Abstract
This review integrates new developments in psychology with updated physiological insight on the complex relationships among chronic psychological stress (arising from weight stigmatization and body shaming), food composition, physical activity and metabolic health for the example of diabetes. We address how visual measures of health, such as body mass index (BMI) and waist-to-hip ratio, do not adequately capture metabolic health and can instead contribute to weight stigmatization, chronic stress, and system-wide impairment of metabolic health. We also emphasize the importance of food composition over calorie counting. We summarize how chronic stress interacts with nutritional deficiencies and physical inactivity to disrupt the stress response, immune response, gut microbiome, and function of fat depots. We specifically address how interactions among lifestyle factors and the gut microbiome regulate whether fat stored around the waist has a negative or positive effect on metabolic health. We aim to provide a resource and updated framework for diabetes prevention and health promotion by (i) highlighting metabolic imbalances triggered by lifestyle changes during the transition to industrialized society and (ii) detailing the potential to support metabolic health through access to modest, but comprehensive lifestyle adjustments.
“Shaming directly interferes with health-supporting behaviors, and shaming-induced chronic stress enhances cravings, blood sugar levels, and allocation of energy to storage as fat.”
Introduction
In both healthcare settings and public discourse, health status is often assessed using visual and quantitative measures. These measures include weight-to-height ratio (body-mass index, BMI), which became a common standard in healthcare to assess a patient’s metabolic health in the mid 1980s. 1 More recently, waist-to-hip ratio has been added as another proxy used to assess metabolic health.2,3 While these measures provide for quick and non-invasive quantitative assessment, evidence summarized in this review shows that these features are unable to assess metabolic health qualitatively and instead contribute to weight stigmatization/body shaming 4 and associated chronic stress. This review details key pathways by which chronic stress erodes metabolic health and causes wide-ranging negative impacts. 5 Based on this evidence, our review argues for a more qualitative approach to understanding metabolic health – not restricted to measures like weight or calories – in order to reduce the negative physiological effects associated with chronic stress resulting from stigmatization and shaming. We provide specific physiological evidence in support of the statement by Chrisler and Barney (2017) that “it is possible to be both fat and fit” 6 and thereby expand the definition of what it means to be healthy. In addition, we explore how modest lifestyle changes can benefit metabolic health even in the absence of significant weight loss. Specific attention is also given to the importance of food composition, thus moving away from the adage that “a calorie is a calorie” 7 and shifting the focus to “what we’re eating.” 8
Overall, this review emphasizes the qualitative nature of metabolic health and how reduction of weight stigmatization (and a focus on food composition rather than calories) may help restore metabolic balance and health. In doing so, this review builds on a call from the perspective of psychology and social science, as well as increasingly also from medical researchers (see, eg,9,10) for paradigm change in how we think about weight and health and a pressing need to end weight stigmatization. We here summarize specific additional arguments from molecular physiology, which we hope to be informative to a broad audience of researchers, healthcare providers, policy makers, individuals experiencing body shame (see 11 ), and the public at large.
We specifically synthesize evidence on the synergistic interaction of chronic stress level with food composition and physical-activity level in modulating the risk for type 2 diabetes as an example for a pro-inflammatory disease.
12
In particular, the case of diabetes serves to identify the key mechanisms through which chronic stress in interaction with other external factors disrupts the stress-response system, the immune system, appetite and blood sugar control, and the functionality of various fat depots (Figure 1). Some commentary is provided on putative evolutionary contexts for these molecular mechanisms and how these evolved programs can trigger disease in today’s environment of industrialized society (see also
13
). This review synthesizes insights that may be useful in addressing body shaming and chronic stress in disease-prevention efforts. Moreover, we highlight potential benefits of comparatively small changes – including intermittent stress relief, addition of micronutrients, and modest physical activity – in improving metabolic health in industrialized societies. Schematic of themes addressed in this review and their relationship with each other. The focus of this review is highlighted in pink: the relationship between body shame and the resulting chronic stress with key aspects of metabolic health.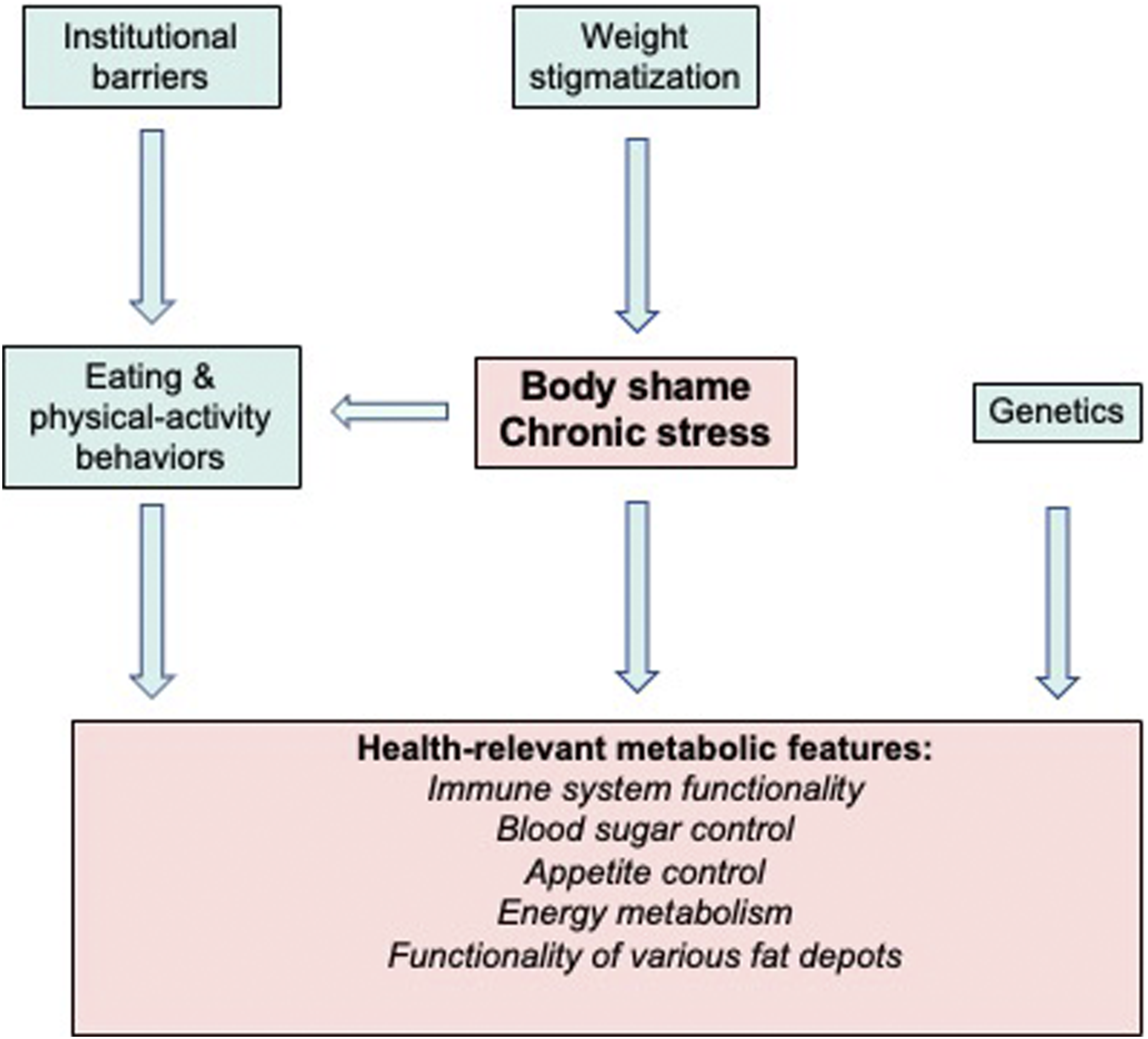
Preview of major review sections
After briefly describing the connection between body shaming and chronic stress, we identify system-wide immune dysfunction as a root-cause of pro-inflammatory diseases including diabetes – and as a result of chronic stress and a target of stress relief. Using India as a case study, we address the transition to industrialized society as a driver of systemic lifestyle changes – with increased chronic stress, a profound shift in food composition, and decreased physical activity. We briefly touch on interactions of environmental/lifestyle factors with genetic risk for diabetes in certain populations in India and elsewhere. We then propose a conceptual framework of how lifestyle factors in industrialized society trigger pro-inflammatory disease by creating concomitant imbalances in immune response and energy metabolism. Several subsequent sections target selected features of this framework, such as blood glucose and appetite control or immune-system function, to track how these processes are modulated by external cues from stressors and other lifestyle factors. Subsequently, these and other individual features are integrated into a comprehensive metabolic framework elucidating both degradation and restoration of metabolic health in industrialized society. A switch is defined from metabolic dysfunction (with food cravings, chronic high blood glucose and insulin resistance, immune-system dysfunction, and a state of fat-storage depots that exacerbates these outcomes) to programming for lessened food cravings, restored blood glucose control, insulin sensitivity, and immune function as well as for a metabolic state of fat-storage depots that actively supports the latter outcomes. Additional sections are devoted to the role of the gut microbiome and mitochondrial infrastructure in the programming of this metabolic transition by external cues. Throughout, this physiological evidence illustrates that metabolic health cannot be adequately assessed visually from physical features. This insight is applied to an evaluation of BMI as well as other, similar measures. For the case of waist circumference or waist-to-hip ratio, physiological evidence for the potential of metabolic transition of existing fat depots around the waist is highlighted. The following sub-section of the Introduction briefly previews relevant advances in the understanding of the biology of fat as background for subsequent sections.
