Abstract
Hormonal disorders like PCOS (Polycystic Ovary Syndrome), autoimmune thyroid disease (AITD) including Hashimoto’s thyroiditis, male hypogonadism are commonly encountered in clinical practice in the US and worldwide, with rising frequency. These typically affect patients during young or middle age, compared with other common chronic illnesses like type 2 diabetes, hypertension, atherosclerotic cardiovascular disease, where onset may usually be in middle or older age. Multiple studies point to the role of disordered lifestyle health behaviors as contributory to these endocrinopathies, and conversely therapeutic lifestyle changes leading to improvement in signs, symptoms, biochemical markers, and sequelae of these conditions. This article presents 3 different real life case studies of the conditions enlisted above and documents the positive impact of lifestyle improvements on their disease condition. Therapeutic lifestyle behaviors are an extremely useful and important component of management of these familiar endocrinologic disorders, and clinicians need to routinely counsel their patients about healthy lifestyle interventions when treating these common syndromes.
Keywords
“Opioids inhibit hypothalamic GnRH secretion, disrupting its normal pulsatility and leading to decreased LH levels; and therefore, testosterone levels decrease.”
Case 1
PCOS (Polycystic Ovary Syndrome)
When she started seeing us, she was counseled about therapeutic lifestyle changes. She started minimizing ultra-processed food (UPF) and stopped consuming it daily (eating those only on very special occasions like a birthday) as well as moving for 30 minutes daily without any exception. Initially, she began physical activity at moderate intensity, but then as she developed more self-efficacy, she titrated the exercise intensity and now consistently does high intensity interval sessions for 30 minutes daily. At age 32 years old (a little over 1 year after initiating lifestyle therapy), she was able to spontaneously conceive and went on to have a healthy full-term pregnancy without any complications, notably no gestational diabetes mellitus or preeclampsia. She continued metformin therapy during the pregnancy but had stopped both spironolactone and finasteride several months before her pregnancy. She delivered a healthy child vaginally. She did not restart the spironolactone or finasteride post-partum. She breastfed her infant and was able to lose pregnancy weight successfully.
