Abstract
Breast cancer is the second most common type of cancer in women worldwide, where nutritional intervention should be part of a multidisciplinary lifestyle approach in oncology, promoting therapeutic success. Insulin-like growth factor 1 (IGF-1), along with estrogen, can promote the development of neoplastic cells in breast tissue. Cancers that develop under IGF-1 stimulation are often resistant to therapy. This case report describes a 47-year-old woman, body mass index 27.4 kg/m2, with HER2-positive breast cancer, as well as elevated blood glucose, total cholesterol, and low-density lipoprotein cholesterol. Soon after her breast cancer diagnosis, she transitioned from a Western pattern diet (WPD) to a predominantly whole-food, plant-based diet (PWFPBD) for 1035 days, followed by 232 days of PWFPBD plus night fasting for 16 hours per day. IGF-1 decreased 22.38%, glycemia and total cholesterol decreased by −55.06% and −36.00% at the end of the first intervention and went up by 6.25%, and 3.87%, respectively, at the end of the second intervention. A PWFPBD, with or without 16-hour overnight fasting, seems to modulate plasma levels of IGF-1 on a 47-year-old woman diagnosed with breast cancer, type HER2-positive. Future research, should explore the physiologic and pathophysiological mechanisms and clarify whether this dietary strategy, may be clinically useful in preventing HER2-positive breast cancer.
‘It is estimated that about 40% of all cancers can be prevented, by sustainable lifelong changes in dietary patterns, . . .’
According to the Global Cancer Observatory (GCO) and GLOBOCAN 2018 database, breast cancer is the second most common type of cancer worldwide, with about 2.1 million cases in 2018. It is the type of cancer with the highest incidence rate in most countries (154 of 185) namely Australia/New Zealand, Northern Europe, and North America, and also the leading cause of cancer death in over 100 countries. 1
In Portugal, in 2018 there were 6974 new cases of breast cancer, representing 27.1% of all cancers in women and an estimated 3.4% increase in incidence rate by 2030.2,3
The risk of developing breast cancer increases with age until menopause, approximately 2-fold higher risk every 10 years. 3
Survival rates vary but have improved worldwide, by early detection in countries where the population has the greatest access to medical care and therapeutic strategies. In some countries, the 5-year survival of early-stage cancers is 80% to 90%, decreasing to 24% for advanced breast cancer, which seems to indicate a critical need for improved treatment in metastatic cases. 4
It is estimated that about 40% of all cancers can be prevented, by sustainable lifelong changes in dietary patterns, reduced alcohol consumption, increased physical activity, and maintaining a healthy body weight. 5
Insulin-like growth factor 1 (IGF-1) is an insulin-like hormone, synthesized in the liver, bone, and muscle cells, stimulated by pituitary growth hormone (GH) and with an anabolic tissue effect responsible for growth and cell proliferation. 6
In childhood, the actions of this growth factor are necessary for proper development, however, in adulthood increased levels of IGF-1 may promote an increased risk of cancer and decreased life span. 7
The role of this hormone and its receptors in the pathogenesis of breast cancer has been widely investigated and appears to have a strong implication in tumor growth and malignant disease progression. 8
IGF-1 signaling pathways play an important role in the regulation of apoptosis (programmed cell death that acts as a protective mechanism). Research, as well as in vitro and in vivo models, indicate that increased levels of circulating IGF-1 are associated with an increased risk of several common cancers.6,8
These 2 mechanisms, accelerated growth and decreased apoptosis, are hallmarks of cancer cells and reasons why increased IGF-1 levels appear to show a carcinogenic effect.9-12
Several therapeutic strategies targeting the IGF-1R receptor have shown impressive anti-carcinogenic activity in laboratory models. 6
There appears to be a well-established link between increased serum IGF-1 levels and increased risk of breast cancer in premenopausal women. 13 Studies show a 60% to 86% increased risk compared to women with low serum levels and risk may increase up to 150%.9,14
A meta-analysis, which gathered data from 17 different prospective studies, found that women with higher IGF-1 levels had a 28% higher risk of breast cancer, regardless of their pre- or postmenopausal status. 15
Cancers that develop under IGF-1 stimulation are often resistant to chemotherapy and radiation, and recent evidence suggests that IGF-1 along with estrogen promotes the development of neoplastic cells in breast tissue.16-18
Some studies report that people with Laron syndrome, a genetic deficiency in growth hormone receptor (GHR) and IGF-1, have a reduced prevalence of type 2 diabetes mellitus (T2DM) and cancers, and otherwise have a longer life span, compared with unaffected relatives.19-23
Insulin, the hormone responsible for glycemic reduction, and its receptors: Insulin receptor (IR), insulin receptor substrates (IRS), and insulin-like growth factor 1 receptor (IGF-1R) promote cell division, proliferation, and migration as well as activation of other signaling proteins such as growth factor receptor-bound protein 2 (GRB2) and mammalian target of rapamycin (mTOR), a cell proliferation and growth mediating enzyme, responsible for tumor progression. 24
Rapamycin is an antifungal agent produced by soil bacteria that inhibits mTOR and prolongs life in animal models.25,26 Thus, inhibition of mTOR may slow down aging and have benefits in preventing chronic diseases such as cancer, diabetes, neurodegenerative diseases, heart disease, and metabolic syndrome. 25
Researchers report that one of the reasons why calorie restriction is effective in increasing longevity is because it triggers a decrease in insulin/IGF-1 signaling pathways.22,27
Given the importance of the role of IGF-1 signaling pathways in general markers of health and comorbidities, the study of their determinant’s bioactivity is relevant. The systematic review and metanalysis by Rahmani et al 29 focused on the influence of dietary changes (excess or low consumption) on circulating IGF-1 levels, concludes that fasting significantly reduced IGF-1 levels, while calorie restriction was only effective if energy intake reduced by more than 50%. 28