The biology of body fat
Fat cells of humans and other mammals, in particular white adipose tissue (WAT) around the waist and under the skin, are capable of storing energy as fat (which packs more energy per volume than any other biological substance) for use during a future time with limited food availability. However, as stated in 2016 by Giralt and colleagues, 14 “the last 2 decades have witnessed a shift in the consideration of WAT as a mere repository of fat to be used when food becomes scarce to a true endocrine tissue releasing regulatory signals … to the whole body,” including signals that regulate the immune response, appetite, and energy metabolism. Wells 13 suggested in 2012 that this contribution to the coordination of numerous physiological functions by fat-storage tissue in response to external cues evolved as part of a “risk management strategy” or safety net against unpredictable changes in the ancestral human environment. However, as Wells 13 also points out, this same “sensitivity of numerous metabolic pathways to ecological cues makes our species vulnerable” to unprecedented global shifts in the human environment.
The connection between fat stores and the immune system links the size of available energy reserves to the activity of the energy-costly immune response.13,15 The insight that it is the visceral fat stores that can be associated with production of pro-inflammatory messengers15-19 contributed to a call to abandon BMI as an indicator for disease risk and to use waist-to-hip ratio instead.2,3 However, since then it has become increasingly evident that even existing visceral WAT has the potential to be transformed to an alternate metabolic state that may improve health outcomes rather than presenting a health risk (see Introduction and sections below). At minimum, this insight suggests that waist circumference and waist-to-hip ratio are also inadequate as indicators of health status. In addition to not capturing fat depot’s metabolic status, they are visual features that can contribute to stigmatization, chronic stress, and the many detrimental effects of chronic stress on health (as summarized in this review).
A fat depot of brown color (from presence of mitochondria), ie, brown adipose tissue (BAT) has somewhat opposite roles to those of WAT; BAT burns fat without exercise (generating heat in what is termed adaptive thermogenesis), dampens inflammation and appetite, and increases insulin sensitivity. 14 Remarkably, WAT can undergo a transformation (“browning,” as a result of added mitochondria) to a metabolic state more like that of BAT 14 in response to external cues from environmental/lifestyle factors (including food composition and modest physical activity20,21 as well as stress dynamics). Several sections of our review summarize the current understanding of how lifestyle factors affect the metabolic state of fat depots.
Body Shame Affects Health-Relevant Behaviors and Causes Metabolic Disruption
This section introduces body shaming and associated stress, as well as their roles in modulating metabolic drivers of disease risk – both indirectly through behavior as well as through direct metabolic disruption.
Body shame causes chronic stress and affects behavior
Carter and coworkers (2021)
22
stated, “physical appearance, particularly body weight, is a dimension that is often a source of social comparison and negative judgement
23
[especially] in Western cultures.” Shame and stigmatization increase psychological distress3,24 and depression,
25
which, in turn, affects eating behaviors24,26 and physical activity. Figure 2 highlights these connections. Chronic stress contributes to an enhancement of appetite/cravings (see section Chronic stress and other external cues control transport of glucose in and out of the blood stream below) and to avoidance of exercise in public spaces where disparaging comments are frequently encountered.27-29 Schematic depiction of the dual effect of chronic psychological stress (as related to body shaming) on physiological processes that are relevant for diabetes risk. This dual effect consists of direct impact on physiology as well as indirect impact via modulation of behaviors that, in turn, affect physiology. The arrows depict mechanistic links that can either lead to adverse outcomes when chronic stress levels are high or lead to desirable outcomes when stress levels are low. Concerning eating behaviors and participation in physical activity, an energy-dense, micronutrient-deficient diet and physical inactivity exacerbate the effect of high levels of stress. Conversely, a low-density, micronutrient-rich diet and participation in some level of regular physical activity synergistically enhance the effect of stress reduction. Created with BioRender.com.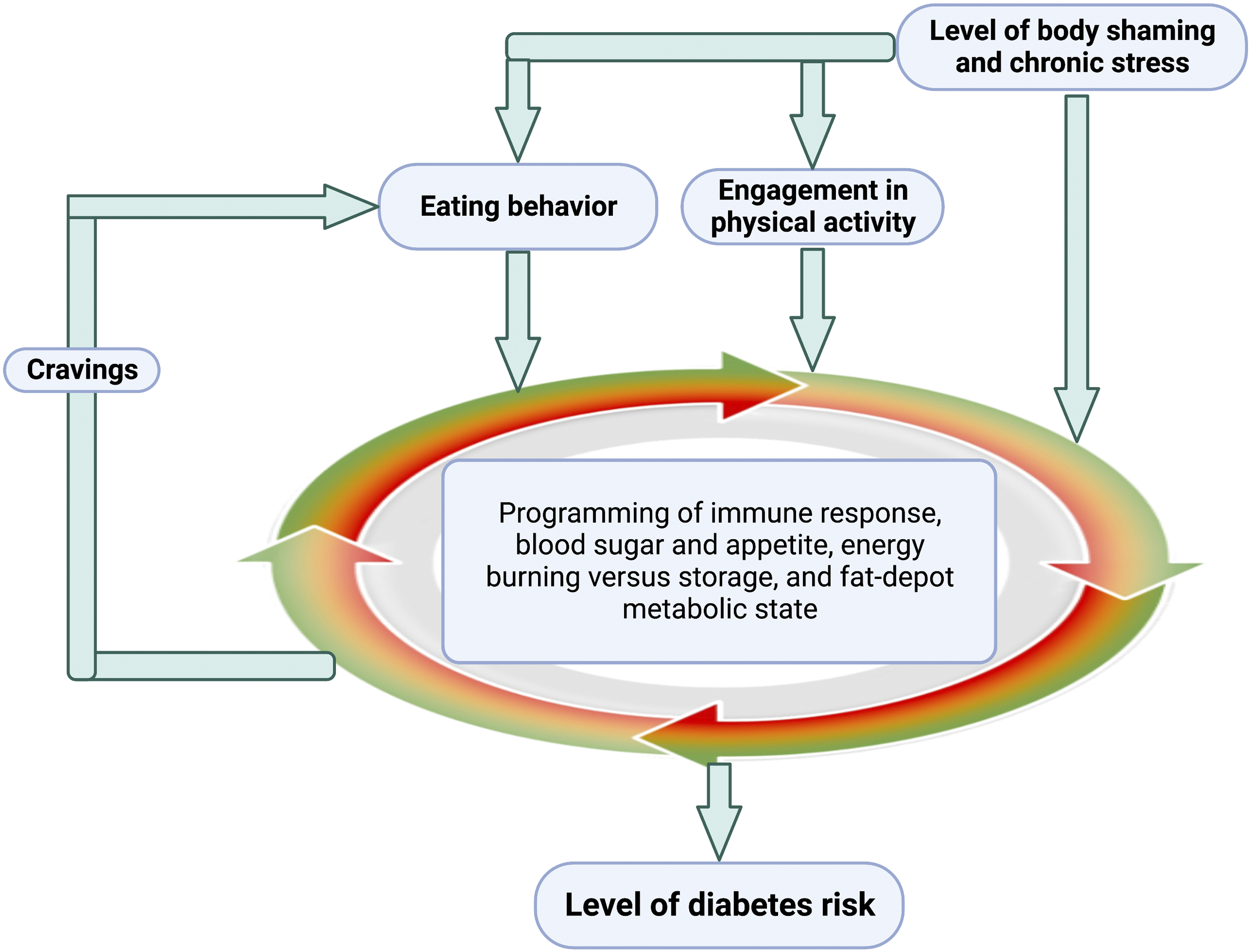
Moreover, chronic psychological stress associated with body shaming 30 manifests as elevated levels of stress markers31-34 (several later sections are dedicated to further detail on this topic). Interventions that reduce body shame and the associated stress are thus called for. 35 Meta-analysis 36 of the outcomes of such interventions thus far indicates their promise in reducing body shame as well as increasing health-promoting behaviors (see also26,37-39).
Notably, psychological interventions that reduced body shame and associated chronic stress are able to ameliorate immune-system function even when accompanied by no or minor weight loss. 36 Similarly, interventions based on diet modification and/or physical activity were also able to substantially improve metabolic health when accompanied by only minor weight loss.40-42 Our mechanistic analysis indicates that the same lifestyle factors that support overall metabolic health also lower cravings and energy storage, and may thus favor weight adjustment over time scales not captured by all interventions. However, overall, these observations point to a direct route from chronic stress to metabolic health that does not necessarily involve weight-related features (as is further elucidated in this review). Immune-system function is intimately linked to the function of the gut microbiome43-46 (see also;47,48 several sections below are dedicated to this topic). Beneficial effects of stress reduction on both immune function and gut-microbiome health are supported by meta-analyses and other systematic reviews on the effect of yoga,49-51 cognitive behavioral therapy, 52 mindfulness-based meditation practice 51 and mindfulness-based stress reduction. 53
These findings show that health evaluation requires comprehensive assessment of metabolic features. Use of features such as BMI actively contributes not only to body shaming and stigmatization3,25 but also to inequities in health care,6,54 which may be a primary mechanism for earlier mortality55,56 (see also 3 ). This effect can be seen as weight stigmatization contributing to institutional barriers to health (in addition to what is depicted in Figure 1). Moreover, weight-related stigmatization has been identified as an instrument of racial discrimination (for an overview, see 56 ). For example, BMI is a particularly poor indicator of metabolic risk in racial and ethnic minorities. 57 Moreover, traditional programs for weight loss can further exacerbate stigmatization and feelings of shame and guilt. 58 The following sub-section focuses on the mechanistic link between chronic stress and immune-system disruption.
