Abstract
First Nations women in Australia diagnosed with type 2 diabetes, co-designed and attended a programmed shared medical appointment that included continuous glucose monitors and culturally responsive food appreciation activities over 8 weekly sessions to improve glycaemic control. The project was a single site, longitudinal change from baseline, mixed methods, feasibility study using HbA1c as the primary outcome measure. Secondary outcome measures included, weight, metabolic health-related blood panels, CGM, Blood Glucose Levels (BGL) time-in-range percentage, Patient Activation Measure (PAM) and Problem Areas In Diabetes (PAID) tools and client satisfaction survey and semi focussed interviews. Forty participants commenced and twenty-five participants completed the program (62,5%). Of the completing participants the mean HbA1c had an absolute reduction of 0.7 percentage points ( from baseline to program completion, P = .013). Eighteen (75%) reduced HbA1c and maintained some reduction for 12 months. Seven (28%) achieved remission, HbA1c <6.4% (46 mmol/l) maintained for 12 months. There were statistically significant improvements in weight, systolic blood pressure, liver enzymes, BGL time-in-range, PAM and PAID scores. It is feasible to use programmed shared medical appointments incorporating CGM aiming to improve glycaemic control and other metabolic measures of health among First Nations women in Australia.
Keywords
“Forty, First Nations women were recruited to the DRAW project and agreed to participate in one of 4 groups”.
Introduction
According to the International Diabetes Federation 1 First Nations People account for 6.2% of the global population, with over 476 million Indigenous Peoples in 90 countries and more than 5000 distinct groups across the globe. Colonisation has disrupted the traditional life practices of Indigenous Peoples through forced assimilation, land and resource displacement, loss of language, restricted cultural practices, changes to eating patterns and historical and intergenerational trauma.1–3 This has contributed to measurable disparate health outcomes that continue to impact First Nations populations across the globe. Some of these include; life expectancy, infant mortality, birth weight, maternal mortality, relative educational attainment, relative poverty and relative nutritional status. 2
Type 2 Diabetes Mellitus (T2DM) is now one of the most common health issues to impact First Nations populations worldwide and is increasing over time.1–3 T2DM is reported to be at rates far greater than their non-First Nations counterparts.2–5
In Australia, First Nations people, are four times more likely to be diagnosed with T2DM, five times more likely to be hospitalised, and six times more likely to die from it. 6
However, it is reported that First Nations people feel mainstream health services reproduce many of the colonising practices that they have experienced since settlement,7–9 and state that they do not feel the services and their models of care are culturally safe or responsive to First Nations people. As a result, First Nations people are often reluctant to access health services for the prevention and management of chronic and lifestyle-related illnesses like T2DM.8–10
A growing body of research is showing it is possible to manage T2DM to a state of remission largely through improved nutrition and movement.11,12 However, to improve outcomes for First Nations people with T2DM in Australia, the barriers to access need to be addressed.
A model of care that is reported to address some of these barriers includes Shared Medical Appointments (SMAs). SMAs are a rare example of a model that can meet health care’s quintuple aim of better cost-effective care and outcomes, better education and high patient and clinician satisfaction.13–17
SMA’s have been shown specifically to improved outcomes for patients with T2DM.13,18–21
In addition, SMAs are reported to align specifically with Australian First Nations peoples’ traditional concepts of health consultations and service delivery. SMAs have many equivalent traits of the traditional yarning circle, for example, which for thousands of years has been a common method of conducting community business, consultation and care. Participants within the yarning generally follow principles of deep listening, respect and equality which is facilitated by a respected person. Participants of the yarning circle are mostly able to contribute without interruption when it is their turn to engage. The method of conversing is a typically collaborative, rather than an adversarial discourse, which is often the more common approach to discussion in Westernised societies.22–24 This familiarity is recognised when Australian First Nations people experience SMAs and have reported improve cultural safety and access as a result. 24 SMAs have been proposed by First Nations people as a model and process for preventing and managing lifestyle-related diseases in First Nations communities.22–24 A curriculum of knowledge, skills development and behaviour change can be delivered as a programmed Shared Medical Appointment (pSMA). pSMAs have been shown to be culturally safe and responsive among First Nations people in Australia.22,23,25
The use of Continuous Glucose Monitor (CGM) systems has been shown in the literature to positively impact behaviour in the management of diabetes in general populations26–29; however, there was no literature to be found for its use with First Nations people specifically. CGM’s read interstitial glucose levels providing real time data and immediate feedback to smart phones or devices. Reports and visual displays from CGMs provide a comprehensive and dynamic overview which fingerstick testing cannot. With an understanding of these data, patients can respond with more precise and informed lifestyle choices, creating behavioural changes to self-manage T2DM.29,30` Promising results have been reported from incorporating CGM with SMAs in the broader population of people with T2DM.31,32
The authors hypothesise that a co-designed, First Nations led, culturally responsive pSMA which includes the use of continuous glucose monitors will encourage engagement, activation and self-management as well as improve glycaemic control and metabolic health outcomes.
Method
Aims and Objectives
This project aimed to examine the feasibility of a co-designed, programmed shared medical appointment (pSMA) that incorporated the use of continuous glucose monitor systems to support First Nations women with sustained HbA1c above 6.5% (48 mmol/l) to manage their T2DM.
Design
The project was a co-designed, single site, longitudinal, change from baseline, mixed methods, feasibility study.
The project design was informed by the Promoting Action on Research Implementation in Health Services (PARiSH) framework 35 to develop practice-based evidence that could provide a roadmap for First Nations health services to more responsively manage T2DM.
The project applied established co-designed principles,33,34 and as such was; First Nations led, culturally grounded in its approach, respectful; beneficial to community (not just the researchers); an inclusive partnership with the service and community, with transparent and objective evaluation.
The community in which this project was undertaken worked with trusted researchers who had developed a relationship with the service over many years. Together the service, the community and the researchers developed and designed the project to ensure cultural safety and responsiveness.* • *The Aboriginal community in which this research was undertaken hold strong views that randomised controlled trials (RTC) have the potential to further subjugate First Nations peoples to oppression and colonising practices and requested that RTC methodology not be used.
Setting
Waminda – South Coast Aboriginal Women’s Health and Wellbeing organisation (Waminda) is located in NSW Australia on Yuin Country. The Aboriginal Health Service has been working with the local First Nations community for 40 years with a strong decolonising framework and social model of health care. First Nations women with a principal diagnosis of T2DM or who are pre-diabetic makeup about 15% of Waminda's 1500 existing clients.
Participants and Recruitment
Participants were recruited using purposive sampling of First Nations women with a diagnosis of T2DM. Some of the women had experienced an existing general well-being program run at Waminda called ‘Dead or Deadly’. Though Dead or Deadly was not run as a Programmed SMA nor was there a focus on type 2 diabetes, the participants however, developed a trust with the service and its staff which helped with recruitment but also had the potential to limit the feasibility outcome of the project because of participant bias.
Inclusionary Criteria
Included First Nations Women registered for Waminda services, aged over 18 years with HbA1c ≥ 5.6% (38 mmol/l) for ≥1 year. The participants would also continue to receive usual care.