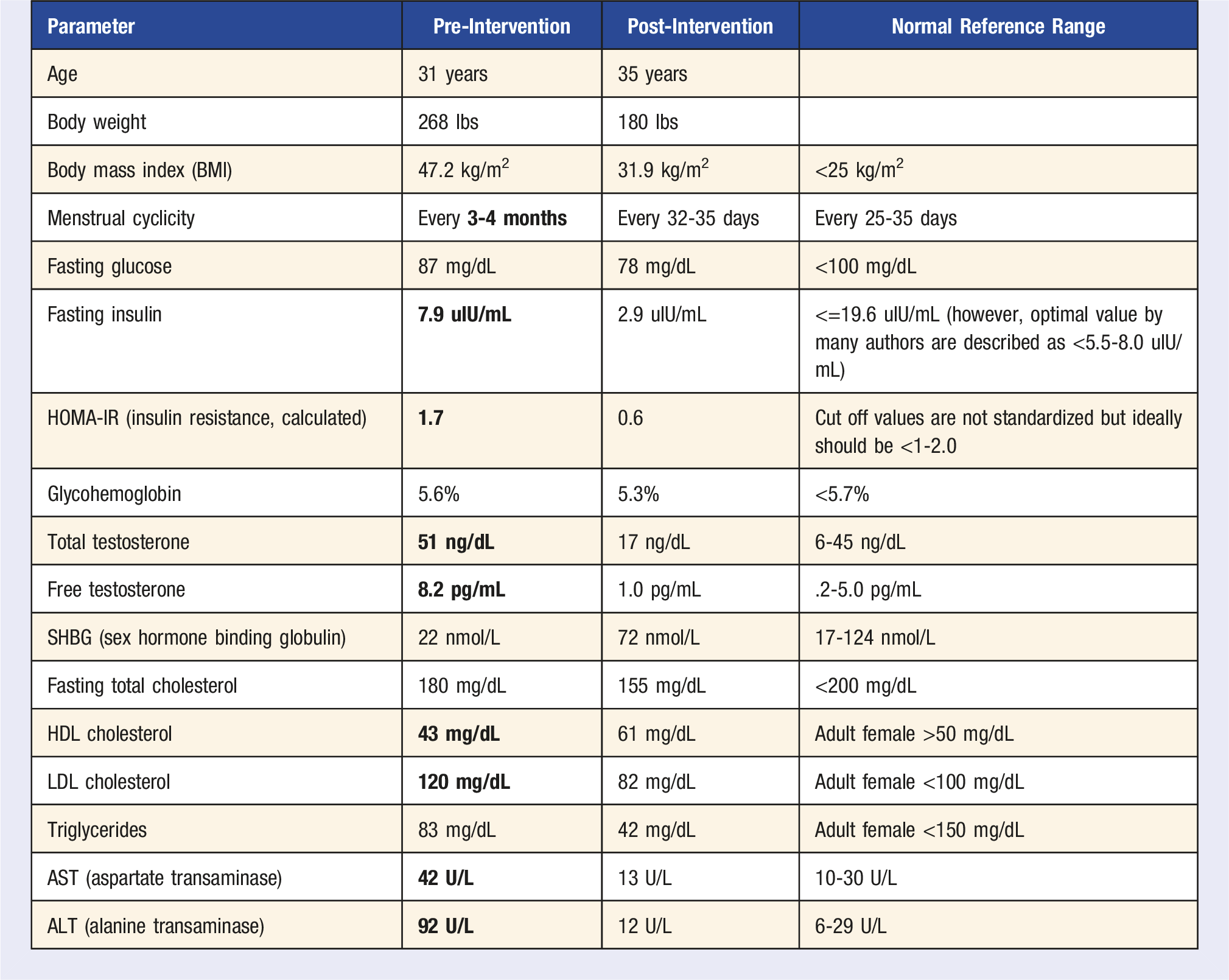
Now at age 35 years old, she weighs 180 pounds with a BMI of 31.9. She has now been menstruating regularly every 32-35 days. She has continued her metformin at the same dose. Her fasting glucose now is 78 mg/dL (previously was 87 mg/dL), fasting insulin 2.9 uIU/mL (previously was higher at 7.9 uIU/mL), calculated HOMA-IR score now .6 (improved from her baseline 1.7) and hemoglobin A1c now improved at 5.3% (baseline was 5.6%). Her androgen parameters now are improved at: total testosterone: 17 ng/dL (this was previously high at 51 ng/dL), free testosterone: now markedly improved at 1.0 pg/mL (previously was high at 8.2 pg/mL), and SHBG now has risen to 72 nmol/L (previously was low normal at 22 nmol/L). Her lipid parameters have also improved: fasting total cholesterol now 155 mg/dL from 180 mg/dL, HDL cholesterol went up to 61 mg/dL from 43 mg/dL previously, LDL cholesterol decreased to 82 mg/dL from 120 mg/dL, triglycerides decreased to 42 mg/dL from 83 mg/dL. Liver enzymes also normalized with AST now at 13 U/L from her prior 42; ALT now 12 U/L from 92 U/L. Due to the decrease in her free testosterone, she is no longer needing the pharmacotherapy for hirsutism and has been able to cut back on the frequency of her LASER treatments significantly.
Case Discussion
PCOS is the most common endocrine disorder in women of reproductive age, with prevalence of 7-13% in the US (an estimated number of 5 to 6 million women). However, some ethnicities may have a higher prevalence approaching almost 20%, particularly south Asian and Pacific Islander 1 who may have a twofold elevated risk for PCOS compared with non-Hispanic whites for the same BMI. The prevalence of PCOS among non-overweight women (BMI <25) is estimated to be 4%, whereas prevalence may be as high as 15-18% among women with BMI >=30. Unfortunately, PCOS remains undiagnosed in 70% of the cases globally. 2 The central diagnostic features of polycystic ovary syndrome (PCOS) typically involve two of the following 3 criteria (Rotterdam criteria 3 ): (i) biochemical hyperandrogenemia (elevated total and/or free testosterone on blood testing) or clinical hyperandrogenism (hirsutism/nodulocystic acne/male pattern balding), (ii) oligoanovulation (resulting in oligomenorrhea), and (iii) polycystic ovaries on ultrasonography.