Chronic stress fatigues the stress axis and disrupts the immune response
Figure 3 illustrates the importance of stress dynamics. Intermittent psychological stress prompts short bursts of immune- Schematic diagram to illustrate contrasting effects of acute vs chronic stress on the immune response. Intermittent, acute bursts of stress turn on stress-defense responses and suppress the immune response (“OFF” state of the immune response). Subsequent stress relaxation allows reactivation of normal immune activity (“ON” state of the immune response). In contrast, chronic psychological stress leads to a disrupted state that is likened here to a broken dial (neither “ON” nor “OFF”), where the immune system can no longer be fully activated (leading to poor immunity against infectious agents) nor fully turned off (leading to a state of chronic, non-resolving inflammation in which the immune system carries out self-attack on healthy tissues). Created with BioRender.com.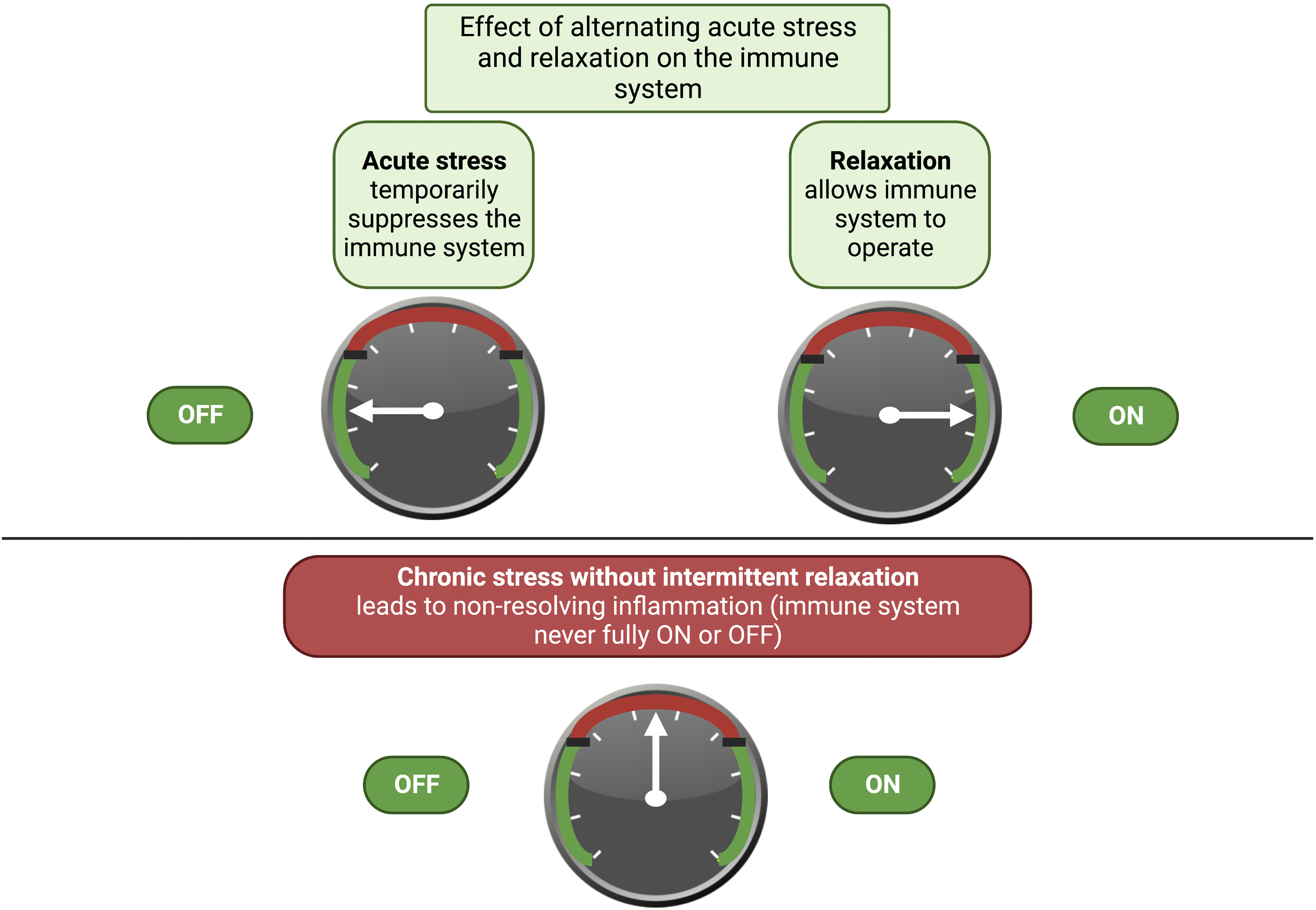
A spiraling cascade, furthermore, exists between dysfunction of the immune system and of the gut microbiome (see47,48). This role of the microbiome, and its modulation by external cues, is further addressed in the section below on Programming of metabolic state by external cues: A role for the gut microbiome. The effect of fat depots to either aggravate or oppose chronic inflammation is detailed in the section below on Programming of mitochondrial infrastructure.
Time to rest and restore is critical
The following perspectives suggest that modernization could be implemented “in a manner that promotes health rather than impairs it and thereby protects the next generation from the inevitability of increased chronic disease” 65 – by design of environments that allow human metabolism to reset and restore on a regular basis. It is becoming apparent that remarkable benefit can be derived through intermittent relief from psychological stress and inflammation as well as from sufficient time for recovery from exercise. Interrupting chronic psychological stress with brief, but regular, daily periods of self-care can help support the stress-response and immune systems as well as the gut microbiome.43,44,66,67 Moreover, exercise without sufficient time for recovery is also unadvisable because exercise, just like psychological stress, prompts a stress-defense response and temporarily suppresses the immune response (see discussion in 47 ). These perspectives shift the focus from viewing stress or exercise as either “bad” or “good” to considering the dynamics of these experiences for an evaluation of their impact on health (see also 68 on the topic of stress). Such a perspective on stress and exercise is also mirrored in the role of dietary factors. For example, dietary micronutrients that support acute inflammation (especially omega-6 fatty acids) are often over-consumed in industrialized societies but are part of a healthful diet – as long as other micronutrients that subsequently help terminate/resolve inflammation (eg, omega-3 fatty acids and antioxidants) are present in adequate amounts.
India as a Case Study
Diabetes prevalence is increasing rapidly around the world, and nowhere more than in India, the “diabetes capital of the world” with more than 70 million people with diabetes. 69 At the same time, weight stigmatization is also rising in India. 70 Whereas weight stigmatization in India has some features specific to the cultural context of India,71-73 the basic tenet that stigmatization is associated with adverse health outcomes applies. For example, associations among weight, weight stigmatization, and depression have been described for populations in India. 74 In the next sections, we use the example of India to highlight how lifestyle transitions caused by industrialization act upon the genetic propensity for certain metabolic responses.
Industrialization caused lifestyle transitions
The following connections illustrate how societal change during industrialization systemically affected lifestyle beyond individual choice. Starting from the 1970s, diabetes prevalence in India rose by a striking 900% over the next 40 years. 75 The majority of diabetics have “lifestyle-dependent” type 2 diabetes mellitus, 76 characterized by an inability to regulate blood glucose by insulin. 77 Diabetes can eventually lead to blindness, amputation of extremities, kidney failure, and heart disease, which causes not only tremendous human suffering, but also loss of billions of dollars annually in India alone that could otherwise go towards economic development. 78 The rapid increase in diabetes prevalence in India prompted numerous studies into how lifestyle in industrialized society triggers diabetes, especially in South Indians who may have a genetic predisposition for the disease 76 (see the following section on environment-gene interaction).
Industrialization-related changes in the human environment led to reduced physical activity, a shift in food composition towards an energy-dense, micronutrient-deficient diet, and chronic psychological stress, 79 which correlated with the rise in diabetes prevalence. The onset of diabetes is shifting to occur at ever younger ages in many countries, 80 especially India.75,76 This earlier age of onset has emerged alongside an increasingly sedentary lifestyle resulting from shifts in working conditions for adults (eg, to an office job) and less playing time outside for children due to increasing urbanization and availability of technology and media. 76 India features a close correlation between physical inactivity and excessive consumption of highly processed, energy-dense, micronutrient-deficient foods and drinks. 75 Modernization also increased the level of continuous low-grade psychological stress – such as that resulting from continuous exposure to technology, news, and social media with an erosion of practices that allow for regular intermittent down-time free from these engagements (see, eg, 81 ).
India’s “thin-fat” syndrome
This section illustrates that elevated health risk can be associated with low weight (especially BMI). Even a low calorie supply from a diet that consists of energy-dense, micronutrient-deficient food triggers elevated energy storage as visceral fat in a syndrome that is common in India and has been termed the “thin-fat” syndrome.82,83 This outcome is consistent with studies showing that a weight-loss diet supplying only a very low calorie level (negative energy balance) still led to elevated visceral fat storage when the diet consisted of particularly energy-dense versions of the major components (macronutrients) of food (certain fats and carbohydrates; see below) and insufficient essential minor food components (micronutrients, such as vitamins, antioxidants, and anti-inflammatory compounds). 16 The section Immune regulation has no simple relationship with fat stores below details how multiple factors interact to cause immune system dysfunction. These outcomes make the case for assessing health risk via comprehensive evaluation of metabolic profiles, food composition, and overall lifestyle and shifting focus (i) from weight to overall metabolic functioning and (ii) from calorie counting to food composition (macronutrient composition and micronutrient content) (see, eg,84,85; see also 86 ).
The next section discusses how lifestyle factors interact with a genetic predisposition for diabetes in certain populations and outlines a food composition that promotes health in the environment in industrialized societies.
