Abstract
The SARS-CoV-2 virus (severe acute respiratory syndrome coronavirus 2), and the disease it causes (COVID-19), have had a profound impact on global human society and threaten to continue to have such an impact with newly emerging variants. Because of the widespread effects of SARS-CoV-2, understanding how lifestyle choices impact the severity of disease is imperative. This review summarizes evidence for an involvement of chronic, non-resolving inflammation, gut microbiome disruption (dysbiosis with loss of beneficial microorganisms), and impaired viral defenses, all of which are associated with an imbalanced lifestyle, in severe disease manifestations and post-acute sequelae of SARS-CoV-2 (PASC). Humans’ physiological propensity for uncontrolled inflammation and severe COVID-19 are briefly contrasted with bats’ low propensity for inflammation and their resistance to viral disease. This insight is used to identify positive lifestyle factors with the potential to act in synergy for restoring balance to the immune response and gut microbiome, and thereby protect individuals against severe COVID-19 and PASC. It is proposed that clinicians should consider recommending lifestyle factors, such as stress management, balanced nutrition and physical activity, as preventative measures against severe viral disease and PASC.
Keywords
“This review thus highlights the potential of lifestyle practices for incorporation into clinical approaches for preventing severe disease manifestation of COVID-19 and PASC.”
Introduction
SARS-CoV-2 is a coronavirus first identified in 2019 1 that led to coronavirus disease 2019 (COVID-19) and the associated COVID-19 pandemic. While debate persists on the evolutionary history of the coronavirus that causes COVID-19, SARS-CoV-2 shares remarkable similarities to coronaviruses found in bats across Asia, 2 and may have arisen from (zoonotic) transmission from animals to humans. 3 Evolving the ability to have its spike protein cleaved by human proteases (furin and transmembrane serine protease TMPRSS2) conferred a greatly increased affinity for binding to a human cell surface receptor (angiotensin-converting enzyme 2, ACE2, receptor) that moves the virus into the cell, and thus enabled SARS-CoV-2 to enter human cells.4,5 Once inside the cell, the virus employs host replication machinery for replication, assembly, and release of new particles (see 6 ). However, and as has been shown for other viruses, the most severe disease manifestations of SARS-CoV-2 are traceable to what Doitsh and Greene refer to as “a form of cellular suicide rather than virological murder” (for the case of infection with human immunodeficiency virus, HIV) 7 based on an uncontrolled immune response leading to massive damage via self-attack (see below). A recent review summarized the involvement of system-wide, non-resolving inflammation, and associated gut microbiome dysbiosis (loss of beneficial microbes) in people living with HIV even after suppression of viral replication by antiretroviral therapy, and explored the use of adjunct lifestyle management with an emphasis on exercise. 8
Our present review summarizes mechanistic evidence that identifies chronic, non-resolving inflammation, and gut microbiome dysbiosis as specific risk factors for SARS-CoV-2 infection, severe COVID-19, and post-acute sequelae of SARS-CoV-2 (PASC). Even PASC symptoms including cardiovascular sequelae 9 and thromboembolic events 10 are likely a result of immune imbalance rather than direct viral pathogenesis. Note is made of the resistance of bats to coronavirus, which is at least in part related to bats’ low propensity for inflammation. It is proposed that the human propensity for systemic inflammation can be, and should be, managed with a lifestyle that restores immune system balance and gut microbiome composition (eubiosis), and may thereby reduce the risk of infection, severe disease, and PASC. This review thus highlights the potential of lifestyle practices for incorporation into clinical approaches for preventing severe disease manifestation of COVID-19 and PASC. Finally, larger-scale systemic interventions are identified that will be needed to (i) provide patients of all backgrounds with access to these lifestyle interventions and (ii) prevent and/or manage future pandemics of zoonotic origin.
Part 1: The Problem
Section 1: Severe COVID-19 is Linked to Compromised Human Immune Defenses
Non-Resolving Inflammation
A catastrophic response of the immune system can be precipitated by SARS-CoV-2 in humans, with substantial organ damage and severe disease.11,12 We summarize below available evidence that pre-existing low-grade inflammation and gut microbiome dysbiosis are further exacerbated by SARS-CoV-2, leading to uncontrolled, systemic inflammation during COVID-19 with poor disease outcomes in predisposed individuals.13-16
A dysfunctional immune system leads to the syndrome summarized in Figure 1, with (i) impaired immune defense, for example, poor viral control by the interferon (IFN) system (that inhibits viral replication) and (ii) simultaneous uncontrolled self-attack (uncontrolled systemic inflammation associated with non-resolving inflammation). In such a dysfunctional state, the inflammatory response upon pathogen infection spirals out of control, with feed-forward cycles of production of messengers (e.g., cytokine hormones and reactive oxygen species, ROS) that trigger excessive programmed cell death and organ failure.17-22 A state of chronic, non-resolving inflammation is also closely linked to chronic conditions, such as obesity
23
and hypertension,
24
and is a causative factor in diseases, such as cardiovascular disease,
25
diabetes,
26
and other pre-existing conditions associated with an elevated risk of severe COVID-19.27,28 Obesity in particular is closely linked to COVID-19 prevalence and severity.
29
Many negative effects of obesity can be attributed to an excess of visceral fat30,31 that is metabolically active and produces pro-inflammatory cytokines.32-34 Pre-existing non-resolving inflammation associated with excess visceral fat accumulation has the same principal features as the additional acute immune system dysfunction provoked by severe COVID-19 (Figure 1), the latter of which can be viewed as a more dramatic version of low-grade chronic (non-resolving) inflammation. Acute infection is thus apparently superimposed upon existing chronic inflammation and microbiome dysbiosis (Figure 1; see also below), and SARS-CoV-2 leads to an excessive response with massive organ damage by compounding pre-existing non-resolving inflammation. PASC is similarly characterized by a prolonged inflammatory host response to the virus,
9
and interventions that address non-resolving inflammation (see section on nutrition, exercise, and stress management) may thus ameliorate both acute and post-acute sequelae of COVID-19. Schematic depiction of the cycle of non-resolving inflammation, production of reactive oxygen species (ROS), microbiome dysbiosis, and impaired antiviral response by the interferon (IFN) system (that inhibits viral replication) in relation to human lifestyle factors (orange box). Departures from immune homeostasis perpetuate the depicted cycle.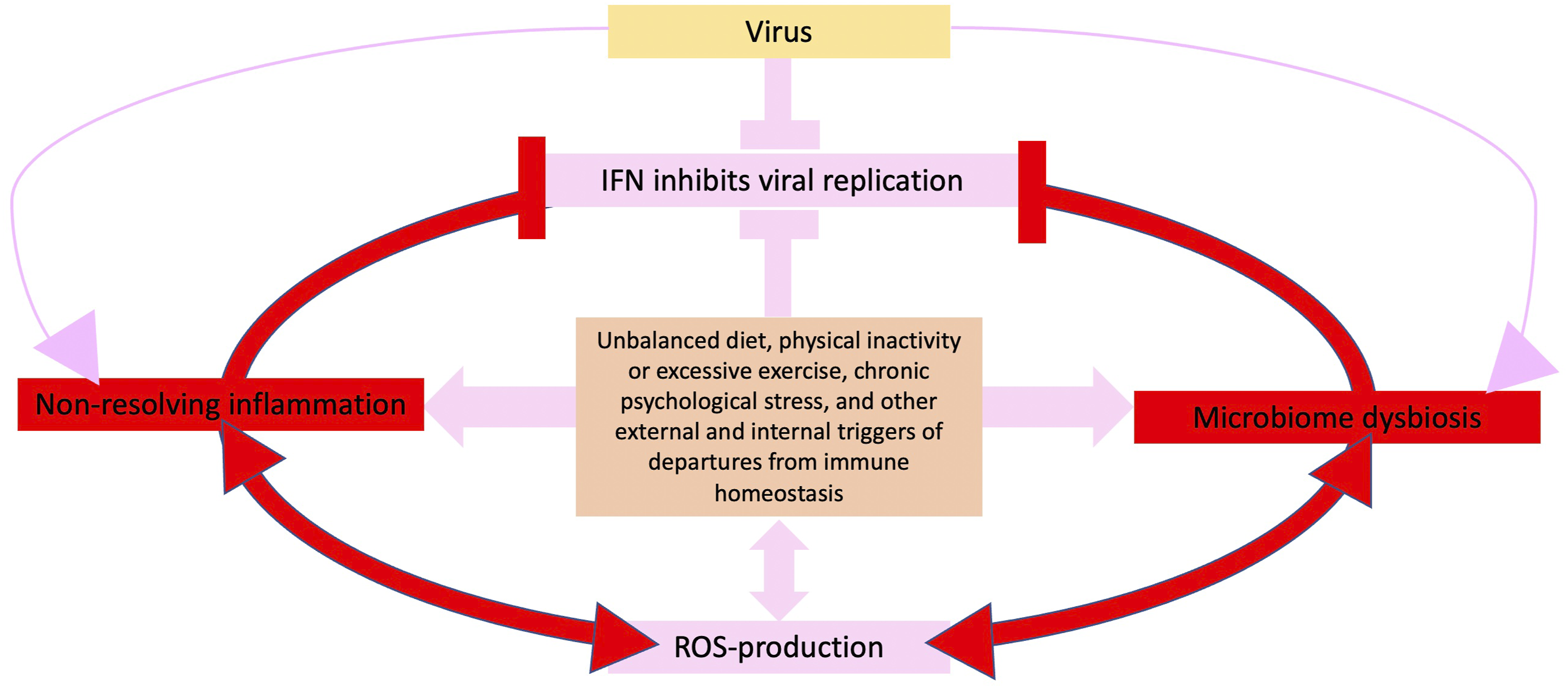
Figure 1 emphasizes interactions between uncontrolled systemic inflammation, in which ROS and destruction of virus-infected cells by programmed cell death, play a major role35-37 and production of antiviral IFN (Figure 1), cytokine protein hormones critical to viral defenses in vertebrates.38-40 In most mammals, including humans, IFN is activated only upon infection. 41 Many viruses have found ways to inhibit the production of IFN protein after IFN gene expression is activated.42,43 For example, SARS-CoV-2 can destroy human IFN messenger RNA and thereby inhibit protein production. 44 As depicted in Figure 1, pre-existing chronic inflammation can also weaken the IFN response. Conversely, inhibition of IFN production and signaling 45 by the virus prevents IFNs from limiting the systemic inflammation response46,47 (Figure 1; see also 48 ). Such a combination of impaired IFN responses and spiraling systemic inflammation (Figure 1) may be key to the triggering of severe disease by SARS-CoV-2 in humans.
