Abstract
Continuous glucose monitoring in a person with diabetes who is embracing lifestyle changes can be greatly impactful in numerous ways. There are many factors that have been identified to affect blood glucose, and for someone who may be implementing the six pillars of lifestyle medicine, their blood sugar may require closer monitoring. Lifestyle medicine interventions may lead to improved glucose levels or even remission. The continuous glucose monitor allows people to see glucose levels, trends, and how rapidly their glucose levels are rising or dropping, empowering them to make connections with how they feel and how their actions are impacting their blood sugar, as well as giving information about how medications may need to be adjusted or deprescribed. CGM, when appropriately used, can help determine how to best manage diabetes, optimize outcomes, minimize risks, and empower the person and healthcare team.
“The pillars of lifestyle medicine can profoundly impact people withdiabetes, leading to a reduction of medication doses, discontinuation of certain medications, or even diabetes remission.”
Introduction
Technology has improved dramatically for the care of a person with diabetes and the role of continuous glucose monitoring in a person who is embracing lifestyle changes may be especially significant. At least forty-two factors have been identified that affect blood glucose—these include medication, food, biological factors such as sleep and stress, physical activity, environmental, and behavior/decisions. 1 While traditional blood glucose monitoring used to be the primary option to check point-in-time glucose levels, continuous glucose monitors (CGM) allow for the person with diabetes to see glucose levels, trends, and how rapidly their glucose levels are rising or dropping. This allows a person to be empowered, making connections between how they feel, their glucose level, and internal or external factors. Self-care behaviors related to the six pillars of lifestyle medicine have shown positive outcomes in people with diabetes.2-4 Having this glucose data and making the connections allows for more real-time decisions to be made in terms of medication or self-care behavior changes.5,6
Lifestyle Changes Improve Health Outcomes in Diabetes Patients
Sufficiently intensive lifestyle interventions have shown to induce diabetes remission and it is recommended that intensive lifestyle changes be the primary modality for type 2 diabetes care with remission as the goal. 7 The 2013 Obesity Guidelines describe a 2%-5% weight loss resulted in lower fasting glucose and a .2-.3% reduction in A1c. Reductions of .6% to 1% were seen in people who lost 5-10% of weight, leading to a reduction in diabetes medications. Additionally, an average weight loss of 2.5 kg to 5.5 kg in adults who were overweight or obese reduced the risk of developing type 2 diabetes by 30% to 60%. 8 In people with type 2 diabetes, as lifestyle changes are implemented, their requirement for glucose lowering medications may change, resulting in needing lower doses or achieving remission. Studies have shown CGM use is associated with reductions in A1c and increased time in range. 9 A mixed qualitative and quantitative survey-based study of people with type 2 diabetes who started using CGM within the previous 12 months found that CGM use led to an increased awareness of glucose levels, encouraging respondents to improve nutrition and physical activity. 10 CGM technology is one modality to evaluate how glucose levels are responding in real time. This paper will describe the CGM devices currently available, how the ambulatory glucose profile can help clinicians adjust and potentially deprescribe medications, and recommendations for CGM use.
