Abstract
Most patients with type 2 diabetes (T2DM) do not meet guideline-driven exercise recommendations. Physical activity (PA) is any form of bodily movement via skeletal muscle contraction. It can include walking, gardening, leisure activity, exercise, or movement for transportation or an occupation. Aerobic and resistance exercise have been well studied and are known to improve glycemic control in patients with T2DM. While data is less substantial, low-intensity PA such as walking, yoga, tai-chi, qigong, and activity for household tasks have also been shown to be effective methods of reducing hyperglycemia. Additionally, PA to break up sedentary behaviors can also have glycemic benefits. Healthcare providers should encourage patients to increase their PA as a more sustainable means of meeting guideline-directed exercise and activity recommendations. Discussing these changes with patients involves identifying motivators, setting goals, removing barriers, monitoring progress, and understanding the patient’s social support and environment.
“PA is more likely to become a lifestyle modification when derived from autonomous motivation rather than from guilt or pressure.”
Introduction
The World Health Organization declares diabetes a global epidemic. 1 In the U.S. the prevalence of diabetes continues to rise, from 10% in 2001 to almost 15% of adults in 2020. 2 It is the most expensive chronic condition in the U.S., with one in every four U.S. health care dollars spent towards people with diabetes. 3 Diabetes leads to many complications, including blindness, kidney failure, and cardiovascular disease such as heart attack and stroke. 1 Poor lifestyle habits, such as low-quality nutrition, weight gain, and lack of physical activity heavily impact the prevalence of diabetes. 4
Large studies, like the National Diabetes Prevention Program (DPP) and the Look AHEAD (Action for Health in Diabetes) trial, have shown the beneficial effects of lifestyle interventions in controlling and reversing diabetes.5,6 The DPP assigned patients an intensive lifestyle intervention for 3 years with the goals of 1) 7% weight reduction, 2) at least 150 minutes per week of moderate physical activity (PA), and 3) less than 25% of daily calories derived from fat. Results showed a 16% decreased risk of T2DM with every kilogram lost and a 44% lower diabetes incidence in those who met the PA goal, even if they did not achieve 7% weight loss.
5
The Look AHEAD trial utilized a similar intensive lifestyle intervention in patients with obesity and T2DM. A1c levels decreased by 0.6% from a baseline of 7.3% after 12 months and 7% of participants had partial or complete remission of their T2DM after 4 years, which was 6 times more than the control participants.
6
Similarly, the China Da Qing Diabetes Prevention Trial showed a 43% lower incidence of T2DM in patients in their intensive lifestyle intervention group even 20 years later.
7
Their lifestyle intervention PA component included increasing leisure activity by at least 1 exercise unit a day, defined as either 1) 30 minutes of mild PA such as slow walking or housework, 2) 20 minutes moderate PA including faster walking, cycling, or ballroom dancing, 3) 10 minutes of strenuous PA such as running, climbing stairs, or playing volleyball, or 4) 5 minutes of very strenuous PA like jumping rope, swimming, or playing basketball.