Furthermore, both non-resolving inflammation 49 and SARS-CoV-2 infection 50 (see also below) are associated with gut microbiome dysbiosis (Figure 1), an impaired gut barrier, and resulting leakage of immunostimulatory bacteria and bacterial products (microbial translocation) into the bloodstream. For example, the bacterial lipopolysaccharide (LPS) is a marker of microbial translocation and an endotoxin that activates immune cells and triggers a system-wide inflammatory response.51,52 Addressing underlying non-resolving inflammation and resulting microbial translocation is thus imperative to prevent severe COVID-19. Fortunately, and as described in the section on nutrition, exercise and stress management, numerous lifestyle factors have the potential to act together in restoring a balanced immune response and preventing non-resolving inflammation.
Gut Microbiome, Viral Infectivity, and Inflammation Response
Figure 2 depicts the gut environment in both its functional state (eubiosis) and its disturbed state (dysbiosis). In the following, evidence is summarized that microorganisms play multiple roles in affecting the outcomes of SARS-CoV-2 infection. Pre-existing gut microbiome dysbiosis is often associated with non-resolving inflammation,
53
which impacts infection risk
54
and may be one reason why even seemingly healthy individuals can develop severe COVID-19
55
(Figure 2). In a nutshell, components of the human microbiome influence (i) the rate of viral entry into human cells and (ii) whether or not the human immune response spirals into uncontrolled inflammation with massive organ damage. SARS-CoV-2 infection is associated with gut microbiome dysbiosis, including reduced bacterial diversity with decreased abundance of multiple beneficial symbionts and increased abundance of opportunistic pathogens.
56
Schematic depiction of the human gut environment and links to immune system function. In a health-promoting gut environment (eubiosis), anaerobic fermenters maintain gut-barrier integrity and prevent endotoxin translocation into the blood stream as well as the resulting constitutive activation of immune cells. In contrast, in a dysbiotic gut microbiome, as may be present during infection with SARS-CoV-2, gut-barrier integrity is weakened. Furthermore, binding of SARS-CoV-2 to the ACE2-B0AT1 receptor complex and subsequent movement of this complex into the enterocyte decreases availability of this complex for its functions in (i) the production of antiviral defenses, (ii) gut-barrier-supporting tight-junction proteins, and (iii) electrolyte and glucose homeostasis. The net effect of these changes is a disturbed gut and immune environment with a self-perpetuating cycle of microbial translocation and uncontrolled immune activation. ACE2, angiotensin- converting enzyme 2. Created with BioRender.com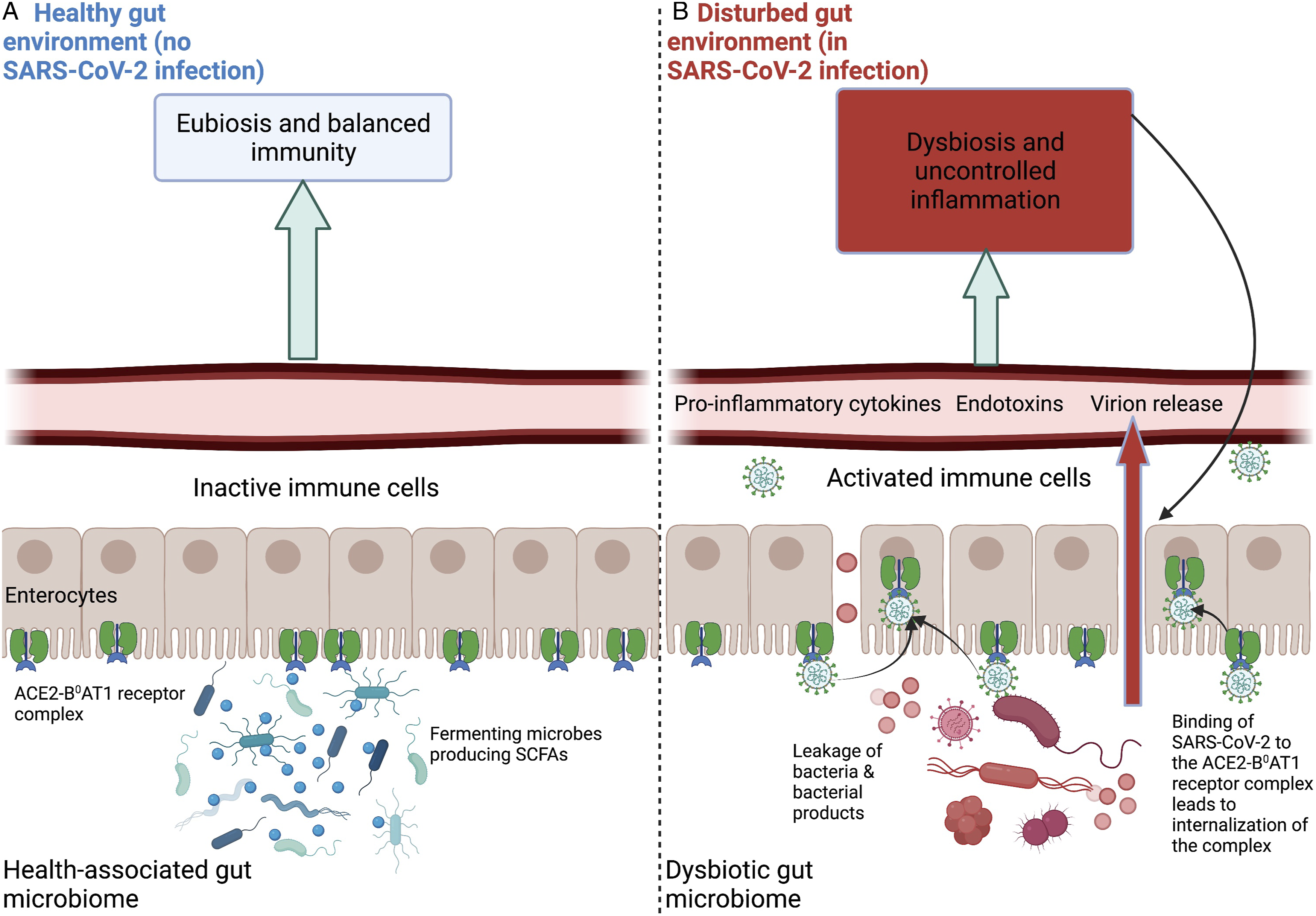
Evidence that COVID-19 involves impairment of the gut barrier includes elevated levels of the bacterial endotoxin LPS in individuals who died from COVID-19 compared to survivors 57 (Figure 2; see also 45). One mechanism of how SARS-CoV-2 impairs gut function involves its use of angiotensin-converting enzyme 2 (ACE2) receptors in the gut (as well as the lungs) for entry into human cells. 58 SARS-CoV-2 binding to ACE2 receptors (Figure 2) leads to movement of this receptor-virus complex into the cell, 59 and thus decreases ACE2’s ability to supply cells lining the gut with nutrition and other support for their function in maintaining gut-barrier integrity.60,61 Specifically, to carry out these functions, ACE2 in the gut must interact with B0AT1,62-64 a transporter of substrates needed for production of antiviral defenses and gut-barrier-supporting tight-junction proteins as well as for electrolyte and glucose homeostasis. 60 When ACE2 is highjacked by SARS-CoV-2 for viral entry into cells, ACE2 is unavailable for its interaction with B0AT1. Furthermore, since ACE2 is a regulator of the renin-angiotensin system, 65 decreased ACE2 in pancreatic beta-islet cells can disrupt insulin secretion and contribute to development of hyperglycemia and diabetic ketoacidosis.9,66,67
One of the substrates affected by ACE2 unavailability is the dietary amino acid tryptophan that is needed for the production of antimicrobial peptides.60,62 Disruption of tryptophan homeostasis via decreased ACE2 availability can also contribute directly to uncontrolled inflammation, as seen in patients infected with SARS-CoV-2.62,68-70 Reduced food intake during sickness may further reduce tryptophan availability (the role of nutrition is explored in detail in the section 4 on severe COVID-19 is linked to external factors that impair human immune defenses below). Impaired tryptophan absorption and metabolism may also be involved in PASC symptoms, such as muscle weakness, headaches, and depression as typical features of tryptophan deficiency. 71
Furthermore, components of the gut microbiome can either increase or decrease ACE2 expression
72
(Figure 2) and thus affect the rate of cellular entry of SARS-CoV-2 and other ACE2-dependent processes. Notably, antibiotics are often prescribed as treatment for COVID-19 despite the viral origin of this disease
73
and the relatively low number of confirmed microbial infections during COVID-19.
