Abstract
Introduction: Nutritional intervention, such as a whole-food plant-based (WFPB) diet, is suggested to improve symptoms of autoimmune disorders. Its effects on neuromuscular junction (NMJ) diseases are less known. Case Description: A 56-year-old female with a combined myasthenia gravis and Lambert-Eaton myasthenic syndrome presented with persistent musculoskeletal weakness and pain, fatigue, and diminished concentration. Methods: A WFPB diet was implemented for 5 months in this case study. Functionality, biometrics, and quality of life were assessed at regular intervals via blood work, vital signs, and patient surveys. Results: After 10 weeks, 2 medications were de-prescribed due to improvement in blood glucose levels and blood pressure readings. Neuromuscular symptoms lessened as measured by the Myasthenia Gravis Illness Index. Weight loss of 18.7 pounds was achieved. No deterioration in physical activity was reported throughout the 5-month trial of WFPB diet. Discussion: Our results have implications that an individualized dietary strategy may be considered as one component of management of NMJ diseases. The effects of WFPB diet on NMJ diseases remain inconclusive.
‘. . . a WFPB [whole-food plant-based] diet may provide clinical benefit to a patient with autoimmune NMJ [neuromuscular junction] disease.’
Neuromuscular junction (NMJ) diseases are autoimmune disorders that cause impairment of conduction at the NMJ, which causes focal or generalized weakness. Myasthenia gravis (MG) and Lambert-Eaton Myasthenic Syndrome (LEMS) are 2 widely known NMJ diseases. MG is an autoimmune disease that results in the blockade of postsynaptic acetylcholine receptors. 1 LEMS is caused by antibodies produced against presynaptic calcium channels, preventing the release of acetylcholine-containing vesicles. 2 While approximately 25% of patients with MG have coexisting autoimmune disorders such as rheumatoid arthritis and systemic lupus erythematosus, 3 only a handful of cases document the coexistence of MG and LEMS.4-7
Lifestyle intervention through nutrition, such as a whole-food plant-based (WFPB) diet, has reportedly been associated with objective and subjective improvements in various medical conditions, ranging from cardiovascular disease to autoimmune disorders.8-14 We hypothesized that a WFPB diet may provide clinical benefit to a patient with autoimmune NMJ disease. Our aims through this case study were to (1) document the clinical course of a WFPB diet trial in a patient with combined MG and LEMS and (2) summarize its findings.
Case Description
A 56-year-old Caucasian female with a rare diagnosis of combined, seropositive MG and LEMS consulted a clinical pharmacist for medication management. Her chief complaints included persistent muscle weakness, pain, and decreased cognitive function. Her goals for the pharmacy consultation were reduction in pain and improvement in symptoms. She also expressed hopes for a possible de-escalation of her complicated medication regimen. She is allergic to penicillin and sulfa, which cause anaphylaxis, and to adhesive tape, which causes rash and raised skin. The list of her prescription and nonprescription medications at the initial visit is summarized in Table 1. Additional ancillary services she utilizes include aquatic physical therapy twice a week, acupuncture, and massage therapy.
List of Prescription and Nonprescription Medication of the Patient at Initial Presentation and Indications Provided by the Patient.

Initiated 2 weeks into the study period, discontinued at 2 months after acquiring influenza virus secondary to suppressed immune system, and re-initiated after her hip arthroplasty.
Patient was unable to confirm the dose of Rituxan.
Past medical history at the onset of this case study includes small fiber neuropathy, migraine without aura, steroid-induced diabetes, thyroid hormone dysregulation and thyroid nodule, iron deficiency anemia, anxiety, rheumatoid arthritis with chronic daily oral steroid use, microscopic colitis in remission, osteoarthritis, fibromyalgia, rotator cuff tears, and chronic low back pain. She had earlier been tested negative for celiac disease. She denied having undergone testing for non-celiac gluten sensitivity or intolerance. She had trialed a gluten-free diet in the past and noted minimal to no symptomatic relief for her neuromuscular symptoms. Relevant past surgical history includes thymectomy.