Fasting then appears to have the potential to delay or prevent the onset of age-related metabolic and neoplastic diseases. 28
Fasting in its various forms, prolonged, intermittent, or periodic, can be an important way of preventing various chronic conditions and increasing longevity by activating cellular protective mechanisms such as mTOR inhibition, autophagy activation, and decrease IGF-1 levels in the body, causing a decrease in cell growth and proliferation.28,29
Cell autophagy or auto-digestion is a cellular pathway involved in recycling amino acids and dysfunctional organelles. Changes in this autophagic protective system are associated with numerous pathologies, including cancer. 30
Most scientific research focuses on what to eat for breast cancer prevention, such as specific foods, food groups, or dietary patterns. Recently it has been shown that when to eat also seems to have equal importance. The right time for food intake influences metabolic health and cancer.31,32 Marinac et al, 31 in a study of 2413 women diagnosed with breast cancer, suggest that those who fasted less than 13 hours at night had a 36% higher risk of relapse, compared with those who fasted for at least 13 hours. In this research, fewer night fasting hours are associated with higher HbA1c levels and less sleep, which may partly explain the risk of relapse as well as the increased risk of diabetes, cardiovascular disease, and other cancers. The study concludes that extending the length of the night fasting interval may be a simple, nonpharmacological strategy to reduce the risk of breast cancer recurrence. Improvements in glucose and sleep regulation may be mechanisms that link fasting to breast cancer prognosis. 31
The report from World Cancer Research Fund (WCRF)/American Institute for Cancer Research (AICR) “Diet, Nutrition, Physical Activity, and Cancer: A Global Perspective—A Summary of the Third Expert Report,” provides a comprehensive analysis, based on the latest scientific evidence, on cancer prevention and survival, through diet and physical activity. 33 According to this report and the World Health Organization (WHO), it is estimated that 30% to 50% of all cancers could be prevented through a healthy lifestyle: by being at a healthy weight; being physically active; eating a diet rich in whole grains, vegetables, fruits, and beans; by limiting consumption of “fast foods” and other processed foods high in fat, starches, or sugars; by limiting consumption of red and processed meat, sugar and sweetened drinks, and alcohol; by not using supplements for cancer prevention; by mothers breastfeeding their babies; by avoiding exposure to carcinogens, environmental pollution, and prolonged infections.33,34
Several studies seem to confirm the importance of adhering to as many recommendations as possible for the prevention of various types of cancer. According to Catsbur et al, 35 in a study that included 49,613 women who were followed over 16 years, following 6 to 7 recommendations is associated with a 31% decrease in breast cancer risk.
Adherence to at least 5 recommendations reduced the risk of breast cancer by 60% in postmenopausal women. 36
Incidence of breast cancer tends to be lower in countries where traditional diets based on plant foods prevail, namely the Mediterranean.37-39
A noticeable increase in the incidence of breast cancer has been seen in immigrants, who abandon traditional foods (such as cereals, vegetables, and soy-based foods) and have adopted Western diets (WD), rich in meat, dairy, and fat. 40
A predominantly whole food plant-based diet (PWFPBD) dietary pattern is similar in many respects to a Mediterranean diet, which is based on regular consumption of olive oil (as the main source of added fat), on the consumption of plant foods (whole grains, fruits, vegetables, nuts, and seeds), moderate consumption of fish and dairy products, moderate consumption of red wine and low and limited consumption of red meat, has been receiving particular attention from health professionals, and may also be associated with a decreased risk of some cancers, especially breast cancer.41-45
According to Catsburg et al, 46 a dietary pattern composed predominantly of plant products seems to be associated with a 27% decrease in breast cancer risk. However, a pattern characterized by meat and potatoes was associated with a 26% increased risk of postmenopausal breast cancer. In conclusion, the study suggests that a plant-based diet that limits red meat consumption is associated with a decreased incidence of breast cancer, particularly in the postmenopausal phase. 46
The Adventist Health Study 2 (AHS-2), a prospective study that included 69,120 participants, reports that a vegan dietary pattern was more effective in preventing breast and gynecological cancers in women and is associated with a 34% risk reduction. 47
In summary, the dietary pattern promoted by the WCRF/AICR, and based on a review of the available literature, which consists predominantly of plant foods and limits hypercaloric and animal products, especially red and processed meat, is associated with a decreased risk of breast cancer, and several studies have shown the benefits of adhering to these recommendations.33,35,36
Materials and Methods
The participant is an active 47-year-old woman, architect by profession, with reported menarche at the age of 14 years, 3 children, the first child at the age of 28 years, and breastfed for 2.5 years total.
In October 2013, a professionally validated diagnosis of breast cancer type HER2-positive, with metastatic right axillary involvement (16 affected axillary lymph nodes) was detected by routine (every 2 years) ultrasound review, had no history of previous illnesses.
She was physically active in her younger years, but after giving birth kept moderate physical activity for 1 to 2 times per week for 1 hour and followed a Western pattern diet (WPD) until 2013, characterized by the ingestion of industrialized products, centered on animal foods, red and processed meat, saturated fat, refined cereals, free sugars, and with a low intake of fruits, vegetables, and legumes. No alcohol intake or tobacco habits were reported.