74
There is thus concern that antibiotics may eliminate beneficial gut microbiota and exacerbate dysbiosis in patients infected with SARS-CoV-2.75,76 Figure 3 places the effects of microbiome dysbiosis or eubiosis into the context of other factors reviewed below, including lifestyle as well as endogenous factors. The role of lifestyle factors as adjunct treatment to address COVID-19 associated dysbiosis is explored further in the section on nutrition, exercise and stress management. Summary of factors that either impair (hobble) or enhance antiviral defenses and immune control (green shading). The modern lifestyle, characterized by an unbalanced diet, chronic psychological stress, sedentariness, and exposure to environmental pollutants, interacts with genetic and other endogenous factors to derail the human immune response and cause non-resolving inflammation (orange shading). In contrast, a rebalanced lifestyle and/or certain endogenous factors (blue shading) can restore balanced immunity and antiviral defenses, while also limiting organ damage. ACE2, angiotensin-converting enzyme 2; GL, glycemic load (a dietary carbohydrate’s glycemic index, i.e., how quickly it is converted to glucose, multiplied by how much is consumed per day); IFN, interferon; sat., saturated; SCFA, short-chain fatty acid; unsat., unsaturated.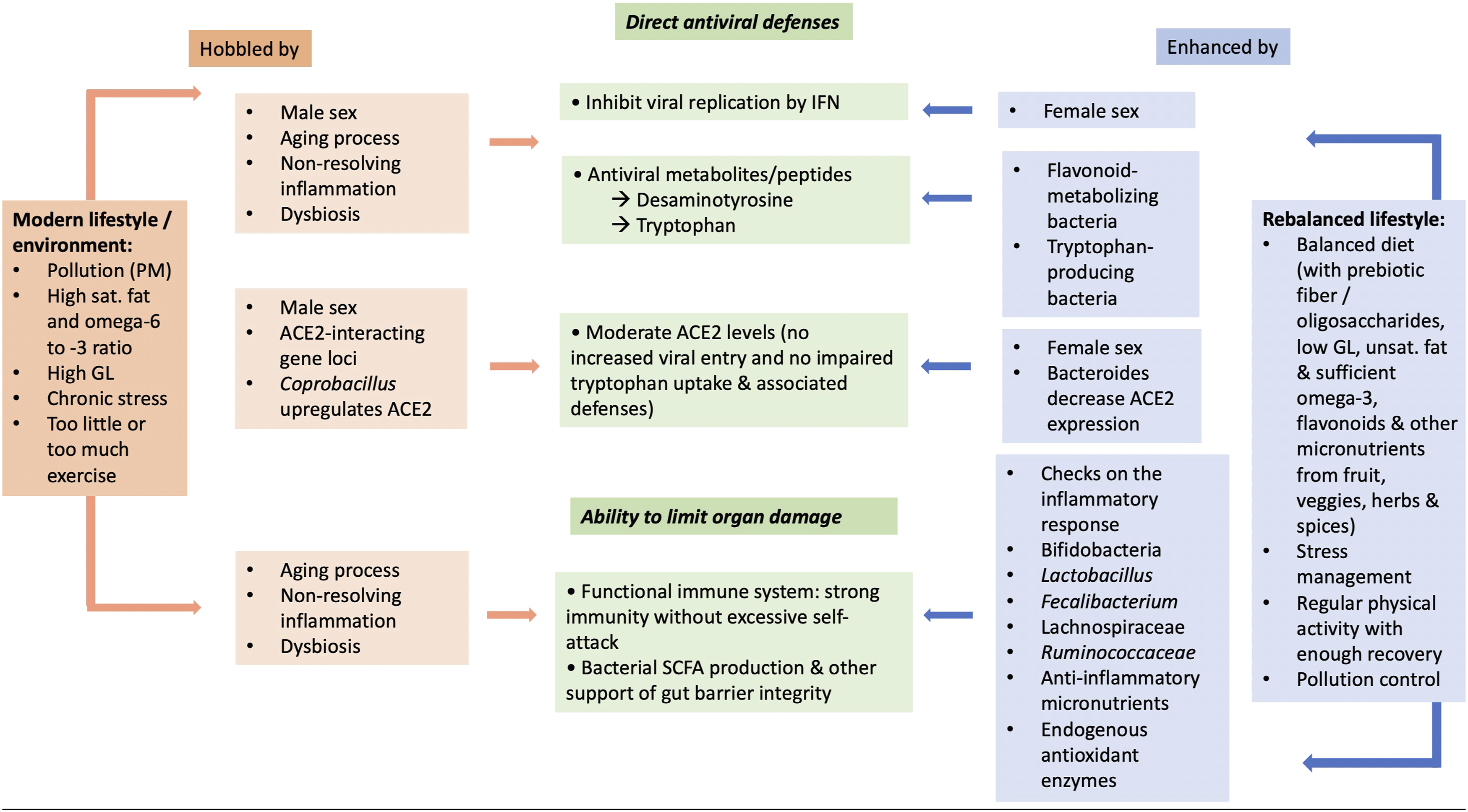
A recent comprehensive review of alterations in microbiome composition provided an exhaustive list of bacterial and fungal genera and species affected by SARS-CoV-2 infection, which can be classified into (1) loss of microorganisms that either directly oppose viral infectivity or prevent uncontrolled inflammation and (2) elevated levels of microorganisms that either enhance SARS-CoV-2 infectivity or promote uncontrolled inflammation. 77 Figure 3 features selected examples of gut bacteria associated either with high or low susceptibility to severe COVID-19 and PASC. Examples of bacteria with protective effects during SARS-CoV-2 infection include (i) those that decrease ACE2 expression in the colon, 78 such as Bacteroides (prevalence negatively correlated with fecal viral load), 79 (ii) those involved in tryptophan metabolism, such as Alistipes (enhanced in feces of patients with low/no COVID infectivity), 80 (iii) maintain gut immune homeostasis (Alistipes onderdonkii), 81 and/or (iv) produce the short-chain fatty acid (SCFA) butyrate (Faecalibacterium prausnitzii; 82 Figure 3). SCFAs are end products of bacterial fermentation with anti-inflammatory functions (dampening the pro-inflammatory action of NF-kB and other effects).83,84 Butyrate also maintains gut-barrier integrity; 85 decreases in the butyrate-producers Ruminococcaceae and Lachnospiraceae were associated with severe COVID-19. 79
Given butyrate’s role in maintaining gut-barrier integrity 8 and keeping inflammation in check, decreased butyrate production during COVID-19 may exacerbate pre-existing gut microbiome dysbiosis and trigger a feed-forward cycle of further gut-barrier weakening, microbial translocation, and systemic inflammation. SARS-CoV-2 does trigger such inflammation (see above) and damages the digestive system. 86 Persistent inflammation has also been observed in individuals with PASC; 87 PASC can also involve persistent gut microbiome dysbiosis 88 and increased markers of intestinal permeability were seen in children with multisystem inflammatory disorder state that can develop after acute COVID-19. 89 A comprehensive review of underlying factors of PASC 87 similarly highlighted non-resolving inflammation and microbiome dysbiosis as well as additional features (including reduced IFN production).
Dysbiosis of the microbiome in COVID-19 extends to the oral microbiome. 90 Patients with PASC “had significantly higher abundances of microbiota that induced inflammation, such as members of the genera Prevotella and Veillonella, which, of note, are species that produce LPS.” 90 It has also been noted that microbiome composition of patients with PASC was “similar to that of patients with chronic fatigue syndrome.” 90
Based on these findings, we propose that dietary support, and possibly bacteriotherapy, aimed at restoring butyrate-producing and other protective bacteria, may promote eubiosis and counteract viral infectivity as well as catastrophic inflammation during, and subsequent to, SARS-CoV-2 infection 77 (see section on nutrition, exercise and stress management).