Impact of Six Pillars of Lifestyle Medicine
Weight and lifestyle (diet, physical activity and behavioral) management are a cornerstone of all therapy in international evidence-based guidelines for PCOS. 4 This meta-analysis 5 of 19 trials (1193 PCOS patients) found several different diets, for example, DASH (Dietary Approaches to Stop Hypertension) diet, calorie restricted (CR) diet, Mediterranean diet, low fat diet, etc. to be significantly related to improvements in HOMA-IR score, fasting insulin, fasting plasma glucose, BMI, body weight, and waist circumference. The longer the duration of adherence to the dietary patterns, the greater the improvement was. Compared with metformin, diet was associated with more weight loss (including both weight/BMI and waist circumference). A review 6 found that vigorous aerobic exercise in PCOS patients improved HOMA-IR, waist circumference (WC), and cardiorespiratory fitness (VO2max). Another review 7 found that resistance exercise reduced body fat, WC and increased lean mass. Sleep disturbances seem to be bidirectionally associated with PCOS. 8 A cross-sectional study 9 of 724 women from Adelaide, South Australia found that PCOS was associated with increasing occurrence of difficulty falling asleep (odds ratio (OR) 1.94, 95% confidence interval (CI) 1.28-2.95); this association was attenuated but still statistically significant after accounting for BMI and depressive symptoms. A prospective cohort study 10 from Taiwan National Health Insurance Research Database found that women with PCOS were associated with an increased risk of developing obstructive sleep apnea (OSA) in later life (hazard ratio: 2.63, 95% CI 1.57-4.04) compared with controls. In this study, 11 serum triglyceride levels were higher in women with PCOS who smoked compared to non-smokers, even when adjusted for BMI. Metabolic syndrome was more common in smokers with PCOS compared to non-smokers with PCOS and smokers who were controls (28.6 vs 3.6%; P = .02). Even rare alcohol consumption (<1 drink/week) in PCOS patients was independently associated with histologic non-alcoholic steatohepatitis (NASH) in 1 retrospective study. 12 In vitro and animal studies 13 show that marijuana may induce disturbance of ovulatory function and menstrual cycle. Similarly, in experimentally treated animals, cocaine caused impaired menstrual cyclicity and increased rate of anovulation. Chronic heroin use is associated with high rates of hypothalamic amenorrhea. Use of these drugs in women with PCOS may further impair ovulatory function. A cross-sectional study 14 from Bangladesh used UCLA loneliness (UCLA-3) scale for psychometric assessment and found an overall loneliness prevalence of 71% among patients with PCOS, with 39%, 18%, and 14% reporting mild, moderate, and severe loneliness, respectively.
Case 2
Hashimoto’s Thyroiditis (Autoimmune Hypothyroidism)
Patient Y started seeing us initially at age 53 years old with a chief complaint of “having a hard time losing weight.” 2 years prior to presentation, her peak body weight was 245 pounds, but through effort (going to the gym 60-90 min daily), she had been able to go down to 206 pounds. However, lately it had crept up to 225 pounds. She had been diagnosed with mild autoimmune subclinical hypothyroidism (thyroid peroxidase (TPO) and thyroglobulin (Tg) antibodies positive) 2 years prior to presentation and had been started on levothyroxine 50 mcg orally once daily. At the time of presentation, she had been taking the levothyroxine consistently and appropriately every day and her thyroid stimulating hormone (TSH) was within normal limits at 1.969 mIU/L (normal reference range: .4-4.5 mIU/L). At the onset of Hashimoto’s thyroiditis 2 years prior, she also developed severe disabling pain in multiple joints, which prevented her from doing as much activity as she previously enjoyed. She used to be able to walk 10 miles a day, whereas now she was barely able to walk 3 miles a day. She sought rheumatology opinion 5 months prior for her symptoms. Her blood biomarkers for various autoimmune syndromes were found to be negative, including rheumatoid factor, cyclic citrullinated peptide antibody, uric acid, Lyme’s serology, etc. In addition, she sought orthopedics consult who recommended physical therapy (PT) but patient did not have adequate pain relief from doing PT.
Immediately prior to her presentation with us, the patient searched the internet and decided to cut out gluten and dairy from her diet, to help her autoimmune thyroid condition as well as pain in joints. She also started Weight Watchers program to help her lose weight and completely cut out refined carbohydrates like sugary beverages, desserts, white bread, etc. When she saw our lifestyle endocrinology clinic, we reinforced the importance of diet and the other pillars to her and encouraged her to keep up with the recent changes she had started making. Within a few weeks of dietary intervention, her joints stopped hurting as much. She was able to increase her exercise and started going to the gym daily after work for strength training for at least 30-45 minutes followed by 15 minutes of vigorous intensity aerobic exercise (elliptical).