Two dietary interventions were performed: the first during chemotherapy and radiotherapy and the second one after all therapeutic protocols ended.
The first intervention consisted of a transition to a PWFPBD, followed for 1035 days, the second intervention consisted of a 16 hours per day fasting followed for 232 days, and after each dietary intervention blood samples and anthropometric data were collected (Figure 1).
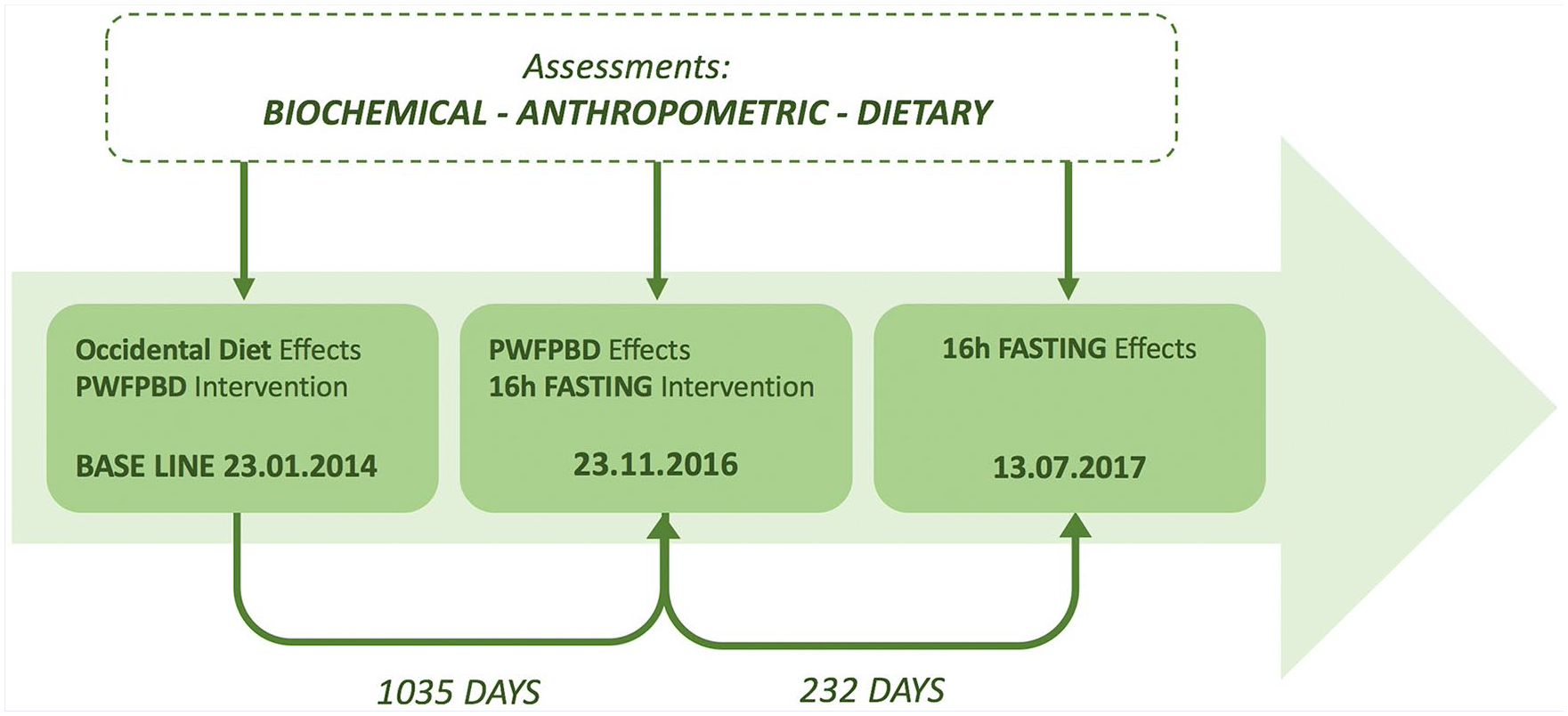
Baseline-occidental diet effects and 2-moment intervention time line charts: predominantly whole food plant-based diet (PWFPBD), and 16-hour fasting.
Baseline
At baseline, blood samples were taken and analyzed by a professional laboratory.
Participant body mass (w; kilograms) and height (h; meters) were measured, through a calibrated scale used throughout the process, before eating and after bladder and bowels voided, and body mass index (BMI) (WPD) was calculated as shown in equation: BMI = w/h2 (kg/m2). The BMI classification was based on WHO (2015) European standards. 48
Diet was assessed by a direct retrospective 24-hour recall, of one typical weekday, conducted by a registered dietitian. Nutritional assessment was conducted to determine energy intake and nutrient composition through “Cronometer,” a nutrition and fitness tracker, using predominantly entries from Nutrition Coordinating Center Food and Nutrient Database (NCCDB). 49
Predominantly Whole Food Plant-Based Diet—First Intervention
In this intervention, a transition to a healthy PWFPBD was performed gradually in 1 week, and followed for 1035 days. The diet was based on vegetables, fruits, whole grains, legumes, nuts, oilseeds, seeds, olive oil, and spices, and minimized animal proteins, saturated fat, eggs, dairy, processed products, and added sugars, adapted to local, and individual preferences. The participant attended a theoretical-practical course, for 3 months, on “The Role of Food in Cancer Prevention,” 50 where she learned about new food benefits and how to cook healthfully.