Notably, cytokines are primarily released from immune cells in the lungs during COVID-19, 91 although this remains to be confirmed for the omicron variant that appears to elicit less lung damage. 92 The following section on the gut-lung axis addresses links between the states of the gut and lung microbiomes. 93
Gut-Lung Axis
As depicted in Figure 4, the gut-lung axis is a bi-directional pathway for passage of gut products, including both beneficial and detrimental microbial metabolites, through the bloodstream to the lungs, and lung products to the gut.
94
For example, immune factors and cells are transported from gut to lungs for roles in protection against respiratory infections.95,96 Gut microbial products such as SCFAs may improve antiviral responses in the respiratory tract,97,98 including the IFN response
99
(cf. Figure 1). Another product, desaminotyrosine, produced by gut bacterial fermentation of dietary flavonoids,100,101 also travels from the gut to the lungs, increases IFN signaling, protects against influenza infection,
102
and may also offer some protection against severe COVID-19.
103
This is one mechanism for the antiviral and anti-inflammatory effects of flavonoids from vegetables, herbs, and spices.
104
Schematic depiction of the gut-lung axis. The gastrointestinal and respiratory tracts are connected, such that non-resolving inflammation in one area may adversely affect immunity and microbiome status in the other. Furthermore, particulate matter and other pollutants can enter directly into both tracts, triggering systemic inflammation.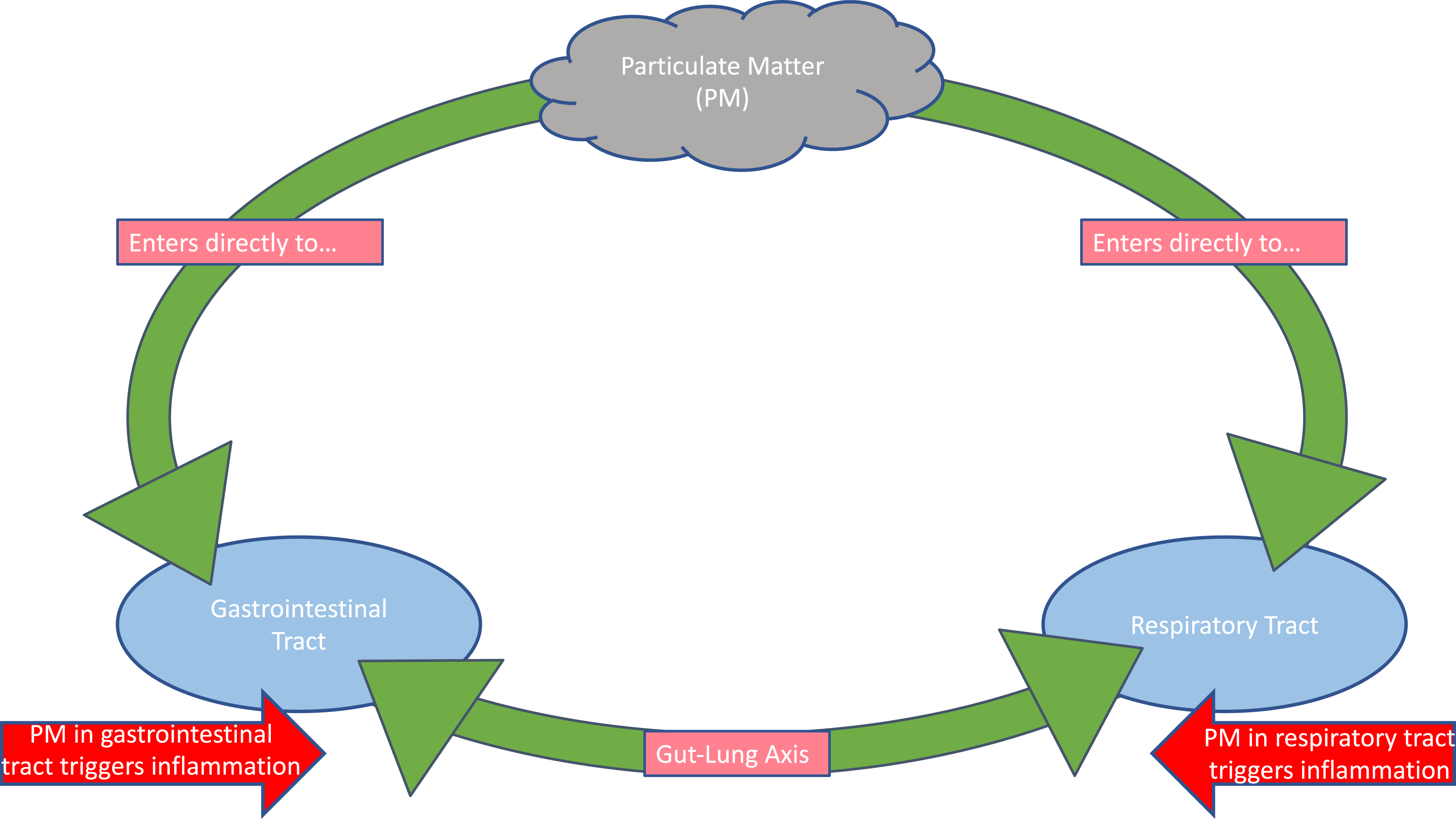
The gut microbiome thus clearly shapes the lung immune response,99,105 and gut microbiome dysbiosis increases the risk of respiratory illness. 95 Lifestyle interventions that restore eubiosis may thereby play a protective role against respiratory infections like SARS-CoV-2. Conversely, lung inflammation can negatively impact the gut microbiota 106 and further weaken viral defenses. Furthermore, environmental pollutants, such as particulate matter (PM), can enter directly into both gut and lungs (Figure 4), trigger systemic inflammation throughout the gut-lung axis, and disrupt both lung and gut microbiomes. 107
Section 2: Bat Resistance to SARS-CoV-2 is Linked to a Low Propensity for Inflammation and a Unique IFN Response
This section briefly summarizes available evidence for why bats are resistant to many viruses and serve as vehicles for viral transmission to humans. An understanding of how bats control viral infection and inflammation may be able to provide directions for the management of non-resolving inflammation in humans through a combination of pharmacological and lifestyle medicine.
Bats are viral reservoirs—subject to few, if any, detrimental effects from viruses that trigger severe disease in humans.39,108,109 Bats exhibit unique immune responses that help explain their role as ancestral hosts for coronaviruses. Decreased inflammatory pathways, coupled with a particularly effective IFN-based direct inhibition of viral replication,110,111 enable bats to control viral infections while avoiding excessive self-attack and resulting organ damage. Bats possess a constitutively active IFN response, which presumably inhibits viral replication immediately upon entry of the virus 110 (Figure 2). By maintaining constant IFN gene expression, bats presumably have IFN protein ready constitutively to control viruses.
In addition, bats possess a mutated version of a stimulator of IFN genes (STING) that activates ROS-producing cascades, which leads to a dampening of such cascades and, again, a lesser risk for massive organ damage in bats. 112 The combination of this dampened response, together with constitutive IFN activation, may help bats live in “peaceful coexistence” with viruses by maintaining effective defenses without excessive, self-destructive responses 112 (Figure 2).
In summary, whereas bats do not exhibit uncontrolled inflammation39,40 and the organ damage characteristic of severe COVID-19 in humans,113-115 humans can be thought of as being at the mercy of external factors that either limit or exacerbate their inflammatory response. The next sections identify endogenous (section 3) and lifestyle features of modernization (section 4) that either enhance systemic inflammation or have the potential to serve in lifestyle management to control inflammation (section on nutrition, exercise and stress management).
It should be noted that the often high human susceptibility to severe COVID-19 also involves mutations in the SARS-CoV-2 spike protein 4 (see also Ref. 5). The spike protein on the surface of SARS-CoV-2 facilitates viral entry into target cells by binding to ACE2.58,116 SARS-CoV-2 spike protein has evolved the ability to become cleaved to a form with superior ACE2 binding by human proteins (proteases such as furin and TMPRSS2).116,117
Section 3: Severe COVID-19 is Also Linked to Endogenous Biological Factors
Aging and Weakened Defenses
The aging process is orchestrated by a genetically programmed shift to a pro-oxidative, pro-inflammatory state that increasingly favors programmed cells death.118-121 As humans age, mitochondria open their “poison cabinet” 122 and increase ROS production, which triggers chronic systemic inflammation and age-related diseases. Aging can thus be thought of as a state of inflammaging, 123 that is, progressively less- and less-resolving inflammation. It thus takes older individuals longer to resolve immune system activation 124 and cytokine release is heightened in older age. 125 This view is also consistent with reports that gene loci associated with non-resolving inflammation126,127 constitute a genetic risk for severe COVID-19. At the same time, aged cells exhibit weakened viral defenses with decreases in the IFN response128-130 (Figures 1–3).
The disrupted immune environment characteristic of aging is also linked to a dysbiotic gut microbiome; older individuals exhibit losses in SCFA-producing Bifidobacteria, Lactobacillus, and others that are essential to maintaining gut-barrier integrity and preventing non-resolving inflammation.131,132 Furthermore, aging may affect ACE2 levels, with aging males, in particular, exhibiting increased ACE2 in heart and lungs. 133 Since ACE2 is critical for cellular entry of SARS-CoV-2, increased ACE2—in conjunction with an inflammatory environment, microbiome dysbiosis, and impaired viral defenses—offers further explanation for why older individuals are at an increased risk for severe COVID-19 134 (Figures 1 and 3).
