Abstract
Musculoskeletal (MSK) pain is highly prevalent worldwide and is associated with high levels of disability. Systemic inflammation may contribute to MSK pain, and diet is a crucial modulator of systemic inflammation. The Dietary Inflammatory Index (DII®) assesses the tendency of diets to cause inflammation. Higher DII scores, indicating pro-inflammatory diets, were hypothesized to be associated with a higher prevalence of MSK pain symptoms and duration. Data from 4118 subjects from the 2003-2004 NHANES survey were included to assess the association between DII scores and the presence and duration of osteoarthritis symptoms and neck and low back pain. After adjusting for potential confounders, the multivariable analysis showed that DII scores were significantly associated (P < .05) with increased risk of osteoarthritis symptoms (joint pain, aching, and stiffness) in the past year (OR = 1.099; 95% CI 1.029,1.174), low back pain (OR = 1.070; 95% CI 1.003,1.142), and any pain lasting longer than 24 hours (OR = 1.128; 95% CI 1.046,1.216). Neck pain and pain duration were not significantly associated with DII scores. These findings suggest a significant association between a pro-inflammatory diet and the presence of different MSK pains (joint pain and low back pain) and with pain lasting longer than 24 hours.
“The overall results offer promising perspectives for exploring potential pain mechanisms in individuals suffering from musculoskeletal pain”.
Introduction
Evidence has been building on the association between musculoskeletal (MSK) pain and inflammation. 1 Specifically, MSK pain presence, severity, duration, and the transition between acute and chronic pain, have been associated with heightened inflammatory status. 2 Both localized (tissue-specific) and systemic inflammation (involving circulatory markers) have been shown to play a role in pain experiences in MSK pain. They have also been found to have a bidirectional interaction, though the mechanisms involved are not currently known.3-5
Diet is a known modulator of inflammation, as specific pro-inflammatory diets are associated with chronic systemic6,7 and tissue-specific long-standing (also referred to as simmering) inflammation.8,9 Overall, the evidence suggests a possible link between diet, inflammation, and musculoskeletal pain, though uncertainty exists on the strength, impact, and mechanisms involved in this interaction. 10
The most prevalent MSK disorders include osteoarthritis (OA), neck pain, and low back pain (LBP). 11 OA is a major source of disability and socioeconomic burden and one of the main contributors to chronic pain, which is associated with even more severely decreased function and quality of life. 12 OA is characterized by joint pain, stiffness, aching, degeneration, and reduced range of motion. Inflammation, both local and systemic, is one of the main contributors to the development and progression of this disease.13-17
LBP is the leading cause of disability worldwide, 18 with an estimated lifetime prevalence between 65% and 80%.19,20 Despite significant resources allocated to LBP research, there is still a lack of knowledge on properly preventing and treating this condition.21,22 An estimated 15% to 20% of the people who experience acute LBP transition into chronic low back pain (CLBP).23,24 CLBP has an even more significant burden on society by heavily affecting healthcare delivery systems and negatively impacting function and cost for individuals who suffer from it. 25 There is rapidly growing evidence of associations between inflammation and acute LBP, chronic LBP, and the transition between the 2.26-34
Neck pain is the fourth cause of disability worldwide.35,36 Though there has been attention to researching the contribution of inflammation to neck pain, the evidence has been largely inconclusive. A recent systematic review on systemic inflammatory cytokines and spinal pain concluded that CRP only was elevated in chronic whiplash disorders (moderate evidence). 37 The same review showed conflicting results for other cytokines. Another review showed no significantly elevated markers in acute neck pain and elevated IL-1β and TNFα when comparing chronic neck pain subjects vs controls. 38 Overall, neck pain remains a common musculoskeletal syndrome with significant impairments, especially when chronic. 39
The Dietary Inflammatory Index (DII®) is a widely used measure for assessing the inflammatory potential of diet.40-46 Diets vary significantly in their inflammatory and systemic impact on the body.7,47-50 For example, dietary patterns are associated with heightened inflammatory status51-53 and global genomic DNA methylation (a crucial epigenetic factor in regulating gene expression 54 ) in a cancer-free population.55,56 Moreover, each food has different properties regarding the inflammatory response elicited in the body, 57 and individual responses may vary, exemplified in lactose intolerance 58 and non-celiac gluten sensitivity. 59 Last, the quantity and frequency of each food’s consumption also play an essential role in the overall inflammatory effect.60-63 While the complex impact of diet on the human body is far from being understood, assessing the inflammatory tendencies of diets has provided encouraging results.64-66
The primary aim of this study was to investigate the association between MSK pain (particularly OA, neck pain, and LBP) and diet-related inflammation, assessed through the DII, in a representative sample of the US population. The secondary aim of this study was to assess whether subjects with a pro-inflammatory diet experienced MSK pain more frequently and for a longer time, thus predisposing them to chronicity. We hypothesized that higher DII scores are associated with higher MSK pain prevalence and duration.
