Abstract
“Due to the increasing demand for animal-free medications, there is potential for the pharmaceutical industry and manufacturers to be inclusive of the needs of diverse patient populations.”
Introduction
Globally, millions of individuals avoid animal-derived ingredients for a multitude of reasons, including health, animal cruelty, dietary preference, environment, religious, or personal beliefs.1,2 While it is easier to find food and clothing without animal origins, these groups often have to compromise when it comes to medications. The main reasons include a lack of transparency in labeling, the complicated ingredient names in pharmaceuticals and the challenges in obtaining information around the origins of an ingredient. 3
Recently, this topic has garnered greater attention due to the increasing popularity of veganism, with an estimated 79 million individuals around the world adhering to this lifestyle. 1 Over the years, interest in veganism has been steadily rising due to more social and global awareness involving religion, animal welfare, health, environment, and medicines. 2 Despite the increasing interest in veganism, there is still a scarcity of information in the public domain surrounding medicine and the availability of medications with animal-free ingredients. It is estimated that approximately 3 out of 4 medications contain animal-derived ingredients. 3 This topic was driven by anecdotal patients’ stories in the general media and inquiries to community pharmacists. The purpose of this comprehensive qualitative review is to assess current literature on the topic of animal-free medications and understand the landscape for key stakeholders (i.e., patients, healthcare providers, and pharmaceutical manufacturers). The topics were determined based on our literature search and a discussion among the authors. Potential solutions for key stakeholders’ pain points will be addressed.
A comprehensive literature search was conducted on PubMed and Google Scholar from inception to June 2022 to identify key articles surrounding the topic of animal-free ingredients and medications. Articles were included if providing novel insight or data into the topic.
Stakeholder Perspectives
Religious Concerns
There are an estimated 1.9 billion Muslims, 4 1.2 billion Hindus, 5 535 million Buddhists, 6 14.7 million Jews, 7 and 6 million Jains in the world. 8 Many of these religions and others have dietary restrictions based on their beliefs. Pork is forbidden in Jewish and Muslim diets, so porcine-derived medications, such as heparin, can present an issue. In Islam, gelatin is only acceptable if it comes from a halal source. In Hinduism and Jainism, there is a strong emphasis on the practice of Ahimsa, or non-harming, as a spiritual and practical tool, which along with beliefs in karma (cause and effect) and samsara (reincarnation), guide Hindus and Jains following a vegetarian or vegan diet. Jehovah’s witnesses forbid the storage and transfusion of blood and its products, such as albumin. 9 There may be some exceptions in cases of an emergency, however, it is important for pharmacists and other healthcare providers to acknowledge the impediment that exists in choosing medications for some religious individuals.
Animal Cruelty Concerns
Animal testing is typically required in the drug development process. In the United States alone, the estimate is about 26 million animals annually, 10 but the total number of animals utilized in drug development is difficult to ascertain. The U.S. Food and Drug Administration (FDA) requires animal testing for all new drugs, which must include 1 rodent and 1 non-rodent, to ensure the medication is safe before proceeding to human clinical trials. 11
Since animal testing is currently required by the FDA in the drug research and development process, 11 certain patients may refuse to take life-saving medications due to their moral commitments. In a survey from VeganMed, 3.6% of respondents would refuse to take an animal-derived drug that would be crucial for survival. This creates a potential conflict regarding medical care. 12 Various long-term medications such as icosapent ethyl (Vascepa), a medication used to lower triglycerides, is made from fish oil, and enoxaparin (Lovenox), an anticoagulant used to treat or prevent blood clots, is made from pork. Additionally, propofol (Diprivan), an aesthetic agent, and certain influenza vaccines contain ingredients derived from chicken eggs, which cause some patients to refuse therapy due to either allergies or moral concerns. 13
The Animal Welfare Act in the United States and the Animals (Scientific Procedures) Act in the United Kingdom are some of the many regulations that address concerns of animal cruelty. 14 Rats and mice are primarily used in most laboratory testings; however, they are excluded from the definition of “animal” under the Animal Welfare Act. 15 Even with regulations in place, the use of animal testing is still a problem since 96% 16 of drugs fail in human trials after having promising results in the animal testing phase. To assert the morals around the continued use of animal testing, the scientific community follows the 3 Rs (replacement, reduction, refinement) campaign. It advocates the search for replacement of animals with non-living models, reduction in the use of animals, and refinement of animal use practices. 17 Although the 3 Rs campaign has been the standard of practice globally for over 50 years, the United Kingdom has started to utilize the 6 principles framework that has a deeper focus on the moral concerns regarding animal research. 18 The 6 principles framework is built on the foundation of 2 core values, social benefit and animal welfare, which include the principle of no alternative method, the principle of expected net benefit, the principle of sufficient value to justify harm, the principle of no unnecessary harm, the principle of basic needs, and the principle of an upper limit to harm. 19