8
These studies show how tightly linked weight, diet, and exercise are to developing and managing T2DM. Physical activity, as defined by bodily movement produced by skeletal muscle contraction to increase energy expenditure, is an area which requires further attention (Figure 1).
9
Americans spend more than half of their day (55%) in sedentary behaviors such as sitting or lying down and 34% of patients with diabetes report less than 10 minutes a week of moderate/vigorous activity.10,11 The most frequently performed PAs are climbing stairs, walking, and household maintenance.
12
PA can be exercise, leisure activity, or movement required for transportation or an occupation.
9
Exercise, including aerobic, resistance, and flexibility training, is a subset of PA specifically done with the intention of improving physical fitness.
13
While most clinical trials focus on dietary approaches in T2DM management and prevention, small increases in activity can aid and improve glucose control significantly. Subtypes and examples of physical activity.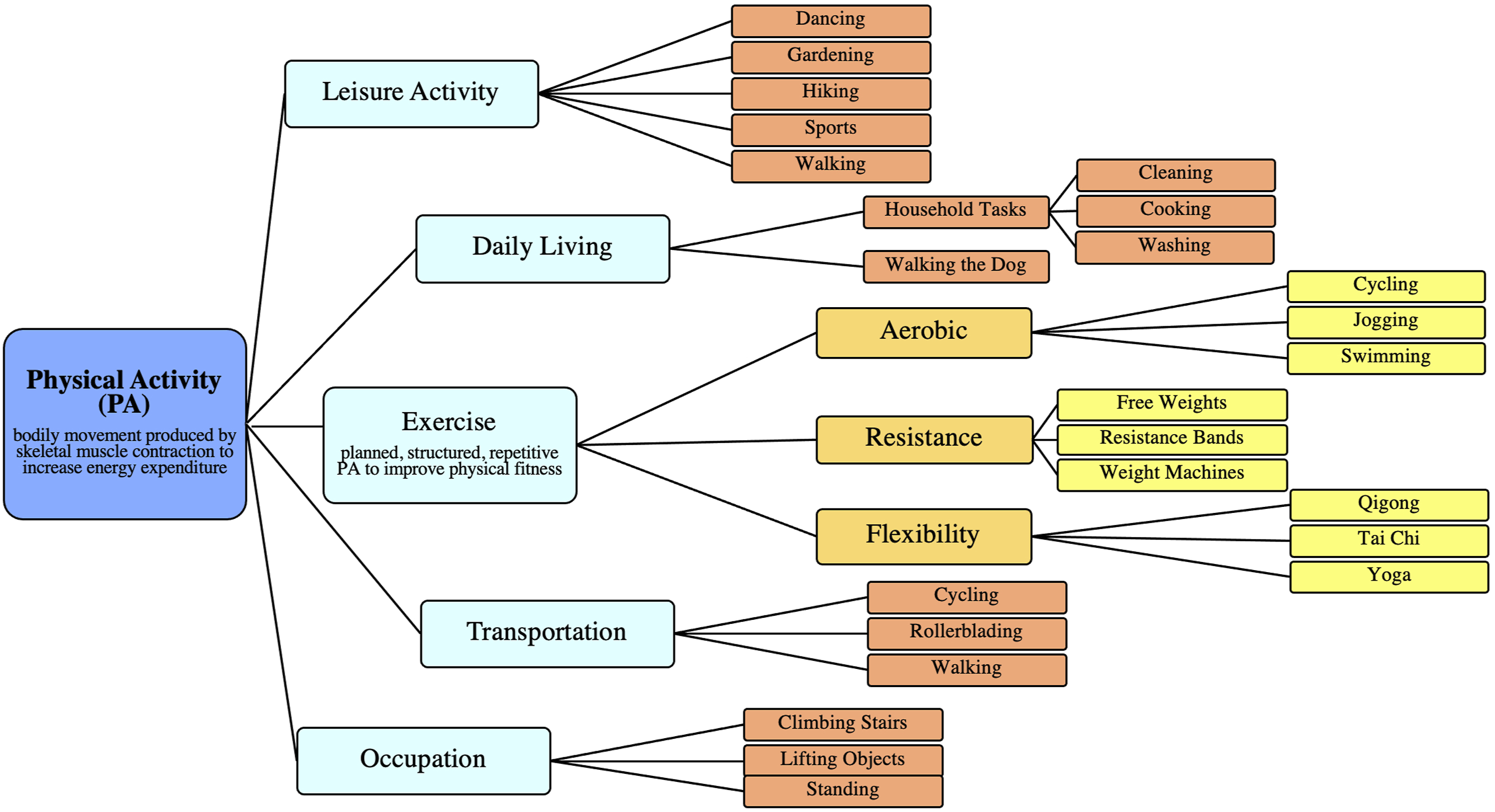
Mechanism of Action
PA is effective in treatment of diabetes and is believed to improve blood sugar control by 1) decreasing inflammation to improve insulin sensitivity, 2) activating insulin-independent glucose pathways, and 3) reducing sympathetic response (Figure 2).12-18 Decreased Inflammation. Elevated blood glucose levels damage vasculature and release inflammatory markers such as C-reactive protein (CRP), tumor necrosis factor-alpha (TNF-alpha), interleukin (IL)-1 within the bloodstream, furthering inflammation and progression of DM2.
15
Build-up of these cytokines has been linked with pancreatic beta cell damage and insulin resistance.
19
PA suppresses these cytokines by increasing levels of IL-6, which decrease TNF-alpha and IL-1 signaling.
15
Through lesser understood mechanisms, PA stimulates muscle enzymes (myokines) to release additional anti-inflammatory markers that in turn reduce inflammatory signals from adipokines, visfatin, and apelin.
20
Utilizing excess calories also prevents the accumulation of pro-inflammatory visceral fat.14,15 These effects decrease systemic inflammation and improve insulin’s ability to activate glucose uptake into the muscles, even hours after the period of activity.
16
Mechanism of Action by which Physical Activity Improves Blood Glucose.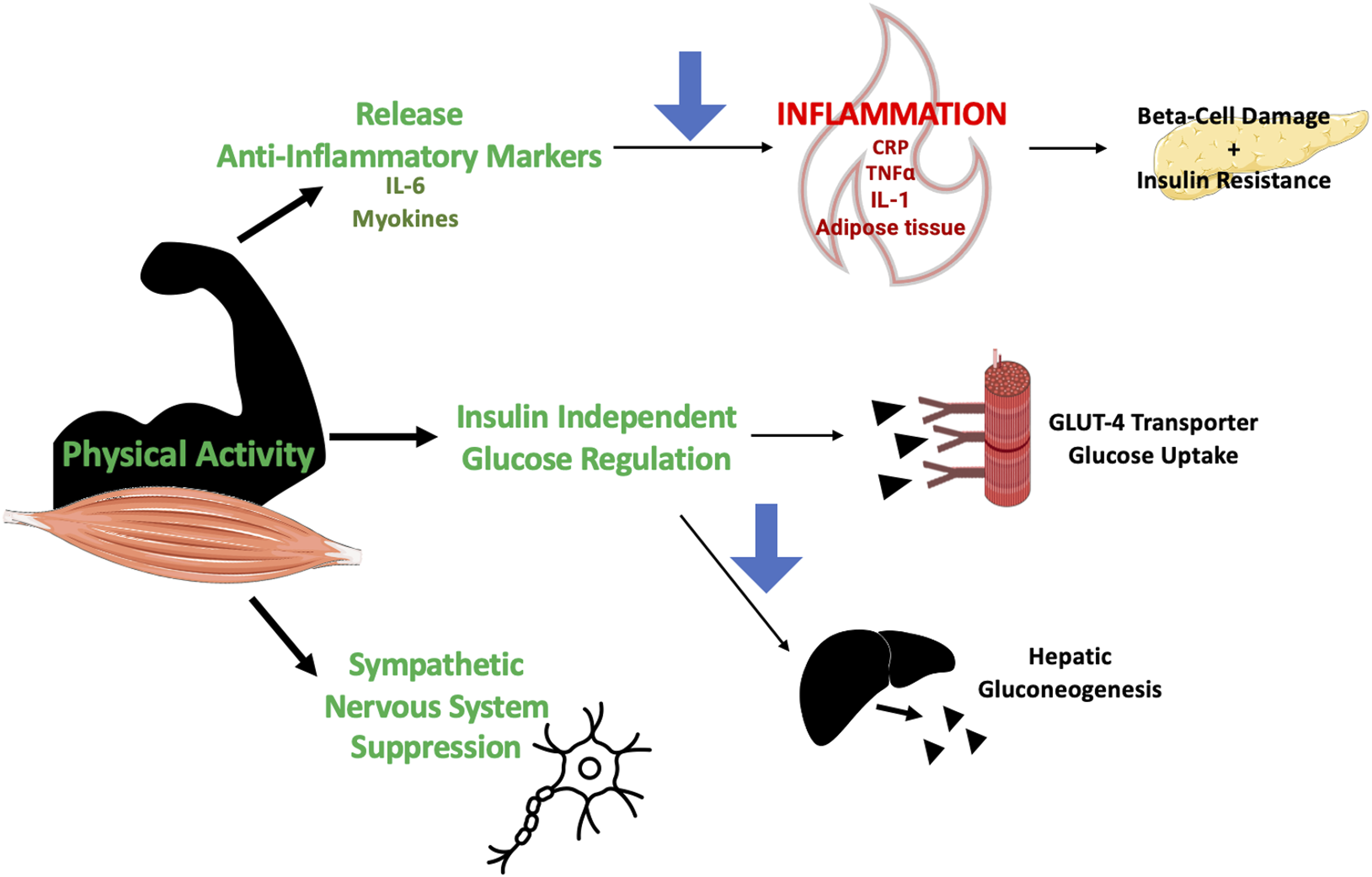
Insulin Independent Glucose Uptake
Not only does PA improve the body’s response to insulin, but it also independently increases glucose utilization. Contraction of skeletal muscles stimulates increased glucose uptake through an insulin-independent process involving glucose transport into muscle cells via the GLUT-4 glucose transporter and hexokinase. Consistent PA promotes euglycemia by 1) upregulating GLUT-4 glucose transporter expression on activated muscle cells to increase the effectiveness of glucose absorption and 2) building muscle mass for a larger glucose reservoir. 16 Additionally, intense PA may suppress hepatic glucose production to decrease fasting blood sugars. A key defect in DM2 is failure of insulin to suppress endogenous glucose production in the liver. PA activates hepatic adenosine monophosphate-activated protein kinase (AMPK), which in turn decreases glucose-6-phosphatase expression and hepatic gluconeogenesis. 21 Through these mechanisms, PA helps lower blood glucose levels.