Methods
Study Design and Participants
The 2003-2004 National Health and Nutrition Examination Survey (NHANES) dataset is the only large dataset (and only NHANES cycle) that had collected specific MSK pain data and had all the dietary data necessary to calculate the DII. The NHANES represents the U.S. population and provides generalizable health and nutritional information. Since 1959, the NHANES has been an ongoing effort to survey the population’s nutritional status, other potentially relevant health determinants, and self-reported health outcomes such as LBP. More information on the NHANES can be found in Appendix 1 and the Centers for Disease Control and Prevention website. 67 Only individuals between 20 and 85 years of age were included in this cross-sectional study because the pain questionnaire was administered only to individuals above 20.
Dietary Inflammatory Index (DII®)
The DII is based on literature published through 2010 linking diet to inflammation assessed through the following blood markers: Interleukin (IL)-1β, IL-4, IL-6, IL-10, Tumor Necrosis Factor-alpha (TNFa) and C-reactive protein (CRP). Details regarding DII are described in detail elsewhere. 57 Higher DII scores represent more pro-inflammatory diets, while lower (i.e., more negative) DII scores indicate more anti-inflammatory diets. 57 The index has been validated in 48 studies with various inflammatory markers in different settings57,68; for example, CRP levels were validated in another NHANES dataset. 69 Other relevant findings for DII calculated for NHANES datasets include positive association with depressive symptoms in women, 70 and leukocyte telomere length. (a biomarker of aging). 71 It has also been associated with different measures of mortality (all-cause, cardiovascular disease, and digestive-tract cancer) in subjects diagnosed with pre-diabetes. 72
Study Variables
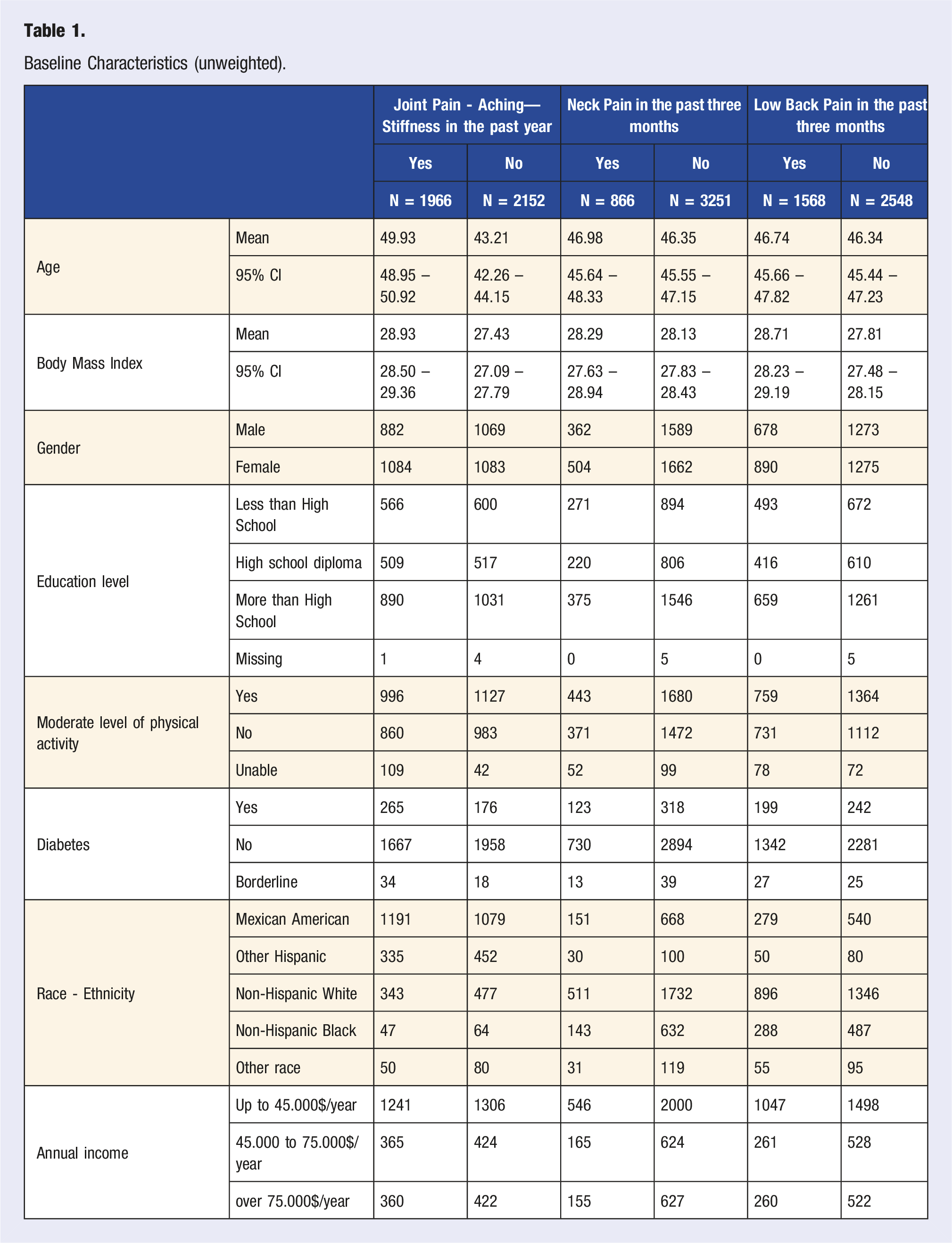
Baseline Characteristics (unweighted).
The NHANES dietary data were collected using the 24 hour food recall interview (24HR) method. The 24HR has been widely used in literature to assess habitual dietary intake and calculate the DII. The data used to calculate the DII were collected as part of the 24HR administered on the first day (day 1) in the Mobile Examination Center (MEC, see Appendix 1 for more details) or at home for those who agreed to the interview only. A second 24HR was administered via phone in a time window between 3 to 10 days after the first one was completed.
The DII (independent variable) was calculated based on the method described by Shivappa et al 57 (see Appendix 2 for more details). The DII was calculated for each 24HR questionnaire, then averaged for each patient for statistical analysis; this approach better captures the intra-individual variability that typically occurs with diet. The DII calculation can include up to 45 items, though fewer dietary items were available and used for analysis here (n = 28); this reduced number of items does not compromise the DII calculation, as shown by previous research,50,74 including a meta-analysis. 75