Several companies are creating alternative pathways to limit animal testing and animal by-product usage in drug development. One example is iBio, a developer of next-generation biopharmaceuticals and a pioneer in sustainable, plant-based biologics manufacturing who uses the FastPharming System to deliver their products. This system utilizes vertical farming, automated hydroponics, and glycan engineering technology to rapidly deliver gram quantities of high-quality monoclonal antibodies, vaccines, bioinks, and other proteins. 20 Another company that aims to reduce the use of animal testing is Genoskin. They use donated human skin for pharmaceutical and cosmetic testing. With this innovation, the generated data is more relevant to drug development and research than what is collected with animal testing. 21 With the increase of advanced technology and methods, the use of animal testing can continue to decrease. 22 During the COVID-19 pandemic, many patients inquired about the presence of animal-derived ingredients in the vaccines. 23 As of June 2022, the first plant-based vaccine has been approved for human use developed by Medicago and GlaxoSmithKline combining coronavirus-like particles in plants with an adjuvant. 24
Health-Related Concerns
Animal-derived ingredients in pharmaceuticals, nutraceuticals, and cosmeceuticals can have severe consequences for consumers. In 2008, many heparin batches imported from China were contaminated, killing 81 people and severely injuring hundreds of others. 25 Heparin is a popular anticoagulant; however, the active ingredient is derived from pig intestines, which is often procured internationally due to the high demand and lack of resources in the United States. 26 Contaminated heparin batches were traced back to 12 companies in China that added oversulfated chondroitin sulfate (OSCS) to their products. 25 OSCS are derived from animal cartilage chondroitin sulfate and can appear to resemble heparin during lab testing, therefore traditional screening tests in the United States cannot differentiate between the affected and unaffected lots. 27 These heparins were made in unregulated labs and farms in rural China, and were suspected to add OSCS to increase volume and drive up sales. 25 Indeed, ingredient procurement from other, less-regulated countries can be detrimental to the health of consumers and increase the risk of illness and disease.
Approximately 3% of the general population has an alpha-gal allergy, which is caused by tick bites. 28 The main management of alpha-gal allergy requires avoidance of red meat to prevent anaphylaxis. In addition to anaphylaxis, affected individuals can present with skin and gastrointestinal symptoms, such as abdominal pain, vomiting, and diarrhea. 29 In a 2020 case report, a 45-year-old woman with an alpha-gal allergy and a history of hypothyroidism was unable to immediately start any common thyroid hormone replacement formulation due to potential traces of nonprimate mammalian meat components in thyroid hormone medication. She was later prescribed a plant-based compounded levothyroxine medication, which exhibited good clinical results. 30 As such, many patients have described in the public domain how minute concentrations of animal ingredients in medications can induce allergic reactions. 31
Environmental Concerns
Animal-derived ingredients in medications are directly and in-directly connected to environmental concerns. One example is the large-scale cultivation and fishing of marine life for the extraction of the fish oil industry worth 2.3 billion USD. 32 The demand for fish oil has increased over the years due to higher intake recommendations from health agencies, leading to over 100 confirmed cases of marine life extinctions in the world. 33 However, studies have shown that increasing omega-3 fats intake, including fish oil supplements, does not significantly improve cardiovascular health or reduce the risk of death from any cause. 34 Aside from the fish that are used in the production of fish oil, bycatch are also contributing to the disruption of marine ecosystems. Bycatch are fish and other ocean wildlife that get caught and killed during the process of fishing for a specific species, and are estimated to be as much as 40% (or 63 billion pounds) globally each year. 35
Although our marine ecosystems are affected, farm animals (cows, pig, sheep) by-products (lactose, magnesium stearate, glycerin, lanolin) are among the more popular sources of inactive ingredients in pharmaceuticals, supplements, and cosmeceuticals. Animal agriculture is one of the largest contributing factors to greenhouse gas emissions and deforestation. 36 Reducing animal consumption would free up resources, such as land and feed, that are used to occupy and sustain these animals. Further, animal agriculture uses approximately a third of the drinking water available worldwide. 37 Livestock manure can also pose a huge threat to humans due to its emission of ammonia which can combine with other air pollutants, such as nitrogen and sulfates, to produce harmful particles. Inhaling these particles can cause a variety of health issues, including heart and lung disease. 38
Animal-Free: Legal and Labeling Landscape
Several countries have tried to address the consumer needs around animal-derived ingredients in pharmaceuticals.
