Abstract
‘. . . sufficiently intensive lifestyle modifications are capable of producing significant clinical improvements in patients with T2D.’
Objectives
The objectives of this position paper are to (1) present the position of the American College of Lifestyle Medicine (ACLM) on type 2 diabetes (T2D) treatment and remission, (2) provide support for this position by reviewing the evidence base and key intervention studies on T2D remission, and (3) issue a call to action for adopting remission as the appropriate treatment goal.
The position of the ACLM, informed by current best research evidence, is that (1) sufficiently intensive lifestyle modifications are capable of producing significant clinical improvements in patients with T2D and (2) that the optimal treatment to bring about remission (defined below) includes a whole food, plant-based (WFPB) dietary pattern coupled with moderate exercise. A WFPB diet emphasizes fruits and vegetables, legumes, and whole grains, and includes nuts and seeds while eliminating or minimizing animal foods such as red and white meat, poultry, fish, eggs, and dairy, as well as refined foods that include added sugars and oils. ACLM is in agreement with the position stated by the group who conducted the Counterpoint Study on the reversibility of T2D, “Diabetes reversal should be [the] goal in the management of Type 2 diabetes.” 1
Furthermore, we propose that remission should always be held as the
Background
Prevalence of T2D globally was 108 million in 1980. 2 By 2014, the global population registered 422 million T2D cases, and prevalence is expected to rise another 50% to 642 million by 2040. 3 Today in the United States, the total of diagnosed and undiagnosed cases is approximately 34.2 million, and as many as 88 million are prediabetic. 4 The total yearly cost of diagnosed diabetes in the United States is estimated to be $327 billion, including $237 billion in direct medical costs and $90 billion in decreased productivity. 5 The highest rates of diabetes are among older, more obese patients, and patients with hypertension; while those with longer diabetes duration, and those with diabetic complications, especially chronic kidney disease, have the highest mortality rates. 6
The current “disease management” approach for patients with T2D is primarily aimed at delaying progression of the disease rather than remission.7,8 In addition to the burden of disease and expense, causes for concern include the development of end-stage organ damage such as retinopathy, nephropathy and end-stage renal disease, nonalcoholic fatty liver disease, and cardiovascular disease.7-9 We are now seeing increased disease and mortality in younger populations with T2D. 10 A shift toward treatment that can not only prevent but also reverse T2D and produce complete remission is the single greatest need in T2D care. The field of lifestyle medicine (LM) is poised to make this contribution.
Several definitions of T2D remission have been circulated. The ACLM uses the definition published by Buse et al in 2009, 11 which requires blood glucose levels on par with a healthy individual without diabetes.
Remission is defined as achieving glycemia below the diabetic range in the absence of active pharmacologic (anti-hyperglycemic medications, immunosuppressive medications) or surgical (ongoing procedures such as repeated replacements of endoluminal devices) therapy . . . of at least 1 year’s duration . . .
Partial remission is subdiabetic hyperglycemia (A1c not diagnostic of diabetes [<6.5%], fasting glucose 100 to 125 mg/dL)
Complete remission is a return to “normal,” measures of glucose metabolism (A1c in the normal range [<5.7%], fasting glucose <100 mg/dL)
Another leading definition of remission comes from the Association of British Clinical Diabetologists (ABCD) and the Primary Care Diabetes Society (PCDS), 12 who propose a similar but simpler and less stringent definition requiring blood glucose levels to fall below the diagnostic criteria of diabetes (what Buse et al define as “partial remission”) for a period of only 6 months. Individuals who meet the stricter definition of remission from Buse et al would therefore also meet the British definition.
A growing number of clinical experts are discussing the concept of remission as a treatment goal,13-16 inspired by the outcomes observed following weight loss, 17 fasting/fasting mimicking diets, 18 bariatric surgery, 19 and, more recently, intensive lifestyle modifications. 20 Given the current upwards trend in diabetes incidence, recognizing the need for sufficiently intensive lifestyle change in diabetes treatment is essential to advance solutions that treat the cause of disease and contain the spiraling cost 5 of diabetes management.