A literature review identified potential confounders related to pain symptoms and inflammation; they were determined a priori and examined in the adjusted models.76-79 The following possible confounders were investigated: age, sex, Body Mass Index (BMI = weight (kg)/height(m)2), physical activity level, education level, diagnosis of diabetes, race/ethnicity, and total household annual. Age (calculated in years) and BMI (kg/m2) were treated as continuous variables, while the other variables were analyzed as categorical.
Statistical Analysis
The statistical analysis was performed following the guidelines provided on the Centers for Disease Control and Prevention (CDC) Website. 80 The official NHANES website suggests using SAS “SURVEY” procedures for statistical analysis because these survey-specific procedures are intended to account for the complex recruitment/stratified population sampling design employed in this survey. Moreover, missing data were treated with the “NOMCAR” option, which uses the Taylor series variance estimation to account for missing values as “not missing completely at random.” SAS® 9.4 for Windows (SAS Institute Inc, Cary, NC, USA) was used for all analyses.
Because of the complex nature of the survey, the NHANES dataset comes with statistical weights to be included in the analysis to make the sample representative of the general US population at the time of data collection. The appropriate weight (“WTDR2D”) was used for every statistical calculation, thus making the results generalizable to the 2003-2004 US population. The weighted population numbers are reported in Appendix Table 2.
The statistical analysis was determined a priori. The independent variable (DII) was tested as a continuous variable against the 4 main outcome variables of OA symptoms, neck pain, LBP, and any pain lasting longer than 24 hours in the prior month (each treated as a dichotomous variable). The same statistical approach was used for the secondary aim variable of pain duration. A logistic regression model (“PROC SURVEYLOGISTIC”) was used to identify the significant factors at a P-value <.05. Odds Ratios (ORs) and 95% confidence intervals (CI) were calculated. Each model was run unadjusted and then adjusted for confounders, as often performed in analyses using complex population samples (such as the NHANES).81-84 A linear regression model was performed to assess the relationship between DII scores and circulating CRP levels; for this analysis, CRP levels were log-transformed to normalize their distribution.