In the United States, the American Medical Association published policy H-100.947 Animal-Derived Ingredients to increase social and cultural awareness of animal-derived medical products. This policy urges manufacturers to disclose all ingredients and components pertaining to both active and inactive ingredients on their product label and annotate any derived from an animal source. 39 The policy encourages more labeling transparency; however, it is not a requirement for manufacturers to follow this practice. Since there isn’t an official manufacturer database currently available, the only way to avoid animal-derived products is to reach out to manufacturers directly.
Other than the AMA policy, current United States federal regulations related to general disclosure of the presence of animal-derived properties are not well defined. However, in March 2019, the FDA released the “Medical Devices Containing Materials Derived from Animal Sources” guidance document to update the policy regarding the use of animal-derived materials in medical device manufacturing. Their objective is to clarify and update information on the use of International Organization for Standardization since animal-derived materials may carry a risk of infectious disease transmission. 40
In the United Kingdom, the Medicines and Health Products Regulatory Agency has a database containing summaries of product characteristics, descriptions of the medicinal product’s properties and conditions attached to its use, but it currently does not include labeling medicines that are suitable for vegetarians or vegans.41,42 On the other hand, the National Pharmaceutical Regulatory Agency in Malaysia requires having a label to declare the source of animal-derived ingredients (active and/or excipient), including starting material and gelatin only if applicable, but it is not practiced frequently by manufacturers. 43
In terms of practicing vegetarians, India is ranked top of the world with 38% of the total population. 42 However, 98% of Indian pharmaceutical capsules are animal-derived.44,45 When it comes to food, current regulations in India require a distinction between vegetarian and non-vegetarian using a green or brown dot label. 46 However, this does not necessarily extend to pharmaceuticals and cosmetics. 47 In 2013, the India Supreme Court ruled cosmetics and drugs cannot be treated the same as food when it comes to labeling them with a red or green mark to distinguish between vegetarian and non-vegetarian ingredients. 48 Later in 2016, Maneka Gandhi proposed to the Health Ministry to make it mandatory to replace animal-based gelatin capsules with cellulose-based capsules and mark them with a green dot. However, due to pricing and stability issues, the expert panel recommended against the proposal 2 years later. 49 India’s Central Drugs Standard Control Organization (CDSCO) states that information for excipient origin should be provided when addressing the quality of finished formulations of bulk medications, but there is no set law to distinguish between animal- or plant-based ingredients. 50
Although different policies and guidelines for labeling ingredients in medical products exist around the world, manufacturers often fail to implement these suggestions due to a lack of strict regulations or requirements for declaring the origins of ingredients. This serves as a problem to patients and HCPs who need to obtain information about animal-derived products for health, religious, or moral reasons.
Vegan vs Animal-Free Labeling
Recently, many different Vegan certifications have been utilized for food, supplements, and cosmetics (Figure 1). As such, the Vegan certification should address the needs of various consumer types. Veganism is a way of living that seeks to exclude any form of animal exploitation, whether it be for food, clothing, or cosmetic products.
51
Vegans refrain from using any partly or wholly animal-derived ingredients.