At the end of this period of intervention, the nutritional assessment was made by a 4 consecutive days retrospective dietary history report, conducted by a registered dietitian. Nutritional assessment, blood samples, and anthropometric analysis were made the same way as the baseline.
Sixteen-Hour Fasting—Second Intervention
A 16:8 intermittent fasting intervention was conducted for 232 days, in which food was consumed during an 8-hour window and fasting for the remaining 16 hours each day, allowing only water intake. The same dietary pattern as in the previous intervention was followed, trying to change only the mealtime frame.
After this intervention, nutrition, blood samples, and anthropometric analysis were performed the same way as PWFPBD, first intervention.
Results
Body Composition
The participant measured 1.63 m in height and, at the end of the 3 phases analyzed, body weight varied considerably from 69 kg, 57 kg, to 54 kg, respectively, representing a 15 kg decrease during the 3.5 years of total both interventions.
BMI went from a baseline of 27.40 to 21.70 kg/m2 after the first intervention, which translates to a reduction of −20.80%, and 20.60 g/m2 representing a −5.07% reduction after the second intervention (Table 1).
Anthropometric and Serum Levels Analysis From 3-Moment Intervention: At Baseline, PWFPBD, 16-Hour Fasting, and Difference Percentage. a
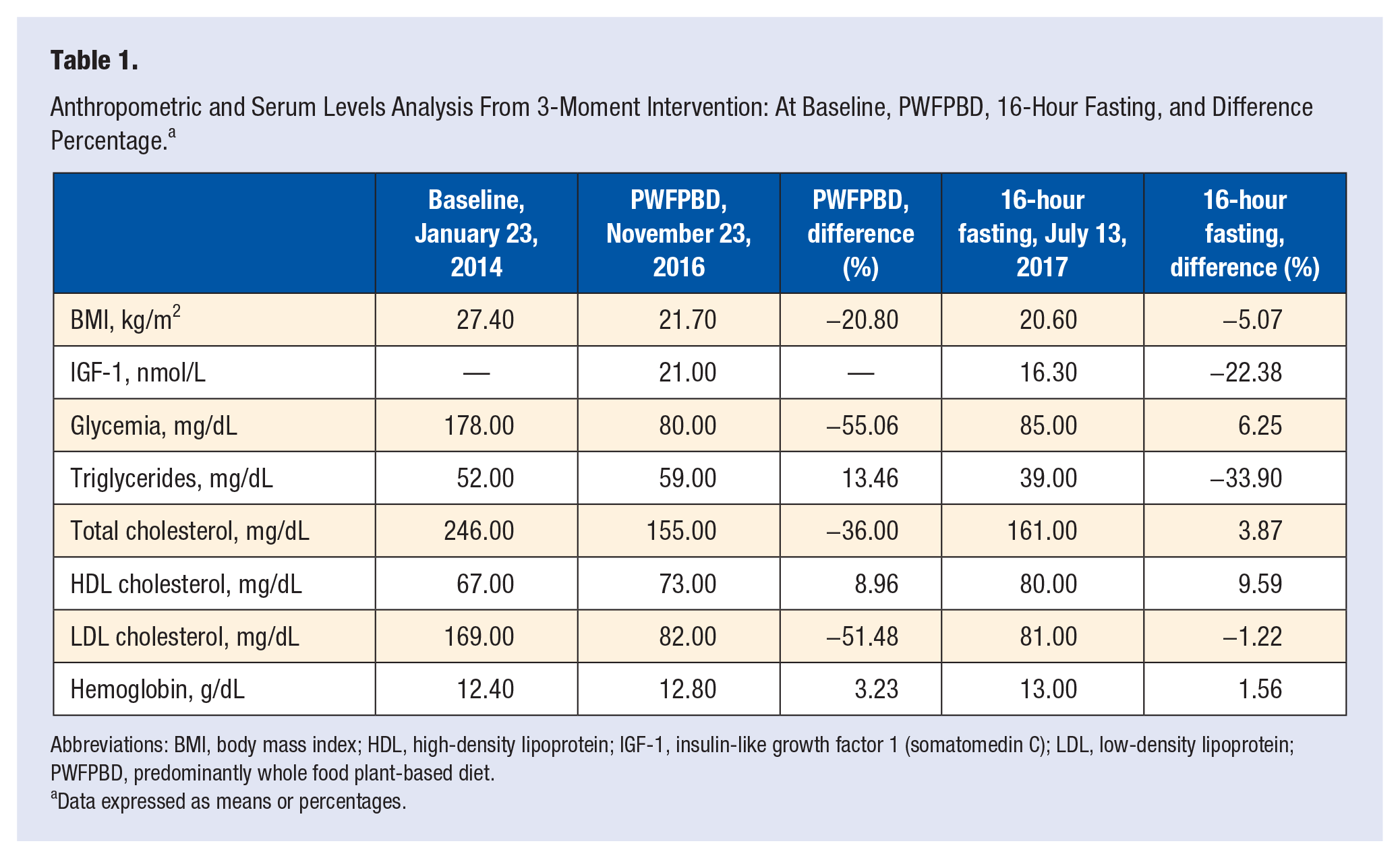
Abbreviations: BMI, body mass index; HDL, high-density lipoprotein; IGF-1, insulin-like growth factor 1 (somatomedin C); LDL, low-density lipoprotein; PWFPBD, predominantly whole food plant-based diet.
Data expressed as means or percentages.
Daily Energy Expenditure
Daily energy expenditure, at baseline, using Harris-Benedict equation, 51 was 1927 cal/d and decreased to 1756 cal/d and 1712 cal/d after the first and second intervention, respectively.