Secondary analyses were planned for anticipated potential effect modifiers such as rheumatoid arthritis (RA) diagnosis potentially driving OA symptoms and for participants experiencing only one type of pain (either OA symptoms, neck pain, or low back pain) compared to multiple types of pain. We also anticipated performing additional analyses for identifying potential effect modifiers and sensitivity analyses as suggested by the results; the intent was to explore associations and interactions that could not be determined a priori.
As anticipated, we ran additional analyses to explore why the unadjusted models of joint pain, aching, and stiffness, as well as the presence of the symptoms for the month, were both not significantly associated with DII, whereas they were in the adjusted models.
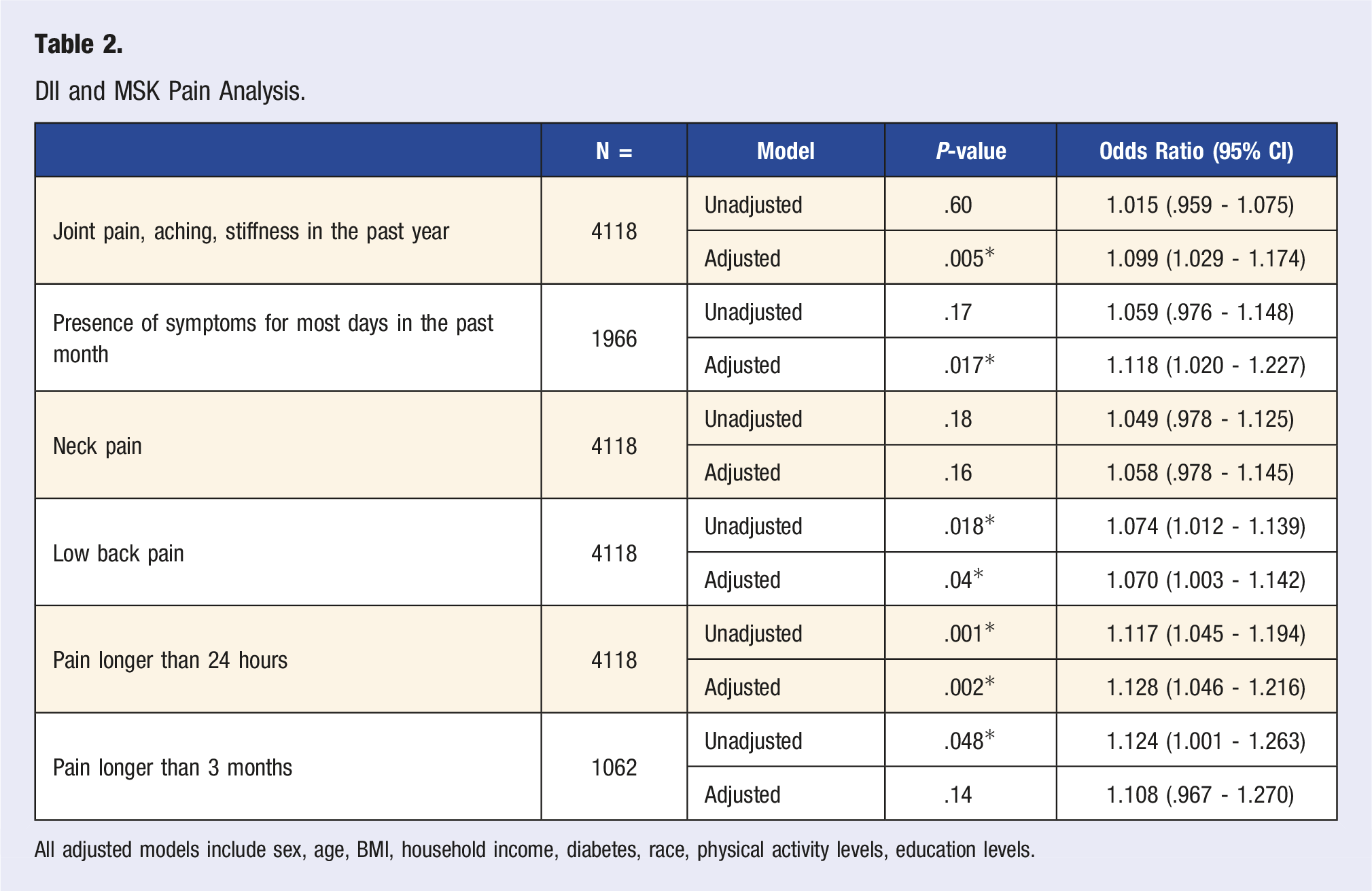
DII and MSK Pain Analysis.