Her family history includes coronary artery disease and cerebrovascular disease in her father, and hypertension, breast cancer, and depression in her mother. She smoked socially during the second decade of her life but quit approximately 30 years ago. She is a former alcohol user and quit 14 years ago. She does not use any recreational drugs. Prior to the case study, she was on a diet that mostly excluded red meat and dairy. She occasionally is physically active beyond what is required for work or home through aquatic therapy and arm exercises, which sometimes cause her pain. Her ambulation is limited due to her NMJ disorder and avascular necrosis of the hip. She was unemployed before and during the case study. When she was diagnosed with combined MG-LEMS, confirmed as seropositive for both conditions based on antibody testing and electromyography at a large academic medical center, it was recommended that she no longer receive immunizations for pneumonia, influenza, or shingles.
Her health care team includes primary care physician, health/nutrition coach at a bariatric center, gastroenterologist, gynecologist, clinical psychologist for weekly counseling, chiropractor, pain specialist, neurologist, endocrinologist, rheumatologist, orthopedist, and neuro-ophthalmologist.
Methods
The study period ranged from January 6, 2020, through June 2, 2020. The intervention was led by a clinical pharmacist, who is a core faculty at a Midwestern community-based family medicine residency program, and a board-certified ambulatory care pharmacy specialist, with an interest in lifestyle medicine.
One week prior to the WFPB diet implementation, the patient was given general recommendations on diet with freedom to choose foods within the guidelines. The patient was instructed to avoid animal products, minimize highly processed foods, focus on consuming whole foods, and limit extraneous sources of fat such as vegetable oils commonly used in cooking. She was allowed to consume gluten-containing products as she had previously tested negative for celiac disease, and her experience with a gluten-free diet prior to the case study made no subjective symptomatic difference.
The patient was to check blood pressure once every day, weight once weekly, and document the findings in the electronic health record. All foods consumed were to be logged in a mobile application (MyFitnessPal). 15 Home visits were made at the onset of the WFPB diet trial and at 3 months, where the clinical pharmacist collected vitals and initial physical assessments.
For follow-up, the clinical pharmacist had regular phone visits to check in on the patient and monitor her progress. The phone visits occurred weekly for the first 2 months, then in 1- to 2-month intervals. At each visit, surveys were administered, vital signs were recorded as measured by the patient, education and/or reinforcement regarding the WFPB diet was provided, and any patient concerns were addressed. An example of patient concerns includes a request for a sample diet plan in the event of a flare of her microscopic colitis, in remission at the time of inquiry, to ensure her adherence to the WFPB diet during the study period.
The survey forms were administered pre-WFPB diet intervention, and 1- and 5-month post-WFPB diet intervention. The forms administered include Satisfaction with Life Scale (SWLS), 16 Generalized Anxiety Disorder-7 (GAD-7), 17 Fibromyalgia Impact Questionnaire (FIQR), 18 and World Health Organization Quality of Life (WHOQOL). 19 The Myasthenia Gravis Impairment Index (MGII) was administered pre-intervention and at 2-months postintervention. 20 Subjective concerns and feelings of the patient were documented when appropriate at each check-in phone visit.
This case study did not meet the criteria of human subjects research as determined by the local institutional review board (IRB Reference #1605419-1). The patient gave verbal and written informed consent for this case study to be published.
Results
During the 5-month study period, the research team conducted 12 check-in phone visits and 2 home visits. Table 2 summarizes the biometric and laboratory data collected before the start of WFPB diet, at 1-, 2-, and 5-month follow-up. Within 30 days of starting WFPB diet, the patient lost 10.4 pounds of weight from her initial reading. By 5 months into the trial, she weighed 18.7 pounds less from her pre-diet weight. Her blood pressure readings stayed within normal limits. Fasting blood glucose and body mass index (BMI), obtained only at 1-month post-diet due to unavailability of data, did not show hypoglycemia or hyperglycemia and had decrease in BMI.
Biometric and Laboratory Data From Pre-WFPB Diet Intervention and 1-, 2-, and 5-Month Post-WFPB Diet Intervention (Check-In Visits #4, #8, #12).