Blood Analysis
The serum levels of IGF-1 were not analyzed at baseline due to lack of availability; however, there was a reduction from 21.00 nmol/L to 16.30 nmol/L at the end of the second intervention, which represents a reduction by −22.38% (Table 1).
At the end of the first intervention: there was a decrease of fasting glycemia and total cholesterol by −55.06% and −36.00% (HDL increased by 8.96% and LDL decreased by −51.48%), and an increase of triglycerides and hemoglobin by 13.46% and 3.23%, respectively (Table 1).
At the end of the second intervention, fasting glycemia, total cholesterol, and hemoglobin went up by 6.25%, 3.87% (HDL went up 9.59% and LDL went down −1.22%), and 1.56% respectively, and triglycerides went down −33.90% (Table 1).
Energy Intake and Dietary Assessment
At baseline typical day average energy intake was 2981 kcal/d after the first intervention changed to 1612 kcal/d representing a −45.93% reduction, and by the end of the second intervention was 1601 kcal/d decreasing only by −0.65% (Table 2, Figure 2).
Diet Analysis From 3-Moment Intervention: At Baseline (1 day), PWFPBD (Mean of 4 Days), and 16-Hour Fasting (Mean of 4 Days).
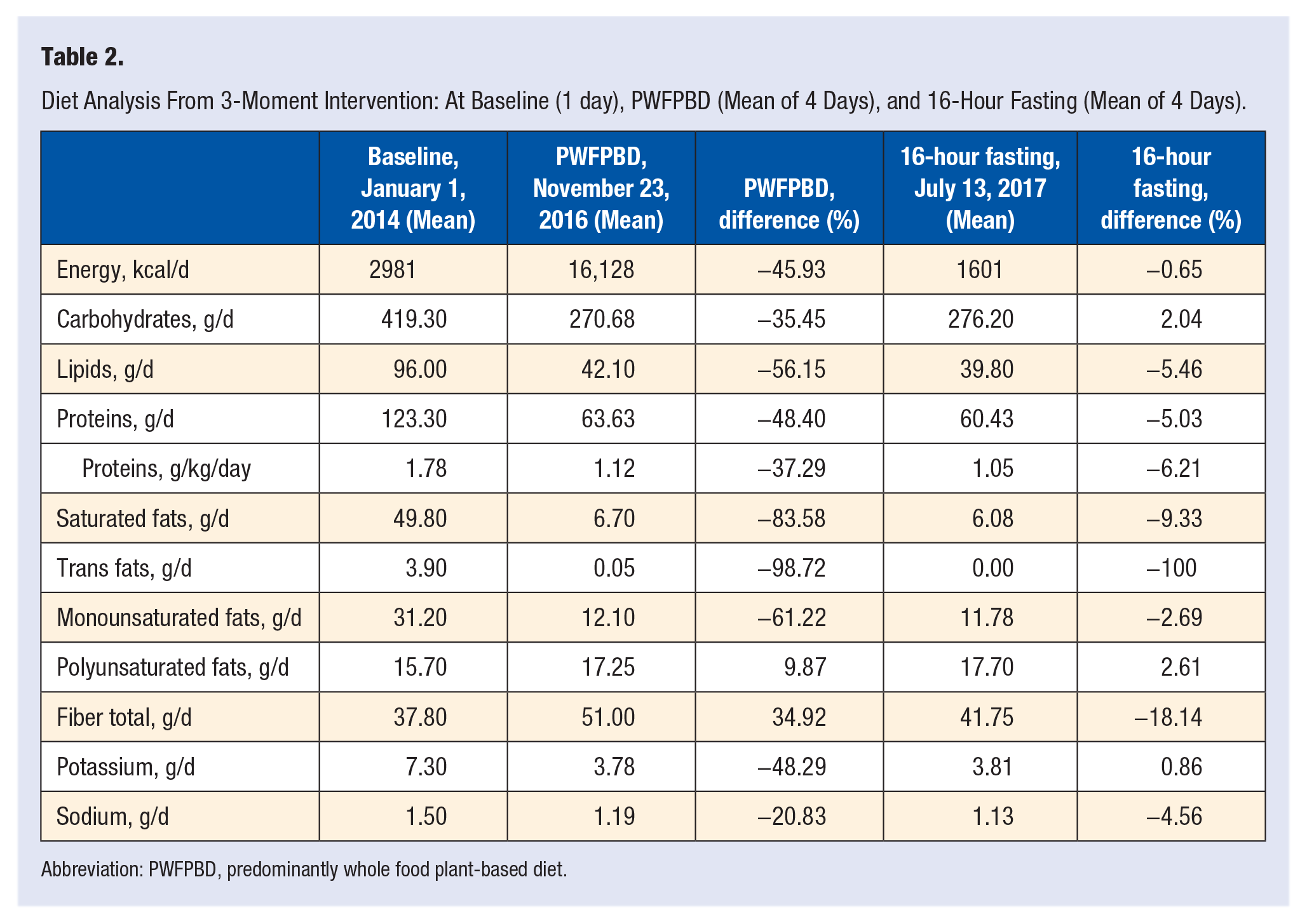
Abbreviation: PWFPBD, predominantly whole food plant-based diet.