Description of CGM Systems
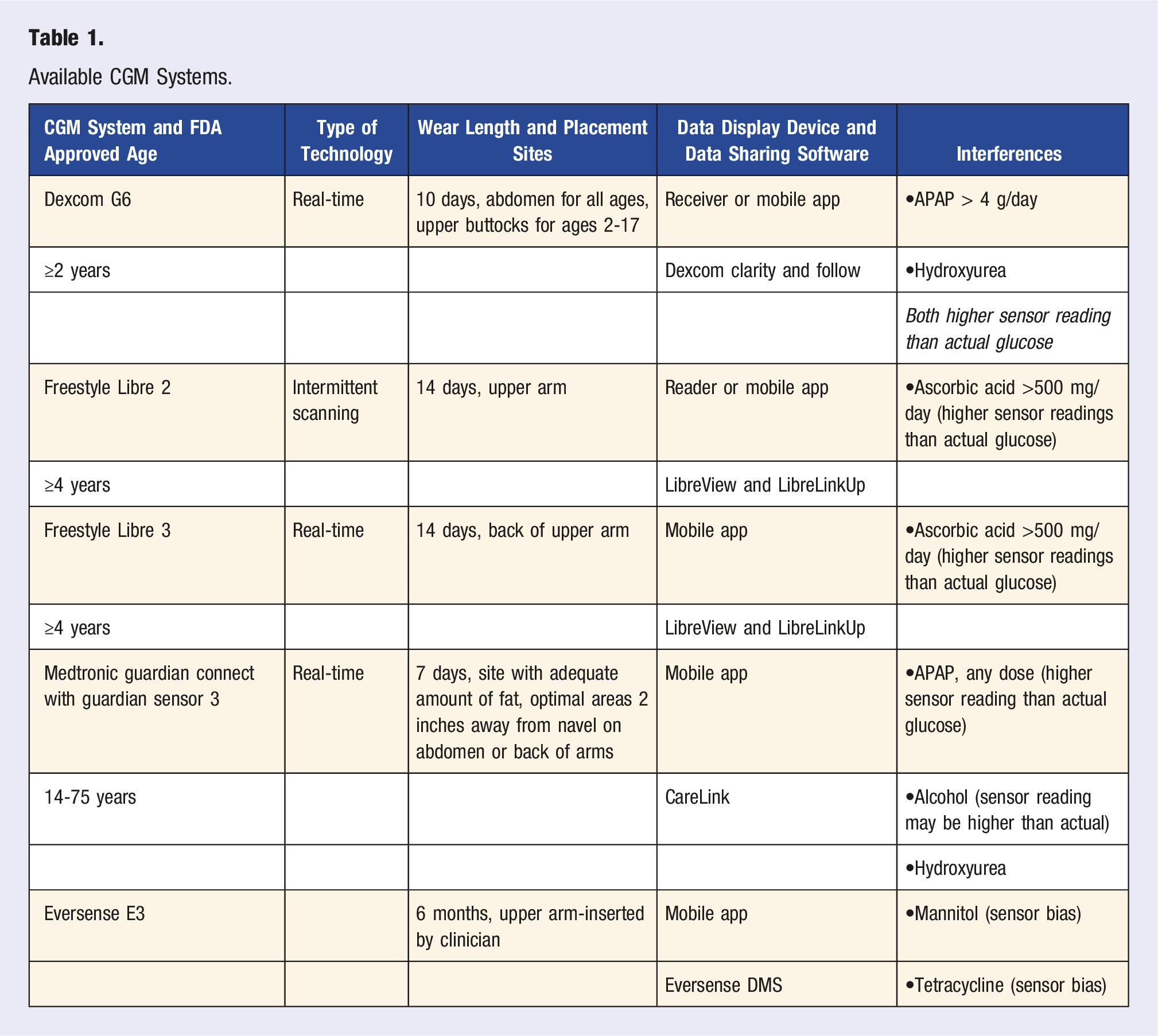
The CGM device measures interstitial glucose concentrations almost continuously (every 1 to 5 minutes) via a sensor that is inserted subcutaneously. 11 An enzyme coated wire measures interstitial glucose through generation of an electrical current when glucose reacts with the enzyme glucose oxidase. 12 The interstitial glucose measurement is close to plasma glucose levels unless glucose levels are rising or falling rapidly. 13 The meters either need to be manually scanned or the information is transmitted wirelessly to a reader or cellphone. Additionally, some devices have the capability to share information with family/friends or medical providers. 14
In addition to professional CGM devices, there are two types of personal CGM devices available: real-time CGM (rtCGM) and intermittent scanning (isCGM). The professional CGM is generally owned by the clinic, is placed on the person with diabetes in the clinician’s office, and is worn for 7 to 14 days. Depending on the type of device, the data may be blinded or visible to the patient. The person then returns to the clinician to download and view the data to assess for patterns and trends. The professional CGM is generally used by people who may not have access to personal CGMs, or by those who prefer to wear the device only for a short time. It can help identify periods of hypo or hyperglycemia and there is some evidence that wearing a CGM intermittently may be useful in lowering A1c.13,15,16 Of the two personal CGM devices, the difference between rtCGM and isCGM is that the isCGM requires the person wearing the sensor to scan their sensor at least one time in eight hours in order to obtain all the data.