The Aboriginal Health Practitioners (AHP), Credentialled Diabetes Educator (CDE) and the participating General Practitioner were responsible for recruitment. The potential participants were provided information about the project verbally and then also given an information sheet designed by the Waminda research Cultural Committee. The process was explained in detail prior to formal consent being obtained by the research project officer. Ethics: Approval for the project was obtained from Aboriginal Health and Medical Research Council (AHMRC) ECN 1876/21. Trial registration: The project protocol was registered webpage can be found at https://www.anzctr.org.au/ACTRN12622000650796.aspx
Program Development and Description
The co-design, iterative process was entered into with the local First Nations community, service staff, researchers and participants. The aim was to develop a program to manage and even bring T2DM into remission, using a culturally safe and responsive approach within a SMA.
The resulting Diabetes Remission in Aboriginal Women program (DRAW) has three elements:
The wearing of a Dexcom G6* Continuous Glucose Monitor (CGM)
*(This company had no role in this study’s design analysis or publication. The CGM devices were purchased at full price using resources provided by the IHP grant).
The Dexcom G6 CGM displays a blood glucose level (BGL) reading on participants' devices – every 5 minutes. By agreement, participant data was streamed to the research team and General Practitioner to observe and collect data.
The insertion of the CGM and familiarisation of the technology occurred in the first week of the DRAW program at the same time baseline measures were taken. The women were advised to continue their normal diet until the commencement of the first pSMA session, approximately 1 week following the initial visit.
Programmed Shared Medical Appointments
Waminda had in the past, successfully integrated Programmed Shared Medical Appointments (pSMA) into their health service delivery for weight management, smoking cessation, and chronic pain.22,23 The SMA element was delivered prior to the Nyully Food session and has two parts: 1) The first part was the education component and included, sharing knowledge, lived experiences, CGM data insights, and health coaching. This component was followed by; 2) individual facilitated medical consultations with the Waminda GP in the presence of the other group members. This was an opportunity for medication changes, new scripts and insulin titration and further learning from others in the supportive environment.
The sessions were delivered by a trained and experienced SMA facilitator who was also a Credentialed Diabetes Educator (CDE) and an Aboriginal Health Practitioner (AHP) as well as the participants’ regular General Practitioner.
Nyully Food
Nyully food is a 7-week food appreciation program developed by Waminda, their dietitians and diabetes educators. The program provides participants the opportunity to explore a diet equivalent to pre-colonisation. This includes the foods that were once consumed in their local region and to find the modern-day equivalents if they no longer exist nor are accessible. Following pre-colonisation food principles naturally lead to reductions or removal of ultra-processed, high-sugar and unhealthy fat content foods, and replacing them with whole foods, protein-rich foods, healthy fats, vegetables and fruits.
The Nyully Food program was delivered after the SMA element of the pSMA, through weekly sessions of demonstration and yarning (conversation) in the Waminda kitchen and food gardens. The dietitian and diabetes educator assisted participants to identify, source and prepare food that is known to be T2DM friendly. The food prepared for the session was then shared and eaten amongst the participants in a socially interactive environment as part of the experience while they could all observe the effects of the food on their BGLs through the CGM.
The DRAW pSMA was scheduled to be delivered in groups of 8-12 participants over 8 weeks. The number of groups would be determined by the number of participants accepting the invitation and meeting the inclusion criteria for this feasibility study. Week 1: a) Informed consent process, Continuous Glucose Monitor (CGM) fitting and familiarisation (1 x ∼ 3hr session) at the Waminda clinic. Baselines measurements (Measure 1). Weeks 2-8: b) Shared Medical Appointments (7 x ∼ 90min sessions). The SMA facilitator delivers the co-designed T2DM curriculum (30-60 min), then facilitates the SMA consultation with the GP (∼60 minutes). (c) Nyully Food Program (7 x ∼ 2hr sessions). (d) Outcome measures, Week 8 *, immediately after the DRAW program delivery (Measure 2). (e) Outcome measures @12 months after completion of the DRAW program (Measure 3). *The HBA1c measure was actually measured at the next naturally occurring 3 month point from the baseline measure, as per the frequency criteria for HbA1c measures, as determined by the certified laboratory. For some participants this was up to 3 weeks after the end of the DRAW program delivery.The DRAW Protocol Schedule
Outcome Measures
HbA1c was the primary outcome measure. According to the Type 2 diabetes remission and lifestyle medicine position statement from the American College of Lifestyle Medicine, a confirmed T2DM diagnosis includes a HbA1c analysis measuring >6.5% (48 mmol/mol). HbA1c moving from above 6.5% (48 mmol/mol) to 6.4% (46 mmol/mol) or below for a period of at least 6 months and in the absence of glucose lowering medications is considered to be remission of T2DM. 11
HbA1c and secondary outcome measures including blood pressure (BP), weight, and other blood panel tests, including those for lipids, liver and renal pathology, were collected as baseline prior to commencement (Measure 1) and then on completion of the 8-week program (Measure 2). HbA1c and weight were also measured again 12 months after the completion of the program (Measure 3). Blood samples were analysed by an accredited laboratory in NSW Australia.
CGM-generated data were collected from patients’ using Dexcom Clarity software to generate the Ambulatory Glucose Profile. The main outcome measure focussed on percentage of Time-In-Range Between 3.9-10 mmol/L is considered a normal range.29,30
Patient Activation Measure (PAM 13) and the Problem Areas in Diabetes Tool (PAID) were first administered at baseline in week 1. PAM is a validated tool measuring the level of patient engagement in the program and correlates to improved overall patient outcomes and reduced health care expenditure. 35 The PAID tool measures changes in psychological distress in living with diabetes. Negative changes indicate increasing confidence in self-managing T2DM. 36
A five-point Likert scale satisfaction survey was administered on conclusion of the program. In addition, all participants were invited to engage in either a semi-structured individual yarn (i.e. interview) or a yarning circle (i.e. focus group) which ever they felt more comfortable in undertaking. The interviews aimed to understand the lived experience of having T2DM as well as the experience of undertaking the DRAW program. This invitation was extended to participants who did not complete the program to identify their reasons.
All interviews (yarns and yarning circles) were audio recorded and transcribed into NVivo 12 for an inductive thematic analysis. The qualitative component of this research is the subject of a Doctorate and only a limited summary is supplied in these results.
Quantitative analysis was undertaken with IBM SPSS X 28. Descriptive statistics were generated to portray the participants' demographic characteristics. Repeated measures ANOVA was used to measure difference between the baseline (Measure 1), on completion of the program (Measure 2) and 12 months after program completion (Measure 3) for HbA1c and weight. Paired t-tests were used to analyse the significance of the difference (set at P < .05) between measures for the other blood panels and mean BGL percentage time-in-range from baseline (Measure 1) to completion of the program at week 8 (Measure 2).
Results
Forty, First Nations women were recruited to the DRAW project and agreed to participate in one of 4 groups. Between group analysis revealed that there were no significant differences at baseline (Measure 1) for age, length of time since T2DM diagnosis, completion rates nor HbA1c measures.
Description of the Participants.