Diabetes: Environment-Gene Interaction
This section addresses key aspects of metabolism that are subject to modulation by environmental factors as well as to genetic variation and epigenetic modulation. Type 2 diabetes mellitus is one of many chronic diseases involving a state of continuous low-grade immune-system activation, with chronic, non-resolving inflammation,87,88 as well as disruption of glucose control.89-91 Both of the latter traits also exhibit genetic variation among human populations with a different predisposition for diabetes. 77
Some populations have a genetic propensity for diabetes
South Indians share with some other populations, such as Akimel O’odham of the Americas (see below), a high genetic propensity for insulin resistance and diabetes76,92-94 as well as other inflammatory diseases and disorders,15,16 and elevated visceral fat storage. 95 Several sections below provide mechanistic insight into external controls acting on blood glucose level, the immune response, and visceral fat storage.
Genetic variants have been noted for both Akimel O’odham and South Indians for genes associated with inflammation-promoting processes,96-99 genes involved in blood-glucose control,100-104 and genes that support burning of energy (via thermogenesis) even when physical activity level is modest105,106 (see Introduction and several sections below). These and other results indicate that diabetes risk has a multi-gene (polygenic) component that is acted upon by the environment/lifestyle.107,108
While the Akimel O’odham possess a genetic predisposition for metabolic features that elevate diabetes risk, lifestyle has a profound effect on whether or not this predisposition manifests itself in disease. This example highlights the power of lifestyle but should not be understood to mean that external factors can cancel out any genetic predisposition or are the sole determinant of health outcomes. Two populations of Akimel O’odham with the same genetic background living either in Arizona, USA, or in the highlands of Mexico exhibit a dramatically different incidence of diabetes. 65 This case is also a poignant example for how such an environment is imposed by society rather than being a consequence of personal choice. The Akimel O’odham of Arizona commonly live in an environment characterized by a fast-food diet, physical inactivity, and chronic psychological stress. This particular environment is linked to diversion of water infrastructure associated with Western settlement and government subsidies for the Akimel O’odham replete with energy-dense, nutrient-poor food. 109 Metabolic programming by environmental cues that elevate diabetes risk is thus common in this Arizona population. In contrast, diabetes is virtually unheard of among the Akimel O’odham of Mexico in the Sierra Madres who live in a different environment characterized by a high level of physical activity, a more traditional (low-density, micronutrient-rich) diet, and what appears to be a low level of chronic psychosocial stress. 110
A recent review 111 summarized the growing evidence for genetic variation in additional genes involved in “appetite control, brain regulation, and thermogenesis.” Moreover, reports of further, epigenetic modifications of these and other genes by environmental cues are increasing.111,112 Epigenetic programming for greater energy intake and storage 111 or for a pro-inflammatory profile 113 has been reversed by cues from food composition, physical activity and/or stress relaxation practices.
Food composition can promote health in industrialized society
Key features of a diet that helps support health in today’s industrialized environment with respect to the composition of dietary fat, carbohydrates, and various groups of essential micronutrients needed to balance energy metabolism and immune response and help resolve inflammation.
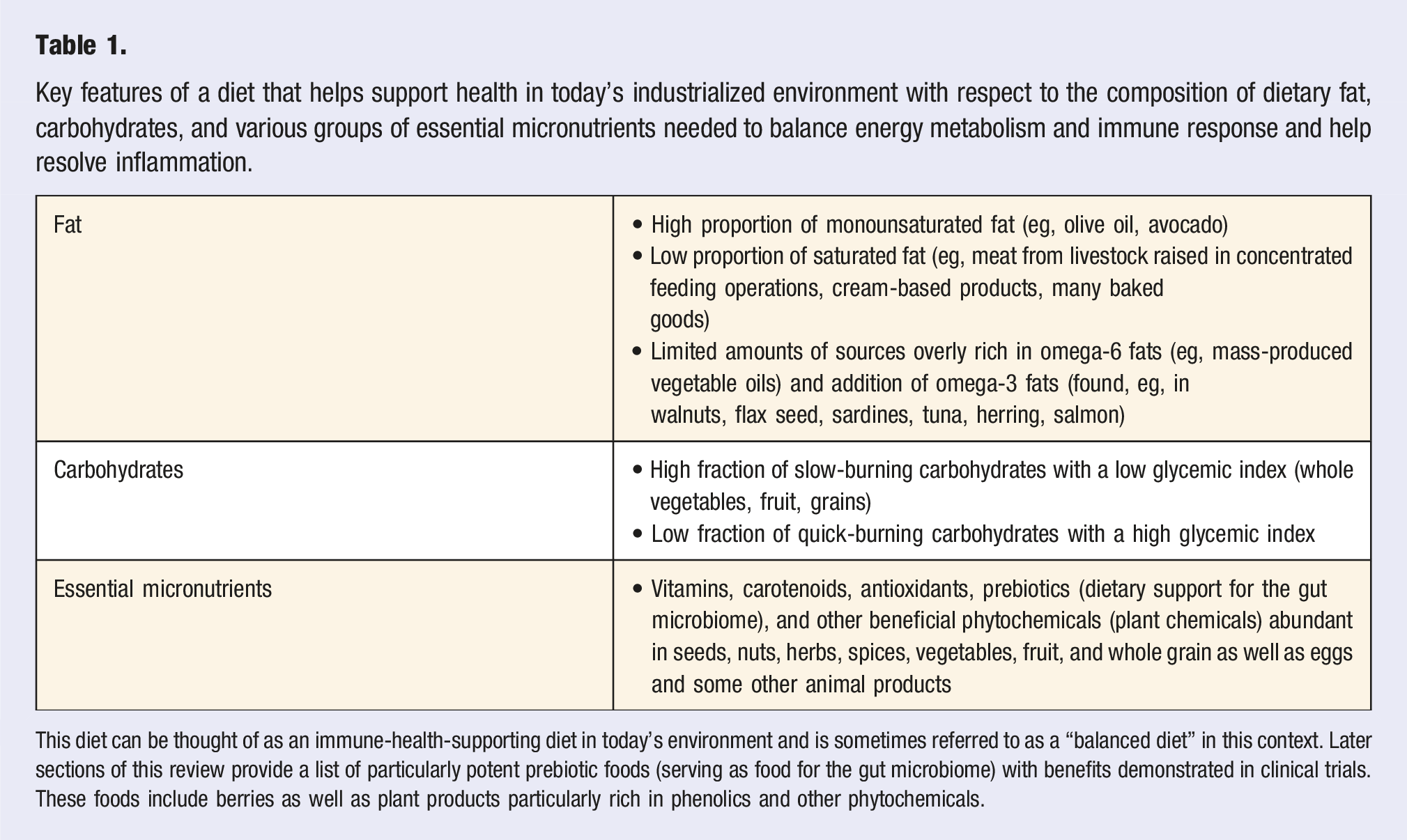
This diet can be thought of as an immune-health-supporting diet in today’s environment and is sometimes referred to as a “balanced diet” in this context. Later sections of this review provide a list of particularly potent prebiotic foods (serving as food for the gut microbiome) with benefits demonstrated in clinical trials. These foods include berries as well as plant products particularly rich in phenolics and other phytochemicals.
We refer to authoritative reviews on interventions based on nutrition 123 or combinations of nutrition and exercise (see, eg,124,125 for details on the role of nutrition beyond the scope of this review, see48,126-129). Notably, in order to prevent systemic immune dysfunction, both dietary anti-inflammatory and additional anti-inflammatory regulators produced within the human body in response to physical activity are needed. 130 However, in the modern environment of industrialized societies, humans typically neither consume enough dietary inflammation-resolving regulators nor do they produce enough supporting endogenous regulators to keep the immune system operating effectively. This insight can help communicate the considerable benefits of a combination of specific dietary features with regular, modest physical activity and regular, modest stress relaxation.
Moreover, the potential of external cues to restore metabolic health and oppose genetic predisposition, and/or epigenetic programing, for elevated diabetes risk supports the need for access to a health-promoting lifestyle for all members of society. The next section focuses on the specific feature of blood glucose level and selected examples for processes that transport glucose into and out of the blood stream. We highlight the effect of chronic stress on the transport of glucose in and out of the blood stream in the context of the effects of food composition and physical (in)activity.
Chronic Stress and Other External Cues Control Transport of Glucose in and out of the Blood Stream
This section describes how chronic stress raises blood glucose level through mechanisms that interact synergistically with the effects of food composition and physical activity. Figure 4 highlights that chronic psychological stress triggers breakdown of sugar stores and release of this glucose into the bloodstream
131
in addition to blocking the reverse process of glucose removal from the bloodstream.