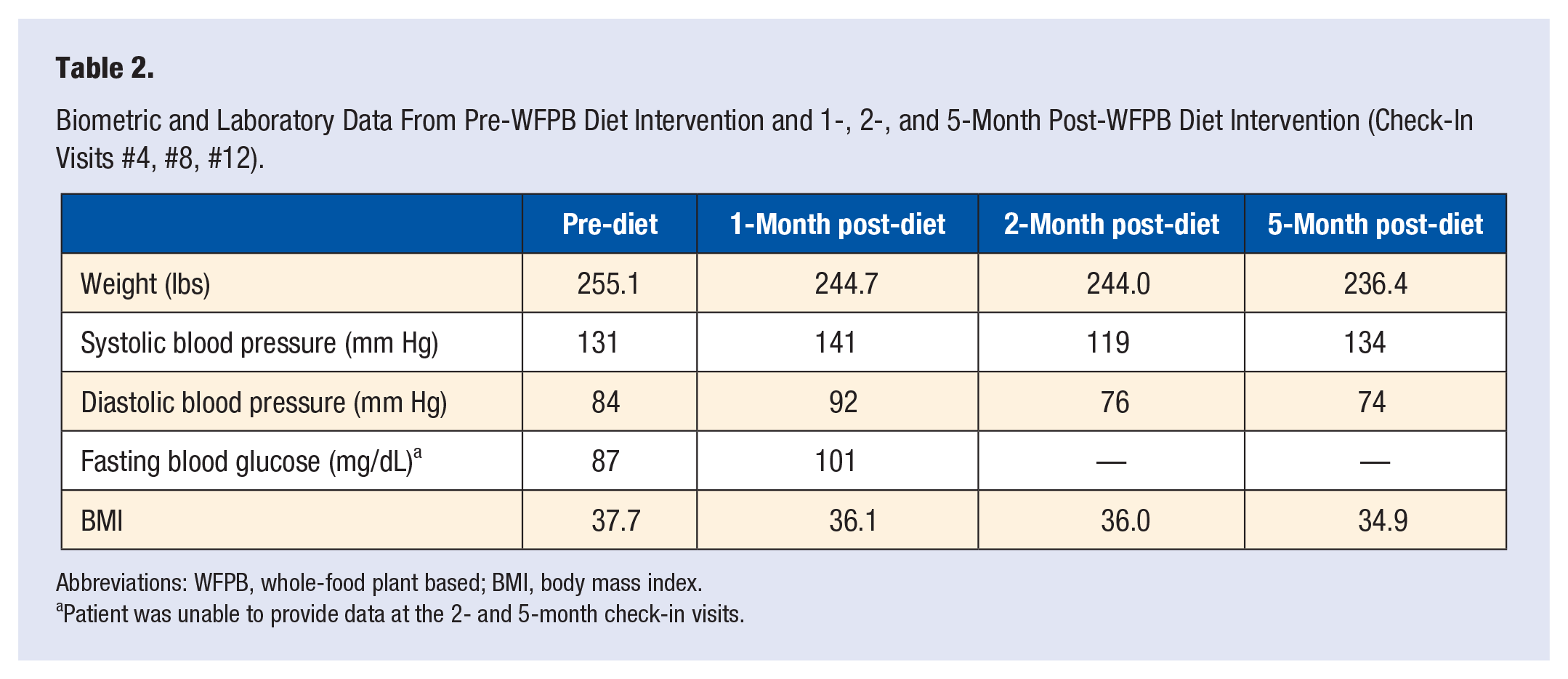
Abbreviations: WFPB, whole-food plant based; BMI, body mass index.
Patient was unable to provide data at the 2- and 5-month check-in visits.
Table 3 summarizes the survey responses recorded in numeric scores before the start of WFPB diet, at 1-, and 5-month follow-up. MGII was administered pre-diet, and 2-month post-diet. Her scores improved with regard to her satisfaction with life, pain reduction related to fibromyalgia, and in all 4 domains of quality of life at 5-month post-diet. Her GAD-7 scores improved at 1-month post-diet but returned to baseline at 5-month post-diet. Her MGII scores related to generalized symptoms of NMJ disease improved, while her ocular symptoms, such as diplopia and ptosis, and lower facial strength remained stable.
Survey Responses From Pre-WFPB Diet Intervention and 1- and 5-Month Post-WFPB Diet Intervention.