Suppressed Sympathetic Response
PA may improve glycemic control not only through skeletal muscle contraction as noted above, but also through suppression of the sympathetic nervous system. Stress responses can worsen insulin resistance by sympathetically stimulating neural vasoconstriction of vessels, leading to less glucose delivery to skeletal muscle and therefore decreased glucose uptake. 17 As measured through heart-rate variability, aerobic, resistance, and high intensity interval training have all been shown to decrease sympathetic activity and increase parasympathetic activity. 18 Similarly, PA such as yoga, tai-chi, and qigong emphasize breathwork and relaxation techniques, which may help decrease sympathetic response and improve skeletal muscle glucose delivery.17,22
Current Guideline Recommendations
The most robust data surrounding physical activity lies within aerobic and resistance exercise. Current guideline recommendations by the American Diabetes Association (ADA), European Association for the Study of Diabetes (EASD), American College of Sports Medicine (ACSM), American Association of Clinical Endocrinology (AACE), and the American Heart Association (AHA) advocate regular resistance and aerobic exercise, as well as physical activity such as increasing daily steps to avoid sedentary behavior.23-27 There is consensus of 30 minutes of moderate intensity aerobic exercise 5 days a week (150 minutes per week) or 20 minutes of intense aerobic exercise 3 days a week (60-75 minutes per week), along with 2 to 3 days per week of resistance training. ASCM further recommends high-intensity resistance exercise over low-to-moderate-intensity resistance exercise to better control blood glucose levels and noted mixed outcomes with low-intensity exercise such as yoga and tai-chi. 24 AACE and the ADA recommend avoiding no more than 2 to 3 days between exercise routines to maintain improvement in insulin sensitivity.23,27
Aerobic vs. Resistance Exercise
Aerobic exercise is physical activity which increases heart rate while resistance training utilizes an opposing force to cause muscle contraction. 28 Regular aerobic exercise in patients with T2DM has been shown to improve A1c, blood pressure, triglyceride levels, and decrease insulin resistance. 29 Systematic reviews and meta-analyses have shown fewer daily episodes of hyperglycemia and a reduction in A1c by .3-.7% with aerobic training.29,30 In those with T2DM, resistance exercise improves muscle strength by 10-15%, increases bone mineral density, as well as reduces A1c, blood pressure, cholesterol levels. 31
Comparison of Hemoglobin A1c Improvement in Aerobic, Resistance, and Combined Aerobic and Resistance Exercise.