132
Why would that be so? Release of glucose from its stores in the liver into the bloodstream can support stress defenses. Conversely, any removal of sugar from the bloodstream for the purpose of storage (in either liver or muscle in an insulin-dependent way) is not advantageous under acute stress when the energy from sugar must instead be used for defensive action supported by brain activity and muscle action. It thus makes sense for acute psychological stress to block sugar uptake into the liver (or into working muscle for the express purpose of storage). However, continuous blockage of glucose removal from the bloodstream (for the purpose of storage) by chronic psychological stress contributes to the undesirable phenomenon of chronic high blood sugar. These effects of chronic stress add to the effect of regular consumption of large quantities of foods that quickly release sugar (“high-glycemic” diet 1;
133
see also Table 1) and of physical inactivity (see next paragraph). Schematic depiction of synergistic effects (red) of chronic psychological stress, an overly energy-dense, micronutrient-deficient diet, and physical inactivity in causing chronic high blood glucose. In addition, it is indicated that reduction of shame and stress (green box and bar) lessens psychological stress, and can enhance the beneficial effects of a low-density, micronutrient-rich diet and physical activity. Arrows represent direction of glucose movement. Created with BioRender.com.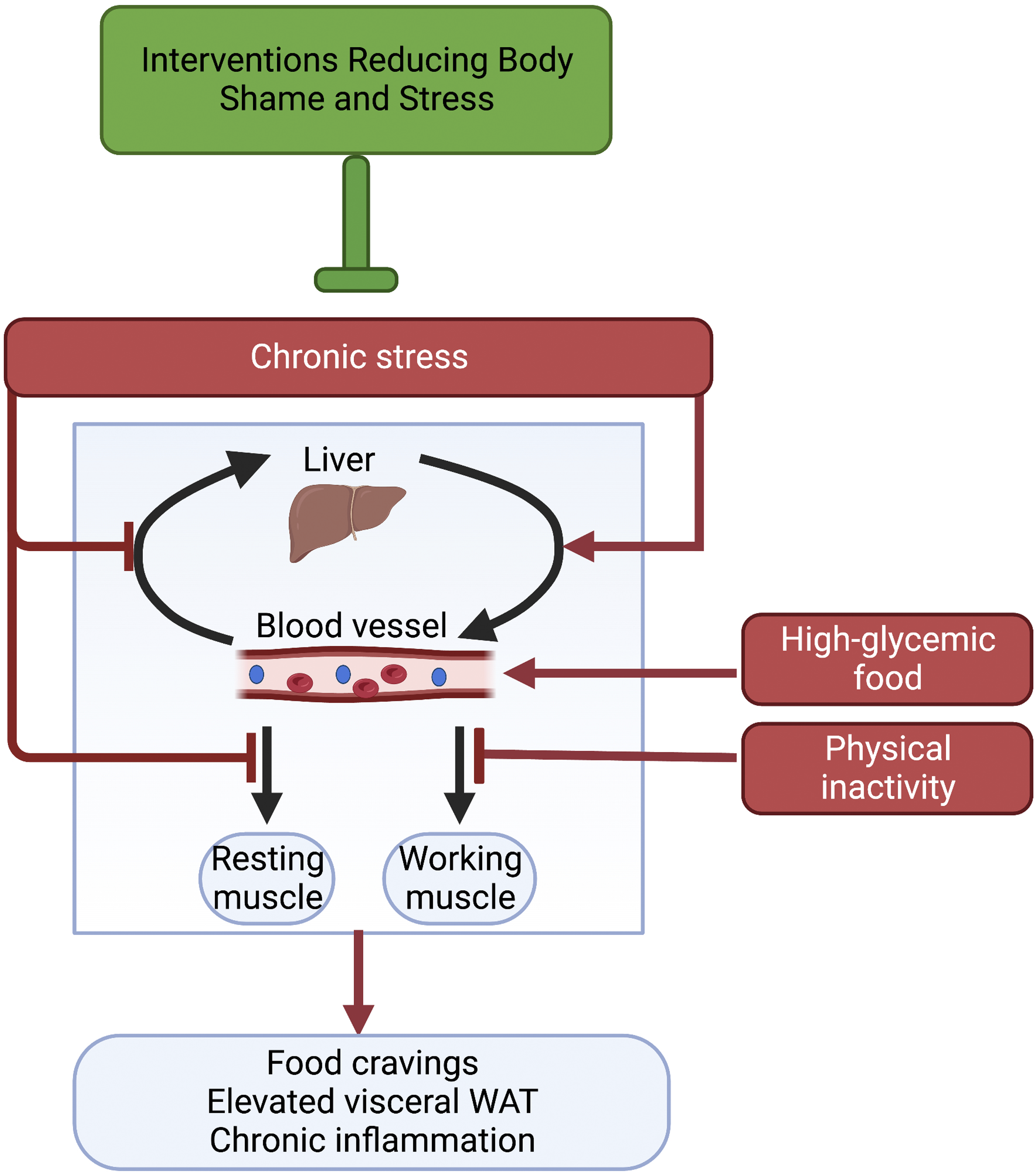
Figure 4 illustrates that both resting and working muscles take in sugar from the blood stream. While resting muscles take in that sugar for the purpose of storage through an insulin-dependent path, working muscles instead operate a second, insulin-independent path to take up sugar for the purpose of immediate use in ATP production. 134 Because this second route for sugar removal operates only in working muscles, physical inactivity fails to draw sugar out of the blood stream this way when chronic stress suppresses insulin-dependent sugar uptake or diabetes leads to insulin insensitivity (Figure 4). Conversely, physical activity improves blood-sugar control even when the response to insulin is compromised in insulin resistance/insensitivity. This mechanistic insight highlights the benefits of concomitant reduction of the chronic stress associated with weight stigmatization (Figure 4) in conjunction with attention to food composition and carefully-dosed physical activity (with its ability to lower blood glucose in individuals with insulin insensitivity or diabetes). 134
In addition to the dietary macronutrient imbalance highlighted in Figure 4, micronutrient deficiency can also contribute to chronic high blood glucose. For the purpose of clarity, Figure 4 does not feature such additional mechanistic effects of the many plant-based micronutrients (phytochemicals) on multiple aspects of glucose transport and signaling. 135 A variety of phytochemicals can stimulate synthesis and engagement of the major insulin-dependent glucose transporter that facilitates uptake of glucose into the liver and resting muscle. 135 Another example is glucose absorption from the intestines into the bloodstream that can be inhibited by various phytochemicals, including those in cinnamon. 136 It is noteworthy that specific herbs, spices, teas, and other phytochemical-rich foods each can have multiple beneficial effects related to various components of the system that controls glucose transport, use, and associated signaling effects, as reviewed for the example of cinnamon by Nuffer and coworkers 137 (2023). Additional sweeping effects of phytochemicals on key metabolic features beyond blood glucose control are detailed in a sub-section below on Micronutrients support metabolic transformation.
The next section highlights selected mechanisms of how chronic stress (or stress relief), modulates the immune response and interacts with effects of food composition and physical (in)activity.
Immune Regulation Has No Simple Relationship With Fat Stores
This section summarizes key principles of how external cues modulate the immune response. Whereas fat stores play a role in some of these pathways, this role is complex and can exacerbate, ameliorate, or have no effect on health outcomes – all dependent on lifestyle factors. This insight further highlights the inadequacy of physical features related to fat stores in predicting health outcomes.
Figure 5 highlights a direct path from the external environment, as affected by institutional barriers and weight stigmatization, to an internal imbalance between the production of pro-inflammatory messengers and the availability of inflammation-resolving messengers, which can trigger pro-inflammatory diseases. Chronic stress (see also Figure 3 above and associated text) contributes to continuous overproduction of pro-inflammatory messengers combined with failure to produce inflammation-resolving messengers. This effect is exacerbated by consumption of an excess of dietary pro-inflammatory omega-6 fats (eg, from today’s mass-produced vegetable oils) and deficiency in dietary inflammation-resolving omega-3 fats (as, eg, found in some types of seafood, nuts, and seeds) and antioxidants (see
138
). Physical inactivity further exacerbates this imbalance by failing to produce internal anti-inflammatory messengers (see also above). This insight can, conversely, be used to design adjustments in the environment that provide concomitant stress relief, tweaks to food composition (see Table 1), and opportunity for physical activity, all of which help restore immune-system function and lower the risk for pro-inflammatory diseases. Schematic depiction of how weight-related stigmatization and institutional barriers to a healthful lifestyle promote chronic stress, a high-density, micronutrient-deficient diet [“unbalanced diet”; see Table 1], and physical inactivity. These factors act together synergistically in causing cravings (loss of appetite control), high blood glucose (loss of glucose control), and visceral fat storage (latter 3 effects visualized in blue boxes on the left) as well as disrupting immune-system balance (visualized by scale image and blue boxes under scales). Specifically, chronic stress and a diet with excessive levels of omega-6 fatty acids produce excess levels of proinflammatory messengers. A diet deficient in omega-3 fatty acids and antioxidants combined with physical inactivity (that fails to trigger endogenous production of anti-inflammatory messengers and antioxidant enzymes) results in insufficient levels of inflammation-resolving messengers. This imbalance promotes system-wide inflammation and proinflammatory disease (red box at the bottom). Created with BioRender.com.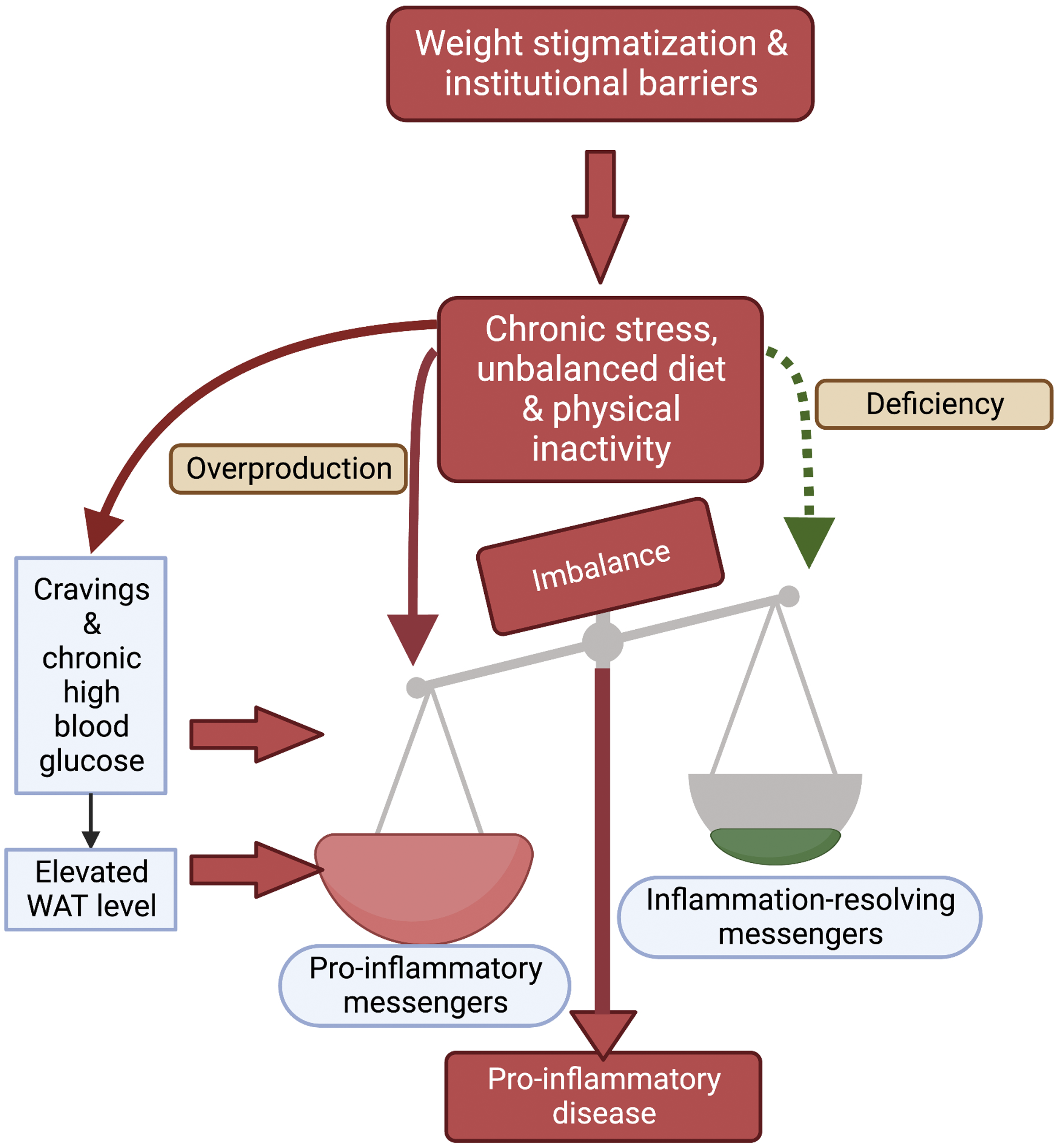