Excessive Visceral Fat and Non-resolving Inflammation
The accumulation of excessive visceral fat is associated with weakened immune defenses to viruses like SARS-CoV-2 as well as uncontrolled, systemic inflammation.135,136 Both features, that is, blunted defenses and systemic self-attack, are likely contributors to the correlation between obesity and severe COVID-19. Specifically, the spiraling immune response to SARS-CoV-2 is apparently superimposed on a chronic pro-inflammatory state associated with an excess of metabolically active visceral fat.137-139 An excess of pro-inflammatory visceral fat may also be a driver of the association between PASC and obesity. 140
Conversely, the link between excessive visceral fat and non-resolving inflammation provides opportunities to reduce obesity-associated disease risk through lifestyle intervention. It has been pointed out that weight is just one among several factors 141 influencing COVID-19 disease risk. From the mechanistic considerations listed above, it seems possible that benefits could be derived even before any major weight loss—by adoption of a diet replete in antioxidants and omega-3 fats and a resulting lessening of uncontrolled inflammation. Similarly, concomitant stress management would be expected to reduce the cycle of immune dysfunction, microbiome dysbiosis, and oxidative stress (Figure 1). This comprehensive review thus focuses on the intersections among diet, physical activity and stress management in shaping the stress response, immune system and gut microbiome. For more details on lifestyle interventions to mitigate non-resolving inflammation and reduce the disease risk associated with obesity, see section on nutrition, exercise, and stress management.
Biological Sex, IFN, and ACE2
A recent review summarized genetic variation in a number of genes that are involved in the interaction between SARS-CoV-2 and humans. 117 For example, males appear to be disproportionately impacted by severe COVID-19 (Figures 1 and 3), with the fraction of reported cases resulting in death for males over twice that for females, despite roughly equal infection rates between sexes.142,143 Male sex is also a risk factor for pulmonary PASC. 144 Antiviral IFN production was higher among females than males, likely due to differences in the role of sex hormones in IFN gene regulation 145 (Figures 1 and 3). The higher estrogen levels in pre-menopausal women than men,146,147 and control of IFN-regulatory factors by estrogen, provide an avenue for increased IFN production and associated protection against severe COVID-19 in women. 148 These findings are consistent with reports that loss of function of certain IFN genes149,150) may constitute a genetic risk for severe COVID-19.
As stated above, aging increases ACE2 levels, and this was especially prominent in older men 133 and men with heart failure 151 (Figure 3). Increased ACE2 may contribute to enhanced disease severity among males due to the reliance of SARS-CoV-2 on ACE2 for entry into cells. Overall, the strongest association of a gene locus with COVID-19 identified thus far is for the 3p21.31 locus, 152 a region with genes that interact with the ACE2 receptor. 153
Moreover, the binding affinity of SARS-CoV-2 spike protein to the ACE2 receptor may be affected by recently demonstrated genetic variation in the human proteins that cleave the spike protein to the form with a high affinity for ACE2. These human proteins, including the proteases furin and TMPRSS2, exhibit substantial genetic variation, which may play a role in the extreme variability of outcomes from no symptoms to extreme disease. 117
Biological Sex and Behavioral Differences
Differences in lifestyle may compound biological differences. Tobacco use and binge drinking both contribute to non-resolving inflammation and impaired viral clearance,154-157 and are more prevalent among males.157,158 Men are also less likely to wash their hands, 159 wear masks, 160 and socially distance. 161 The greater likelihood of males engaging in risk-taking behavior 162 thus applies to the COVID-19 pandemic as well. 163 Notably, the number of cases of COVID-19 appears to be roughly the same for men and women; given the high transmissibility of the virus, risk-taking behaviors may lead to infection not only of the risk-taker, but also of those who interact with the risk-taker. Furthermore, women are more likely to be frontline workers than men, especially frontline healthcare workers, 164 which may also contribute to the comparable infection rates of women and men.
Section 4: Severe COVID-19 is Linked to External Factors that Impair Human Immune Defenses
As depicted in Figure 3, an unbalanced diet, either physical inactivity or excessive exercise, chronic psychological stress, and environmental pollutants all contribute to non-resolving inflammation with resulting impaired immunity against pathogens and excessive self-attack on body systems. Diet, physical activity level, and stress level thus act synergistically with each other and SARS-CoV-2—presumably by targeting the same molecular players. One example is NF-kB and/or its signaling pathways (e.g., p38 MAP kinase).165-175 While an imbalanced lifestyle with respect to the above external factors presumably predisposes to various co-morbidities as well as severe COVID-19 in humans, lifestyle is modifiable (see section on nutrition, exercise and stress management), and personal and policy level changes towards implementing a healthful lifestyle have the potential to play a role in preventing severe disease.
Unbalanced Diet
Regular consumption of processed, overly-energy dense, micronutrient-deficient foods promotes non-resolving inflammation
176
(Figure 3). A diet low in whole fruit, vegetables, seeds and whole grains fails to provide anti-inflammatory micronutrients. In addition, foods high in free sugars, quick-burning starches and/or saturated fat not only provide excess calories, but simultaneously serve as gene regulators that cause cravings and promote excessive fat storage in a deep layer around the waist176-181 (visceral fat). By being metabolically active, continuously producing inflammation-triggering hormones, visceral fat cells promote non-resolving inflammation when present in excess numbers. Foods with a high ratio of pro-inflammatory omega-6 to inflammation-resolving omega-3 fats are also linked to non-resolving inflammation.181-187 The modern diet provides an excessively high ratio of omega-6 to omega-3 oils, largely in the form of mass-produced vegetable oils.184,185,188 Moreover, processed foods are deficient in prebiotic dietary components, such as fiber and oligosaccharides, needed as food for communities of fermenting colon microbes whose waste products serve as human gene regulators, such as butyrate,189,190 with inflammation-resolving effects. Figure 5 depicts how free sugars (such as the table sugar sucrose) and quick-burning starches are removed early during their passage through the human gut, leaving no food for fermenting colon microbes. Without dietary components that pass all the way to the colon (insoluble fiber, such as cellulose, soluble fiber, such as pectin and fruit pulp, resistant starch, and short-chain sugars or oligosaccharides), fermenting microbes are unable to produce SCFAs like butyrate that are needed to maintain gut-barrier-integrity,191,192 oppose non-resolving inflammation, and support other vital functions. Metabolism of quick-burning starch (high glycemic index; rapidly broken down into glucose) vs fiber, resistant starch, and oligosaccharides. Quick-burning starch is digested in the small intestine to glucose and can increase appetite and promote fat-storage. In contrast, fiber, resistant starch, and oligosaccharides are only digested by colonic microorganisms into sugars that are then fermented by other microorganisms into short-chain fatty acids, such as butyrate. Created with BioRender.com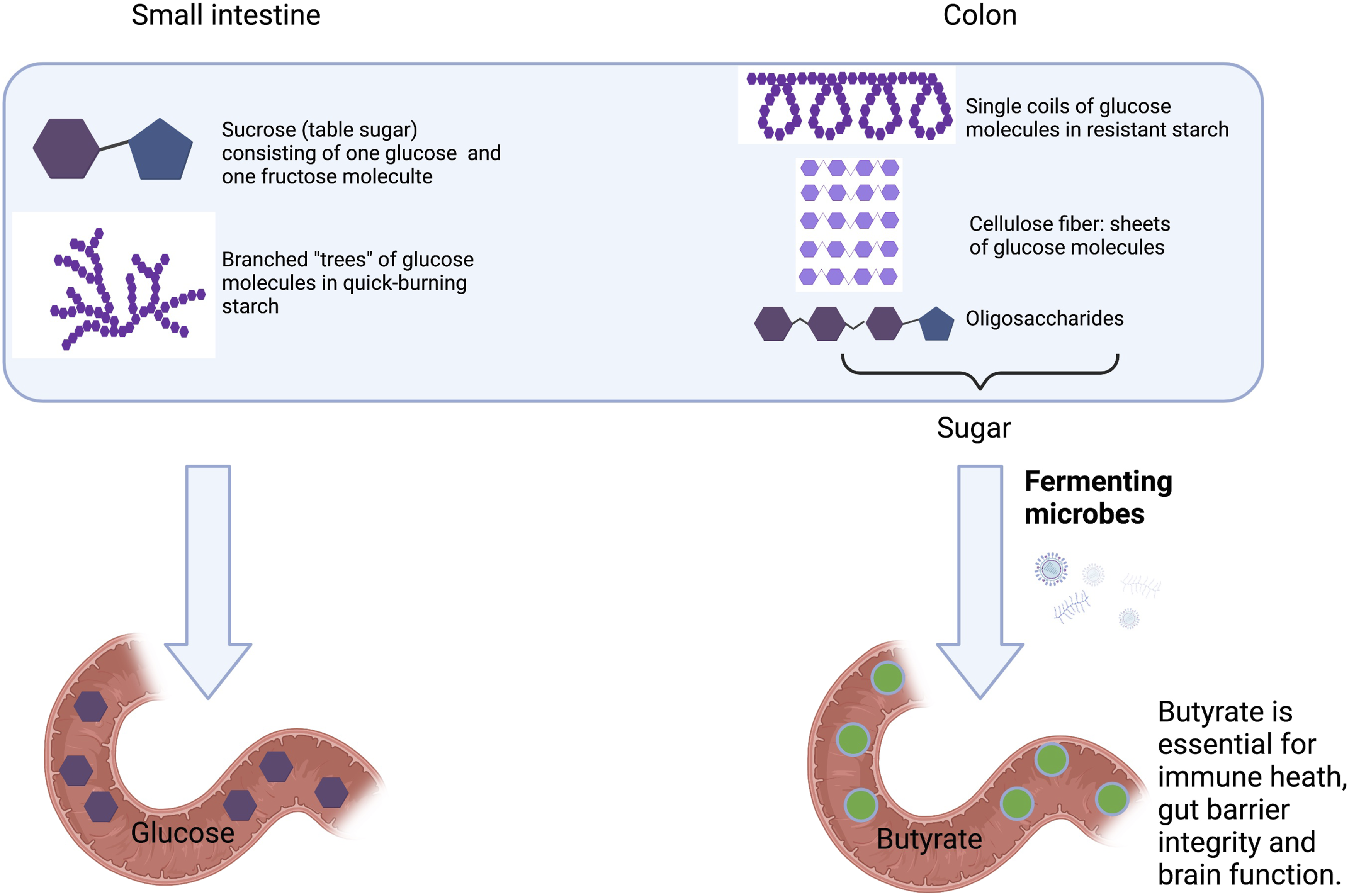
Chronic Psychological Stress Fatigues the Otherwise Immune-Suppressive Stress Response
Whereas intermittent acute psychological stress produces bursts of immune-suppressing stress hormones, chronic stress eventually fatigues this stress-response system 193 and contributes to non-resolving inflammation.194,195 Chronic psychological stress also disrupts the gut microbiome.81,196 Chronic psychological stress in combination with a diet high in omega-6 fatty acids and low in inflammation-resolving antioxidants and other micronutrients stimulates non-resolving inflammation especially strongly. Similarly, while acute stress can lead to a temporary inhibition of appetite/eating, chronic stress can apparently stimulate appetite/eating, especially with cravings for an energy-dense diet rich in sugar and fat. 197 Higher stress levels were thus associated with an unbalanced diet and higher body weight, 198 and elevated levels of the stress hormone cortisol were positively associated with obesity in some studies. 199 Therefore, managing chronic psychological stress may also counteract obesity. Figure 3 summarizes these effects in the context of other lifestyle factors.