51
Because the U.S. FDA regulatory process for drug approval typically involves animal testing,
52
no medication can truly be labeled as “vegan.” Hence, vegan certification would be inappropriate for pharmaceuticals. It would be more appropriate to classify these products as “animal-free,” which implies that a product does not contain any animal-derived ingredients. The term animal-free is also more inclusive of all consumer types. It also stands to legally protect the manufacturer from any misbranding related claims. Animal-Free vs Vegan Certification.
Independent Certification
Independent certification allows products to stand out and facilitates consumers to make informed decisions based on their medication needs. Some examples of independent certification include animal-free certification, halal certification, and kosher certification. For companies that want to give their vegan products a competitive edge, the animal-free certification is an effective way to do so. The label is easy to understand and helps products differentiate themselves from the competition. Examples of Certified Animal-Free products include Lysulin and Cinnulin PF. 53
For a product to be halal certified, each of its ingredients must be traceable and cannot contain pig, alcohol, blood, predatory animals, human parts, or insects. Animal-derived ingredients must come from permissible animals that were slaughtered per a specific method according to Islamic law. In countries with Muslim majorities, individuals can easily find medications that are halal certified. However, other countries need to continue to develop the halal certification practices by outlining a clear, well-regulated halal accreditation procedure. With the increasing awareness of halal pharmaceuticals, countries such as Singapore, Australia, and the United States are beginning to invest in halal pharmacies. 54
Kosher certification implies that the product is prepared in adherence to the dietary laws of Judaism. 55 One of the most well-known agencies that certifies Kosher products is the Orthodox Union (OU), which granted kosher certification to Pfizer’s Elelyso for Type 1 Gaucher and whose logo appears on approximately 70% of America’s kosher certified foods. Products with a kosher symbol perform 20% better compared to competing products that lack the kosher symbol. 56
Implications for Health Care Professionals
Physicians, pharmacists, and other health care professionals (HCPs) play a crucial role in being a reliable source of information for patients and their medications. The American Medical Association Code of Medical Ethics states that physicians “must recognize responsibility to patients first and foremost” and “shall respect the rights of patients.” 57 Similarly, the American Pharmacists Association Code of Ethics for Pharmacists, states that a pharmacist should “respect personal and cultural differences among patients” and “encourages patients to participate in decisions about their health.” 58
More measures need to be taken to better teach and embrace these “Code of Ethics” at the professional education level. Queen’s University Belfast (QUB) in the United Kingdom surveyed its students in their final year of pharmacy school on their knowledge and opinions of veganism. 59 Most students acknowledged that learning about veganism is important for their future role, however, only a minority believed they had received adequate training on the matter at hand. Some faculty members at QUB also acknowledged that they focus more on drug contraindications and allergies, rather than personal and religious beliefs when educating students on how to select appropriate medications for patients. They recommended providing more guidance on topics such as considerations for excipients for vegan patients, 60 implications of religious and cultural beliefs, 9 ethical and practical considerations, 61 and databases that include details on product ingredients and nutritional information.