In addition, Figure 5 depicts the path from the same lifestyle cues (chronic stress, unbalanced diet, and physical inactivity) to the induction of cravings, elevated blood glucose and elevated WAT levels (see sections above on these topics). It is depicted how chronic high blood glucose can contribute directly to systemic non-resolving inflammation. 139 For example, when blood glucose level is continuously high, glucose attaches to blood proteins such as the cholesterol-transporting low-density lipoprotein, which activates immune cells that mount a self-attack on arterial walls leading to plaque formation. 140 Furthermore, chronic high blood glucose promotes conversion of glucose to fat and storage of this fat. 141 However, whether or not these fat stores contribute to immune imbalance through production of pro-inflammatory messengers 142 depends, once again, on the environment/lifestyle factors. The section below Programming of metabolic state by external cues: A role for the gut microbiome illustrates how external cues directly – and indirectly through effects on the gut microbiome – regulate system-wide metabolic state, including WAT browning and whether visceral fat depots contribute to immune imbalance or oppose it. Specifically, chronic stress (eg, associated with body shaming), together with unbalanced food composition and physical inactivity, not only increase visceral fat storage but also make these WAT stores a potential health hazard. Conversely, stress relief (eg, from a reduction of body shaming), in conjunction with a balanced food composition and some physical activity, can convert WAT stores to a potential asset for health promotion (see sub-sections below on Thermogenesis as a marker of the metabolic state of fat depots, Micronutrients support metabolic transformation, and Exercise dynamics and metabolic state of fat depots).
In summary, Figure 5 illustrates that systemic inflammation and pro-inflammatory diseases involve an imbalance between overproduction of pro-inflammatory messengers and deficiency of inflammation-resolving messengers that is driven by external cues. The contribution of visceral fat stores to this balance depends on their metabolic state, which cannot be assessed visually. This scenario provides a molecular example for how it could be “possible to be both fat and fit” – as well as healthy. 6 This example thus also supports the need for a shift in focus from body weight or composition to the effect of the external environment on function and impact of fat depots.
The next 2 sections address how external cues can program a sweeping metabolic shift (affecting appetite, energy storage, energy burning, and the functionality of fat stores and the immune system). The first of these sections focuses on the involvement of mitochondrial infrastructure in the programming of this metabolic transition and the second section focuses on the role of the gut microbiome and dietary micronutrients.
Programming of Metabolic State by External Cues: A Role for Mitochondrial Infrastructure
This section focuses on the programming of both appetite/energy intake and the size of the mitochondrial infrastructure available for burning of consumed calories as opposed to storage for later use.16,114,143-145
Programming of appetite
Humans and other mammals carry metabolism-controlling programs that trigger cravings for energy-dense food in response to availability of such foods in the environment (for a review, see 142 ) as well as to seasonal cues. 146 These responses can be thought of as survival mechanisms that presumably only started leading to adverse health outcomes after humans adopted a continuously sedentary lifestyle with a continuous supply of overly energy-dense, micronutrient-deficient food. 147 Originally, ancient humans would presumably not have had continuous access to a bounty of food (eg, fruit with high sugar levels and/or animals with large fat stores) that continuously stimulates appetite and storage of energy in visceral adipose tissue.
Chronic stress further contributes to a stimulation of appetite. After acute stress leads to the above-described emptying of energy stores to support stress defenses, appetite stimulation presumably supports refilling of these stores. Under chronic stress, emptying of energy stores and appetite stimulation in support of refilling of these stores are likely to occur concurrently.
Programming of mitochondrial infrastructure
Occasional availability of a local windfall of energy-dense food in ancestral human environments was presumably associated with a reduced need for physical activity to traverse large distances in search of food. This represents a combination of increased energy intake (of energy-dense, micronutrient-deficient food) with a simultaneous decrease in energy usage for physical activity. Both of these cues serve as signals to dismantle (Figure 6A) a considerable portion of the mitochondrial infrastructure needed for calorie burning (Figure 6B). In other words, the number, size, and activity of mitochondria can either be dialed down or dialed up by external cues. Figure 6 illustrates that this metabolic switch occurs both in skeletal muscle cells (where the mitochondrial capacity for ATP production is modulated; see, eg,148,149) and in WAT (where mitochondria can either be virtually absent or produced in sufficient quantity to cause browning1,14 see also following sub-section). This switch can be thought of as a switching station that, rather than prompting only storage of excess energy, automatically sends a fixed portion of consumed calories down the path to storage when only few mitochondria are present (Figure 6A) – and even when only few calories are consumed. This mechanism further explains the striking thin-fat syndrome, with a significant fraction of energy becoming stored as visceral WAT82,83 even on a calorie-limited diet if this diet has an unbalanced composition with energy-dense macronutrients and insufficient micronutrients (see Table 1 above). This mechanism also further rejects a tacit assumption that elevated visceral WAT must be a result of excessive calorie intake.
150
Schematic depiction of metabolic programming for either (state A, with few, small mitochondria) predominant storage of food calories in central adipose tissue or (state B, with many, large mitochondria) predominant burning of energy from food in mitochondria to ATP and heat (in muscle and other energy-utilizing tissue) or just heat (in BAT). Programming for predominant energy burning involves synthesis of more and larger mitochondria in both muscle cells and in those fat cells that are metabolically active (see section below). Diet, physical activity and psychological stress all exert control over this metabolic programming. This metabolic transition from A to B is accompanied by a sweeping transition in additional key aspects of metabolism (bullet points at the bottom). State B provides health benefits in the context of industrialized society. Created with BioRender.com.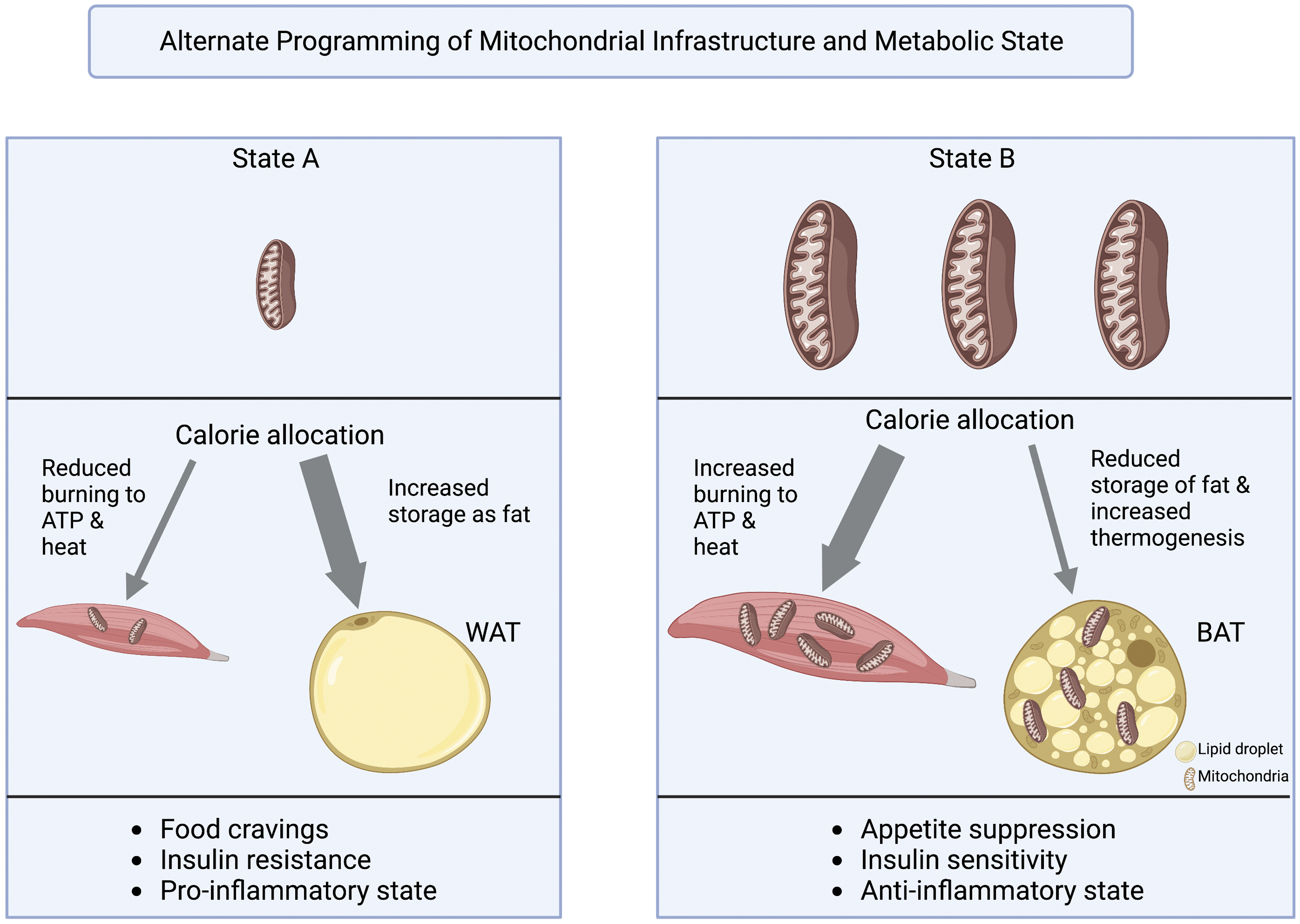
Conversely, low-density, micronutrient-rich foods as well as even modest physical activity both send signals to increase mitochondrial infrastructure and burn incoming calories to ATP and/or heat rather than routing them to storage for later (Figure 6B). State B provides health benefits in the context of industrialized society. The contribution from physical activity as a regulator of such programming offers an explanation for the remarkable benefits of even minimal physical activity that, in and by itself, burns only few calories 151 and may not contribute to visual body changes. Specifically, Lathia and coworkers 151 concluded that “small, cumulative, ‘non-exercise activity,’ such as standing and walking in the course of daily functioning, contributes to avoiding … negative outcomes and increasing general health.” By providing a gene-regulatory stimulus for maintaining the prerequisite mitochondrial infrastructure for energy burning, physical activity is thus effective even when done at a modest level. The potent effects of even small signals promoting robust adjustments in metabolic infrastructure could also help construct a narrative that supporting health by moving regularly does not have to be an imposition (and could be fun; see 152 ) and that visual assessments fail to capture the metabolic benefits produced by modest lifestyle change.