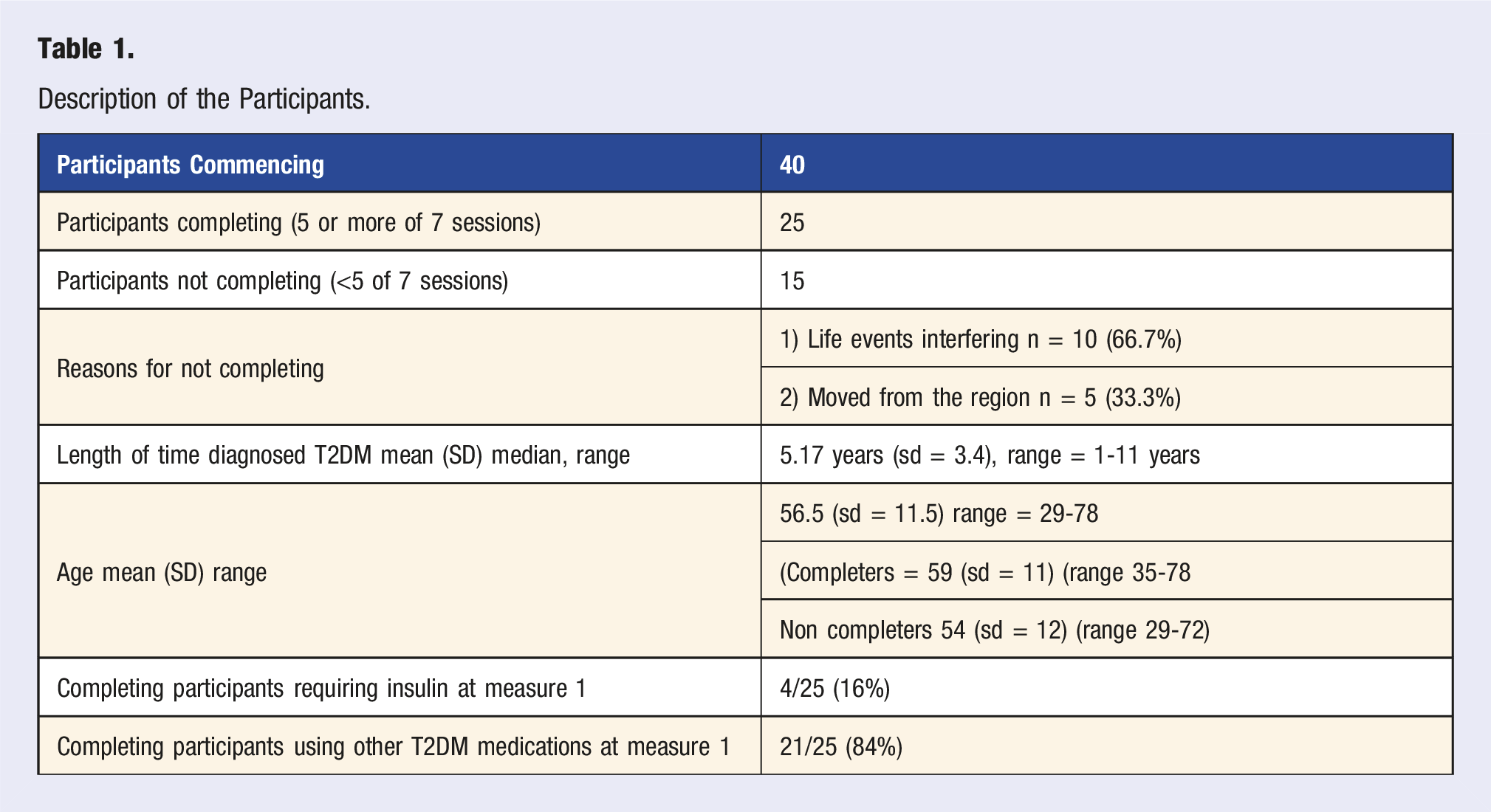
The two main reasons 15 participants did not complete the program were: 1) overwhelming life events and or 2) moving away from the region.
As this was a feasibility study and not a methodologically or statistically powered trial, the data was then separated for completing and non-completing participants. The non-completing participants data were excluded from the quantitative analysis (with the exception of their 12-month HbA1c routine, standard care measure, which was extracted from medical records and compared with those who had completed the program at the third measure (Measure 3).
Table 1 shows that the mean age of all 40 participants was 56.5 years and the median duration of their T2DM diagnosis was 5 years.
Comparison of HbA1c and Weight for Patients Who Completed DRAW (≥5 of 7 Sessions) at Baselines (M1), Post-Program (M2) and 12 Months Post Program (M3).
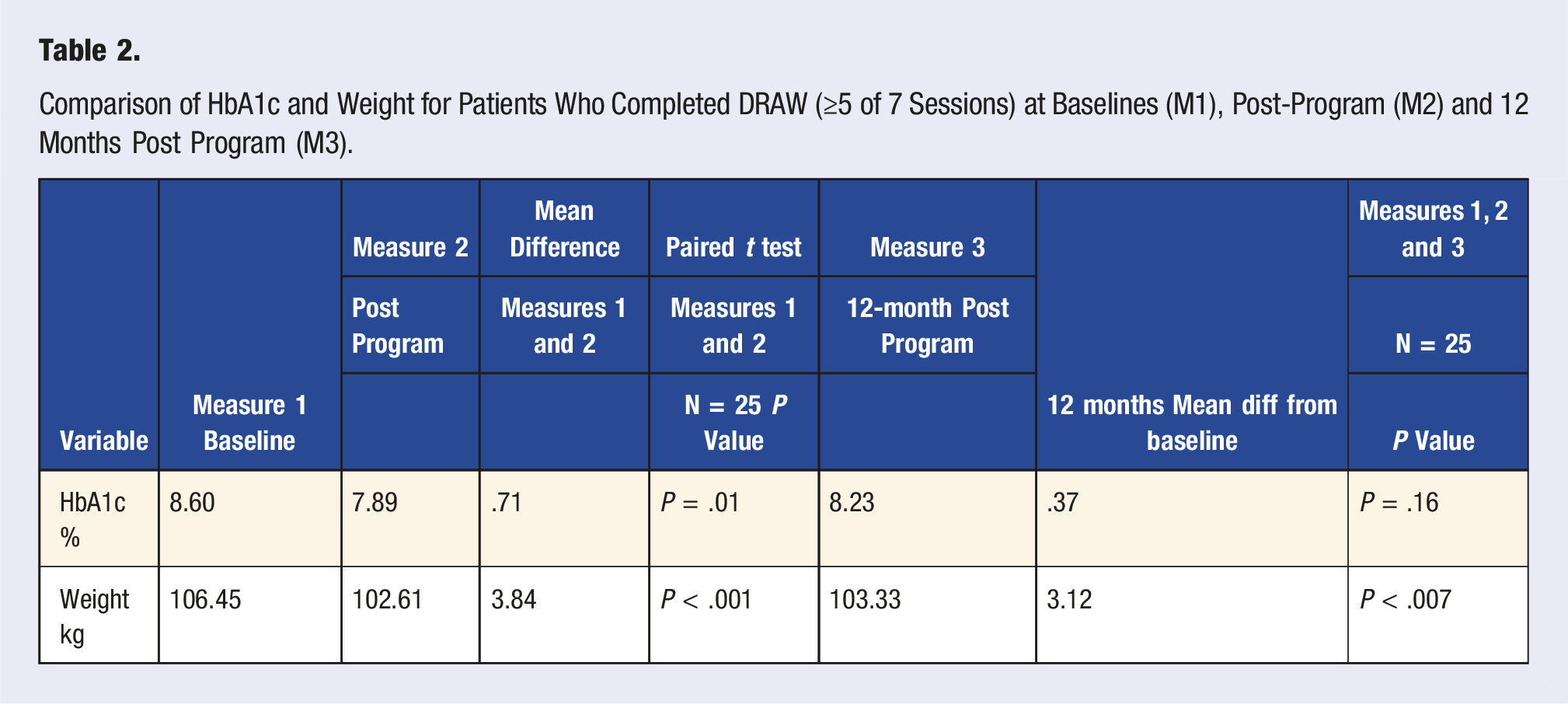
There was a statistically significant, mean reduction in HbA1c (.71%, P = .01) for the cohort between baseline (Measure 1) and post-program measure (Measure 2) with 18 of 25 completing participants (72%) recording some decrease in HbA1c.