Furthermore, a vicious cycle has been described where (i) an unbalanced lifestyle increases the risk for severe COVID-19 and, conversely, (ii) the pandemic aggravated lifestyle imbalances by further elevating chronic stress, reducing physical activity, and dietary imbalances.140,200 People with obesity reported experiencing a pronounced further reduction in diet quality and physical activity levels as well as worsened mental health. 200 As stated above, our review links negative effects of obesity to excessive visceral fact and its negative metabolic effects. As such, excessive visceral fat is more closely linked to negative health outcomes than traditional markers of overweight like body mass index141,201,202 (BMI). Recent discussion also suggests that a focus on features such as BMI is contributing to body shaming and weight-related stigmatization, which further intensified an obesity-inducing imbalanced lifestyle during the pandemic.141,200 Specifically, weight-related shame and stigmatization further increased psychological distress 141 and depression, 200 which exacerbated poor diet choices, 203 decreased motivation for physical activity, 204 and increased psychological stress. 203 Moreover, stigmatization can also lower the quality of medical care by causing providers to focus on BMI rather than other measures of health, and leading people with obesity to avoid medical encounters for fear of potential shaming. 205 In fact, weight-related stigma has been linked to earlier mortality, 206 is thought to represent a primary mechanism by which body size can impact health, 207 and may exacerbate COVID-19 risk for individuals with excess visceral fat. 141 These links suggest that psychological interventions should specifically address body shaming.
Women were more likely than men to experience psychological stress, and an associated exacerbated poor diet and decreased physical activity, during and after COVID-19-associated quarantine. 140 Psychological interventions described in the section on nutrition, exercise and stress management may therefore offer important relief especially for women navigating the negative psychological impacts of the pandemic, and the benefits may include lesser visceral fat accumulation and less non-resolving inflammation particularly in women.
Physical Inactivity and Excessive Exercise
Because dietary antioxidants must work in tandem with antioxidant enzymes produced internally during physical activity, 208 physical inactivity further contributes to non-resolving inflammation.8,176 In the modern environment, humans neither consume enough dietary antioxidants nor do they produce enough antioxidant enzymes internally to keep the immune system operating effectively. Notably, either physical inactivity or excessive exercise (especially exercise without sufficient time for recovery) can cause gut microbiome dysbiosis, impairment of gut-barrier integrity, and non-resolving inflammation.8,209 Insufficient or excessive exercise thus compounds the adverse effects of the modern diet and chronic psychological stress (Figure 3).
Pollutants
Environmental toxins further contribute to the feed-forward cycle of non-resolving inflammation and microbiome dysbiosis (Figure 1) that increases the risk of developing severe COVID-19. One such pollutant is particulate matter in the air (Figure 4), which can contain a wide variety of substances including nitrates, sulfates, and clay particles, 210 all of which stimulate ROS production.211-217 Particulate matter is linked to impairment of lung development and decreased overall lung function 218 and can also be deposited into the digestive tract 219 where it alters the composition of the gut microbiome and decreases butyrate production (Figure 2). Overall, urban air pollution triggers inflammatory and oxidative stress pathways.220,221 Other pollutants may have similar adverse effects222-225 and may thus also increase the risk of severe COVID-19.
Section 5: Modernization-Associated Inequality in Access to a Health-Promoting Lifestyle
Urbanization has promoted the imbalanced lifestyle described above, that is, excessive consumption of energy-dense, micronutrient-deficient food, a sedentary lifestyle, chronic psychological stress, and increased exposure to environmental toxins.226,227 While nearly every community is impacted by these factors, not all communities are affected to the same extent. Low-income communities are less likely to have access to healthful, nutrient-dense foods,172,228,229 or safe spaces for physical activity.230,231 Ethnic minorities and/or other individuals of lower socioeconomic status are also disproportionately impacted by chronic psychological stress232,233 and exposure to environmental toxins.234,235 Because health-promoting lifestyles are less accessible to underserved communities, individuals in these communities are more likely to experience the syndrome of non-resolving inflammation and microbiome dysbiosis 172 (Figures 1–3) and the resulting higher risk of severe COVID-19. For example, 35% of adults with incomes below $15,000 are at risk of developing severe COVID-19 if infected with SARS-CoV-2, compared to 16% of adults with incomes of $50,000 or more. 236 Furthermore, hospitalization rates due to COVID-19 are two to five times higher for underrepresented groups than non-Hispanic white persons. 237 COVID-19 thus disproportionately burdens people of color and lower-income individuals. Consequently, addressing inequities in access to a health-promoting lifestyle (with balanced food, stress reduction, physical activity, and clean air and water) would allow practitioners to effectively recommend lifestyle practices as preventative measures and thus play a pivotal role in preventing severe disease manifestations in individuals of all backgrounds.
Another feature of modern life is the spread of misinformation that can prevent individuals from engaging in scientifically supported strategies to reduce COVID-19 infection risk and severity (i.e., vaccination). Individuals with lower levels of education are more susceptible to such misinformation238,239 and COVID-19 vaccination rates are lowest among communities with low levels of education. 240 Boosting population education level may thus be an additional avenue for preventing and/or controlling pandemics like COVID-19.
Part 2: Solutions Based on Mechanistic Insight
A mechanistic understanding of how a modernization-associated lifestyle triggers severe viral disease offers directions for interventions (Figures 1–3). Given that balanced nutrition, regular physical activity, and psychological interventions all target similar molecular players and reduce non-resolving inflammation (see above), such lifestyle factors have the potential to reduce the risk of severe SARS-CoV-2 manifestations and PASC. Moreover, the synergy among various lifestyle components has the potential to allow modest improvements in each area to compound into a powerful positive effect, with notable reductions in inflammatory signaling and dysbiosis. However, access for currently underserved populations to even modest interventions will require community-level changes.