Prescribing Medications
In a survey of 500 urological patients, 40% had dietary restrictions avoiding animal products, yet, up to 50% of this subgroup had been prescribed gelatin-containing medications. 62 Often physicians are not aware that their patients are consuming an animal-derived product that goes against their dietary, religious, or moral stances. 3 Another survey showed that 70% of psychiatrists were aware that certain prescription drugs may contain animal-derived excipients, but the majority do not know if it applies to the drugs they are prescribing. 61 In situations where prescribing medication containing animal-derived ingredients is unavoidable, addressing the patient’s dietary, religious, and personal beliefs can aid medication adherence and informed decision-making. 63 When giving their medical opinion, it should be factual and also conscious of the characteristics of their patient. 64
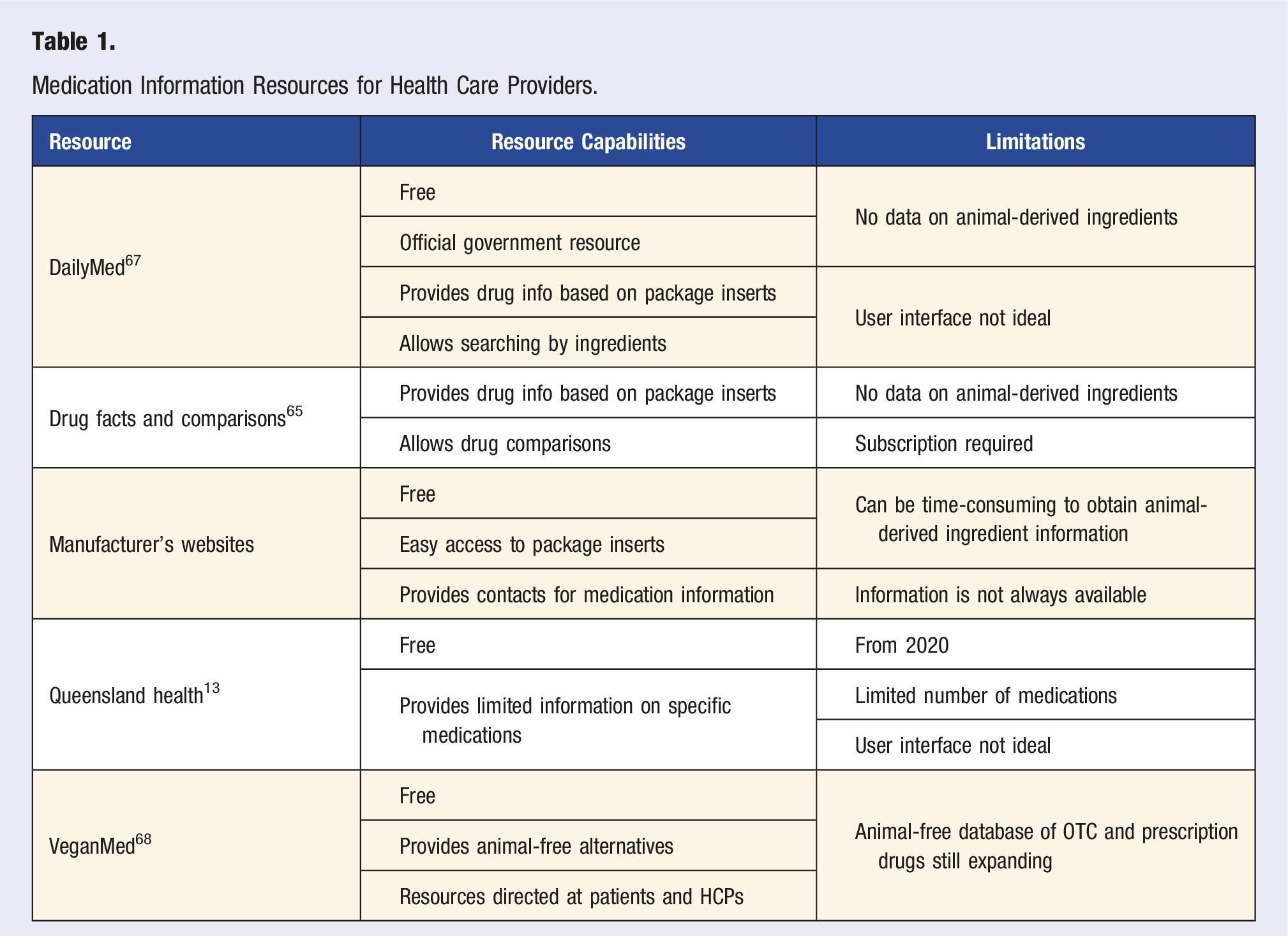
Resources for HCPs
The current approach to confirm the ingredient origins in a pharmaceutical product is to contact the manufacturer. To obtain a manufacturer’s contact information, one can use a Drug Facts and Comparisons manufacturer index 65 or the manufacturer’s website. Providers should be aware that generic medications are produced by multiple manufacturers, and each manufacturer may use different excipients. 66 As such, calling the manufacturer could be time-consuming and one may not always get the correct answer in a timely manner, as the manufacturer may need to conduct further inquiry.
DailyMed 67 is a United States government resource that provides package inserts for reviewing drug ingredients. It can allow the determination of common animal-derived ingredients such as lactose. However, the platform is not user-friendly, and one would not be able to determine the origins of ingredients that can be from both, an animal or plant source (e.g., magnesium stearate).