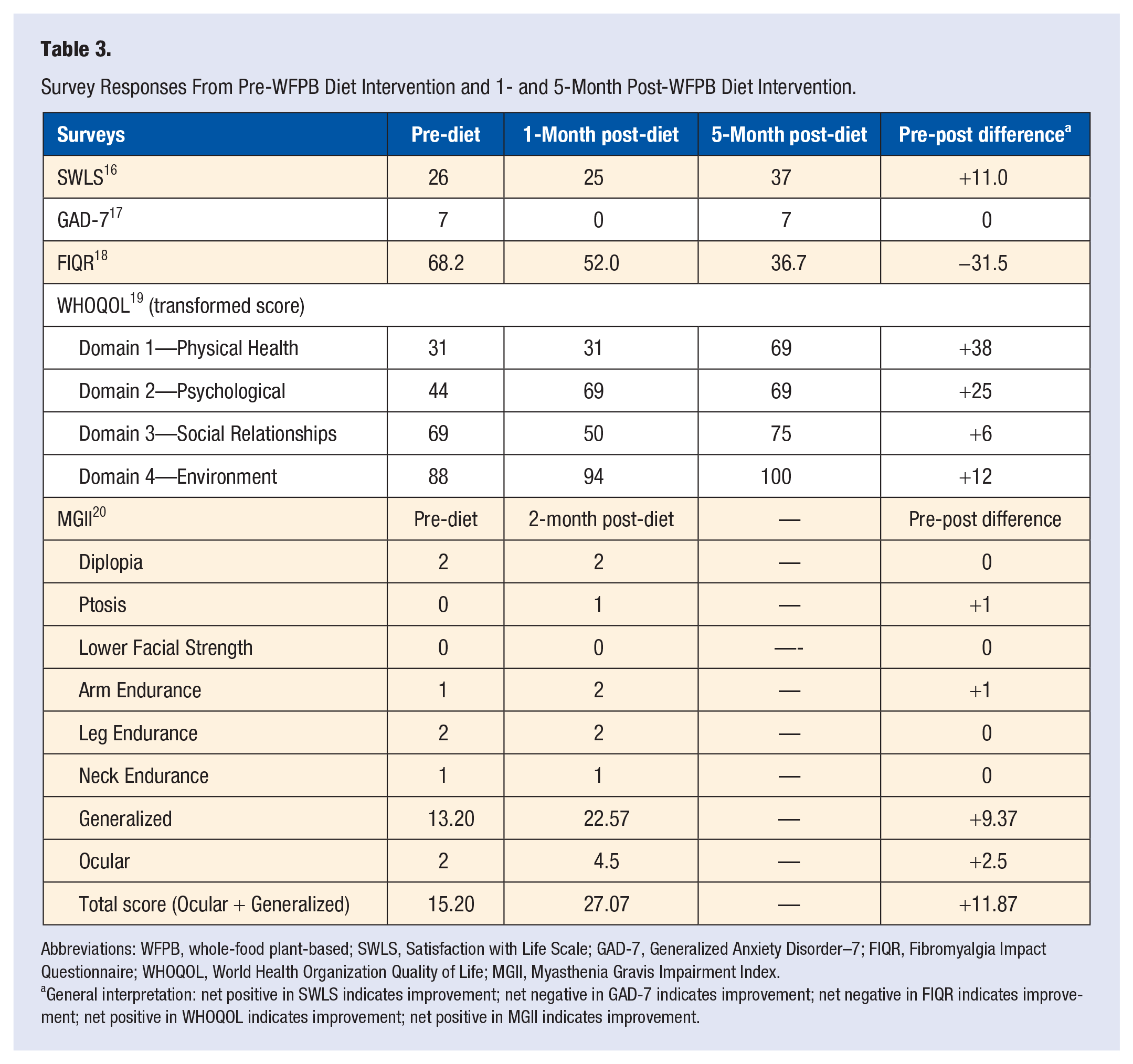
Abbreviations: WFPB, whole-food plant-based; SWLS, Satisfaction with Life Scale; GAD-7, Generalized Anxiety Disorder–7; FIQR, Fibromyalgia Impact Questionnaire; WHOQOL, World Health Organization Quality of Life; MGII, Myasthenia Gravis Impairment Index.
General interpretation: net positive in SWLS indicates improvement; net negative in GAD-7 indicates improvement; net negative in FIQR indicates improvement; net positive in WHOQOL indicates improvement; net positive in MGII indicates improvement.
The patient achieved de-prescription and de-escalation of her medications during the study period. By 1 month, she was able to discontinue spironolactone and Januvia while maintaining fasting blood glucose levels and blood pressures at goal. By 5 months, she was able to discontinue lisinopril, saccharomyces boulardii (probiotic), polyethylene glycol 3350, low-dose naltrexone, turmeric, tart cherry, and cannabidol/tetrahydrocannabidol (CBD/THC) gummies. Fexofenadine had been replaced with fluticasone nasal spray for her seasonal allergies to avoid potential counteraction between amifampridine, which is a cholinergic agonist for treatment of myasthenia gravis. Medications that were de-escalated since the start of the WFPB diet include decreased dose of levothyroxine, decreased frequency of lidocaine patch use, and decreased frequency of diclofenac-misoprostol use.