Of the four participants who required insulin at baseline (Measure 1), none required insulin or other T2DM-related medications on completion of the program (Measure 2) nor at 12 months after the program (Measure 3). Too many data sets were incomplete to calculate variations in other T2DM medication consumption.
Chart 1 illustrates individual mean HbA1c% changes over the three measures: baseline (Measure 1); post DRAW program (Measure 2); and post program plus 12 months (Measure 3). Mean Individual HbA1c% Measures at Baseline, Post Program and Post Program + 12 months. 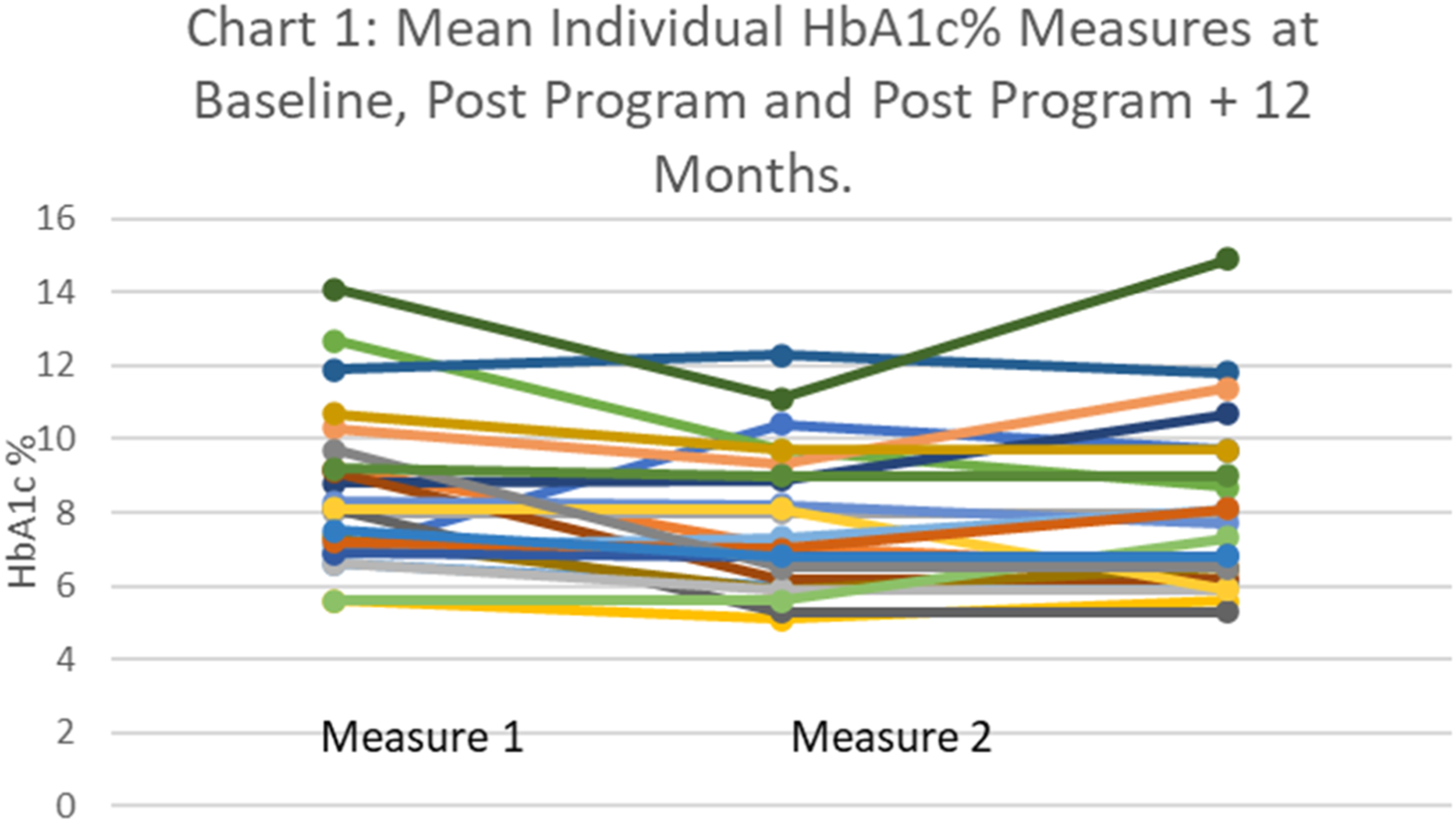
Chart 2 shows the extracted data from Chart 1 that represent the 7 participants who achieved remission (HbA1c < 6.5%). The chart demonstrates that 6 of the 7 participants achieved remission by the completion of the program and maintained it for 12 months. One participant was still above the threshold for remission after the program but managed to meet the threshold of <6.5% by the 12-month measure (Measure 3). Mean Individual Measure of Participants In Remission at Post Program + 12 Months.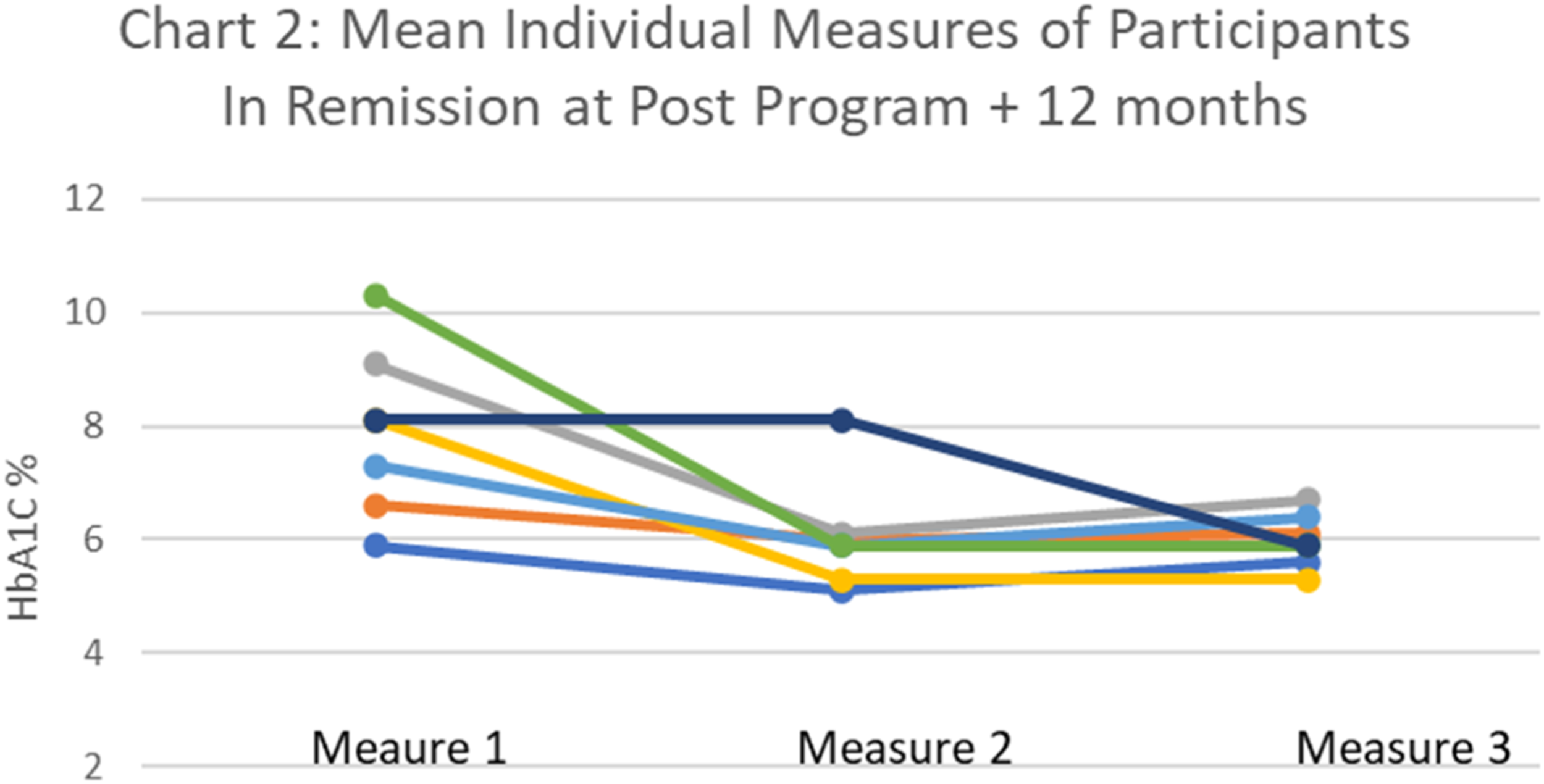