Medication Information Resources for Health Care Providers.
Need for Interdisciplinary Collaboration
Physicians and pharmacists can collaborate to identify medications suitable for their patients and provide drug information to resolve patient-specific problems until this can be provided by pharmaceutical manufacturers in an efficient manner. 69 With physicians and pharmacists practicing good communication, trust, respect, and mutual recognition of each other’s professional expertise, patients will be able to receive accurate and up-to-date information regarding their medication therapy. 70
Motivational interviewing promotes collaborative conversation with the patient and encourages active participation to develop better understanding of the medication. 71
Implications for Patients
A study revealed that nearly 63% of patients wanted their physicians to notify them if their prescribed medications contain ingredients derived from pork or beef. 72 However, another study found that only 20.5% of vegetarian patients would specifically ask either the doctor or pharmacist about their prescribed drug composition to determine if it goes against their dietary practices. 73 It is speculated that patients fail to ask about their medication formulations due to a lack of knowledge of their medications, not reading or being able to read the patient information leaflet, or believing that their physician or pharmacist would disclose the information to them. 73 It is important for patients to address any dietary restrictions with their doctor, especially if not asked initially. 74 The best practice for patients is to be honest in order to receive the appropriate treatment. 75 Being involved in the therapeutic decision-making process may increase patient compliance to treatment regimens. 61
Consulting healthcare providers 76 is a quick way for patients to identify if their medication is animal-free. Additionally, other resources may be utilized if patients are looking for more in-depth information. The most direct approach would be to contact the manufacturer. 76 Another method includes obtaining information from websites such as vegan.com or PETA, which contain multiple resources ranging from lists of animal-derived ingredients to other helpful websites for vegans.77,78 Veganissimo A to Z: A Comprehensive Guide to Identifying and Avoiding Ingredients of Animal Origin in Everyday Products written by Reuben Proctor and Lars Thomson expands the knowledge of animal-derived products in a simple and easy-to-read way. 79 Throughout the United States, vegan festivals serve as a place for activists, restaurants, and other companies to network and learn more about the vegan lifestyle. 80 These tools/communities may serve as an aid for anyone seeking reliable resources regarding their medications while practicing a vegan lifestyle. Identifying animal-derived excipients using Patient Information Leaflet or Summary of Product Characteristics remains a challenge to patients and providers. 60 With increased awareness, collaboration with pharmaceutical companies, and further research into effective alternative ingredients, there is potential for animal-free medications to become a cornerstone of therapy. 81
Vegan Medicine Marketplaces
A study narrowed down 3 factors that consumers choose when purchasing a pharmaceutical product: label, awareness, and trustworthiness. 82 Although labeling remains an issue, patients should be directed to verified marketplaces to purchase medications suitable to their needs. Amazon is the top online retailer for many products, including medications. 83 Patients can search for vegan medicine and receive over 10,000 results. 84 Aside from large corporations such as Amazon, there are several other smaller startup companies that specialize in veganism. Vejii, a digital marketplace located in Delaware, offers plant-based products such as groceries, vitamins, and pet supplies, 85 as does a similar online retailer named Vegan Essentials. 86 Another startup, called VeganMed, based in California and founded by pharmacists, provides patients with the right resources to discover animal-free options and aims for more transparency when it comes to labeling animal-free medications. 68 These companies provide a quality e-commerce experience when searching for vegan medications and alternatives, but this list is not all-inclusive.
Implications for Pharmaceutical Industry and Manufacturers
Rising Interest in Veganism and Potential Growth for Manufacturers
As of 2022, an estimated 1.5 billion people identify as vegetarians, accounting for 22% of the world’s population. 87 In 2020, 13% of consumers in Asia and 4% of consumers in Europe stated that they were vegan. 88 Approximately 6% of consumers in the United States are vegan. 89 Similarly, a large population does not consume animal products due to religious or other reasons (See STAKEHOLDER PERSPECTIVES section). Based on Google AdWords, vegan-related searches shot up by 47% during 2020. 90 With the increased advocacy for natural nutrition, veganism is driving the demand for more animal-free medications (See Abstract). It is estimated that the market for plant-based products in the United States alone is worth approximately 7 billion USD. 91 The global market for vegan softgel capsules generated revenue of around 218.7 million USD in 2020 and is anticipated to reach around 423.4 million USD by 2027. 92 The exponential growth of the vegan market creates a lot of opportunities for research and development of animal-free medications.