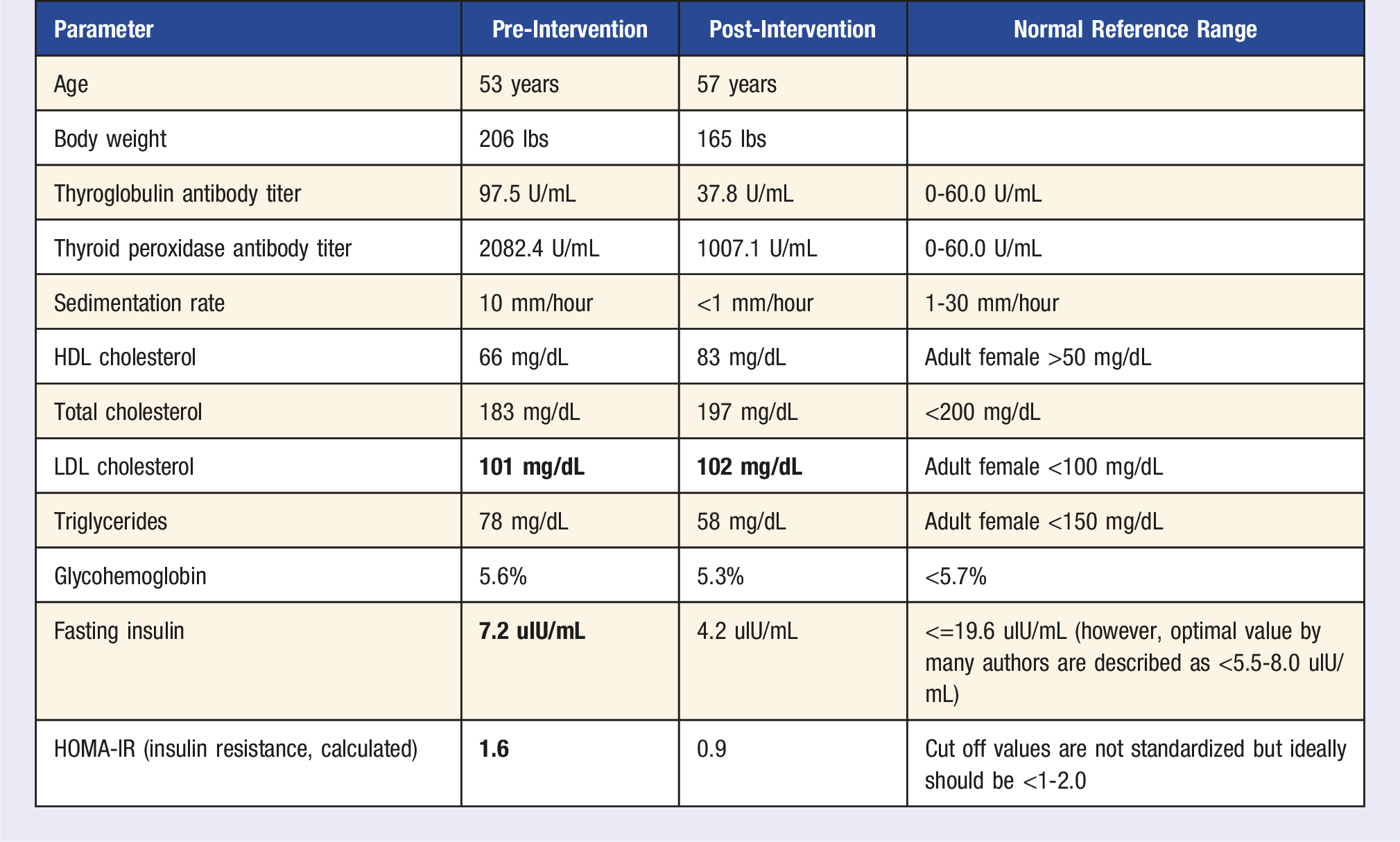
Gradually, over a period of 40 months, the patient has now been able to lose 60 pounds from her initial weight of 225 pounds at presentation to 165 pounds now at age 57 years old, superseding her target body weight of 180 pounds. Importantly, her thyroid antibody titers improved. Her thyroglobulin antibody which was 219.2 U/mL at the time of presentation went down to 37.8 U/mL (normal reference range: 0-60 U/mL) and her thyroid peroxidase antibodies went from 2082.4 U/mL to 1007.1 U/mL (normal reference range: 0-60 U/mL). Her erythrocyte sedimentation rate decreased from 10 mm/hr at presentation to <1 mm/hr (normal reference range: 1-30 mm/hr).