All adjusted models include sex, age, BMI, household income, diabetes, race, physical activity levels, education levels.
Results
Baseline demographic characteristics of the included population are provided both unweighted (Table 1) and weighted (Supplemental Appendix Table 1).
OA Symptoms
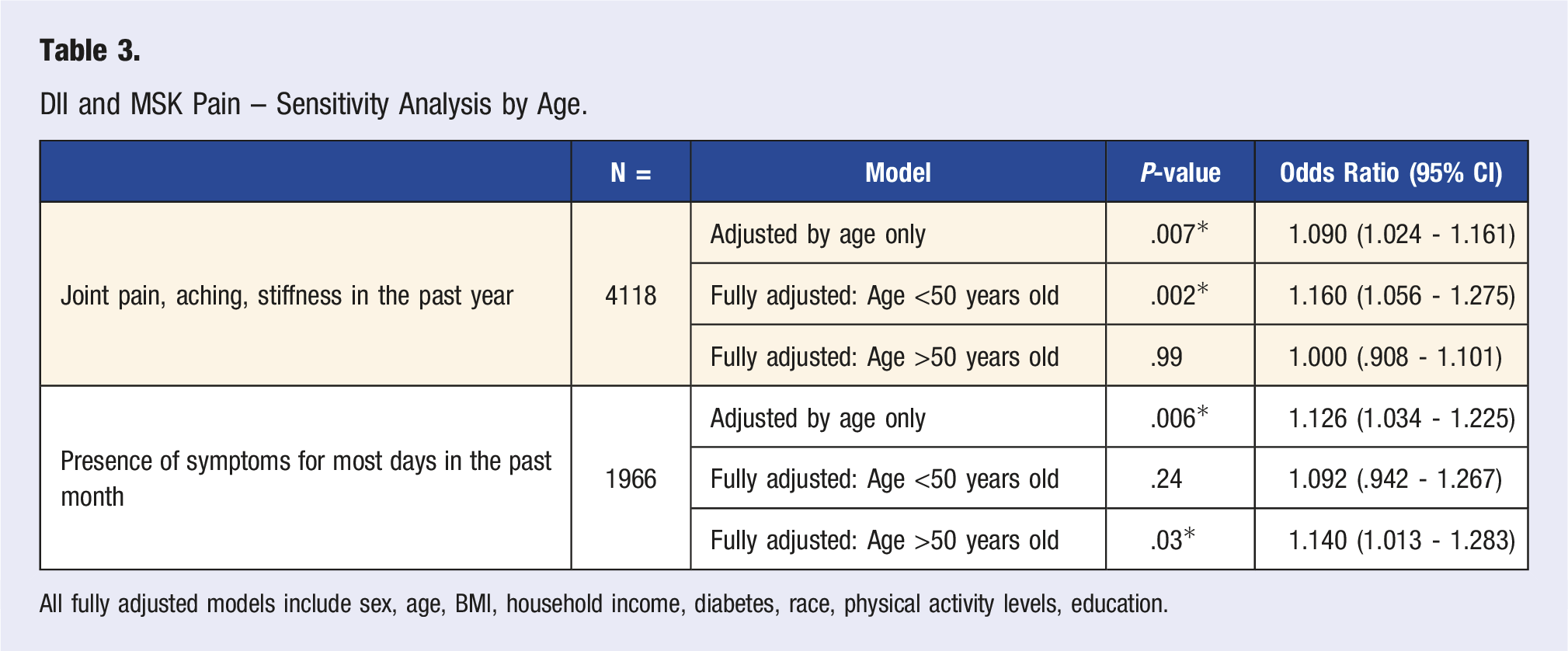
DII and MSK Pain – Sensitivity Analysis by Age.
All fully adjusted models include sex, age, BMI, household income, diabetes, race, physical activity levels, education.