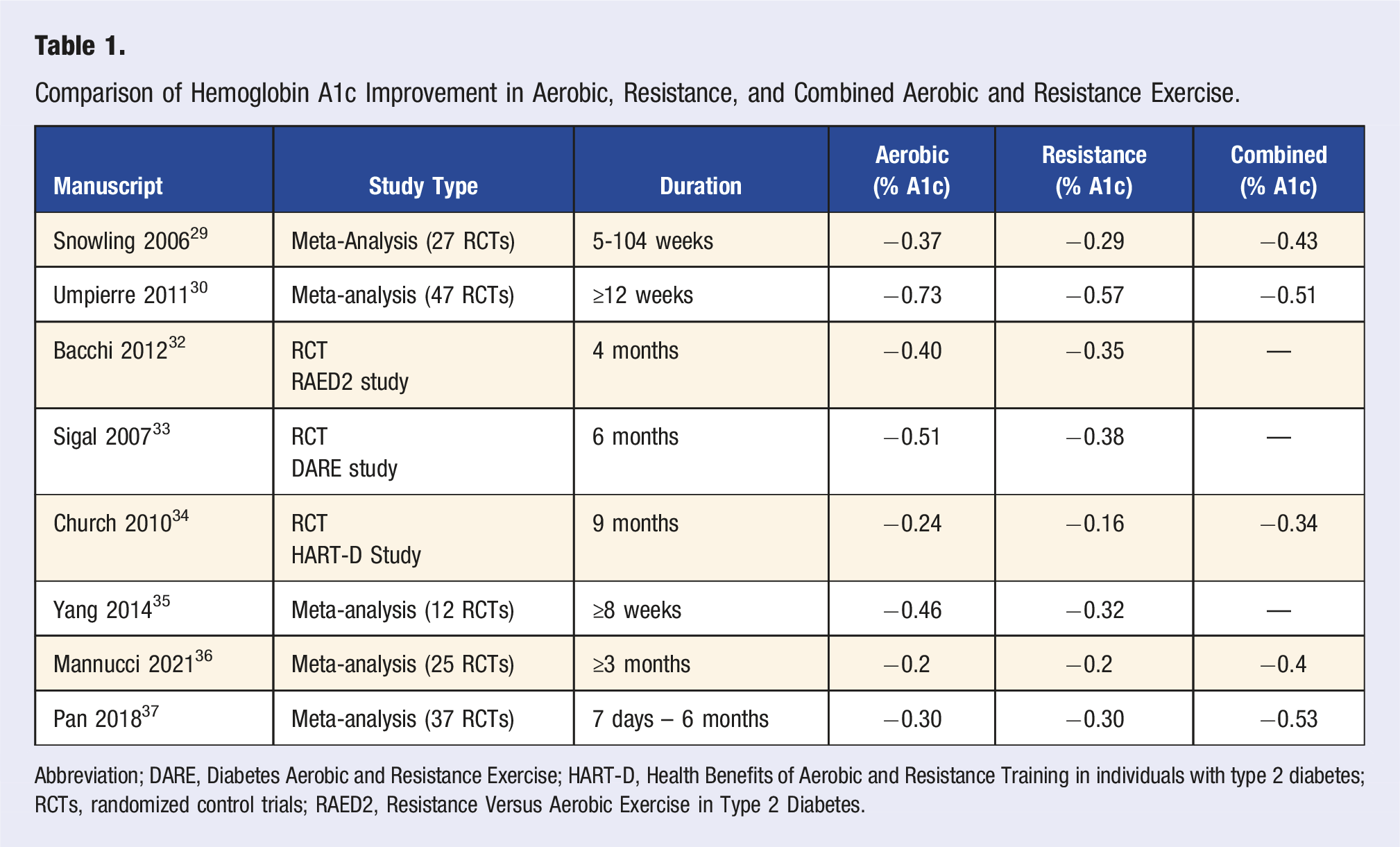
Abbreviation; DARE, Diabetes Aerobic and Resistance Exercise; HART-D, Health Benefits of Aerobic and Resistance Training in individuals with type 2 diabetes; RCTs, randomized control trials; RAED2, Resistance Versus Aerobic Exercise in Type 2 Diabetes.
On the other hand, combined aerobic and resistance training has greater improvements in A1c compared to each form of exercise individually. The Health Benefits of Aerobic and Resistance Training in individuals with type 2 diabetes (HART-D) trial compared the change in A1c after 9 months in 145 diabetic patients who completed either 150 minutes weekly of aerobic training (treadmill), 3 days per week of resistance training (2-3 sets of 12 repetitions for 9 muscle groups), or combined aerobic and resistance training (150 minutes weekly aerobic training and 2 days per week resistance training of 1 set of each of the 9 exercises). 34 Results showed no significant difference in A1c either group (aerobic −0.24%, resistance −0.16%) compared to the control group, but a significant improvement in A1c of 0.34% in the combined aerobic and resistance exercise group compared to the control group. 34 Multiple studies have shown significant improvements in hemoglobin A1c (0.5-0.7%) of patients with T2DM through a combination of aerobic and resistance training.36-39 After an average of 3-4 months, a meta-analysis of 25 studies identified a decrease in A1c of 0.4% with combined aerobic and resistance training and a decrease of 0.2% for aerobic and resistance training individually. 36 Another meta-analysis showed improvement in A1c of 0.53% with combined exercise and an improvement of only 0.3% with either aerobic or resistance exercise separately. 37
Long-term, consistent exercise has greater effects on glucose levels than short term exercise adherence.30,33,34,36,40 One study had 30 T2DM patients perform 90-minutes of regular exercise 3 times a week for 8 years, with this group having an A1c 1.06% lower than the non-exercise group. 40 Shorter studies, like the DARE and HART-D studies mentioned above, showed improvements in A1c of 0.51% after 22 weeks and 0.34% after 9 months, respectively, in patients who participated in combined exercise.33,34 Of note, the combined exercise group in the DARE study completed both the full aerobic and resistance exercise programs each week. 33 One meta-analysis of 47 studies spanning from 3-12 months in duration noted an A1c improvement of 0.7% with structured exercise (combined aerobic and resistance) for more than 150 minutes a week, and individually an improvement of 0.7% with structured aerobic exercise and of 0.6% with structured resistance training. 30 This discrepancy between improvement in glycemic control from 0.34% to 0.7% amongst the described studies could also be due to the benefit of structured classes to improve adherence, as one study revealed compliance with resistance training when supervised was greater than 88% but compliance decreased to 66 to 72% when self-monitored. 31
Progressively increasing the duration and intensity of aerobic exercise can lead to further improvements in A1c.41-43 The Italian Diabetes and Exercise Study (IDES) compared glucose control in 300 participants with DM2 participating in a twice weekly mixed aerobic and resistance training program at either low- or high-intensity training. 41 While both groups had equivalent resistance training, those in the low-intensity group achieved 55% maximal oxygen consumption during aerobic training and those in the high-intensity group performed aerobic exercise at 70% maximal oxygen consumption. The duration of the exercises varied to keep caloric expenditure equal in both groups. After 1 year, those in the high-intensity training group had a 0.17% greater improvement in A1c compared to those in the low-intensity training group. 41 Similarly, a meta-analysis evaluating progression of aerobic training in duration and intensity, initially from 20-60 minutes and increased to 30-90 minutes by the end of the intervention, noted an improvement in A1c of 0.84% compared to the no exercise group after an average of 12-16 weeks, while no change in aerobic training yielded a significant decrease in A1c of 0.45% compared to the control group. 42 Another meta-analysis showed every additional 30 minutes per week of moderate to vigorous aerobic exercise could improve A1c by 0.22%, though further increases above 100 minutes per week did not seem to correlate with further reductions in A1c. 44 Increasing the intensity of resistance training can also improve A1c as a recent metanalysis showed improvement in A1c by 0.61% with high intensity resistance training and by only 0.23% with low-moderate intensity resistance training. 45
Adherence With Exercise
While many may know the benefits of regular exercise, the National Health and Nutrition Examination Survey (NHANES III) of 1600 patients with DM2 found almost 70% of interviewees had either no regular physical activity or insufficient physical activity. 46 Even in the general population, less than 10% of Americans meet current exercise recommendations. 47 The biggest barrier to exercise has been reported as lack of time. 48 While advocating for patients to meet guideline recommendations of exercise five times a week is encouraged, it is likely an overwhelming goal for patients with diabetes to achieve. Especially when so many have lower physical activity durations and number of daily steps compared to those without diabetes. 49 Therefore, more attainable forms of PA should be also be recommended to help accommodate patient’s various baseline activity. Even independent of time spent in moderate to vigorous activity, low intensity PA, defined as 15 minutes per day or 90 minutes per week, can help improve fasting glucose, cholesterol, and increase life expectancy by 3 years.50,51 Low intensity PA and using PA to break up sedentary habits may be more sustainable alternatives to improve compliance with PA. 52
Low Intensity Physical Activity
Walking