Moreover, the benefits of an augmented mitochondrial infrastructure go beyond energy-burning capacity and include production of antioxidants and other inflammation-resolving messengers by mitochondria. Furthermore, the transformation from state A to state B (Figure 6) includes a switch (i) from appetite stimulation to appetite suppression, (ii) from induction of insulin resistance to promotion of insulin sensitivity, (iii) from emphasis on energy storage to energy utilization (including increased thermogenesis), and (iv) from continuously stimulating inflammation to resolving inflammation when no longer needed. The next sub-section focuses on thermogenesis as a marker for this metabolic state of fat depots.
Thermogenesis as a marker of the metabolic state of fat depots
This section provides further background on the feature of thermogenesis as a marker for the transition between WAT and BAT. This background is used in the remainder of the review to summarize the available evidence for how specific lifestyle factors can affect the transition between WAT and BAT. As stated above, even minimal physical activity stimulates production of mitochondrial infrastructure with its multiple features (see Figure 6) that can protect health in industrialized contexts. This effect of even minimal physical movement (that does not quite qualify as exercise) has been termed non-exercise-activity thermogenesis (NEAT; see153,154 and other chapters in the volume by Leitzman et al 1 155 on Epidemiology and Health). Levine and McCrady-Spitzer 154 posit that “the human being is designed to walk” and that “urbanization and modernization … have reduced NEAT” by acting on “central mechanisms [that] regulate NEAT.”
Figure 6 illustrates that mitochondria of working muscle cells produce ATP and heat as a byproduct in thermogenesis, and that fat cells have the potential to burn fat exclusively to heat (adaptive thermogenesis) without exercise. Brown adipose tissue, BAT, contains many mitochondria that perform such thermogenesis, which may provide support for “long-term body weight regulation” 85 (see also156-161). Moreover, long-term activation of BAT thermogenesis was associated with a lower risk for heart disease, diabetes, and other conditions independent of BMI. 85 As stated in the Introduction, even visceral WAT has the potential to produce mitochondria that perform adaptive thermogenesis after metabolic transformation (“browning”). 14 However, it should be noted that more research is needed to clarify the relative roles of adaptive thermogenesis (as a marker of WAT to BAT transition) vs the other metabolic features listed in Figure 6 (and see also below) in producing health benefits.
In animal models, a diet rich in saturated fat and sugar induced “whitening of BAT and inhibit[ed] beiging of WAT.” 21 In other words, a diet high in energy-dense macronutrients eliminated the mitochondria that confer health-promoting roles to BAT especially in an industrialized context and maintained the features that make WAT a health hazard in this context. Conversely, as a result of a more balanced diet (see Table 1) “transformation of visceral fat tissue [from WAT to BAT] alone without weight reduction [was] sufficient to reduce proinflammatory predominance in adipose tissue” 21 (see also 20 ). This finding is also in line with results of interventions of body-shame-reduction and other interventions that significantly reduced inflammation with no or minimal weight changes (see section above Body shame affects health-relevant behaviors and causes metabolic disruption). As stated in the Introduction, WAT to BAT transformation is associated with system-wide, sweeping effects that include lessened appetite, increased insulin sensitivity, and decreased inflammation. 14 A similar association was found between mitochondrial number/activity and insulin sensitivity in skeletal muscle in human subjects. 149 The finding that external cues can prompt metabolic transformation of WAT to BAT once again shows that the health impact of visceral WAT cannot be adequately assessed from waist-to-hip ratio or waist circumference. This insight could be incorporated into narratives aiming to lessen body shaming.
There is considerable current interest in the therapeutic potential of conversion from WAT to BAT.162-165 Because pharmaceutical manipulation can be associated with harmful side effects, 163 lifestyle measures are an attractive alternative method to stimulate this transformation 166 (see also below). The following sub-sections address the role of food composition – with and without the involvement of the gut microbiome – as well as physical activity and stress relief (also through support of gut-microbiome function) in the adjustment of mitochondrial infrastructure and metabolic health (again with some emphasis on thermogenesis as a relatively well-studied marker for this adjustment).
Programming of Metabolic State by External Cues: A Role for the Gut Microbiome
Overview
Figure 7 depicts examples of the interaction among external cues in the programming for energy burning in muscle and fat cells. We first address the contribution of overall food composition, as well as stress and physical activity, to the programming of metabolic state as mediated by the gut microbiome (and in the following sub-section add further detail on the programming of metabolic state by micronutrients). As stated above, the gut microbiome produces regulators, such as butyrate (see
112
), that control the activity of human genes involved in key aspects of energy metabolism and immune function (see47,48). Due to the interdependence between immune function and gut-microbiome function, external cues (chronic stress, physical inactivity, and energy-dense, micronutrient-deficient food) that disrupt immune function simultaneously impair microbiome function. Conversely, certain foods (box on left) as well as stress relief and regular physical activity (box on right) simultaneously support gut-microbiome function (Figure 7) and immune function.` Schematic depiction of the effect of lifestyle factors on metabolic programming through direct effects of food, as well as effects mediated by the gut microbiome, on WAT browning and metabolic programming. See Table 1 for a description of food composition. Butyrate, short-chain fatty acid; Phytochemicals, see next sub-sections. Created with BioRender.com.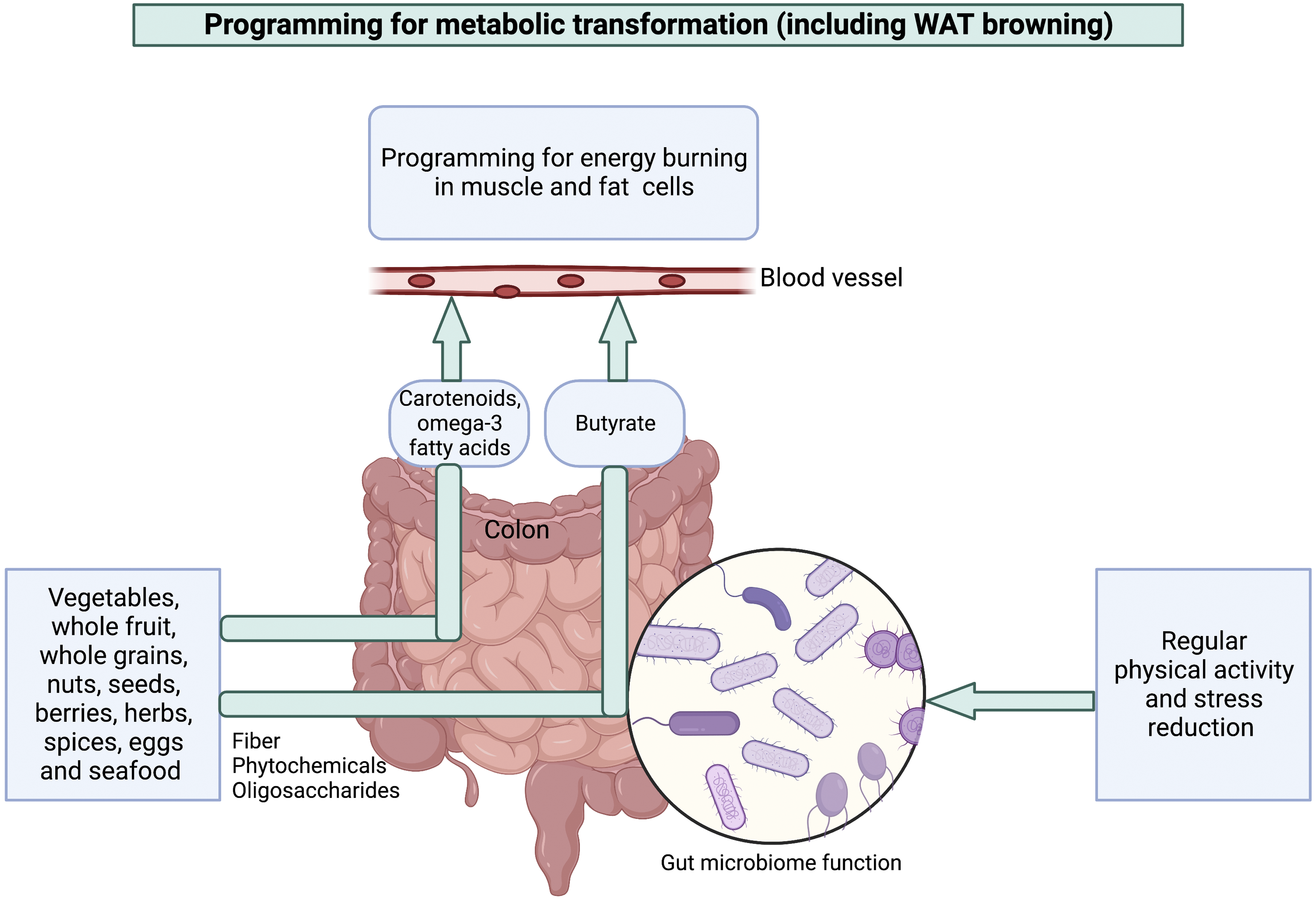
Both macronutrient balance and micronutrient content affect gut microbiome composition. For example, whereas quickly digested carbohydrates (absorbed early in their passage through the intestinal tract) leave no food for beneficial microorganisms in the colon, prebiotic food components (fiber, short-chain sugars, and phytochemicals; see Table 1) support these beneficial microorganisms residing in the colon 48 and their functions in appetite control, energy metabolism, and immune health. 112 Some diets, such as the Mediterranean diet, stand out in being rich in both health-promoting macronutrients and micronutrients and thus promoting WAT browning and fat breakdown as well as lowering fat storage and chronic inflammation. 167 Moreover, and as further detailed in the next sub-section, Figure 7 illustrates multiple effects of micronutrients in supporting metabolic health in an industrialized context.