OA symptoms for most days in the past month were also not significantly associated with DII in the unadjusted model. Nonetheless, DII was significant in the adjusted model (P = .017, OR = 1.12). Upon performing another sensitivity analysis (Table 3), DII was again significant in a model with age and DII only (P = .006), with no significant interaction between these two variables. No other variable showed changes in the significance level of the DII’s relationship to pain. In the unadjusted model stratified by age, DII was found to be non-significant (P = .14) in younger than 50 but significant (P = .039) in the older group. The adjusted model replicated the results highlighted in the unadjusted model, with DII being non-significant (P = .24) in the group younger than 50 years old and significant (P = .03) in the older group.
Neck Pain
Unadjusted and adjusted models showed no significant association between DII and neck pain (P = >.05).
Low Back Pain
DII was significantly associated with low back pain in the unadjusted model (P = .018; OR = 1.074) and in the adjusted model (P = .04; OR = 1.070).
Pain Duration
DII was associated with pain lasting longer than 24 hours in the month prior to data collection in the unadjusted (P = .001; OR = 1.117) and the adjusted model (P = .002; OR = 1.128). Regarding whether DII was associated with pain experienced more or less than 3 months, DII was found to be significantly associated in the unadjusted model (P = .047; OR = 1.124), but it was not significant in the adjusted model (P = .14).
DII and CRP Association in the Current Population
DII and CRP levels were significantly associated (P = <.0001) in both the unadjusted and the adjusted models.
Secondary Analyses
Since the symptoms of OA can overlap with symptoms from Rheumatoid Arthritis (RA), a secondary analysis was performed to assess if RA was driving the observed finding. A second fully adjusted model was run for this analysis, excluding the subjects with a reported RA diagnosis (n = 223). DII retained its significant association with OA symptoms in this second model, though the P-value decreased from .005 to .011.
A t-test was performed to assess the difference between the mean DII value of those who reported only one type of pain and multiple types of pain. This analysis aimed to explore if there is an effect of diet-related inflammation on the number of symptoms reported. For this analysis, the mean value of DII in those participants who reported pain consistent with either OA symptoms, neck pain, or low back pain (n = 2828) was compared to those who reported more than one of those pain (n = 1289) types. No statistical significance was found between these groups (P = >.05).
Discussion
The study results were largely consistent with the hypothesis that diet-associated inflammation increases the risk of musculoskeletal pain. This finding was true for the common OA symptoms of joint pain, aching, and stiffness and their presence in the fully adjusted models for most days. Furthermore, it also was significant in the final model for LBP and the presence of any pain lasting longer than 24 hours. The overall results offer promising perspectives for exploring potential pain mechanisms related to the associations found for individuals suffering from musculoskeletal pain. If replicated, these results could potentially be applied to developing anti-inflammatory dietary interventions for individuals affected by musculoskeletal pain.
An unexpected finding was age’s role in the presence of OA pain symptoms. The fact that joint pain, aching, and stiffness are associated with higher DII in younger people may be interpreted as a contributing role of diet in early symptom onset. It could also mean that the effect of a pro-inflammatory diet on OA pain diminishes when degenerative changes to the joint have already occurred. Furthermore, it could mean that the manifestation of diet-related inflammation in people over 50 years old (with more established OA degeneration of the joints) may present differently, for example with higher pain intensity or frequency. The literature on the topic is only present, scarcely, in animal models. A few studies have highlighted that systemic inflammation may have a role in the early onset of OA.86,87 In line with those findings, the present study adds some evidence that will need further evaluation in future research. In any case, the overall adjusted model shows a significant association between DII and pain presence for most days in the past month, which points to a relevant contribution of DII in the frequency of pain and not only its presence.
Interestingly, DII was not found to be associated with neck pain. These findings open the door for speculations on the reasons behind the difference in neck pain, LBP, and OA pain. One possible interpretation is that neck pain is less affected by systemic inflammation than OA pain and LBP. Another possible interpretation is that different cytokines characterize neck pain other than those considered in the DII calculation (IL-1β, IL-4, IL-6, IL-10, TNF-α, and C-reactive protein (CRP)). It could also mean that only specific phenotypes of neck pain are affected by systemic inflammation; thus, the association is not significant in a large general population sample. Though the study design and available data do not allow for testing these hypotheses, they also do not allow for ruling them out and thus require further research on the topic.