This rise in interest has pushed more pharmaceutical manufacturers to seek out natural materials to encapsulate active ingredients. 93 Currently, the global pharmaceutical market is estimated to be worth 1.27 trillion USD, 94 and the vegan-friendly pharmaceutical market has a huge potential for economic growth. Large manufacturers can easily lose out on profit to compounding pharmacies if they do not keep up with new trends. Compounding pharmacies have the ability to capitalize on this profit by catering to an individual’s needs and formulating medications specifically for each patient. 95
Animal-Free Excipients Exist
Top Animal-Derived Ingredients and Their Use. 97
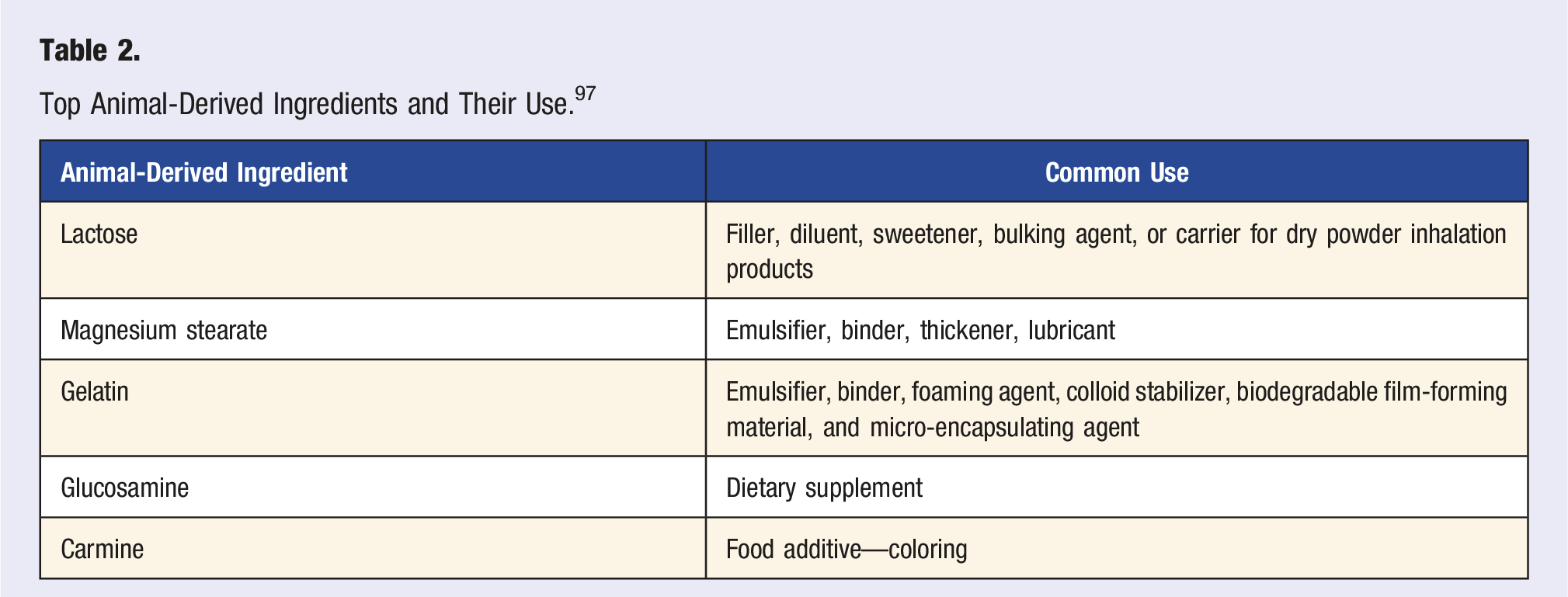
Following the trend of reducing the use of synthetics and chemicals, pharmaceutical firms are looking towards the use of more natural constituents in the development of medications. 93 Compared to a gelatin capsule, plant-based alternatives such as hydroxypropyl methylcellulose (HPMC) capsules have better physical stability, better stability at different temperatures, lower moisture content, and lower hygroscopicity. 98 Currently, HPMC capsules cost approximately 4 times as much as gelatin capsules, however, the price difference is expected to decrease over time as more companies opt for HPMC. 81 Capsugel, a company that specializes in capsule manufacturing, claims that there is an increase in demand for vegetarian capsules despite their cost. 99 HPMC capsules are generally preferred by vegan and vegetarian consumers and are better suited in moist environments. Overall, plant-based excipients, such as HPMC, have been attracting more attention due to their safety and abundance in comparison to animal proteins. 100
Companies have also successfully developed alternatives to other common excipients. JRS Pharma developed EMDEX as a substitute for spray-dried lactose, which is now marketed as Reactine.