Other Beneficial Effects
Case Discussion
Autoimmune thyroid disease (AITD), commonly presenting as the eponymous Hashimoto’s thyroiditis (HT), is one of the most common autoimmune disorders in humans. The incidence of HT is estimated to be .3-1.5 cases per 1000 people, with female to male predominance of 7-10:1. 15 Its prevalence rises with increasing age, 16 especially in patients diagnosed with other autoimmune conditions, such as pernicious anemia, celiac disease, type 1 diabetes, vitiligo. HT typically presents clinically with hypothyroidism, when thyroid hormone (tetraiodothyronine or T4, triiodothyronine or T3) production by the affected thyroid gland is insufficient due to thyroid follicular/parenchymal damage +/− fibrosis. However, in many cases it can present with euthyroidism when preserved thyroid cells compensate for destroyed thyrocytes and serum T4, T3, Thyroid Stimulating Hormone (TSH) levels remain within normal limits; or rarely thyrotoxicosis (Hashitoxicosis) that typically tends to be short-lived (1-2 months duration usually) and is characterized by release of pre-formed thyroid hormones into the bloodstream due to thyroid follicular inflammation and damage. Pharmacological treatment of autoimmune hypothyroidism with oral levothyroxine replacement therapy is well-studied. However, what is less well-studied is the impact of different pillars of LM on signs and symptoms of HT.
Impact of Six Pillars of Lifestyle Medicine
Multiple observational studies have linked the intake of lower amounts of vegetables, fruit, fiber, poly- and mono-unsaturated fatty acids, protein with higher risk for Hashimoto’s thyroiditis. In this Italian case-control study 17 of 200 subjects (mostly young women), HT subjects reported higher intake frequencies of animal foods (meat, P = .0001; fish, P = .0001; dairy products, P = .004) compared with controls, who reported higher intake frequencies of plant foods (legumes, P = .001; fruits and vegetables, P = .030; nuts, P = .0005). In logistic regression analysis, meat consumption was associated with increased odds ratio of developing thyroid autoimmunity, while Mediterranean diet traits were protective. Another South Croatian case-control study 18 enrolled 1887 participants and evaluated food intake using a food frequency questionnaire containing 58 food items. Using logistic regression analysis, they found that frequent consumption of animal fats and butter was associated with positive plasma thyroid peroxidase antibody and/or thyroglobulin antibody (P = .01), whereas frequent consumption of vegetables, dried fruit, nuts, muesli were associated with negative findings of thyroid peroxidase antibody and/or thyroglobulin antibody (P = .048 and P = .02, respectively). Pro-inflammatory foods (ultra-processed foods, e.g., deli meats, bologna, ham, white bread/flour, added sugar, high saturated fat foods like processed cheese, deep-fried foods, etc.) are associated with gut dysbiosis, “leaky gut” (increased intestinal permeability) allowing antigens to pass more easily and activate the immune system or cross-react with extraintestinal tissues, respectively. 19 There is accumulating data that a strong “reciprocal” thyroid–gut axis exists. 19 Gut microbiota also influences the absorption of minerals that are important to the thyroid, including iodine, selenium, zinc, and iron. HT patients also frequently inquire about the role of restricting dairy, gluten, soy, etc. from their diets. This meta-analysis of 4 observational studies 20 with a total of 87 HT patients (all female), with mean period of gluten-free diet (GFD) of 5.5 months showed statistically significant reduction in TSH and increase in free thyroxine (FT4) levels. Out of the 87 HT patients, 47 had gluten-related conditions (GRC) (i.e., non-celiac gluten/wheat sensitivity—NCGS; incidental finding of positive anti-tissue transglutaminase antibodies—anti-tTG—without clinical symptoms or histological confirmation of Celiac disease). In this subset of HT patients with GRC, there was statistically significant reduction in TPO and Tg antibody titers. In terms of dairy, these authors 21 performed lactose load test (LLT) and found that lactose intolerance was present in 75.9 % of the patients with HT. They also tested an 8-week lactose-free diet in the group of patients with HT and lactose intolerance and found that the TSH value decreased significantly (P < .05) compared with TSH pre-intervention. However, in the group of lactose-tolerant patients with HT, the results before and after were similar (P > .05). The authors suggested that, in the case of a high dose of levothyroxine used by HT patients or resistance to its treatment and difficulty in regulating TSH, it is worth considering testing for lactose intolerance. Similarly, another group of authors 22 suggested that “it may be worthwhile