Chart 3 shows the extracted data from Chart 1 that represents the measures of the 18 (of 25) completing participants who reduced and maintained a reduction of their HbA1c from baseline (Measure 1) to 12 months after program completion (Measure 3). Mean Individual HbA1c % for Participants Maintaining Some Reduction Post Program + 12 Months.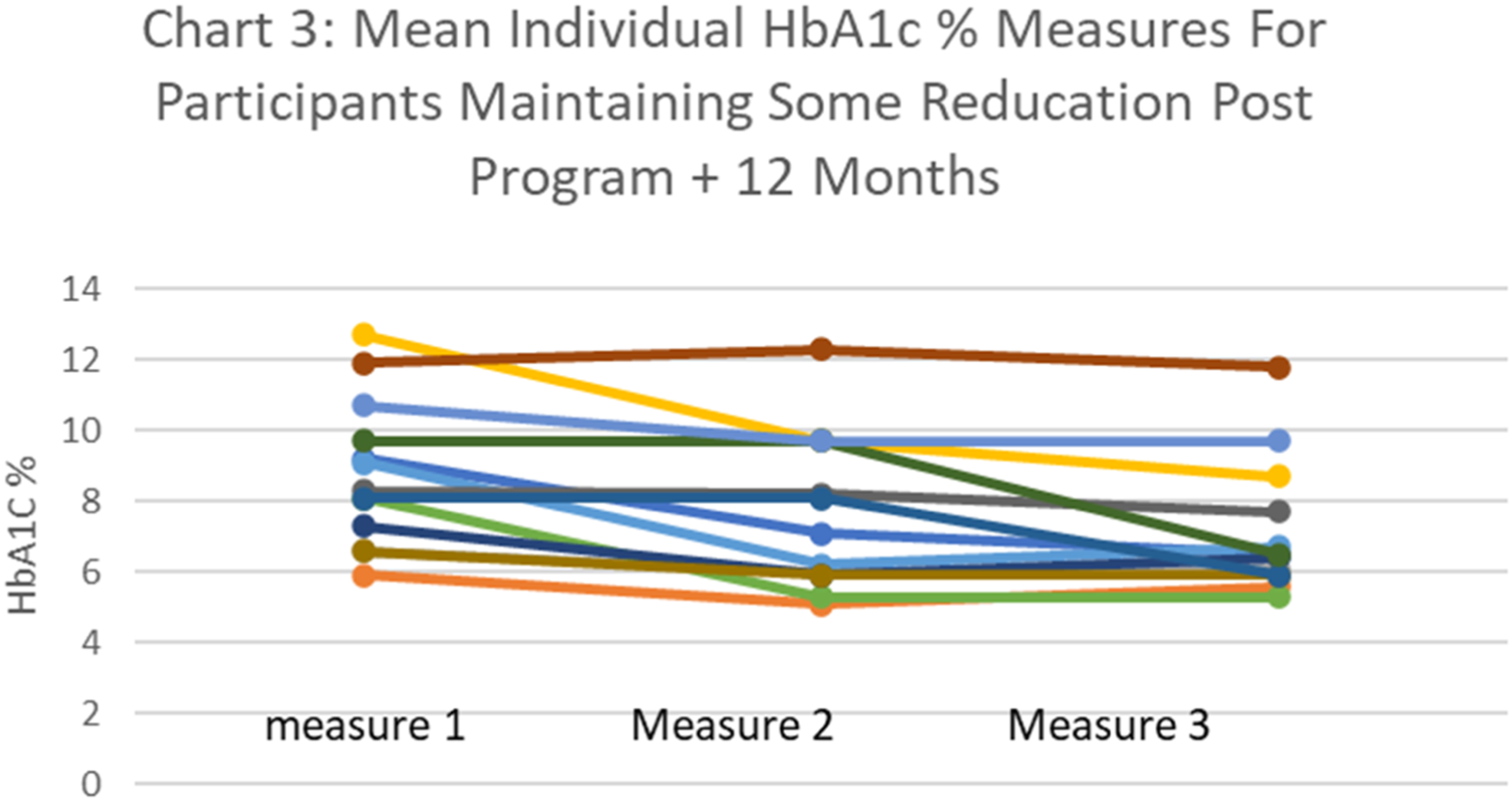
Of the completing participants Table 2 shows that there was statistically significant reduction in weight between baseline (Measure 1) and post program (Measure 2) of 3.84 kgs (P < .001). Between baseline (Measure 1) and 12 months post program (Measure 3) there was a mean loss of 3.12 kgs (P = .007). Twenty-two (91.7%) lost some weight at the completion of the program (Measure 2) and maintained some level of weight reduction compared to baseline at the 12-month after completion of the program (Measure 3). Eight women (32%) lost 5% or more of their original weight between baseline (Measure 1) and the 12 month (Measure 3).