Recently, there has been a new category of CGM for systems that meet strict accuracy requirements. These CGM devices can be designated as integrated continuous glucose monitoring (iCGM), and requirements include the following accuracy requirements: when glucose is between 70 and 180 mg/dL, at least 70% of sensor reading are within +/− 15% of laboratory-confirmed glucose levels; when glucose readings are <70 mg/dL, at least 85% are within +/− 15 mg/dL; and when the glucose is >180 mg/dL, at least 80% are within +/−15%. Freestyle Libre 2, Freestyle Libre 3, and Dexcom G6 have been approved with the iCGM designation thus far.13,17,18
CGM System Features
Available CGM Systems.
Ambulatory Glucose Profile Report
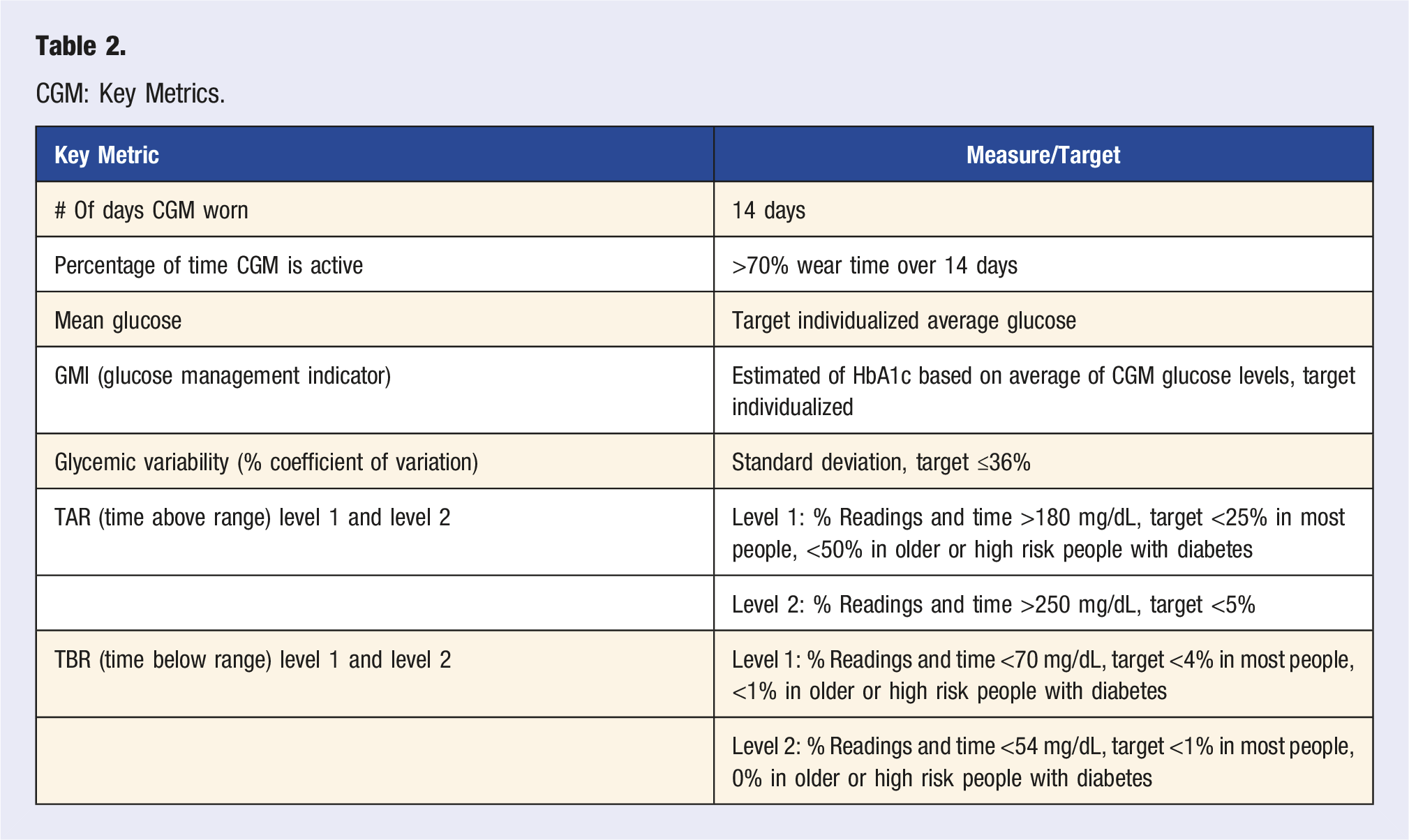
CGM: Key Metrics.