to pursue hydrogen breath testing in patients with Hashimoto’s thyroiditis with significant gastrointestinal symptoms given the overall frequency of the condition.” For soy, this systematic review and meta-analysis 23 of 18 RCTs including soy as an intervention showed no significant changes in FT3, FT4 while an elevation in TSH levels was observed (weighted mean difference (WMD): .248 mIU/L, P = .049) with soy supplementation. The authors concluded that “soy supplementation has no effect on the thyroid hormones and only very modestly raises TSH levels, the clinical significance, if any, of the rise in TSH is unclear.” Similarly, diets poor in micronutrients like iron, copper, iodine, can directly affect thyroid health by decreasing T4 and T3 synthesis. Selenium and zinc are required for converting T4 into T3. Studies showed that having either hypothyroidism or hyperthyroidism can increase the symptoms of pre-existing Restless Legs Syndrome in patients. 24 Similarly, there are inconsistent findings that hypothyroidism (even subclinical) may contribute to (not cause) OSA. Patients who suffer from thyroid dysfunction tend to have higher rates of insomnia and sleep deprivation (may come from symptoms like muscle aches/cold or heat intolerance, anxiety, pain in multiple joints etc.). A Korean study 25 (KNHANES) published in 2019 showed that chronic sleep deprivation or excess sleep was associated with subclinical thyroid dysfunction (hyper and hypothyroidism). This study 26 used the U.S. National Health and Nutrition Examination Survey (NHANES) to investigate the relationship between daily physical activity and both thyroid and immune activity. The authors assessed daily physical activity through accelerometry or accelerometry-validated survey responses. Active adults had a reduced slope of the TSH:T4 relationship. Similarly, greater physical activity was associated with lower CRP levels, and lower white blood cell, basophil, monocyte, neutrophil, and eosinophil (but not lymphocyte) counts. Daily physical activity was also associated with lower prevalence of clinically elevated CRP, WBC, and lymphocytes in a dose-response manner. An RCT from Greece, 27 on 60 women with Hashimoto’s thyroiditis, aged 25-76 years, assigned 30 women each to intervention (stress reduction techniques) or control. The stress reduction intervention comprised 8 weeks of successive teaching of techniques like diaphragmatic breathing, progressive relaxation, cognitive reconstruction, guided imagery. Patients in the intervention group demonstrated statistically significant decrease in the rate change of anti-TG titers. A Saudi Arabian cross-sectional study 28 of 231 patients (mean age: 43 years, >90% female) with long standing hypothyroidism (>18 years duration), used phone interviews with the validated online revised University of California, Los Angeles (R-UCLA) Questionnaire, to screen for loneliness and found that 2.2% and 16% of the participants reported a high and moderately high degree of loneliness, respectively. Unfortunately, this study did not have a control group. The hypothalamo-pituitary axis, (through dopamine and serotonin), is intricately linked with the development and continuation of dependence of all chemical substances, 29 and therefore it is not surprising to find that most substances of abuse in return have multiple effects on the hypothalamo-pituitary-thyroid (HPT) axis and thyroid function. Alcohol has been reported to cause direct suppression of thyroid function by cellular toxicity, and indirect suppression by blunting thyrotropin-releasing hormone (TRH) response. 30 There is evidence for moderate suppression of peripheral serum thyroxine (T4) levels with more significant suppression of triiodothyronine (T3) levels in chronic alcohol users. Peripheral thyroid hormones are suppressed during withdrawal and the degree of suppression of their levels has been associated with the severity of withdrawal. This phenomenon is clinically interesting as thyroid suppression may accentuate the withdrawal dysphoria, thereby increasing the relapse risk in alcoholism. 30 This study 31 used the thyroid hormone and urinary cotinine dataset from sixth Korean National Health and Nutrition Examination Survey in 2014 and 2015, and a total of 4249 participants were included. Urinary cotinine level was negatively correlated with TSH in both sexes; and TPO Ab titer was positively correlated with cotinine levels in males (P < .0001). The authors concluded “a significant dose-related effect of urinary cotinine level on thyroid function, and thyroid autoimmunity.” This fascinating case report 32 described a 56-year-old woman with chronic pain syndrome on opioids, who was treated for myxedema coma which was found to be from hypothalamic thyrotropin-releasing hormone (TRH) suppression from long term opioid use.
