduced risk and a c-statistic of 0.68. Another study that used a self-reported version of the Framingham Stroke Risk Function showed that compared to the same tool that utilizes medical examination measurements, the version derived from participant responses did not underperform in predictive validity in the same cohort. 42 Their results suggested that using a version derived from participant responses might even improve risk prediction, as shown by a higher c-statistic in the version derived from participant responses. When looking at TIA or a composite outcome of stroke or TIA, the models had a notably lower C-statistic (0.53 for both) than other models in the current literature. 43 This may be due to accounting for age by categorizing it into tertiles rather than adjusting for it continuously in the Cox proportional hazard model, the relatively short follow-up time, and a low number of events.
The strengths of our study include the prospective validity of a BCS derived from participant responses, the utilization of a large cohort with multiple years of follow-up, and extensive analyses, including sensitivity analyses. This is also the first study to validate the BCS in a population whose median age was over 70 years at baseline.9,37,39 Our stratified analysis showed that for both those younger than 75 and those older than 75 at the time of enrollment, a higher baseline BCS is associated with a lower incidence of stroke, suggesting that it is never too late for risk factor management to reduce the risk of age-related brain disease.5,7 Furthermore, this is the first study to assess the predictive validity of the BCS for incident TIA during follow-up, although the discriminatory ability was low.
Limitations exist when interpreting our results. First, the generalizability of data from the COSMOS study is limited, as nearly 90% of participants were Non-Hispanic White, and the majority were highly educated, which is not fully representative of the older American population. Prior research indicates that Non-Hispanic Whites and individuals with higher education levels tend to have a higher level of awareness and health literacy of cardiovascular risk factors.44-46 As a result, findings from this cohort may not extend to more diverse or underrepresented groups, who often encounter additional barriers to preventive care, risk factor management, and access to health information.46,47 Future studies should include more representative samples to better inform equitable public health strategies. Furthermore, these demographic characteristics may have contributed to the low incidence of stroke observed in the study, as compared to the rest of the U.S. 48 The potential for volunteer bias has previously been described for this clinical trial and might have influenced the representativeness of the sample. 27 Therefore, the selection biased introduced by utilizing this cohort may have overestimated the predictive validity of our findings. We excluded nearly 19% of participants due to missing BCS data. Those people were slightly older and more often male, however there was no difference in outcome rate. While this pattern could indicate missing at random, we chose complete-case analysis to align with previous methodology,9,37,38 and adjusted for age and sex in our models to minimize potential bias. 49 While we show the predictive validity of a BCS derived from participant responses in its association with incident stroke, we cannot conclude non-inferiority compared to a BCS that uses medical examination measurements. Ideally, we would compare a version derived from participant responses of the BCS with a version that utilizes quantitative measurement in the same database, 42 but those data, specifically actual measurements of blood pressure or HbA1c, were not available in COSMOS. While we were unable to perform direct validation within our sample, prior research from a nationally representative U.S. cohort has shown that self-reported hypertension and diabetes are reasonable proxies for measured values. 21 Future research should aim to replicate our approach in cohorts that include both health information derived from participant responses and clinical data to allow for more robust validation. Furthermore, while the COSMOS-derived BCS became accessible through common health questions, some of these questions are non-modifiable and don’t provide any information on current treatment (e.g., “Have you ever had diabetes”?). This characteristic contrasts with the intent of the BCS as a score to help people and practitioners take actionable steps to take care of their brain health through modifiable risk factors only. 8
The findings of this study represent an initial step toward developing accessible risk assessment tools to aid in the prevention of age-related brain diseases. 5 While knowing actual values for blood pressure and other stroke risk factors could offer more opportunities for detailed risk stratification, 50 self-reported risk factors and tools may serve as an initial mechanism to increase self-efficacy, improve accessible knowledge, and possibly even to engage individuals in brain health promotion and stroke prevention, both in clinical and community settings. Future research should prioritize validating these results in diverse populations to enhance generalizability and extend to other age-related brain conditions with similar pathophysiology and risk factors, such as dementia and late-life depression, 51 and compare a BCS derived from participant responses vs quantitatively measured BCS within the same cohort.