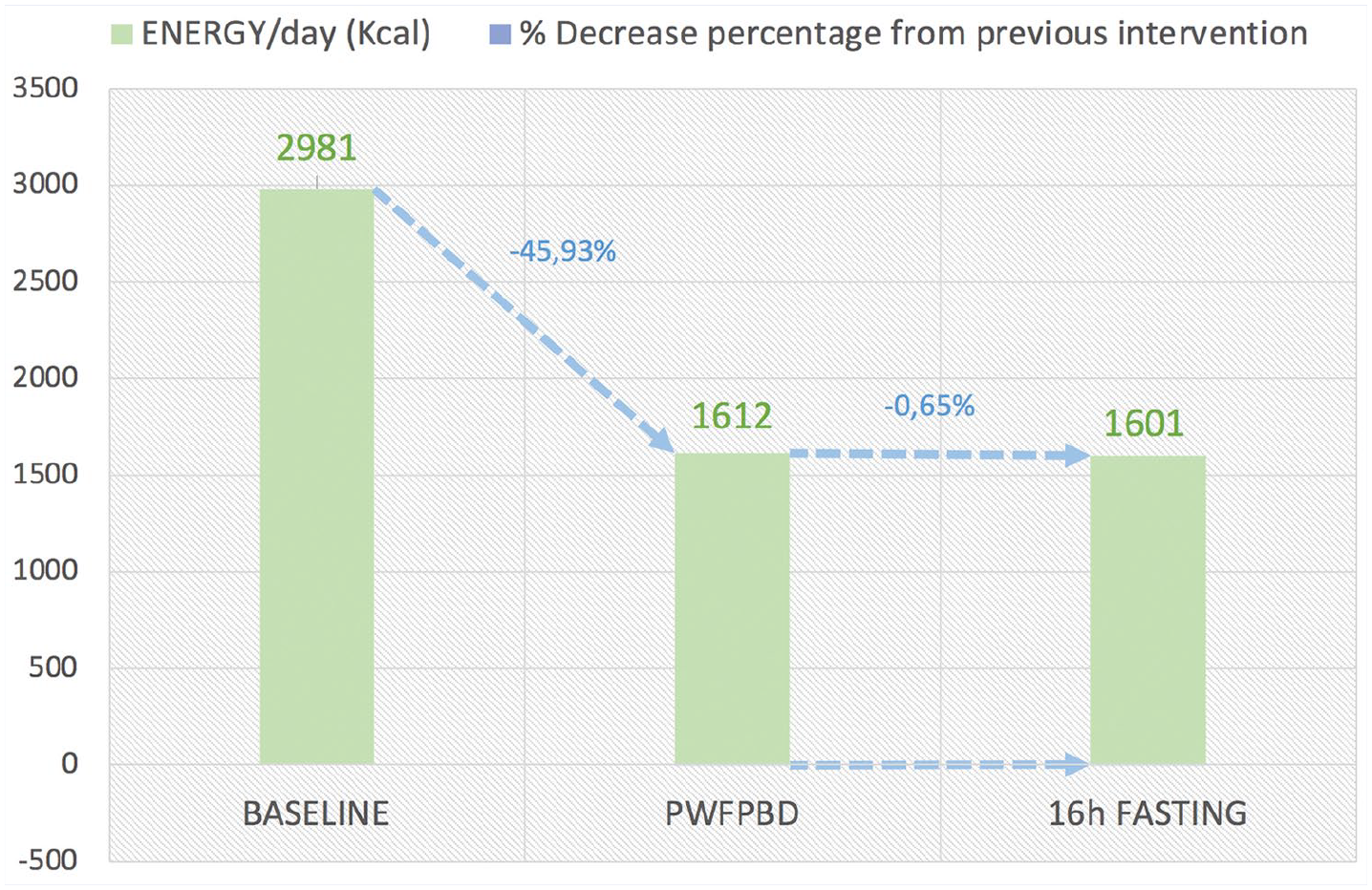
Diet analyses, total kilo calories per day, according to 3-moment intervention: baseline, predominantly whole food plat-based diet (PWFPBD), and 16-hour fasting.
Macronutrients
Up to baseline, the average intake of carbohydrates was 419.30 g/d and composed mostly of the refined type. At the end of the first intervention, the type became whole carbohydrates, and average consumption decreased by −35.45% to 270.68 g/d. At the end of the second intervention, the type of carbs remained the same, with an average consumption of 276.20 g/d, representing a decrease of −2.04% (Table 2, Figure 3).