Walking can vary from low- to moderate-intensity physical activity based on the pace (slow, brisk, fast). Observational data from 70 000 female nurses in the Nurse’s Health Study showed women who did not regularly engage in vigorous physical activity still had a 20-30% reduction in risk of diabetes when regularly walking at a normal pace (3.2-4.8 km/hr).53,54 A meta-analysis of 20 randomized control trials showed a decrease in A1c by 0.50% with walking interventions in diabetic participants, particularly in supervised walking programs (A1c decrease 0.58%) and non-supervised walking interventions using motivational strategies (A1c decrease 0.53%). 55 Similarly, a more recent meta-analysis of 18 trials noted an improvement in A1c by 0.46% in those with diabetes with mild to moderate intensity aerobic exercise (primarily through walking interventions). 56 Other studies have shown improvements in glycemic control and insulin sensitivity with walking for an hour a day or walking at least 10 000 steps a day.54,57,58
Small, randomized control studies suggest that interval walking may be more effective than continuous walking in patients with T2DM.59,60 In one study, participants either continuously walked at the same pace for 60 minutes or alternated every 3 minutes between low and high intensity walking paces for 60-minute sessions five times a week. After four months, fasting insulin levels decreased by around 20% compared to the continuous walking group and mean glucose concentrations, as measured by a continuous glucose monitoring system, decreased by 8.5%. 59 Oral glucose tolerance testing post walking showed lower spikes in blood sugar in the interval group (1.2 mmol/L) compared to the continuous walking group (1.7 mmol/L), suggesting greater improvements in postprandial blood sugars with interval walking. 60
The benefits of walking span further than glycemic control in patients with diabetes. Based on all-cause mortality and CVD events in diabetics from the Health Professionals Follow-Up Study and Nurse’s Health Study, 3-7 hours of walking a week can result in decreased mortality and CVD events by 40-55%.61-63 These benefits were also observed in patients with diabetes who walked to work every day. 64 Even an additional 500 steps a day may decrease cardiovascular and all-cause mortality by 2-9%. 65 Of note, these studies were observational trials, and therefore, the impacts of walking cannot be directly causational to the mentioned outcomes.
Yoga
Yoga aims to integrate the mind and body through the practice of active and restorative physical movements and postures. A review of 23 studies showed improvements in fasting sugars of 7-33% and postprandial sugars from 6-39% from baseline after participating in yoga interventions. 66 On average, these studies were 12 weeks in duration with a 1-hour yoga session, 6-7 times a week. Another systematic review of 45 randomized control trials noted a decline of 26 mg/dL in fasting sugars and 0.5% in hemoglobin A1c. 67 Multiple reviews have shown improvements in sugars and insulin resistance, as well as in blood pressure control and cholesterol profile.66-68 However, challenges to interpreting this data remain as there are different forms and intensities of yoga. Current studies vary in study quality and involve small sample sizes. Further randomized control trials with larger participant groups are required.
Tai-Chi
Like yoga, tai-chi involves centering the mind through a combination of physical activity and respiration. It is a form of traditional Chinese exercise consisting of a flow of several movements throughout the practice. 69 A meta-analysis of 14 studies identified an average decrease in fasting blood sugar of 1.4 mmol/L and decrease in A1c by 0.7%. 70 These declines were seen with tai-chi exercise for 80-120 minutes per week. Another review showed overall improvement in quality of life and BMI (average decrease of 1.5 kg/m2) in patients with T2DM who participated in regular tai-chi. 71
Qigong
Qigong is a Chinese fitness form utilizing multiple repetitive movements with a focus on breathing. A review of 16 studies showed a decrease in A1c of 0.8% compared to the non-exercise group but no significant difference compared to those in other aerobic exercise, like jogging or walking. 72 Studies averaged 3-6 months and involved 30-60 minute sessions during the week. These forms of light PA are useful also for their improvements in balance and gait, minimizing fall risk. 13
Household Tasks
Minimal data exists to evaluate the impact of PA through household tasks such as cleaning, vacuuming, and repairs. However, further studies should be pursued given the frequency of PA performed as walking (80%), household tasks (69%), and gardening (65%), and the positive association between health-related quality of life and increased physical activity. 73 Metabolic equivalents (METs) are a useful measurement of activity intensity and are based on the amount of oxygen uptake. METs allow household activities to be compared, for example, washing dishes is 1.8-2.5 METs, cooking is 2.0-3.0 METs, and cleaning is 2.3-3.8 METs. 54 One observational study of 15,000 Chinese men found an inverse relationship between increased hours performing household PA and incidence of T2DM. 74 Other observational studies show no specific correlation between hours performing household tasks and risks of diabetes, but that increased daily METs, which household activities contribute to, are inversely related with incidence of T2DM. 75 A study of elderly persons noted those who took more than 8-10,000 steps a day and reached an intensity of greater than 3 METs for more than 20-30 minutes a day had a lower risk of metabolic syndrome. 76 Additionally, a study of French adults with T2DM noted increased physical activity, primarily from domestic chores (an average of 630 MET minutes per week), was associated with fewer cardiovascular complications and better self-perceived health. 77 This suggests that recommending physical activities equating to a certain METs threshold could be beneficial for metabolic health.