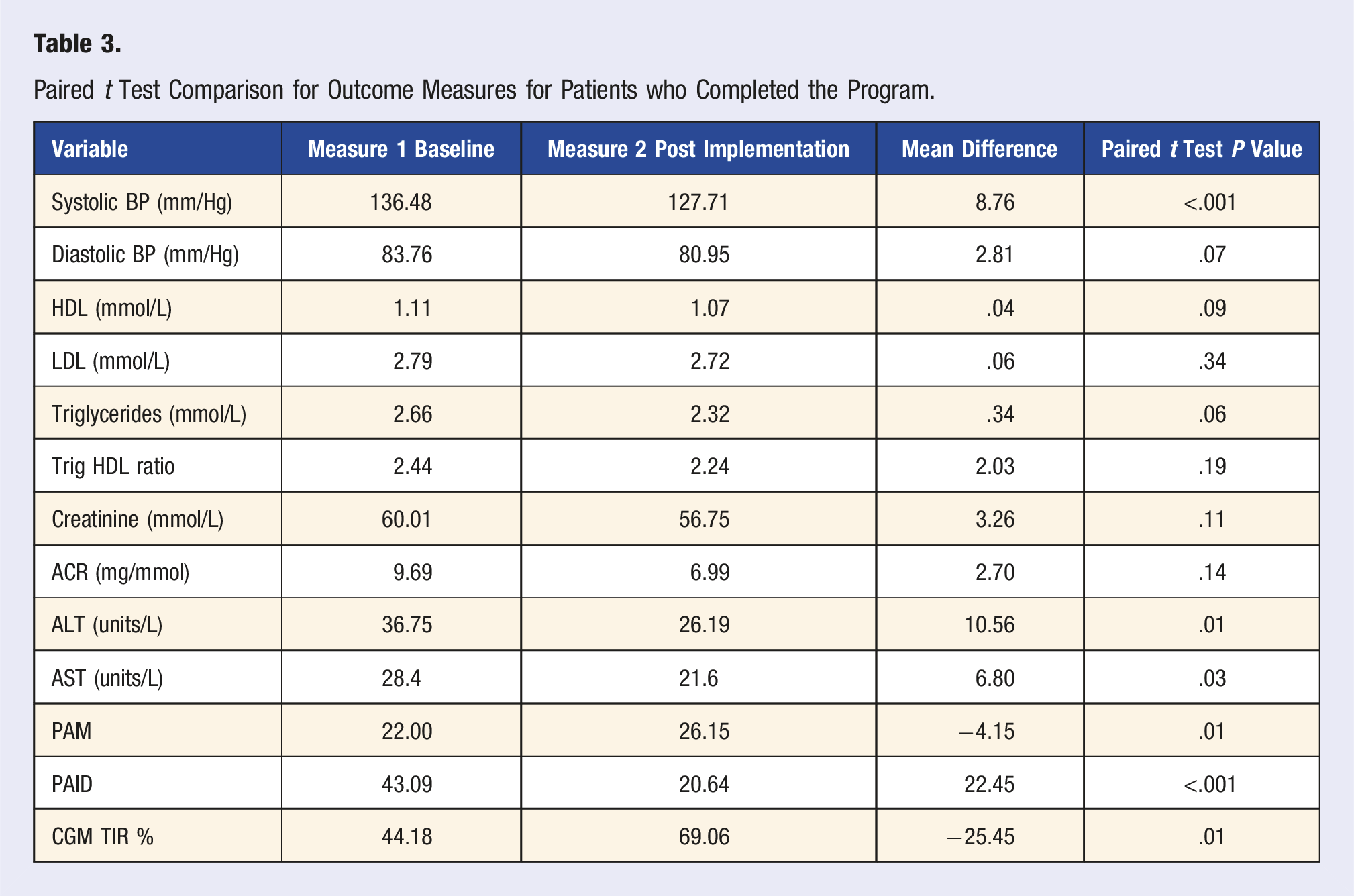
Paired t Test Comparison for Outcome Measures for Patients who Completed the Program.
Percentage of time-in-range improved by 25.45% (P = .011). Six participants achieved TIR >90% by week 8 of the program, where there were none achieving 90% in week 1.
Comparison of Completing Participants With Non-completing Participants for HbA1C Results Baseline (Measure 1), and 12 Months Post-Completion (Measure 3).

Chart 4: Provides a visual representation of the trend for those who completed the program, towards lowering their HbA1c and for those who did not complete the program, towards increasing their HbA1c. A Comparison of Program Completers and Those Who Did Not Complete for HbA1c % at Baseline and Post Program Plus 12 Months.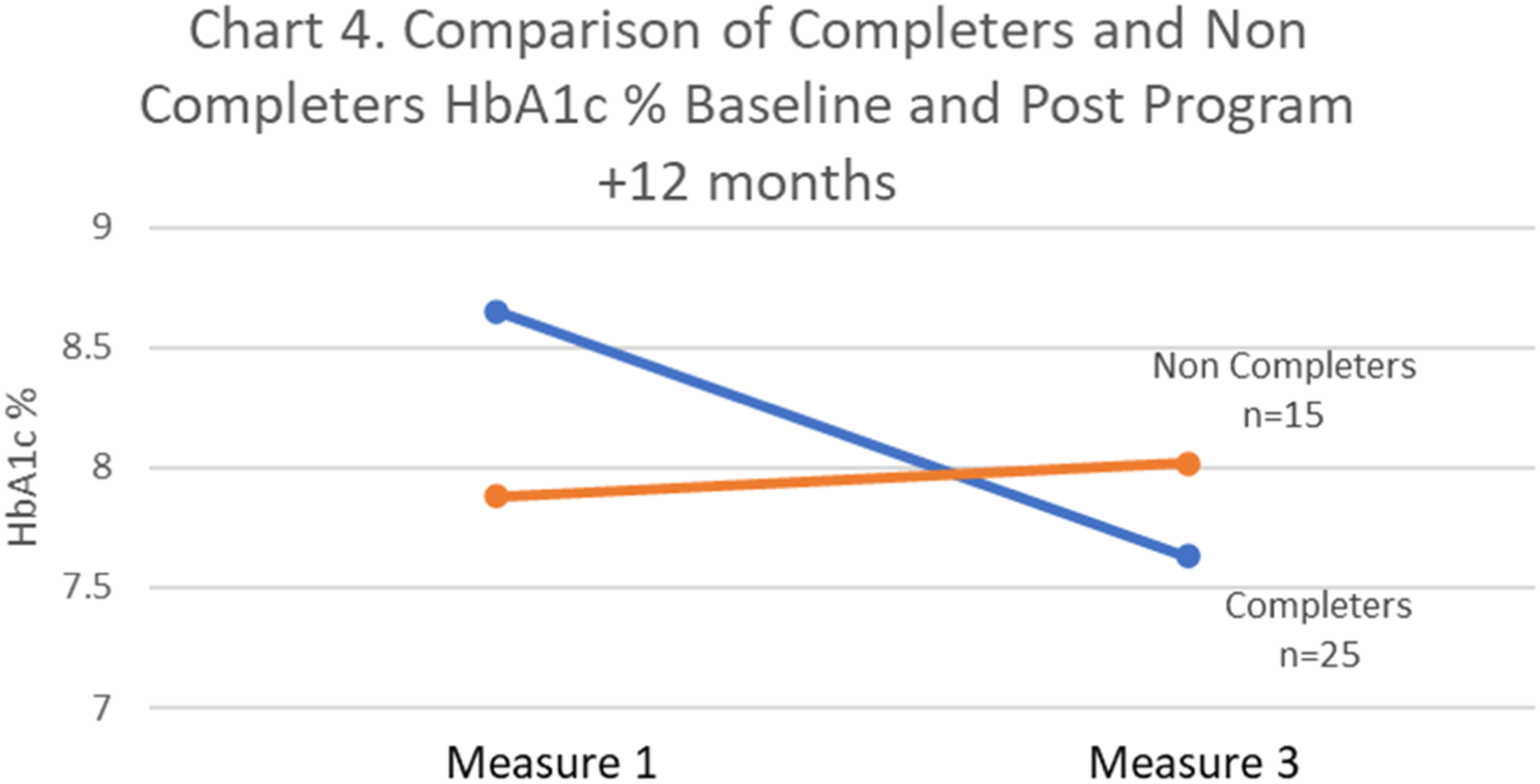
Program Evaluation
The program was evaluated using a co-designed questionnaire that was administered after the completion of the last pSMA session. Only those who completed the program therefore, are represented in the analysis of this questionnaire.