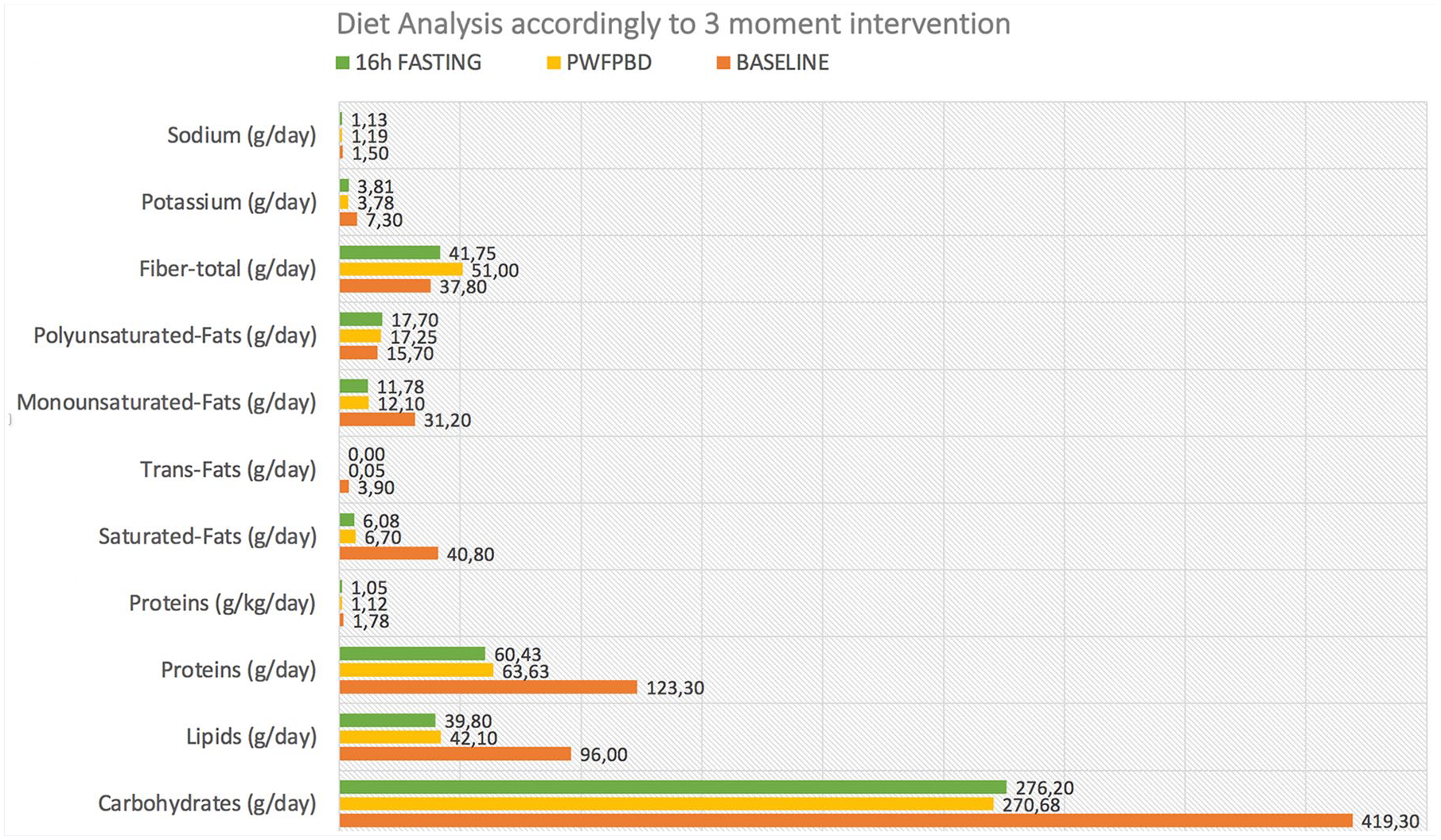
Diet analyses according to 3-moment intervention: baseline, predominantly whole food plant-based diet (PWFPBD), and 16-hour fasting.
Total fiber average intake increased by 34.92% after the first intervention and decreased by −18.14% on the second intervention (Table 2, Figure 3).
The average consumption of lipids at baseline was 96.00 g/d, which after first intervention decreased by −56.15, and on second intervention by −5.46% (Table 2, Figure 3).
Saturated fat average consumption initially was 49.80 g/d, which after the first intervention decreased considerably by −83.58% to 6.70 g/d (Table 2, Figure 3).
Trans fats average intake at the baseline was 3.90 g/d, descended abruptly by −98.72% after the first intervention, and by 100% after the second intervention to zero consumption.
The average protein consumption initially was 123.30 g/d, which at the end of the first intervention decreased by −48.40%, and at the end of the second intervention by −5.03% (Table 2, Figure 3).
Micronutrients
The average intake of potassium was 7.30 g/d at baseline, which decreased by −48.29% after the first intervention, and increased by 0.86% to 3.82 g/d after the second intervention (Table 2, Figure 3).
Sodium consumption initially was 1.50 g/d and decreased by −20.83% and −4.56% after the first and second intervention, respectively (Table 2, Figure 3).
Discussion
The starting point for this case study was based on the change in the participant’s behavior after receiving the diagnosis: moving from a more passive state to an active state, empowered and focused on her health care, transforming an obstacle into an opportunity for change. The possibility of improving health status through nutritional care was the driving factor, in an attempt to assist the ongoing medical protocol therapy with the objective of a positive pathological outcome. The WPD consumed by the participant until 2013, when breast cancer was diagnosed, was reflected in the serum and anthropometric data reported. At baseline, the BMI analysis of 27.40 kg/m2 classified the participant as “pre-obesity level” (25.00-29.90 kg/m2), according to the WHO/Europe | Nutrition body mass index - BMI 52 (Table 1). A glycemia of 178.00 mg/dL (≥126 mg/dL) could be classified as “diabetes,” however and according to Portuguese Diagnosis and Classification of Diabetes Mellitus - General Directorate of Health, it should be confirmed in a second analysis after 2 weeks 53 (Table 1). Total cholesterol of 246 mg/dL (>190 mg/dL) classifies the participant at baseline, as “high cardiovascular risk,” according to the criteria of the Portuguese Cardiology Foundation 54 (Table 1). IGF-1 (somatomedin C) values have not been tested at baseline.