Physical Activity to Break up Sedentary Behavior
Sedentary behavior is defined as sitting or reclining with low energy expenditure of less than 1.5 METs. 78 More time spent in sedentary behaviors was associated with an increased risk of developing diabetes and poorer metabolic and inflammatory profiles in those with T2DM.78-82 Multiple studies have looked at brief interruptions of sedentary behavior with PA, and identified improvements in post-prandial sugars, insulin levels, cholesterol, and weight circumference.83-86 Adults with T2DM had improvements in post-prandial sugars and triglyceride levels when interrupting prolonged sitting with 3-minutes of light walking or simple resistance activities every 30 minutes. These values were measured as area under the curve over the 7-hour intervention, with a mean decrease in post-prandial sugars of 39%, insulin levels of around 36% in either activity group compared to continued sedentary behavior, and a decrease in triglycerides of 40% after simple resistance activities. 83 Insulin (23% less) and post-prandial glucose levels (0.5 mmol/L or 0.8 mmol/L less), as measured by glucoses after an oral glucose tolerance test, were also improved in patients who did low-intensity or moderate-intensity walking for 2 minutes every 20 minutes of sedentary behavior over a 5 hour intervention. 84 Similarly, light walking for 5 minutes every 30 minutes improved post-prandial glucose (0.3 mmol/L) and blood pressure (4 mmHg). 85 Every additional set of ten sedentary breaks in a day was associated with improvements in blood glucose (0.57% lower), insulin (4.19% lower), high density lipoprotein (HDL) cholesterol (0.01 mmol/L higher), and waist circumference (0.83 cm lower). 86 Even prolonged standing rather than sitting may be beneficial as it involves postural muscle contraction and causes changes in vascular tone and blood pressure. 87 One study utilizing continuous glucose monitors found blood sugars after standing for an afternoon (185 minutes) was reduced by 43% in those standing compared to sitting. 87 Further studies are needed, but evidence suggests breaks in sedentary behavior throughout the day improves glycemic control.
PA and Postprandial Hyperglycemia
As noted above, PA has been shown to improve postprandial hyperglycemia.83-85,88-94 Postprandial hyperglycemia can impact micro- and macro-vascular complications of diabetes more than elevated fasting glucose levels, making adequate glucose control after meals beneficial. 91 Six episodes of 1-minute high-intensity walking 30 minutes before meals, referred to as “exercise snacking,” reduced postprandial sugars (−0.7 mmol/L) and 24-hour glucose concentrations (−0.6 mmol/L), compared to moderate exercise for 30 minutes prior to dinner. 92 A meta-analysis of 27 studies evaluated the effects of brief breaks (1-5 minutes every 20-30 minutes) of PA throughout the day against a single continuous bout of exercise (20-60 minutes) before a meal on post-prandial sugars in diabetic and non-diabetic participants. 93 Brief PA breaks throughout the day proved to be more effective than continuous exercise at reducing postprandial hyperglycemia (standardized mean difference −0.36 mmol/L). 93 A narrative review of 11 studies evaluating diabetic patients found equivalent or superior post-prandial glucose control with multiple short 10 to 15 minute PA breaks just before or after meals compared to one episode of 30 to 60 minutes of continuous exercise. 94 These data suggest accumulating PA, whether through multiple breaks throughout the day or PA breaks just prior to or after meals, to disrupt sedentary behavior can improve post-prandial sugars.
PA Duration Over Intensity
The total duration of time involved in physical activity may be more effective than the intensity of physical activity. One study compared patients sitting for 14 hours daily against those replacing some of the sitting time with cycling for 65 minutes a day or those performing a combination of light-intensity walking and standing for 4.5 hours a day. Glucose levels over 24 hours, measured as area under the curve, improved in both groups with light-intensity walking and standing (−711 min × mmol/L) and those who cycled for an hour (−591 min × mmol/L) compared to the control. However, insulin resistance was only significantly reduced in the light-intensity group (HOMA-IR2 = 1.89) compared to the exercise (2.06) and sedentary groups (2.16). 88 Similarly, light exercise for 60 minutes daily reduced hyperglycemic episodes within 24 hours by around 50% compared to no exercise, while high intensity cycling for 30 minutes daily did not have a significant reduction in number of hyperglycemic episodes compared to the same control. 89 Another study showed participants had improved glucose concentrations by performing 5 minutes of moderate intensity exercise every hour for 12 hours compared to those who performed moderate intensity exercise for 60 minutes. 90 For glucose control, increased physical activity duration and frequency rather than intensity of activity can help attenuate glucose concentrations.