Methods
A literature search was performed in PubMed with no date restrictions to identify papers with search terms “diabetes” and either “remission” or “reversal” in the title through April 9, 2020. A total of N = 914 results from this literature search were returned, which were title-screened by a single investigator (MK) for relevance to diabetes remission in populations with T2D. A total of n = 52 potential papers were identified after title screening the results from this literature search. In addition, the literature search was combined with hand searching, reviewing reference lists of review papers, and prior knowledge of the authors. Following title and abstract screening, the list of papers of potential relevance were confirmed by a second investigator (JK). Full-text papers were reviewed as needed.
Key intervention studies relevant to the topic of T2D remission were selected (n = 9). Studies were included as key examples of either effective interventions with appropriate, therapeutic dosing levels (n = 5), or ineffective interventions with subtherapeutic dosing levels (n = 4). The distinction between therapeutic versus subtherapeutic dosing was not solely based on treatment goals but rather on whether the intervention was able to produce remission results among a substantial proportion of participants. Studies were included if they met the following minimum criteria: (1) they were intervention trials of any design conducted in humans, (2) they examined remission of T2D, (3) diabetes medication use was reported, and (4) the definition of “remission” incorporated fasting plasma glucose, hemoglobin A1c (HbA1c), and/or oral glucose tolerance test.
Studies that were conducted in animal models or were not focused on remission of T2D were excluded. Studies were not assessed for risk of bias or level of evidence.