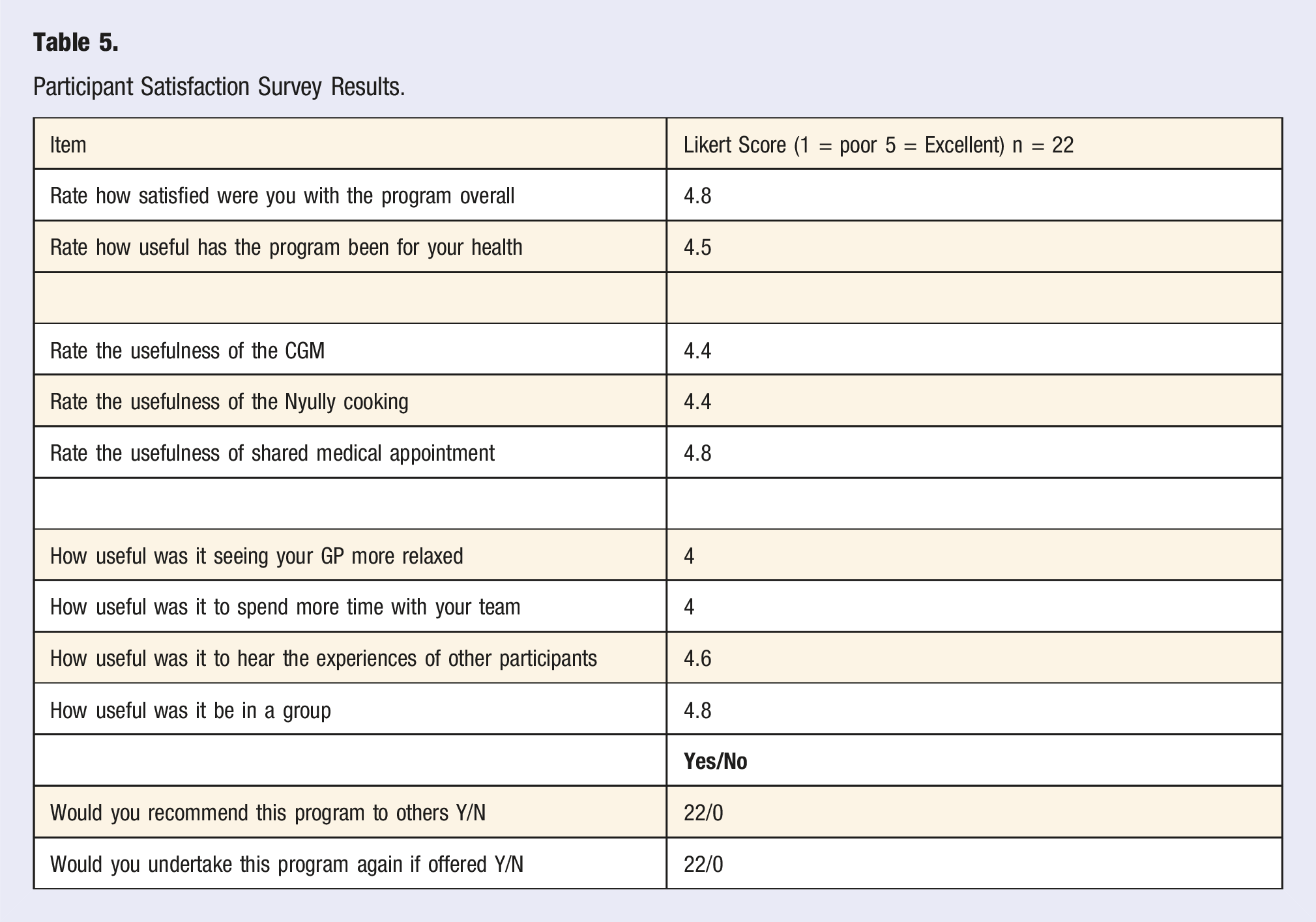
Participant Satisfaction Survey Results.
A Representative Sample of Comments From The Satisfaction Survey.

Discussion
This feasibility study addressed its hypothesis by demonstrating that a First Nations led, culturally safe program that included advanced technology can be successfully combined with contemporary practices leading to sustainable improvements in T2DM. The DRAW program included the use of a continuous glucose monitor, a curriculum of T2DM self-management within shared medical appointment sessions and a series of food appreciation workshops with the aim of influencing patient engagement, activation and self-management as well as improving glycaemic control and metabolic health outcomes.
These data did show that patient activation via the PAM tool, increased significantly between measures (P = .01), indicating improved patient engagement in behaviour change. The literature reports a correlation between increased PAM scores, improved health outcomes, and decreased health costs. 35 The satisfaction scores overall for those who completed the DRAW program were 4.8/5 and together with the qualitative comments indicated that the program was culturally safe, self-determining, as well as fun, informative and outcome producing. This also indicated that participant engagement was high.
There was also a statistically significant decrease in the PAID tool scores (P < .001) that indicated less psychological distress associated with living with T2DM. This change is linked to improvement in confidence in the self-management of T2DM and was also reflected in the qualitative comments (Table 6) indicating improved confidence and self-management. 36
Metabolic Outcomes
Through the positive engagement encouraged by the CGM driving self-determination, Nyully cooking and peer support in the SMA’s this study also showed significant improvement in metabolic health outcomes.
Analysis of the primary outcome measure (HbA1c) show that completing participants significantly lowered their HbA1c between baseline (Measure 1) and program completion (Measure 2) by .71% (P = .01). Seven (28%) achieved remission by lowering their HbA1c levels to below 6.4% (46 mmol/mol) and two of these participants (8%) decreased to below the prediabetes level of < .5.5%. (37 mmol/mol).
Though there was not a statistically significant change between baseline (Measure 1) and 12 months after completing the program (Measure 3) there was a mean HbA1c decrease .42% (P = .16). The seven participants achieving remission at Measure 2 remained in remission 12 months after the program was completed (Measure 3).
This data shows that eighteen (75%) participants who completed the program maintained some reduction in their HbA1c levels between baseline (Measure 1), program completion (Measure 2) and 12 months after completing the program (Measure 3). This compares with the fifteen participants who were not able to complete the program, where there was an overall mean increase in HbA1c from baseline of .4%. This is important as the literature reports that each 1% reduction in mean HbA(1c) is associated with reductions in risk of 21% for any end point related to diabetes. 37 Though this measure was not statistically significant the difference in the trends suggest a fruitful avenue for future well designed controlled trials. The Ambulatory Glucose Profile reports showed improved glucose variability as measured by HBA1c, noted above, and mean percentage of time-in-range which increased statistically significantly between measures by 25.45% (P = .011).