Clinical Recommendations
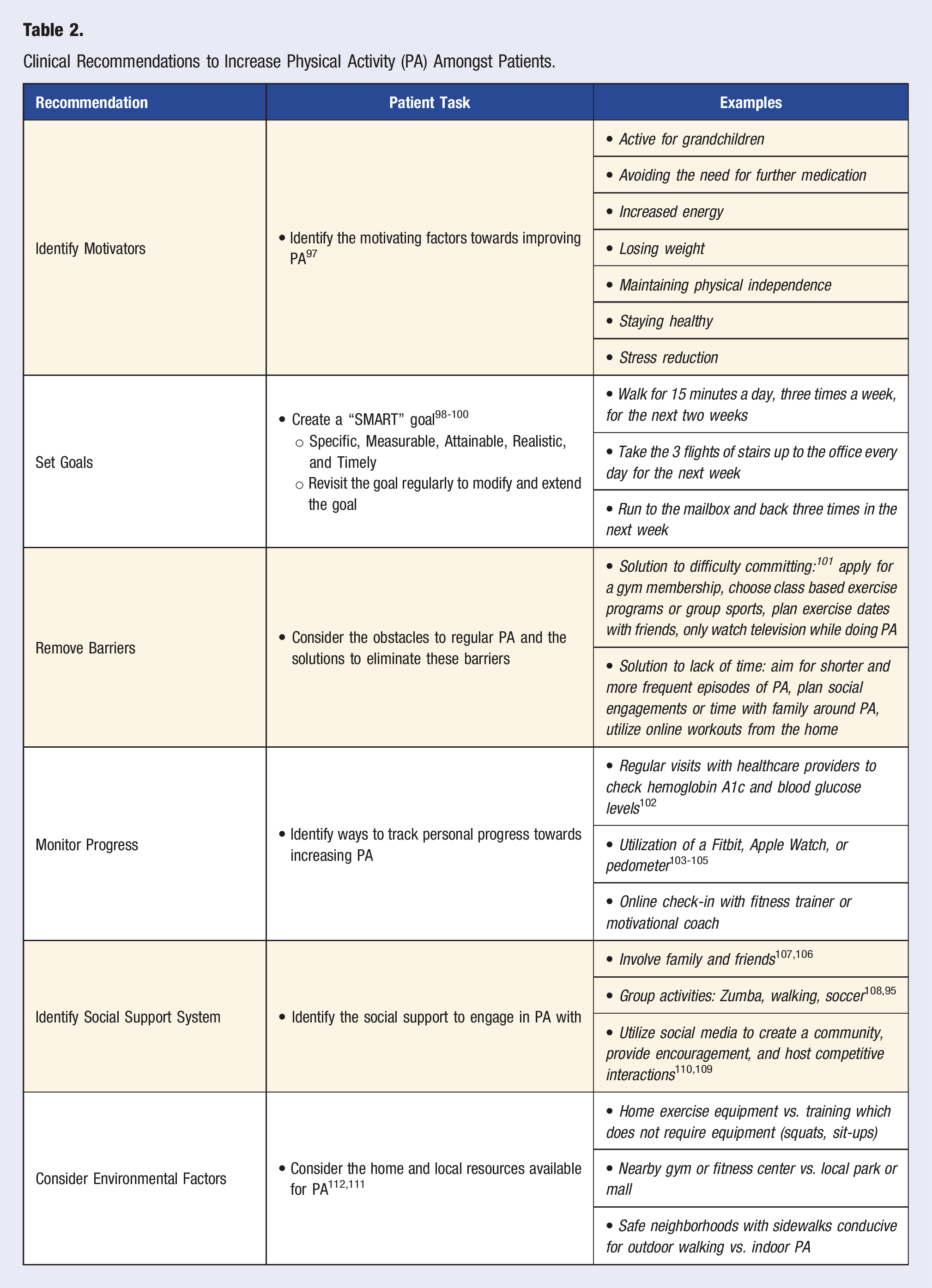
Clinical Recommendations to Increase Physical Activity (PA) Amongst Patients.
Identify Motivators
Identifying a patient’s motivating factors for physical activity can help improve adherence. PA is more likely to become a lifestyle modification when derived from autonomous motivation rather than from guilt or pressure. Patients with diabetes who successfully included PA in their daily routine often noted that they eventually developed an intrinsic drive to be more active and no longer felt it to be a chore. 113 Motivating factors include: staying healthy, avoiding further medications, losing weight, stress reduction, maintaining independence, increased energy, and remaining active for younger family members. 97
Setting Goals
Discussing a specific aim or goal for patients to achieve can make increasing physical activity more tangible. Helping patients establish a specific “SMART” (specific, measurable, attainable, realistic, and timely) goal has been shown to improve A1c and make them more likely to achieve their aim.98-100 Recommendations for counseling patient SMART goals are that 1) the goal is generated by the patient rather than the provider. Providers can guide areas to focus on, but the specific goal should be determined by the patient. 2) The goal is a behavior rather than a result. For example, choosing to “exercise more often” does not offer a means to achieve the task, so patients should be encouraged to determine a controllable goal such as “walking for 15 minutes a day three times a week for the next 2 weeks.” 3) As noted in the prior example, the goal should be specific in terms of the behavior, duration, how, when, and where it will be done. 4) The goal should be one that the patient is confident they will be able to achieve. Studies have shown increased PA behaviors through interventions which promoted self-efficacy, or a patient’s confidence in their ability to achieve their goal. 114 The task can start as a short term goal such as the example above to improve adherence and then reassessed in the future to continue.115,116 Patients should share these goals with all their healthcare providers. Interestingly, in a study of SMART goals developed during a diabetes self-management program, only 20% of participants shared their physical activity or dietary goals with their outside healthcare provider. 100 Clinicians can create an environment where patients can share their PA goals by openly asking patients if they may already have lifestyle goals they are aiming to achieve.
Removing Barriers
To make healthy lifestyle changes, such as increased physical activity, identifying and removing barriers towards these changes is essential. Commitment devices, such as purchasing an annual gym membership, scheduling exercise dates with friends, or only allowing vices such as watching television while doing PA, are tools which can eliminate obstacles such as lack of motivation. 101 Directing patients towards reliable online resources and local opportunities available can also improve adherence. 98 Examples include calls or video visits with motivational coaches, personal trainers, online workouts, and community PA programs like yoga or soccer. Developing a routine and reducing the number of active decisions towards a change can also help ease the transition. 117 For example, wearing gym clothes to sleep to overcome the obstacle of changing into exercise clothing in the morning can help make PA more attainable. While developing PA goals with patients, it is important to consider barriers to achieving the aim and problem solve solutions.