Case 3
Male Obesity Related Secondary Hypogonadism
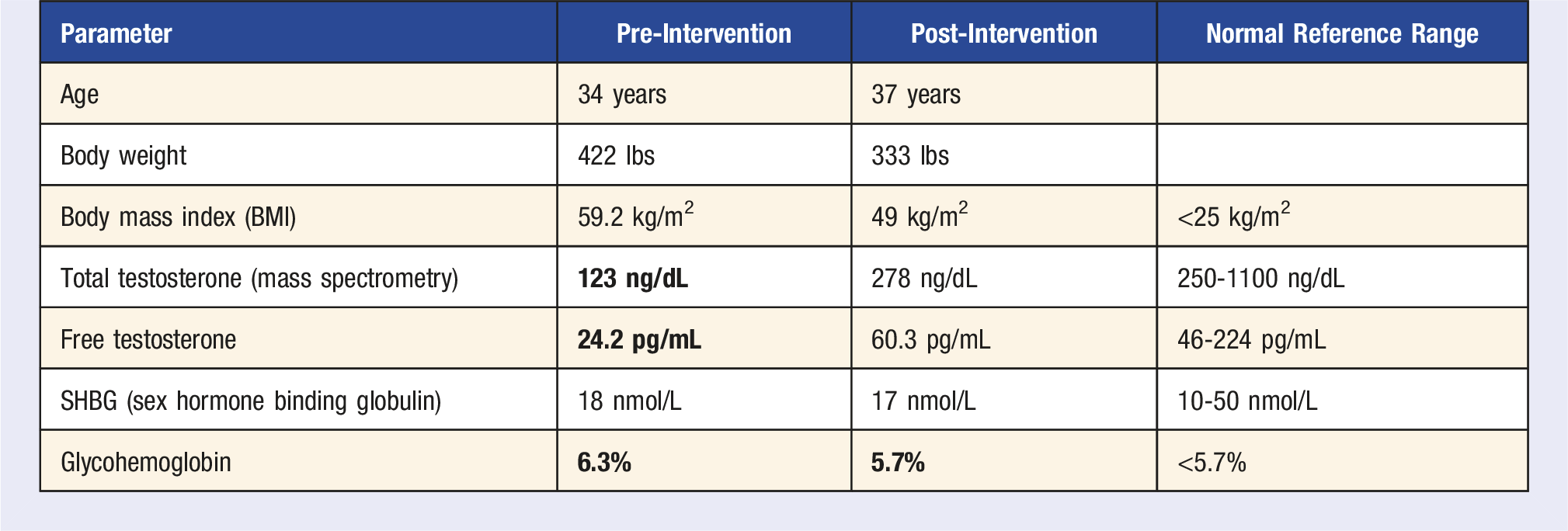
Patient Z was referred to us 2 years ago at age 35 years old when he was found to have a low fasting total testosterone level of 134 ng/dL (normal reference range: 250-1100 ng/dL) and repeat was 123 ng/dL. This was checked because he complained of erectile dysfunction and fatigue. His free testosterone levels were also quite low at 30.3 pg/mL and 24.2 pg/mL, respectively (normal reference range: 46-224 pg/mL). His follicle stimulating hormone (FSH) and luteinizing hormone (LH) levels were low normal. The rest of the workup for secondary hypogonadism revealed normal serum prolactin, DHEA sulfate (dehydroepiandrosterone sulfate), IGF-1 (Insulin like Growth Factor-1), and serum ferritin. His hemoglobin A1c was high at 6.3%, consistent with prediabetes. On physical exam, his body weight was 422 pounds and BMI: 59.2. His hypogonadism was attributed to obesity. Dietary history: He ate limited vegetables, no fruit, no legumes. He drank many servings of soda and at least a 6-pack beer daily. Exercise history: he lifts tires at a warehouse all day but did not have a formal exercise regimen outside of work. Sleep history: he suffered from obstructive sleep apnea and wore CPAP (Continuous Positive Airway Pressure) device consistently every night. In addition, he also suffered from gouty arthritis.