Guidelines Recommendations
The American Diabetes Association (ADA) and The American Association of Clinical Endocrinologists (AACE) provide guidance on utilizing CGM in certain patient populations. Their recommendations are similar but not exactly the same. The ADA states CGM should be offered for diabetes management in youth and adults taking multiple daily injections or continuous subcutaneous insulin infusion who are capable of safely using the device, and should be used daily if possible. Additionally, the ADA recommends that CGM can be used for adults taking basal insulin and can help achieve A1c targets in people who are pregnant. 13 AACE recommendations are similar, with CGM recommended in people taking intensive insulin therapy, youth with type 1 diabetes, individuals with frequent, severe, nocturnal hypoglycemia or hypoglycemia unawareness, and may be recommended in people with type 2 diabetes taking insulin or women with gestational diabetes who are not taking insulin. 5
Lifestyle Medicine, CGM, and Medications to Treat Diabetes, and the Potential for Deprescribing
When selecting pharmacotherapy in a person with type 2 diabetes, as well as when considering deprescribing, an individualized approach taking into consideration comorbidities, treatment goals, and patient preferences should occur. Diabetes medications with cardiorenal benefits may need to be continued in adults with established or high risk of atherosclerotic cardiovascular disease, heart failure, and/or chronic disease. Additionally, weight management goes hand in hand with diabetes management, and the likelihood that a diabetes medication will cause weight gain or the addition of medication that helps contribute to weight loss should also be considered. Other factors, including patient preferences, complexity of treatment regimen, and cost are important as well.13,25
Lifestyle medicine interventions may lead to remission in people with certain health conditions including diabetes. As a patient’s health conditions are improving, there is a likelihood that medications need to be judiciously evaluated, and more than likely doses will either need to be reduced or the medication may need to be discontinued. Understanding the pharmacology and clinical evidence sets the foundation for safe prescribing practices. Deprescribing is defined as the “planned and supervised process of dose reduction or stopping of medication that might be causing harm, or no longer be of benefit.”26,27 In a paper describing best practices to consider when a person with diabetes is embracing a low carbohydrate diet, the stop light approach to adjusting medications is described. This approach states that certain medication may be safe to continue (biguanides, GLP1RA, and DPP4 inhibitors), basal insulins and thiazolidinediones doses may need to be decreased, and medications including sulfonylureas, meglitinides, SGLT2 inhibitors, alpha glucosidase inhibitors, and bolus insulins may need to be stopped. 28
A qualitative case series discusses protocols for medication deprescribing in people with type 2 diabetes who are making lifestyle related modifications. 29 While there is minimal guidance how to approach deprescribing, more resources are being published to help guide clinicians on how to approach this.
Conclusion
The pillars of lifestyle medicine can profoundly impact people with diabetes, leading to a reduction of medication doses, discontinuation of certain medications, or even diabetes remission. CGM technology is a powerful tool that allows real-time data to be seen by the wearer and shared with the patient’s healthcare team, allowing for meaningful conversations around the connection between glucose levels and various aspects of the patient’s life. While additional research is necessary to further illuminate the relationship between CGM technology, lifestyle changes, and diabetes management, CGM when appropriately used can help determine how to best manage diabetes, optimize outcomes, minimize risks, and empower the person and healthcare team.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