The DII showed a weak association with pain lasting more than 3 months in the unadjusted model; this association was not significant in the adjusted model. The fact that only a subset of the whole population (roughly 25%) was included in this analysis underlines the need for further clarification of these results in future studies. Nonetheless, the current evidence does not point to diet-related inflammation as a significant predictor of pain duration in excess of 3 months in this population. Also, the question in the survey was related to any pain lasting longer than 24 hours. This large category may include pains that are not musculoskeletal, thus potentially impairing the interpretation of such results. Future studies will need to address the role of diet-related systemic inflammation in transitioning from acute to chronic pain.
To better understand the significance of the DII results, it is important to remember that odds ratios measure a change in the unit of the variable in question. The DII range in the included population is between −5.19 (most anti-inflammatory diet) and +3.75 (most pro-inflammatory diet), thus having a range of almost 9 points. For example, the odd ratio calculated for an 8-point difference in DII among people with LBP would equal 1.720. This result highlights the full potential of the different impacts that a maximally anti-inflammatory diet can have compared to a maximally pro-inflammatory diet.
Even with the promising evidence of the advantages of an anti-inflammatory diet, translating the DII into individualized dietary suggestions may be challenging. For example, the “Western diet” is commonly acknowledged as pro-inflammatory. 88 This is due to its high intake of red and processed meat, high in saturated and trans fats, cholesterol, and protein, 89 all pro-inflammatory nutrients in the DII calculation. It is also important to note that the Western dietary pattern commonly lacks fruits, vegetables, nuts, whole grains, and spices, thus lacking many vitamins, mono- and poly-unsaturated fats, minerals, and fibers that have anti-inflammatory effects for the DII calculation. Additionally, a diet with a higher proportion of fruit and vegetable consumption, in contrast to higher consumption of meats, refined sugars, and processed foods, typically results in lower overall caloric intake, which is anti-inflammatory as per the DII calculation. Nonetheless, these broad guidelines based on nutrient consumption could be easily misinterpreted by the general public, thus removing the direct application of the DII from clinical practice and everyday use.
The main strength of this study is its contribution in showing that a pro-inflammatory diet is associated with increased OA pain symptoms and their daily presence. It also shows that, in this sample of US adults, LBP presence and the presence of pain lasting longer than 24 hours are associated with diet-related inflammation. Furthermore, the actual inflammatory status of the participants, as predicted by the DII, was consistent with the observed strong association between DII and CRP values. This finding is consistent with previously published data on a 2003-2008 NHANES cohort which also demonstrated an association between DII and CRP (dichotomized). 69 These findings align with growing evidence linking diet (e.g., specific dietary patterns or consumption of specific foods) 90 with chronic low-grade systemic inflammation and various chronic health conditions. More specifically, it supports the evidence that systemic inflammation may contribute to pain causation and duration, though this should be confirmed through more appropriate study designs (e.g., extensive longitudinal studies, intervention trials, and a more significant number of biomarkers included).
If confirmed, these findings could help reinforce the trend towards more comprehensive therapeutic approaches to prevent and manage pain. For example, diet could be integrated as part of a multidisciplinary approach to treating chronic pain, as seen in the DII-based IMAGINE intervention, which has shown to improve health and reduce inflammatory biomarkers.91,92 In this intervention, participants attended 21 classes over a year where they were taught approaches known to help with inflammation reduction. These approaches included diet (with anti-inflammatory properties based on DII scores), exercise, and stress reduction. 93
Limitations
Despite its strengths, this study also presents some limitations. Many of those limitations were linked to the timeline and nature of the collected and coded data. For example, the dataset used was the only one identified that included all the data necessary for calculating DII and the data regarding non-arthritic and idiopathic pain. Because the dataset is from 2003-2004, it might not be a valid representation of the current US population or dietary habits. Generalizing the conclusions to other countries may also be limited.
Nonetheless, the interpretation of the DII does not change in light of potential differences with the US population or its current dietary choices. Even if the dietary intakes may have changed since the data were collected, the fact that there was an association between DII scores and pain outcomes remains relevant and highlights a significant association that should be investigated further. Moreover, DII scores are comparable across different large populations and diets, regardless of when the data were collected.