Monitoring Progress
Regular visits with healthcare providers help motivate patients to continue with lifestyle changes. Testing blood glucose and hemoglobin A1c provide visible feedback regarding changes in lifestyle and develop trust in the validity of these PA modifications when improvements are made. 102 Providing tools to track progress, such as a pedometer or Fitbit, a wearable accelerometer, may improve total daily steps. One study of patients in Sweden noted an increase of around 990 steps a day after 6 months utilizing a pedometer, but only an increase of 144 steps per day after 12 months. 103 However, this was significantly improved compared to the control group, who averaged 890 fewer steps after 12 months compared to their baseline. Similarly, a 6-month study of 200 patients who participated in a Fitbit based game found participants walked 1700 steps more than the control group, though only 50% of participants were consistently wearing their Fitbit by the end of the trial. 104 Another study showed sending patients with diabetes or pre-diabetes daily text reminders or asking for a daily step goal made participants 17% more likely to wear their Fitbit and increase daily steps (791 more steps) on the days they submitted a step goal. Despite these temporary improvements, the overall average number of steps by participants over 6 months did not change. 105 These studies show simply wearing a Fitbit or pedometer may not be enough to increase physical activity, but when combined with regular check-ins with healthcare professionals and with a set goal, may improve physical activity. However, most studies evaluating the impact of wearable devices on health outcomes are small and further studies and standardized evaluation are required.118,119
Social Support Systems
Social support from friends and family plays a large role in physical activity. Studies showed regular PA was more likely in a family when the children or pets were also involved in the activity.106,107 Group activities, such as tennis, zumba, walking, and soccer can encourage commitment by creating a sense of community.95,108 In-person interactions are not always feasible and studies have shown support through social media and mobile applications are effective alternatives.109,110 A systematic review of 53 publications found studies using interventions through social media, most commonly Facebook, increase PA through support, encouragement, and competitive interactions amongst friends or group members. 109 A review of 27 studies utilizing smartphone applications, which are more easily accessible on-the-go to offer motivational reinforcement, self-monitoring, and physical activity games, found significant improvements in health-related outcomes (blood pressure, cholesterol, overall fitness) and an average attrition rate of only 17%. 110 Social support, whether through direct involvement with family, friends, and community, or via social media can help increase and maintain physical activity.
Environmental Factors
Studies have shown a link between the environment and people’s levels of physical activity. Physical environmental factors, such as home exercise equipment, proximity of a PA facility, and walkability of the neighborhood, account for up to 18% of the variance in amount of PA in amongst T2DM.111,112 Suburban, higher income residents are more likely to achieve PA recommendations compared to rural, lower income residents due to accessibility of exercise spaces such as parks, sidewalks, and malls, as well as decreased fear of injury or concern for safety. 120 Resistance training may require more equipment, facilities, and involvement of professional trainers, which could impact accessibility. 36 Therefore, recommendations could include physical activity like group sports, walking, or exercises which do not require props or leaving the home, such as squatting and push-ups. It is important to discuss a patient’s home environment to specifically tailor recommendations for their physical activity.
Safety Considerations: Hypoglycemia
With increased physical activity leading to improvements in weight and A1c, patients who are on medications to treat T2DM should also be monitored for hypoglycemia. Particularly, insulin and insulin secretagogues such as sulfonylureas should be monitored, and a medication dose reduction should be considered. Taking these medications prior to exercise can lead to further decline in blood sugar.24,26 Other oral medications, which have become the gold standard of treatment for T2DM, such as metformin, sodium-glucose co-transporter 2 (SGLT-2) inhibitors, dipeptidyl-peptidase 4 (DPP4) inhibitors, and glucagon-like peptide 1 (GLP-1) agonists do not have the same risk of hypoglycemia and can be continued. 26
Limitations
Most of these studies were done on small patient populations with data presented as pooled meta-analyses. Studies which looked at low-intensity PA in the home, such as through household tasks were causational. Those which were randomized control trials evaluated glucose levels over 24 hours and the long-term benefits of physical activity need to be elucidated. 24 More continuous glucose monitoring studies to evaluate impacts of timing of exercise are needed since A1c is not sensitive enough to determine daily fluctuations in glucose. 121 Additionally, given the nature of free living PA, improvements in blood sugar may be small if each individual PA is evaluated independently, but when patients are encouraged to increase their PA through multiple avenues, there may be large improvements in glucose control.
Conclusion
PA consists of leisure activities such as gardening and sports, activities of daily living such as cooking and cleaning, activity for occupation or transportation, and exercise. While guideline recommendations advocate for 150 minutes of aerobic exercise and 2-3 days of resistance training, most patients with T2DM are unable to meet these guidelines. Current data shows improvement in glycemic control with low-intensity PA such as walking, yoga, tai-chi, qigong, and activity involved in household tasks. Short periods of PA, such as standing or walking, to break up sedentary behavior has also had glycemic benefits. Data is limited, but there is significant support that any form of movement can be helpful in management of T2DM. Healthcare providers should encourage patients to be more active through increasing PA and help them achieve their goals by understanding their social support and environment, identifying motivators, removing barriers, and monitoring progress regularly.
CME/CE Article Quiz
American College of Lifestyle Medicine (ACLM) members can earn FREE CME/CE credit by reading this approved CME/CE article and successfully completing the online CME/CE activity. Non-members can earn CME/CE for $40 per article. Visit lifestylemedicine.org to join the ACLM.
AJLM CME/CE Articles and Quizzes are offered online only through the American College of Lifestyle Medicine and are accessible at lifestylemedicine.org/store. ACLM Members can enroll in the activity, complete the quiz, and earn this CME/CE for free. Non-members will be charged $40 per article.
A Passing score of 80% or higher is required in order to be awarded the CME/CE credit.
Supplemental Material
sj-pdf-1-ajl-10.1177_15598276231180541 – Supplemental Material for The Impact of Physical Activity in Patients With Type 2 Diabetes
Supplemental Material, sj-pdf-1-ajl-10.1177_15598276231180541 for The Impact of Physical Activity in Patients With Type 2 Diabetes by Sandhya Rao Bassin and Reshmi Srinath in American Journal of Lifestyle Medicine
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
XXX.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