The first intervention consisted of changing to a PWFPBD pattern and attending a 3-month course on the role of food in cancer prevention and healthful cooking. With this intervention, energy intake was reduced naturally by 45.93%, the participant reported greater food awareness and greater satiety despite consuming fewer calories. Body weight dropped −15 kg, and a new BMI of 21.70 kg/m2 implied a requalification of the nutritional status from “preobesity” to “normal.” according to the European classification. 48 This substantial decline seems to have had a positive impact on the participant’s health (Table 1). Glycemia, at this phase, dropped to 80.00 mg/dL, values now considered “normal” (60-110 mg/dL), 55 which reveals a protective effect of the PWFPBD (Table 1). Total cholesterol decreased to a healthy 155.00 mg/dL (<200 mg/dL), which fits the participant outside the standard of cardiovascular risk, now to “normal,” 54 also showing a protective effect of this dietary intervention (Table 1). Serum IGF-1 was 21 nmol/L and framed and placed “normal,” in the normal range for an adult woman, between 11.6 and 48.4 nmol/L according to the Laboratory of Clinical Analysis and Medical Diagnosis | “SYNLAB” Portugal 56 (Table 1). High circulating levels of this hormone, appear to promote the growth of tumors and the progression of malignant diseases, 8 and increase risk of breast cancer in women in pre- and postmenopause.9,13-15
The second intervention, 16:8 intermittent fasting, was based only on changing the feeding schedule and trying to maintain a similar dietary pattern, as well as energy intake (with a minor difference of −0.65%), as in the first intervention (Figure 2). Body weight dropped by 3 kg, maintaining the BMI classification as “normal.” However, blood glucose rose 6.25%, which may suggest an effect on glucose metabolism or, possibly, be a reflection of daily variation without clinical significance since it refers to a single reading and still within a healthy range.
Some researchers suggest that IGF-1 behaves similarly to insulin, by the association to the family of six carrier proteins, called insulin-like growth factor binding proteins (IGFBPs), and reactive affinities for both receptors (insulin receptor and IGF-1 receptor). Some studies suggest that IGF-1 has beneficial effects on blood glucose levels, on the other hand, while others suggest the opposite. 57 . Marinac et al31,58 suggest that extending the night fasting length interval, may be a simple nonpharmacological strategy to reduce breast cancer recurrence risk and that improvements in glucose levels and sleep regulation may be mechanisms that link fasting to breast cancer prognosis. The impact of intermittent fasting on blood glucose levels seems uncertain and more research is needed on the functioning of the IGF-1/insulin axis as a whole, its role in pathophysiology, as well as the implications of manipulating its components in cancer prevention.
Total cholesterol rose by 3.87%, but driven predominantly by an increase in high-density lipoprotein (HDL) by 9.59%, which may be positive according to Cedó et al 59 (Table 1). IGF-1 levels dropped by −22.38% (Table 1), suggesting a possible modulating effect of the second intervention on serum levels. As mentioned, fasting can be a relevant way to prevent chronic diseases, and increase longevity through the activation of protective cellular mechanisms such as inhibition of mTOR, the promotion of autophagy, and the decrease of serum IGF-1 levels acting as “brakes” of cell growth and proliferation.28,29 Thus, this case study seems to reflect what Marinac et al 31 and Hutchison et al 32 refer to: Both the type of food and time of intake may be equally important. The right time to eat appears to influence metabolic health and cancer prognosis.
Strengths and Limitations
During the study, the participant started a professional bachelor’s degree course in nutrition sciences, which allowed her to have access to information and tools that promoted her adherence to the plan in an effective and sustained manner. Future participants with less training in nutrition could find adherence to the intervention more challenging.
This participant was eating a healthier-than-average Western diet when she started; the results might have been even more pronounced in someone with a more typical Western diet at baseline.
Also, a single-day food assessment is not representative of a typical baseline dietary pattern.
Another limitation was the use of dietary recalls, affected by memory bias of self-reported dietary intake variability. Further studies should include multiday food records or multiple 24-hour dietary recalls. The study could have greater conclusive weight if the sample analyzed had more than 1 participant. IGF-1 was not tested at baseline, which could have represented a greater detail on the modulatory effects of the first intervention on circulating levels of IGF-1.
Suggestions for Performing New Works on the Topic
Ideally, these interventions would be repeated with a larger sample and for a shorter time intervention, to further elucidate the relationship between the modulatory role of diet on circulating levels of IGF-1 and its role in primary prevention as well as prevention of breast cancer relapses.
Conclusion
A healthy dietary pattern, PWFPBD, followed initially for 1035 days, and then with overnight fasting of 16 hours daily for another 232 days, seems to modulate plasma levels of IGF-1 from 21.00 nmol/L to 16.30 nmol/L, representing a decrease of −22.38% on a 47-year-old woman diagnosed with breast cancer, type HER2-positive.
Although case reports cannot be considered as proof of efficacy, when added to other cases of patients approached with the same diet method, they suggest that this could be a viable coadjuvant intervention for breast cancer patients to improve prognosis and reduce the risk of relapses. Future research, through randomized double-blind clinical trials, is needed to confirm these results on serum IGF-1 levels, explore the physiologic and pathophysiological mechanisms through which PWFPBD and night fasting affect IGF-1, and determine whether these dietary, non-pharmacological strategies may be clinically useful in preventing HER2-positive breast cancer or as an adjuvant treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Cíntia Ferreira-Pêgo is funded by the Foundation for Science and Technology (FCT) Scientific Employment Stimulus contract with the reference number CEEC/CBIOS/NUT/2018. This work is funded by national funds through FCT–Foundation for Science and Technology, I.P., under the UIDB/04567/2020 and UIDP/04567/2020 projects.
Ethical Approval
Written informed consent was obtained from the patient, for the publication of this case report.