Micronutrients support metabolic transformation
This section summarizes available evidence on the transformation of fats cells (ie, WAT browning) by dietary micronutrients. This topic is of particular interest due to the potential to make beneficial tweaks to food composition in the presence of systemic barriers by incorporation of more micronutrients. It should be noted that supplements in pill form (i) typically do not provide the mix of micronutrients available in foods 168 and (ii) can have adverse effects when taken as high-dose single supplements. 169 The next 2 paragraphs summarize effects of several micronutrients in increasing adaptive thermogenesis as the feature of fat cells’ metabolic transformation that has received considerable attention in mechanistic work and can thus serve as a marker for this transition. The subsequent last paragraph of this section summarizes broad-ranging effects of a mix of these micronutrients as an add-on to the Mediterranean diet.
Dietary micronutrients that increase thermogenesis in both WAT and BAT fall into 2 classes for direct or indirect effects (Figure 7). The first class can enter the blood stream directly and unaltered (carotenoids,170-175 vitamins, and omega-3 fatty acids 1 176 ; Figure 7). Even though these micronutrients do not need to be modified by the gut microbiome, they nevertheless support the gut microbiome as a result of their anti-inflammatory effects.48,168 The other class of micronutrients has an indirect effect that is mediated by the microbiome; this class of micronutrients serves as prebiotic food for gut microbes and becomes digested to products that enter the bloodstream (eg, butyrate; Figure 7).
Examples of prebiotic dietary factors serving as food for gut microbes that trigger thermogenesis 177 include fiber 178 and a diverse group of phytochemicals. These phytochemicals include phenolics,179,180 such as those found in bitter hops, 181 anthocyanins, 182 as well as turmeric (curry powder), catechins (green tea and dark chocolate), resveratrol (red wine), menthol (mint), and many others.183,184 These examples demonstrate that foods with striking health benefits can also be flavorful and appealing – in stark contrast to common perceptions. 122 This insight demonstrates that health-promoting eating neither has to leave one hungry nor left wanting for pleasurable eating experiences.
Additional phytochemicals with similar functions include sulforaphane (eg, in broccoli and other cabbages) 185 and compounds in citrus fruit zest. 186 In addition to microbiome-mediated effects, such phytochemicals also have direct effects on fat cells. For example, many of these phytochemicals are bitter-tasting and activate bitter receptors on fat cells, which increase thermogenesis – whereas sweet receptors on these cells solicit the opposite effect. 187
As stated above (see also Figure 6), metabolic transformation involves a suite of metabolic features beyond thermogenesis (for a review, see 14 ). There is evidence for direct communication between products of the gut microbiome and the mitochondria in human muscle and fat cells that were the focus of the previous section and Figure 6. Such gut microbial metabolites regulate the production of oxidants and antioxidants in these human mitochondria,188,189 which may play a role in the sweeping metabolic transition described in Figure 6. Recent clinical trials have demonstrated a corresponding variety of metabolic benefits from addition of phytochemical-rich whole foods to a Mediterranean diet. We summarize in the following paragraph significant benefits beyond those of a regular Mediterranean diet from a green-MED diet amended with a combination of green tea (rich in phenolic phytochemicals) and a plant product (Mankai, containing a mix of multiple phytochemicals, including phenolics, carotenoids, antioxidant vitamins, and anti-inflammatory omega-3 fatty acids; for a review see121,168). This body of work included specific attention to the amelioration of gut-microbiome composition. Micronutrient-rich food components (including herbs, species, vegetables, and other foods; see Figure 7) each support multiple interacting clades of gut microbes in a way single supplements cannot.48,168
Compared to a regular Mediterranean diet, the green-MED diet significantly improved patients’ blood levels of phenolics, 190 gut-microbial composition, 191 and appetite control as well as glucose control. 192 This diet also significantly decreased insulin resistance, visceral fat storage 193 (see also 190 ), and chronic inflammation. 194 These and related findings 195 emphasize the key role of interaction between a gut microbiome rich in health-promoting (fermenting) microbial members and a diet that provides phenolics and other dietary components required to support these microorganisms (Figure 7). Moreover, the demonstrated health benefits of berries, that are rich in prebiotic oligosaccharides and phenolics196,197 (see Table 1), are consistent with effects of the green-MED diet. Future studies should examine whether supplementation with these particularly potent prebiotic foods (berries, Mankai, and/or green tea) may also have significant benefits when added to a less balanced diet and may thus be useful in jump-starting metabolic reprogramming and immune-health restoration in scenarios where immediate more comprehensive lifestyle adjustment is unrealistic.
Exercise dynamics and metabolic state of fat depots
It is known that free radicals and other oxidants (oxidative stress) reduce BAT level and thermogenesis. 198 Exercise affects oxidative stress level in a way that varies with exercise dynamics. 47 Excessive exercise, without sufficient rest, enhances oxidative stress and may thus decrease BAT level and thermogenesis. 199 Physical inactivity likewise enhances oxidative stress by failing to induce production of essential antioxidant enzymes. 130 In contrast, regular physical activity with enough time for recovery induces production of antioxidant enzymes and lessens oxidative stress, 47 which may increase BAT level and thermogenesis. Lastly, and while more research is needed in this area, 200 the effect of exercise on fat-cell metabolic status apparently also varies with genetic heritage.201,202
Take-Home Messages From Mechanistic Insight for Intervention Narratives
The overall conclusion from the evidence summarized above is that substantial change is urgently needed in how metabolic disease in industrialized and industrializing societies is framed. Specifically, the insight that fat depots are neither inherently good nor bad – but can instead have either adverse or beneficial effects on metabolic health – warrants a broad-ranging rethinking of health assessment in healthcare settings as well as in personal perspectives. Moreover, the profound role of society-imposed lifestyle in determining metabolic health must be acknowledged. Specifically, recognition of the serious adverse effects of weight stigmatization (eg, in causing chronic stress that interacts with today’s food composition and sedentary environment) can be used to inform efforts to ameliorate public health.
The following specific insights could be used to inform narratives aiming to improve public health. (1) Health status cannot be adequately assessed from visual features but instead depends on the collective function of vital systems (including stress and immune response, the gut microbiome, and all aspects of energy metabolism), all of which are controlled by input from cues provided by the external environment (eg, stress dynamics, food composition, and physical activity). (2) Use of BMI and measures related to waist circumference not only fails to adequately assess metabolic health but can instead impair metabolic health by contributing to body shaming, chronic stress, and their adverse health impacts. (3) Shaming directly interferes with health-supporting behaviors, and shaming-induced chronic stress enhances cravings, blood sugar levels, and allocation of energy to storage as fat. (4) And most remarkably, fat stored around the waist can affect overall metabolic health in either negative or positive ways, also depending on external cues. (5) Because size and function of fat depots is strongly dependent on cues like stress and food composition, stress reduction should be paired with an emphasis on eating differently rather than eating less. (6) The new insight summarized here indicates that subtle cues from the environment trigger remarkably robust adjustments in the physiological infrastructure that supports metabolic health. Concomitant moderate tweaks in several key areas should thus have multiplicate effects. (7) Systemic, society-wide support is needed to enact the full extent of the transitions to a health-promoting metabolic state of fat depots (which is paired with restoration of stress and immune responses, control of appetite and blood sugar, and balance between energy burning and storage).
Future research should ascertain how the information synthesized here can be framed in the most effective way for various audiences.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