After the initial consultation with us, he was referred to the dietitian for education about dietary changes, which he slowly started making. He was also initiated on antiobesity pharmacotherapy with dulaglutide (off label, due to insurance formulary he was unable to get either semaglutide or liraglutide), which was first started at a low dose of .75 mg once a week and then gradually titrated up to 3 mg once a week. He worked well with dietetic advice and since he had a background of having trained at a culinary school, he was able to start working in some vegetables slowly onto his plate. He also started incorporating frozen fruit (did not like texture of fresh fruit). Most importantly, he started replacing the soft drinks and beer with flavored sparkling water. He has now cut out the alcohol completely and drinks only one can of regular soda every week. He is now minimizing UPF, particularly deep-fried foods. In addition, he has also started using elliptical for 30 min on weekends and remains active at work with tire lifting.
Case Discussion
Male obesity related secondary hypogonadism is a well-described syndrome. 33 Obesity is one of the most important risk factors for secondary hypogonadism in men. A meta-analysis of 18 studies (comprising 4546 men, aged 27.9 to 61.9 years, and BMI 24.8 to 50.3 kg/m2) found prevalence of hypogonadism very high at 50-80%, 34 indicating that obesity is more closely linked with low testosterone than aging. Conversely, intentional weight loss through either lifestyle or bariatric surgery can improve testosterone levels. 35 Testosterone deficiency in obese men is associated with reduced physical activity, sarcopenia, decreased BMD and visceral obesity. 33
Impact of Six Pillars of Lifestyle Medicine
A randomized controlled trial (RCT) from Italy 36 in relatively healthy obese men without diabetes, hypertension, or hyperlipidemia, who had erectile dysfunction, showed that intensive lifestyle modification achieved a 15% weight loss and improved the validated International Index of Erectile Function (IIEF) score by 3.1 (as a comparison, tadalafil improves this by 6.5). Out of the 55 men randomized to the intensive lifestyle intervention, 17 men in the intervention group reported an IIEF score of 22 or higher (as compared to only 3 in the control group {P = .001}), leading authors to conclude that “lifestyle changes are associated with improvement in sexual function in about one-third of obese men with erectile dysfunction at baseline.” A high consumption of bread and pastries, dairy products, and desserts, eating out, and a low intake of homemade foods, noodles, and dark green vegetables independently predicted hypogonadism 37 (odds ratio: 5.72; 95% confidence interval: 1.11-29.51, P < .05). This South Australian group 38 proposed the GELDING theory (Gut Endotoxin Leading to a Decline IN Gonadal function) and found that endotoxemia (circulating LPS-binding protein {LBP}) was negatively correlated with serum testosterone. In a second interventional study 39 from Germany, low-dose endotoxin challenge in lean men produced a transient inflammatory response that was followed by a decline in serum testosterone, without changes in LH or FSH, providing further evidence that endotoxin-driven inflammation may result in impaired Leydig cell function. This indicates the role of gut microbiota driving a pro-inflammatory state in decreasing gonadal testosterone production, a condition that can happen commonly from UPF-dominant diets. This study 40 enrolled a small sample of ten young, healthy males in their 20s and exposed them to 1 week of sleep restriction at 5 h per night, a condition experienced by at least 15% of the US working population. Daytime testosterone levels were decreased by 10% to 15% in this small sample. By comparison, normal aging is associated with a decrease of testosterone levels by 1% to 2% per year. This testosterone decline was associated with lower vigor scores. Substance and drug abuse is a recognized cause of possible alterations of steroidogenesis and spermatogenesis. Alcohol, opioids and anabolic-androgenic steroids tend to reduce a man’s testosterone production interfering with testicular and/or hypothalamic-pituitary function. 41 Other substances such as nicotine, cannabis, and amphetamines alter spermatogenesis inducing oxidative stress and subsequent apoptosis in testicular tissue. Both testosterone production and spermatogenesis seem to be affected by alcohol abuse in a dose-dependent manner: heavy drinkers are more likely to have a poor testicular function than moderate consumers. 41 Opioid-induced androgen deficiency (OPIAD) 42 is a recognized syndrome characterized by decreased levels of testosterone, reduced libido and muscle mass, fatigue, and osteopenia. Opioids inhibit hypothalamic GnRH secretion, disrupting its normal pulsatility and leading to decreased LH levels; and therefore, testosterone levels decrease. Exercise augments the effects of testosterone replacement therapy and the durability of response after cessation of treatment. 33
Conclusion
The above cases and literature review illustrate the importance of discussing lifestyle medicine pillars with patients in routine clinical practice who present with these extremely common hormonal conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