Section 6: Lifestyle as Adjunct Treatment: Nutrition, Exercise, and Stress Management
Balanced nutrition, regular physical activity, and psychological interventions to reduce chronic stress act in synergy to ameliorate non-resolving inflammation and promote a eubiotic gut microbiome composition 241 (Figure 2), which has the potential to reduce severity of COVID-19. 55 Clinicians can recommend these lifestyle factors as adjunct treatments for prevention of severe disease manifestations of COVID-19. Furthermore, given the exacerbated inflammatory state and dysbiosis during COVID-19, the potential of these same lifestyle factors as part of prevention and treatment of PASC (which some estimates suggest is experienced by over half of individuals infected with SARS-CoV-2) 242 warrants further attention. Addressing these symptoms is imperative for reducing the burden experienced by millions of people 242 and healthcare systems tasked with caring for patients experiencing both acute and post-acute sequelae of COVID-19. Lifestyle interventions that reduce dysbiosis, gut barrier impairment and non-resolving inflammation may thus serve as valuable adjunct treatments for PASC. A recent analysis in search of “an evidence-based, multidisciplinary management approach” to PASC concluded that, “interdisciplinary monitoring with holistic management that considers nutrition, physical therapy, psychological management, meditation, and mindfulness in addition to medication will allow for the early detection of post-acute COVID-19 sequelae symptoms and prevent long-term systemic damage.” 9
1. Balanced Nutrition
The hallmark of a balanced diet is a combination of low levels of appetite- and fat-storage-promoting free sugar, quick-burning starches, and saturated fat with a balanced ratio of omega-6-to-omega-3 fatty acids, balanced levels of antioxidants, and prebiotic foods such as fiber, resistant starch, and oligosaccharides (Figure 3). Examples for diets with all of these elements include the anti-inflammatory traditional Mediterranean and traditional Asian diets.176,243 The MIND diet (Mediterranean-DASH Intervention for Neurodegenerative Delay diet, where DASH stands for Dietary Approaches to Stop Hypertension), furthermore, includes a high proportion of berries. Berries, fiber, and other plant compounds in fruit and vegetables support the lung and gut microbiomes244-248 and enhance defenses against viral disease84,249 (Figure 3). These foods may thus serve as adjunct treatments for supporting gut microbiome eubiosis before, during, and after COVID-19. In addition to the above evidence for benefits of whole food groups, benefits of individual components have been documented. For example, a high-fiber diet may ameliorate symptoms in individuals with PASC. 250 In addition to the direct antiviral effects of flavonoid derivates described above, phenolics in dark green leafy vegetables such as kale and spinach activate a transcription factor that terminates inflammation. 251 Foods with high levels of omega-3 fatty acids also promote termination of inflammation. 252 Finally, foods containing high levels of certain carotenoids decrease inflammation markers.243,253-255 Given the unchecked inflammatory state characteristic of severe COVID-19, these foods could be used in conjunction with pharmacological treatment to restore immune balance before, during, and after SARS-CoV-2 infection.
Specific affordable, non-perishable foods that support wellness 172 include canned kippered herring, sardines, and tuna, which are good sources of omega-3 fatty acids. Dried herbs and spices, onions, and shallots all offer a potent antioxidant boost, and canned corn can provide potent anti-inflammatory carotenoids. Seasonal fruits and vegetables also contain many antioxidants, and organic options should be emphasized to avoid pollutants. For example, non-organic strawberries and spinach are at the top of the Environmental Working Group’s list of pesticide-containing foods. 256 Finally, whole-grain products and nuts and seeds (preferably not salted or caramelized) contain fiber and oligosaccharides for microbes to digest and are often affordable options. A recent review with an emphasis on the Mediterranean diet provides “a practical guideline to nutritionists to tailor dietary interventions for patients recovering from COVID-19 infections.” 257
Whereas the focus above is on use of nutrition in immune management and prevention of uncontrolled inflammation, dietary components also have the potential to directly interfere with virus infectivity. One example is prebiotic food that supports microbiome eubiosis, including microorganisms that directly impede viral infectivity (see above). Another principal mechanism is a direct effect of dietary components on viral infectivity. For example, the dietary carotenoid zeaxanthin—that has well-documented, potent anti-inflammatory effects (for recent reviews, see255,258)—has recently been reported to also inhibit the human furin protein 259 that cleaves SARS-CoV-2 spike protein to the form with superior ACE2-binding capacity. 116 The ability of natural products to inhibit furin is also fueling efforts to develop new therapeutic furin inhibitors. 5 Notably, zeaxanthin260-262 also stimulates emergence of mitochondria in fat cells and resulting thermogenesis (fat burning while generating thermal energy), which directly counteracts obesity. Zeaxanthin is not alone in this ability; capsaicin (hot pepper), resveratrol (red wine), curcumin (turmeric root), catechins (green tea), and menthol (mint) have also been reported to stimulate thermogenesis. 263
Another example from among a list of plant-based compounds with both anti-inflammatory and direct antiviral properties is the flavonoid antioxidant quercetin that is currently undergoing testing for the ability to suppress SARS-CoV-2 cell entry, replication, and other desirable effects 6 —in addition to the well-known properties of phenolics in dampening uncontrolled inflammation. 243
2. Regular Exercise with Sufficient Time for Recovery
Whereas regular physical activity can reduce inflammatory markers and boost immunity, exhaustive exercise without sufficient recovery can actually promote non-resolving inflammation8,264,265 and may exacerbate COVID-19 disease risk 266 (Figure 3). Oxidants (ROS) produced in working muscles trigger endogenous synthesis of antioxidant enzymes. 267 Exercise with sufficient recovery supports gut-barrier-integrity and gut microbiome eubiosis268,269 as well as immune system function 270 (Figure 3) and may not only prevent pre-existing non-resolving inflammation, but also counteract the uncontrolled inflammation associated with severe COVID-19. Effective lifestyle-based intervention thus does not only require an extensive commitment to exercise, but regular physical activity with adequate time for recovery is actually preferable. Taking the stairs, walking, and doing stretches and exercises at home all offer substantial benefit. A recent review summarized evidence in support of using regular exercise, with sufficient time for recovery, as an adjunct treatment for individuals living with the human immunodeficiency virus (HIV). 8 The potential of such exercise to prevent severe COVID-19 and manage PASC symptoms warrants attention. For example, exercise training can be more effective than standard drug therapies 271 in managing PASC-associated neurological symptoms such as postural orthostatic tachycardia.272,273 Moreover, combining antioxidant-enzyme-producing physical activity with an antioxidant-replete diet (containing both oil-soluble and water-soluble antioxidants) should be particularly effective (see Ref. 243)—because it takes advantage of the synergy among multiple lifestyle components.
3. Psychological Interventions
Systematic reviews and meta-analyses of available studies indicate that interventions involving yoga,274,275 cognitive behavioral therapy, 276 mindfulness-based meditation practice, 277 or mindfulness-based stress reduction 278 can each lower non-resolving inflammation. While most original studies report reduced markers of non-resolving inflammation as a result of mindfulness-based interventions, results can vary depending on the target population and the environmental stressors in effect. 279 Acupuncture may also be a candidate for the treatment of chronic-inflammation-related disorders. 280 An inexpensive avenue for improving health in as little as 5 to 10 minutes per day is incorporation of stress-reduction techniques, such as deep-breathing sessions, mindfulness practices like a gratitude journal, intentional focus on being in the moment, or taking time for reflection during a walk or run. Moreover, mindfulness practice can serve as a “low-cost beneficial method of providing support” with anxiety and other psychological problems arising from lockdown, quarantines, and other challenges during a pandemic 281 (see also 282 ). It should be noted that “online-based mindfulness programs may be used as complementary interventions for clinical populations, healthy individuals, and healthcare workers with psychological problems due to the COVID-19 pandemic.” 283
Based on the mechanisms stated above, stress management also has the potential to mitigate obesity—by decreasing stress-associated unhealthy eating behaviors with excessive consumption of high-calorie foods. 197 Stress management is indeed associated with weight loss and greater restraint in eating behaviors 284 along with reductions in depression and anxiety.285,286 Consequently, psychological interventions may restore immune defenses by counteracting chronic psychological stress and excessive pro-inflammatory visceral fat accumulation. Psychological interventions may prove particularly important for lessening weight-related stigmatization as exacerbated by the COVID-19 pandemic. Improved support, awareness, and messaging could allow use of “COVID-19 as a teachable moment” to make people feel less overwhelmed and stigmatized. 287
There is evidence that traditional programs for weight loss often result in feelings of shame and guilt, 288 which hinder program success and worsen, rather than improve, self-regulation of behaviors. 289 Addressing and ameliorating stigmatization-related psychological distress has been called for as a necessary first step in achieving positive changes in diet and physical activity level. 290 Psychological interventions emphasizing self-compassion, which counters feelings of shame, can reduce weight-associated shame and promote healthful behaviors. 291 Self-compassion may thus play an important role in perseverance during weight-loss setbacks 292 and management of binge-eating. 293 Indeed, acceptance-based mindfulness was associated with a reduced appetite for highly palatable, energy-dense foods.294,295 In addition to having positive effects on eating behaviors, 296 psychological interventions may also directly counteract stress-related release of glucose from energy stores into the bloodstream 297 and the subsequent conversion of glucose to stored fat. 298
Section 7: Bacteriotherapy as an Adjunct Treatment
Because gut microbiome dysbiosis is associated with severe COVID-19, 299 and may play a role in post-COVID-19 symptoms, 88 ameliorating dysbiosis should help improve clinical outcomes in patients with COVID-19. Probiotic interventions, in the form of oral bacteriotherapy299,300 or fecal gut microbiota transplants 301 may restore eubiosis during COVID-19 and improve clinical outcomes (see also 77 ). A recent study found that commercial probiotic formulations of Lactobacillus rhamnosus reduced the risk of developing symptoms subsequent to SARS-CoV-2 exposure. 302 “Lactobacteria are the most important [beneficial] bacteria in the gut microbiome.” 77 They oppose opportunistic pathogens, improve nutrition, and support an effective, yet balanced immune response throughout the lifespan. Additional research into oral probiotics and fecal transplants as an adjunct therapy during SARS-CoV-2 infection is needed. In contrast, antibiotic use can exacerbate dysbiosis 303 and can be inappropriate given the low frequency of bacterial co-infections during COVID-19. 304 Furthermore, antibiotic use prior to infection may correlate with COVID-19 severity. 305 These recent findings further underscore the impact of the gut microbiome on disease outcomes.