Descriptive data were extracted by a single investigator (KC) and checked by a second investigator (MK), with assistance from 2 additional investigators (JK and GS). The data extraction form was approved by all coauthors. Data tables were formatted through consensus-discussion and revised from the data extraction form to their final form as presented in Tables 2 and 3. If more than one paper was published on the same study, the most relevant publication to the topic of remission was selected.
Weighted mean remission rates were calculated by multiplying the remission rate for each study by the number of subjects in the study, then dividing the sum of those calculations by the sum of the number of subjects in the studies combined. A complete remission rate of zero was assumed for those studies only reporting partial remission. A partial remission rate equal to the complete remission rate was assumed for studies only reporting complete remission.
Results
Literature Review
Included interventions used a variety of lifestyle modifications to achieve T2D remission, typically involving diet and exercise. The definitions of remission varied, including using serum glucose measures alone, or in combination with other measures and being medication-free. Summary characteristics of included studies are presented in Table 1.
Summary Characteristics of Intervention Studies Examining T2D Remission.
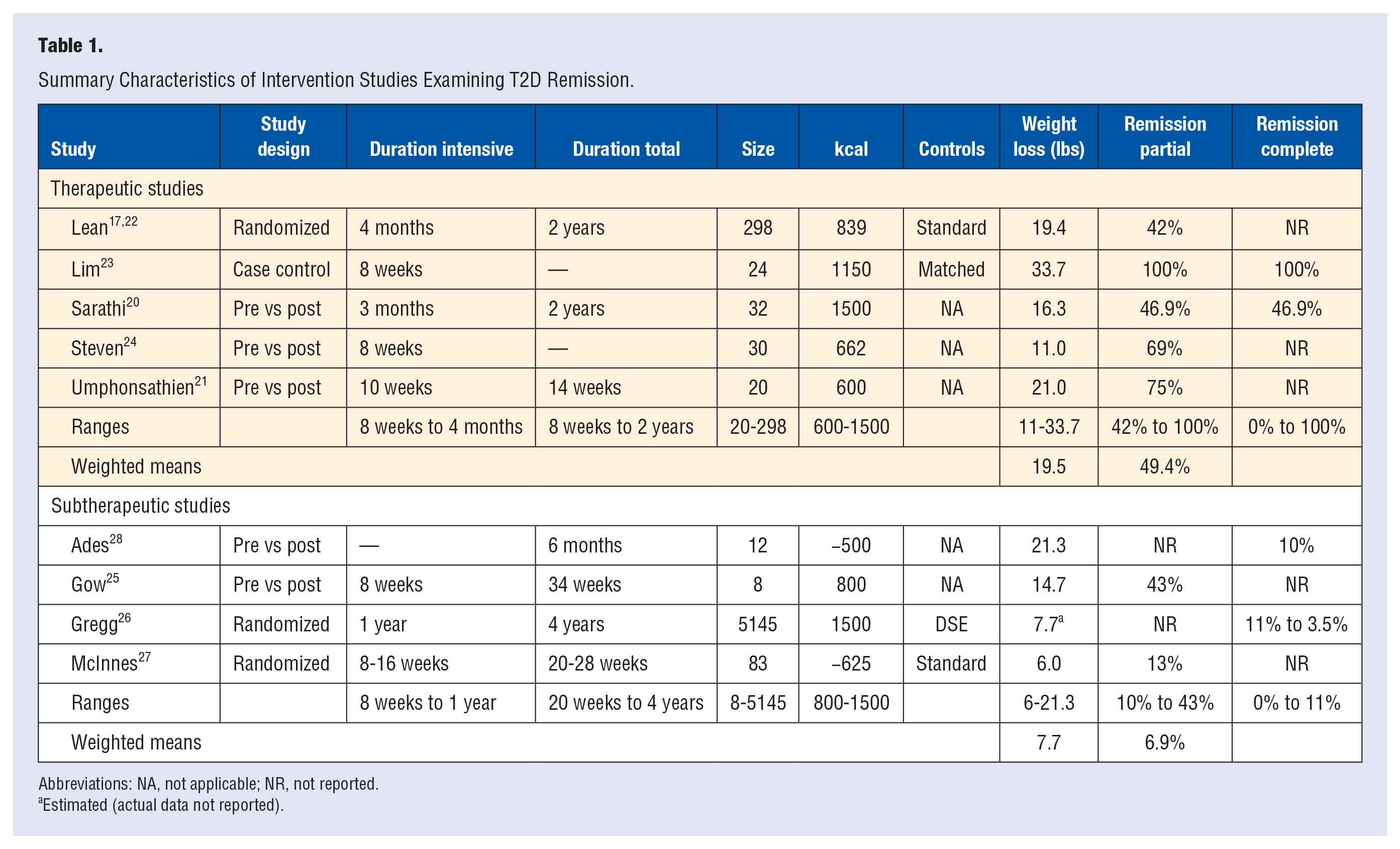
Abbreviations: NA, not applicable; NR, not reported.
Estimated (actual data not reported).
The 5 therapeutic studies (Table 2) ranged in sample size from 20 21 to 29817,22 subjects. Designs included one cluster-randomized trial, 22 one case-control pilot study with a single arm, 23 and 3 single-arm, pre-post interventions.21,20,24 The rates for partial remission ranged from 42%17,22 to 100%, 23 with a weighted mean of 49.4%. The rates for complete remission ranged from 0% to 100%. 23 Weight loss ranged from 11 24 to 33.7 23 lbs with a weighted mean of 19.5 lbs. The caloric content of the intervention diets ranged from 600 21 to 1500 20 kcal.
Type 2 Diabetes Interventions With Therapeutic Dosing.