At each follow-up, interval history of the patient was explored and documented. Subjectively, the patient did not feel she had notable improvement in her joint pain or muscle strength during the first month but did report improvements at 2 months and beyond. One month into the WFPB trial, she did mention she had improved mental clarity, which improved her motivation to engage in activities. The patient noted she was able to read books more easily and for longer duration than pre-WFPB diet intervention. She continued the medical management of her NMJ diseases during the study period. The patient did suffer from steroid-induced avascular necrosis of the hip and underwent hip replacement at month 3 of the study period but wished to continue the WFPB diet. Figures 1 and 2 illustrate the individualized lifestyle intervention prescribed to address patient concerns related to her comorbidity, such as microscopic colitis, and perioperative caloric intake. She did not experience a microscopic colitis flare during the study period.

A paraphrased example of individualized WFPB diet action plan for prevention of and plan during microscopic colitis flare.

A paraphrased example of individualized action plan for calories supplementation with snacks.
Discussion
The results of this study provide implication on the role of WFPB diet in the management of NMJ diseases. Lifestyle choices of adults in the United States continue to contribute to many leading causes of death.21,22 Growing evidence highlights the potential use of nonpharmacologic, lifestyle intervention strategies for the management of various medical conditions.23,24 Challenges remain in elucidating the mechanism behind the therapeutic effects of lifestyle medicine, such as the WFPB diet. 25 The concept of gut-brain axis allows for plausible explanation. 26 Current medical management of NMJ diseases focuses on immunomodulation.27-29 Myasthenia gravis appears to create a unique intestinal microbiome profile. 30 Correlation between autoimmune-mediated gastrointestinal disease and NMJ disorders, and the influence diet has on intestinal microbiota and disease susceptibilities, further suggest that WFPB diet may affect the neurogastrointestinal system.3,31-35
The MGII appears to be useful in monitoring subjective changes in the overall symptoms related to myasthenia gravis while on a WFPB diet. This case study found that our patient experienced subjective improvements in the generalized symptoms during the study period. No significant improvements, however, in the function of specific muscle groups such as the ocular muscles were reported. The MGII was created using a qualitative study method by interviewing 20 patients with myasthenia gravis. 36 This led to the discovery that previous tools, which assessed the function of specific muscle groups at single time points, may be limited in capturing the overall quality of life of a patient with MG. Based on these interviews, the authors of MGII implemented a more holistic approach to capture how patients with myasthenia gravis subjectively felt with regard to their illness in addition to the single point assessments of specific muscle groups. 20 In conjunction with the result of our case study, it is reasonable to consider MGII as a monitoring tool in patients with MG who implement a WFPB diet.
This report does not produce generalizable knowledge. The patient was highly motivated; thus, the Hawthorne effect cannot be ignored. The role and boundaries of a nonphysician prescribing lifestyle medicine, the interdisciplinary collaboration between prescribers of lifestyle medicine and traditional medical professionals, proper and accurate documentation, appropriate billing and reimbursement, and efficient delivery of services within the primary care setting in the United States require further evaluation beyond the scope of this article.
Conclusion
A 56-year-old female with combined MG and LEMS underwent 5-month trial of a WFPB diet. She experienced subjective improvement in overall satisfaction with life, quality of life, pain reduction related to fibromyalgia, and generalized symptoms of neuromuscular disease. We saw improvements in the control of her other chronic medical conditions, such as hypertension and diabetes, in addition to a near 19 pounds weight loss during the study period. Subsequently, several medications were successfully de-prescribed and de-escalated. The mechanism behind the observed therapeutic effects of WFPB diet on NMJ diseases remains inconclusive. We nonetheless report herein that the WFPB diet brought several clinical benefits to this patient without worsening her NMJ disease for the duration of the trial.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This case study did not meet the criteria of human subjects research as determined by the local institutional review board (IRB Reference #1605419-1).
Informed Consent
The patient gave verbal and written informed consent for this case study to be published.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