Section 8: Large-Scale Interventions Need to Support Individual Treatment
Access to Balanced Nutrition, Exercise, and Stress Management for all Members of Society
Curbing infection rates and the widespread transmission that promotes emergence of vaccination-resistant mutants of SARS-CoV-2 will require coordinated public health improvements within countries and on a global scale (Figure 6). Structural change in the management of public health is necessary to make these interventions accessible to all members of society and enable clinicians to effectively incorporate such lifestyle interventions into preventative care for all of their patients. For example, because cost and geographic inaccessibility are barriers to the purchase of health-promoting foods,
306
reduction of cost may positively impact fruit and vegetable consumption, although more research is needed.
307
Education on affordable, health-promoting foods, coupled with expanded access to such foods, may produce the most positive effects.
306
Similarly, increased green space (natural environments with green vegetation) and blue space (bodies of water) is associated with higher levels of physical activity
308
and physical and mental wellbeing,
309
and new, accessible walking paths correlated with increased physical activity.310,311 Expanding access to safe spaces for outdoor physical activity may thus serve as one avenue to increasing physical activity level. Systematic implementation of stress-reduction programs can also improve health; school-based mindfulness interventions provided lasting stress reduction and improvements in attention.
312
Expanded training for implementation of these programs may allow for widespread access to stress-reduction techniques and their beneficial effects on mental and physical health. Depiction of interactions among various components of modern human society that could act synergistically to decrease disease severity and persistence. By managing human-wildlife contact, enacting policies that improve public awareness of pandemics and ensure global surveillance and cooperation, and expanding access to a health-promoting lifestyle to boost human immunity, zoonotic disease emergence, and severe disease manifestations, may be prevented.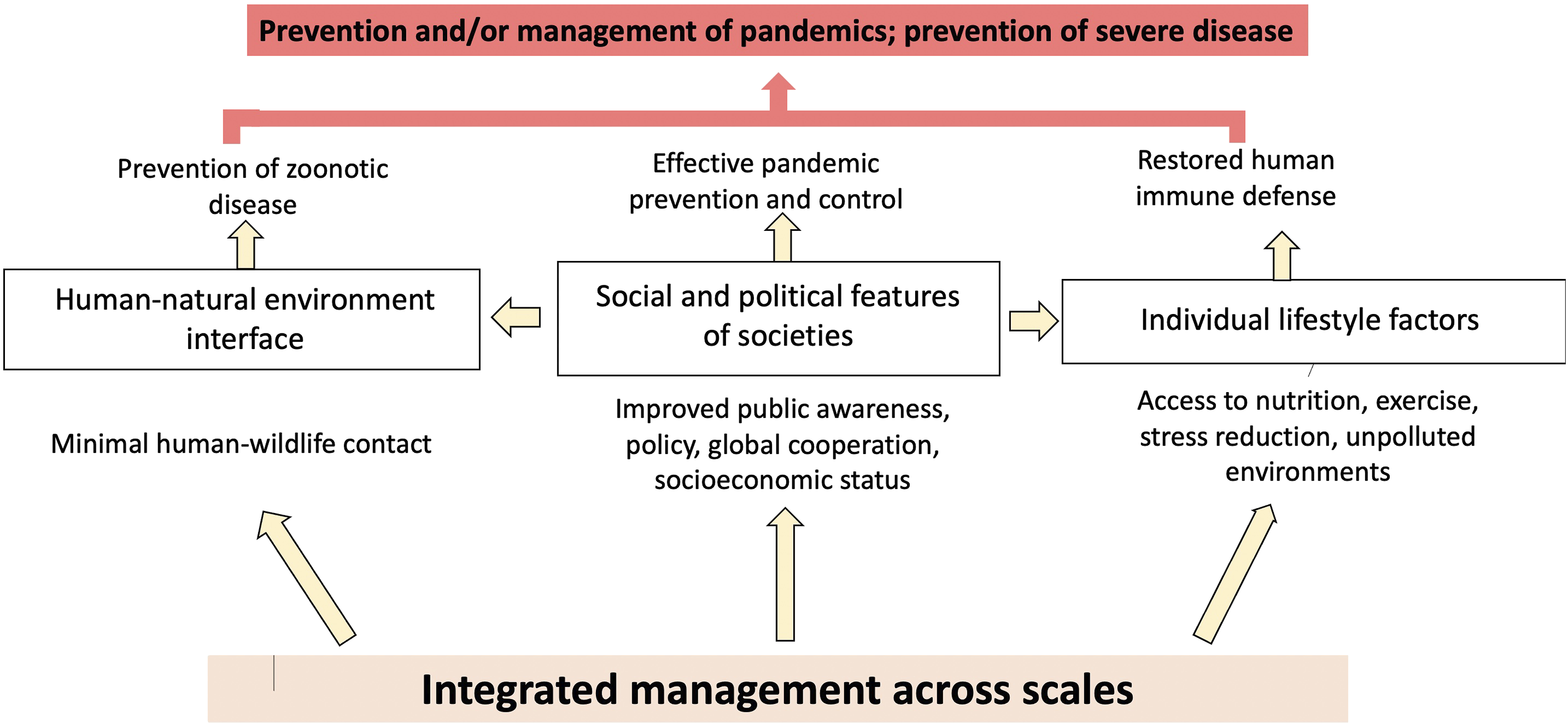
Managing public health additionally requires systematic approaches to combatting misinformation. Misinformation about the COVID-19 vaccine, 313 masks, 314 and social distancing 315 have all hobbled the global response to SARS-CoV-2 and likely led to unnecessary suffering and death. Managing pandemics like COVID-19 therefore requires effective dissemination of scientifically backed data coupled with protective measures to prevent the spread of false claims. Policy changes mandating vaccination for other diseases, fluoridation of water, regulation of tobacco, and sanitation requirements have been instrumental in achieving many of the foundational health improvements of the 20th century. 316 Similarly, policy change that expands access to healthcare and a health-promoting lifestyle is needed now to boost public health and reduce the impact of diseases like COVID-19.
Finally, the larger issues that impact public health overall also determine the ability to control new emerging infectious diseases. The high cost of healthcare and health insurance to individuals, coupled with difficulties in navigating the associated bureaucratic systems, continue to make care least accessible for those who often require it the most. Universal healthcare, better unemployment benefits, and pollution control by expanded environmental regulation will thus likely contribute to a populace/workforce that is both less sick and more productive/innovative, as is demonstrated in societies using this approach.317-322 Decreased poverty rates could improve health by expanding access to healthy foods and physical activity163,323-325 and reducing chronic psychological stress, 326 thus creating an environment in which lifestyle recommendations can be effectively implemented.
Management of Natural Resources and Wildlife
Pandemic prevention and/or management will require not only public health and lifestyle practices aimed at bolstering human immune defenses and countering underlying non-resolving inflammation (sections on nutrition, exercise and stress management and bacteriotherapy as an adjunct treatment above), but also coordinated policies to reduce human contact with wildlife, such as bats and their pathogens (Figure 6). Anthropogenic changes to the natural environment,327-331 as well as wild-meat trade,332,333 and international trade and globalization334,335 have increased human-wildlife interaction336-341 and facilitated rapid emergence of pandemics. Preventing (zoonotic) pathogen transmission from wildlife to humans thus requires management of natural resources, 342 wildlife, 343 and animal trade. 344 For example, public investment in wild-animal-health infrastructure would benefit producers of animal products (by reducing the negative impact of sick animals) as well as consumers of these products (by preventing transmission of pathogens). 345 Furthermore, improved global-disease surveillance systems across human and animal populations could enable early detection of zoonosis and save billions of dollars in after-the-fact spending necessary to mitigate effects of epidemics and pandemics. 345 The earlier a potential pathogen is detected, the easier it typically is to mitigate its effects. 346
Figure 6 summarizes the principal conclusions of this review, for example, that preventing severe disease manifestations of COVID-19 will require individual lifestyle interventions aimed at reducing underlying inflammation and restoring gut microbiome composition. In addition, public health measures are needed to expand access to lifestyle interventions and allow clinicians to effectively and equitably incorporate these prophylactic recommendations into care for all patients. Similarly, global management of natural resources and wildlife trade is needed to prevent future pandemics.
Footnotes
Acknowledgments
Thank you to Robert Adams for his feedback on the structure and content of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