101
ThermoFisher and Applied Biotechnology Institute created TrypLE and TrypZean, a recombinant bacteria, to replace the enzyme trypsin.102,103 Results from a 2011 comparative study demonstrated that non-animal source trypsin performed as effectively as the porcine-derived product.
104
As such, animal-free alternatives exist for common animal-derived ingredients. Figure 2 shows the 13 most common animal-derived medication ingredients and the plant-based alternatives that exist for each of them. However, the use of animal derivatives in pharmaceuticals is still a mainstay (likely due to financial reasons), with the need for more research and development further facilitating alternative formulations to produce fully animal-free products.81,105 Common Animal-Derived Ingredients and Sources.
68

Diversity and Inclusion Concerns
The aforementioned concerns pose a D&I concern as the needs of these consumers are often ignored. 106 Businesses benefit from increased diversity and inclusion, and diverse companies deliver stronger results. 107 From a patient perspective, it is crucial for pharmaceutical companies to evaluate diversity, equality, and inclusion. 108 This applies to not only their workforce, but also the patients that they serve. Pfizer has exemplified these values in the production of some of its medications. Some of their capsule shells are derived from a plant source that is kosher and halal certified, thus making them suitable for Jewish patients, Muslim patients, and vegetarians. 95 Many companies like Johnson & Johnson even state in their credo the need to “maintain in good order the property we are privileged to use, protecting the environment and natural resources.” 109 However, there is significant work to be done in this arena to address the needs of multiple stakeholders.
Limitations
This publication is limited for being a non-systematic review. It garners expert opinion and utilizes peer review publications based on the authors’ discretion. Some aspects may have been missed or truncated due to limited knowledge or word count restrictions.
Conclusion
Millions of individuals avoid animal-derived ingredients for a multitude of reasons. Several organizations and advocacy groups have raised concerns to enable access to medications without animal-derived ingredients, but more regulatory guidance or oversight is needed to address the needs of these patients. Regulations and policies exist in many different countries but are often ignored. Some resources are available for patients and healthcare providers to obtain information on medications and its ingredients, but each has its own limitations. Currently, no single, comprehensive, user-friendly database of medication ingredients exists that consumers can use to quickly determine whether a medication is animal-free.
When appropriate, manufacturers are encouraged to utilize animal-free alternatives for common inactive ingredients. Due to the increasing demand for animal-free medications, there is potential for the pharmaceutical industry and manufacturers to be inclusive of the needs of diverse patient populations. For the 25% animal-free medications available in the market, industry initiated but independent certification marks are a viable solution. This will allow HCPs to become more aware of the medications that they prescribe and patients to have more control over the medications that they take.
Supplemental Material
Supplemental Material - Embracing Medication Needs of Patients based on Ethical, Dietary, and Religious Preferences
Supplemental Material for Embracing Medication Needs of Patients based on Ethical, Dietary, and Religious Preferences by Lena Tieu, PharmD, Jasmine Uchi, PharmD, Nirva Patel, JD, Mihir Meghani, MD, Padmaja Patel, MD, and Yen Nguyen, PharmD in American Journal of Lifestyle Medicine
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors JU and YN are employees of VeganMed. All other authors have no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