Conclusion
Our findings demonstrate that a BCS derived from participant responses is associated with incident stroke in a U.S. cohort. The BCS derived from participant responses showed similar predictive performance and discriminatory ability compared to previous validation studies that use physical and laboratory measurements. Given its accessibility, the BCS derived from participant responses may serve as a practical tool in settings where medical evaluations are not readily available, helping individuals and clinicians identify risk and promote healthy behaviors to reduce the risk of brain disease. Future studies should aim to validate the self-reported BCS in more diverse and underrepresented populations.
Supplemental Material
Supplemental Material - A Self-Reported Brain Care Score in Its Association With Incident Stroke
Supplemental Material for A Self-Reported Brain Care Score in Its Association With Incident Stroke by Jasper R. Senff, Reinier W. P. Tack, Benjamin Y. Q. Tan, Tamara N. Kimball, Savvina Prapiadou, Devanshi Choksi, Tanzeela H. Ranman, Taylor M. McVeigh, Aladdin H. Shadyab, H. Bart Brouwers, JoAnn E. Manson, Howard D. Sesso, Christopher D. Anderson, Jonathan Rosand, Sanjula D. Singh, Pamela M. Rist, and Nirupama Yechoor in American Journal of Lifestyle Medicine.
Supplemental Material
Supplemental Material - A Self-Reported Brain Care Score in Its Association With Incident Stroke
Supplemental Material for A Self-Reported Brain Care Score in Its Association With Incident Stroke by Jasper R. Senff, Reinier W. P. Tack, Benjamin Y. Q. Tan, Tamara N. Kimball, Savvina Prapiadou, Devanshi Choksi, Tanzeela H. Ranman, Taylor M. McVeigh, Aladdin H. Shadyab, H. Bart Brouwers, JoAnn E. Manson, Howard D. Sesso, Christopher D. Anderson, Jonathan Rosand, Sanjula D. Singh, Pamela M. Rist, and Nirupama Yechoor in American Journal of Lifestyle Medicine.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The COcoa Supplement and Multivitamin Outcomes Study (COSMOS) is supported by an investigator-initiated grant from Mars Edge, a segment of Mars dedicated to nutrition research and products, which included infrastructure support and the donation of study pills and packaging. Pfizer Consumer Healthcare (now Haleon) provided support through the partial provision of study pills and packaging. COSMOS is also supported, in part, by grants AG050657, AG071611, EY025623, and HL157665 from the National Institutes of Health, Bethesda, MD. The Women’s Health Initiative (WHI) is funded by the National Heart, Lung, and Blood Institute, NIH, US Department of Health and Human Services through contracts 75N92021D00001, 75N92021D00002, 75N92021D00003, 75N92021D00004, and 75N92021D00005. Neither Mars Edge nor Pfizer Consumer Healthcare (now Haleon) provided input regarding the design of the study, data analyses, interpretation of results, or manuscript development nor did they review, comment, or approve the manuscript.
Disclosures
B.Y.Q.T. receives research support from the National Medical Research Council, Singapore, and Abbott Laboratories (Singapore). C.D.A. receives sponsored research support from the US National Institutes of Health, the American Heart Association, and Bayer AG, and has consulted for ApoPharma. M.C. is supported by the Wellcome Trust [grant number 205339/Z/16/Z]. J.R. receives sponsored research support from the US National Institutes of Health and the American Heart Association, receives payments for consulting and expert testimony from the National Football League, and has a leadership or fiduciary role at Columbia University and Lancet Neurology. HDS and JEM reported receiving investigator-initiated grants from Mars Edge, a segment of Mars Incorporated dedicated to nutrition research and products, for infrastructure support and donation of COSMOS study pills and packaging, and Pfizer Consumer Healthcare (now part of GSK Consumer Healthcare) for donation of COSMOS study pills and packaging during the conduct of the study. HDS additionally reported receiving investigator-initiated grants from Pure Encapsulations and Pfizer Inc. and honoraria and/or travel for lectures from the Council for Responsible Nutrition, BASF, NIH, and the American Society of Nutrition during the conduct of the study. PMR receives sponsored research support from the US National Institutes of Health and the American Heart Association.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