Some limitations existed related to the dataset itself. For example, the definition of OA was based on participants’ recollections of their symptoms rather than a clinical diagnosis. This limitation, though, offers a more inclusive approach to the patient experience since most individuals receive a diagnosis in the later stages of the disease. 94 Another limitation related to how the data were collected is that it was impossible to determine different pain groups and mechanisms. For example, it was not possible to differentiate subjects in acute, sub-acute, and chronic phases. Also, the question on pain lasting longer than 24 hours included any type of pain, which can include non-musculoskeletal pain. Likewise, it was not possible to differentiate between different pain mechanisms, so, for example, it was impossible to understand the degree of contribution from different types of pain, such as neuropathic (both central and peripheral), nociceptive, nociplastic, visceral, or other. Last, the information on the use of anti-inflammatory drugs did not provide enough details to be incorporated into the analysis. This limited the understanding of the impact of this obvious potential mediator on inflammation and pain.
Another study limitation is that the dietary information was collected solely from the 24HR. Even though this method has been validated and is a reliable tool for the data collection on dietary consumption, the 24HR depends on the ability of subjects to recall their dietary intake and is thus subject to inevitable recall bias. 95 Also, the recall period is limited to 24 hours before questionnaire completion; thus, it does not represent long-term habitual intake. The mean value of 2 different 24HR collections was used for statistical analysis to address this limitation.
Additionally, the DII does not consider the individual variability that plays a part in digestion. Growing evidence points to different factors that influence the individualized response of the digestive system to different foods. 96 The variability in the gut microbiome, genetics, and epigenetics plays a role in the individual response to food. In other words, while one specific food may be anti-inflammatory for an individual, it may cause an immune and inflammatory response in another. A classic example would be foods from the nightshade family that are generally anti-inflammatory but can have a pro-inflammatory effect in certain individuals. 97 The extent of this inter-individual variability is currently unknown, thus limiting the generalizability of results from DII.
The cross-sectional study design limits causal inference. Further research should attempt to fill this gap. Future studies in this area should also be tailored to the MSK population’s specific characteristics and how to adequately stratify such a heterogeneous population. For example, studies have shown the importance of sub-classification in treating sub-acute and chronic pain populations. 98 Providing a specific therapeutic approach for different sub-groups within the LBP population has heightened response to treatment. This previous finding should be kept in mind—not only for the LBP population, which is known to have very different combinations of contributing factors—but for all MSK pain populations. Diet-related inflammation may be more relevant to specific sub-groups of MSK pain populations than others. A sub-classification approach should be considered when designing or analyzing the results of studies on MSK pain.
Conclusions
This study contributes to the challenging and multifactorial topic of MSK pain when considering the currently limited understanding of pain mechanisms and treatments. It demonstrated a significant positive association between diet-related inflammation and OA symptoms, duration, presence of LBP, and presence of pain lasting longer than 24 hours in a general sample of the adult US population. In conclusion, even though causality could not be established, this study highlights a promising path for future research assessing the role of diet in the presence of MSK pain, its severity, and its duration.
Supplemental Material
Supplemental Material - Assessing Diet and Musculoskeletal Pain in Adults: Results From a Cross-Sectional Analysis of the National Health and Nutrition Examination Survey (NHANES)
Supplemental Material for Assessing Diet and Musculoskeletal Pain in Adults: Results From a Cross-Sectional Analysis of the National Health and Nutrition Examination Survey (NHANES) by Valerio Tonelli Enrico, James R. Hebert, Gerald Mugford, Zhiwei Gao, Peter Wang, Nitin Shivappa, and Gwendolyn Sowa in American Journal of Lifestyle Medicine
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (U19AR076725-01S1).
Author’s Note
Dr James R. Hébert owns controlling interests in Connecting Health Innovations LLC (CHI). This company has licensed the right to his invention of the dietary inflammatory index (DII) from the University of South Carolina to develop computer and smartphone applications for patient counseling and dietary intervention in clinical settings. Dr Nitin Shivappa is an employee of CHI.
Disclosure
The first and corresponding author, Valerio Tonelli Enrico, had the time used in performing this research covered by a supplemental grant (U19AR076725-01S1) that is part of the Back Pain Consortium (BACPAC) consortium.
Ethical Considerations
The study was based on the public-use data files available to both American and international researchers via the NHANES database. The public-use data are completely de-identified and publicly available with necessary suppression methods to protect confidentiality; thus, according to the HREA, the ethics committee board for the province of Newfoundland and Labrador, the study was cleared from needing the actual IRB clearance.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