As additional indicators of metabolic health, data analysis of secondary outcome measures found statistically significant changes in weight with a mean reduction of 3.84 kgs (P < .001) between baseline (Measure 1) and on program completion (Measure 2). Statistically significant weight loss of 3.2 kgs (P = .007) was sustained for at least 12 months by the cohort of participants who completed the program.
Table 3 shows that between baseline (Measure 1) and program completion (Measure 2), there were statistically significant changes (improvement) in; systolic blood pressure, 8.76 mm/hg (P = .001), liver enzymes ALT (P = .012) and AST (P = .026). All other blood panel measures changed in a favourable direction for improved health, though not statistically significantly.
Satisfaction and Feasibility
The score of 4.8 out of 5 on the satisfaction survey (as noted above) indicated that the completing participants were satisfied with the program overall and suggested with a score of 4.5 out of 5 that they perceived that it assisted them to improve their health. Each element of the program was positively reviewed by the participants. In line with findings from other studies29,30 participants reported positively on their experience of using the CGM. The CGM system provided the participants with an understanding of their glucose responses to different foods, stressors, sleep and activities, empowering them to make personally relevant changes to their lifestyles and behaviours.
The shared real time CGM biofeedback also allowed the Waminda service team to access data remotely to provide health risk management in real time as well as incidental coaching along the way. The real time data provided evidence to the CDE and GP allowing for safe and timely titrating and deprescribing in response to lowering BGLs.
As supported by the literature 30 the CGM appears to be a powerful behaviour change tool enabling participants to make lifestyle changes that improve blood glucose and other measures of metabolic health.
As this appears to be the first study of its kind examining the feasibility of CGM use with First Nations people, there is no other literature to compare with. The First Nations women in this study, one as old as 78 years old, expressed confidence in wearing of the CGM as well as managing the data systems and devices. This was preferred over fingerstick testing across all participants.
The ease of access to their BGL data resulted in increased interaction with it, and improved understanding of their glucose excursions. The ease and regularity of reading their own data re-enforced the learning they gained from other elements of the program. The participants could see and measure the immediate effects of their food choices which nudged them towards adopting the principles of the Nyully food program. The CGM data were also used as a responsive learning tool in the SMAs to reflect on and reinforce behaviour that improved BGL.
The Nyully food program was evaluated highly with an overall rating of 4.4 out of 5. Other diabetes remission programs involving a nutrition component have demonstrated success.12,38 Nyully food did not have a specific macronutrient or calorie restriction objective, rather it privileged a framework of existing First Nation People’s cultural knowledge about their local food and community cooking. Together with the use of the CGM this resulted in food choices by the participants that aided a reduction in glucose variability (GV) in the short term with improved percentage of TIR. Long term HbA1c levels and other determinants of metabolic health such as weight, blood pressure and liver function were also improved.
Some of the reasons for the high satisfaction scores on the 5-point Likert scale included: seeing the GP more relaxed (4), spending more time with their health team (4), hearing the experiences of others (4.6) and undertaking these activities in a group (4.8). The qualitative responses indicated that the SMA component of the program provided a supportive, culturally safe and responsive environment for the participants to share and develop their knowledge and self-management skills for T2DM and the associated co-morbidities. These are similar to findings reported on by previous research.13,21 It would seem that Shared Medical Appointments can be considered for use as a First Nations model of Care.22,25
Limitations
The relatively small numbers of participants, together with the decolonising research framework requested by the community did not allow for a randomised controlled trial, which surrendered the studies statistical power to confidently measure more generalisable outcomes.
This was a single centre study and without randomisation of CGM, Nyully Food, SMAs and a standard care control group, so these findings cannot be generalised. In addition most of the participants had previous experience with a health and well-being program ‘Dead or Deadly’ previously run by the Service and staff. Though the Dead or Deadly program was neither a pSMA nor did it focus on T2DM and its management. This recruitment strategy had the potential of biasing the feasibility outcome of this DRAW project.
The difference in trends between participants who completed the program (and showed a trend towards lowering of HbA1c measures) and those who did not complete the program (and showed a trend towards increasing HbA1c) may provide an incentive to find a path to undertake more robust co-designed clinical trials. Given the increasing rates of T2DM among First Nations people further research examining ways to empower communities to self-manage, using tools like CGMs and SMAs while acknowledging existing local knowledge and science through co-designed approaches would likely be effective and informative to managing, and even reversing T2DM.
The study was limited to exploring the feasibility of the DRAW program among just one of the hundreds of First Nations cultural groups in Australia. The feasibility of the DRAW program among other groups is unknown and could prove to be a fruitful area of further research.
Conclusion
This feasibility study is the first study known to report on outcomes from the use of CGM coupled with a lifestyle medicine approach in managing T2DM in First Nations people. The co-designed DRAW program resulted in high levels of engagement and satisfaction of patients as well as improved health outcomes.
These data show that patient activation via the PAM tool, increased significantly between measures (P = .012), indicating improved patient engagement in the behavioural change activities. There was also a statistically significant decrease in the PAID tool scores that indicated less psychological distress associated with living with T2DM and improved confidence in self-management.
Before and after program measures also showed a statistically significant mean improvement in many of the metabolic measures including the HbA1c of .71% (P = .01), weight 3.8 kg (P < .001), systolic blood pressure reduction of 8.79 mm/Hg (P < .001) ALT reduction of 10.56 u/L (P = .01), AST reduction 6.8 u/L (P = .03) and BGL Time-in-Range increased by 22.45% (P = .01).
Seven of twenty-five (28%) completing participants in this feasibility study were in remission by the end of the program, with four no longer requiring insulin or other T2DM-related medications. These changes were maintained for at least 12 months after completing the program.
In addition, eighteen of the twenty-five completing participants reduced their HbA1c after the program and maintained a reduction for 12 months. Wearing a CGM whilst engaging in an eight-week lifestyle focussed program indicates an exciting potential use of Lifestyle Medicine and technology among First Nations people with diabetes.
This study reveals the feasibility for positive health outcomes when projects are First Nations led and include a genuine partnership approach with communities and health providers. When First Nations knowledge is prioritised, and respectfully integrated with technology and culturally safe health service delivery, favourable self-determining outcomes can be the result.
Footnotes
Acknowledgements
To the Country and people where this research was undertaken: We acknowledge the custodians of Wandi Wandandian, Jerrinja, Cullunghutti, Wodi Wodi from the 13 clans, where our mountains and rivers meet the sea. Honouring and paying respects to our Elders & Ancestors who have walked this country since the creation of time. The authors and researchers wish to acknowledge the generosity and the spirit of the 40 participants of the DRAW project and their families who have chosen not to be named. Waminda – South Coast Women’s Aboriginal Health And Well-Being Organisation, the Australasian Society of Lifestyle Medicine (ASLM) the Aboriginal Health and Medical Research Council Ethics Committee, Southern Cross University. The Commonwealth Government of Australia for the IAHP grant (
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Commonwealth Government of Australia; GO-4300 - 4-G1A1Z8O.