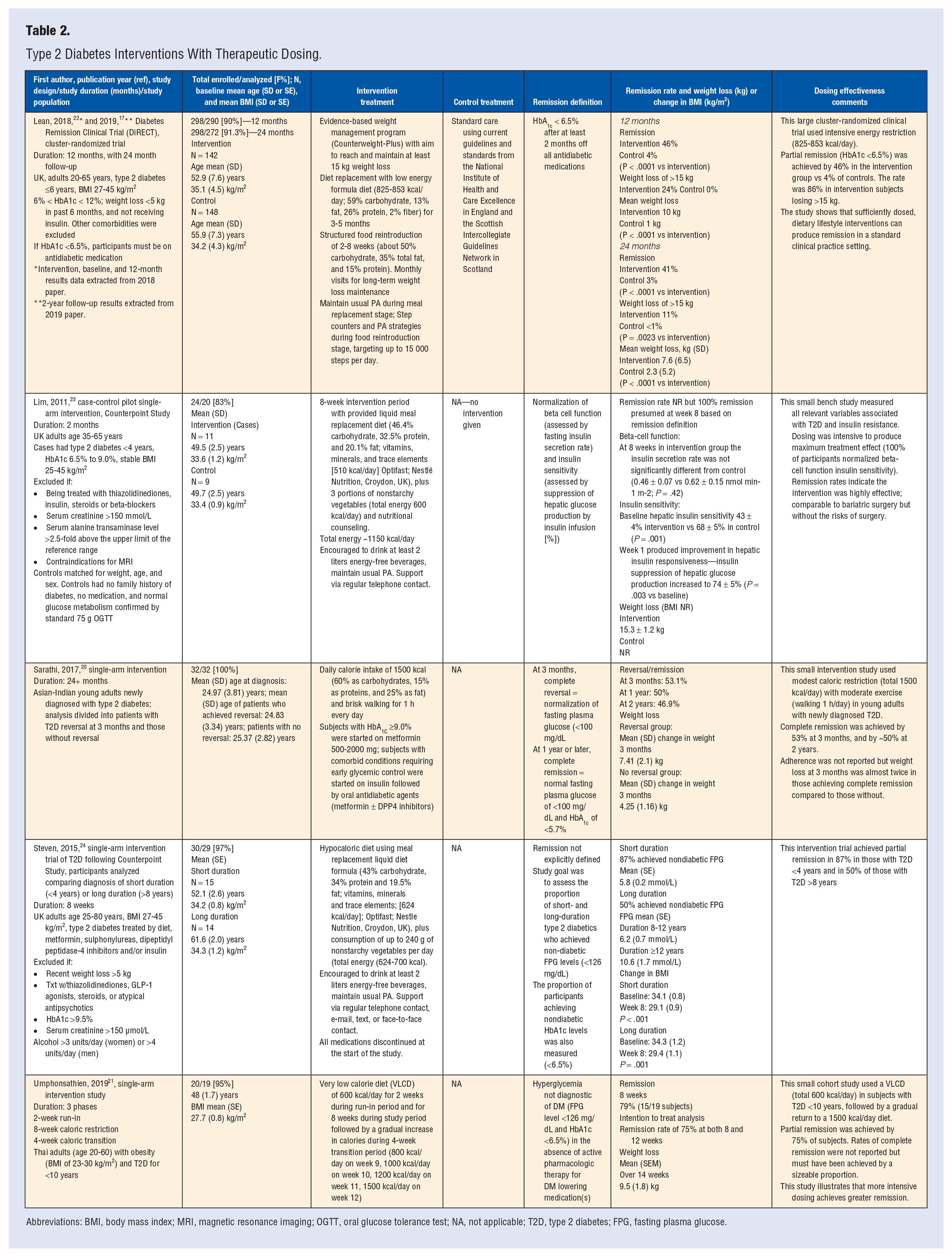
Abbreviations: BMI, body mass index; MRI, magnetic resonance imaging; OGTT, oral glucose tolerance test; NA, not applicable; T2D, type 2 diabetes; FPG, fasting plasma glucose.
The 4 subtherapeutic studies (Table 3) ranged in sample size from 8 25 to 5145 26 subjects. Two studies were randomized,26,27 and 2 studies used a single-arm pre-post25,28 design. The rates for partial remission ranged from 13% 27 to 43%, 25 with a weighted mean of 6.9%. The rates for complete remission ranged from 0% to 10%. 28 Weight loss ranged from 6 27 to 25 29 lbs, with a weighted mean of 7.7 lbs. The caloric content of the intervention diets ranged from 800 25 to 1500 26 kcal in 2 studies, and stated a goal of reduction in total caloric content by 500 kcal 28 to 625 kcal 27 in 2 other studies.
Type 2 Diabetes Interventions With Subtherapeutic Dosing.
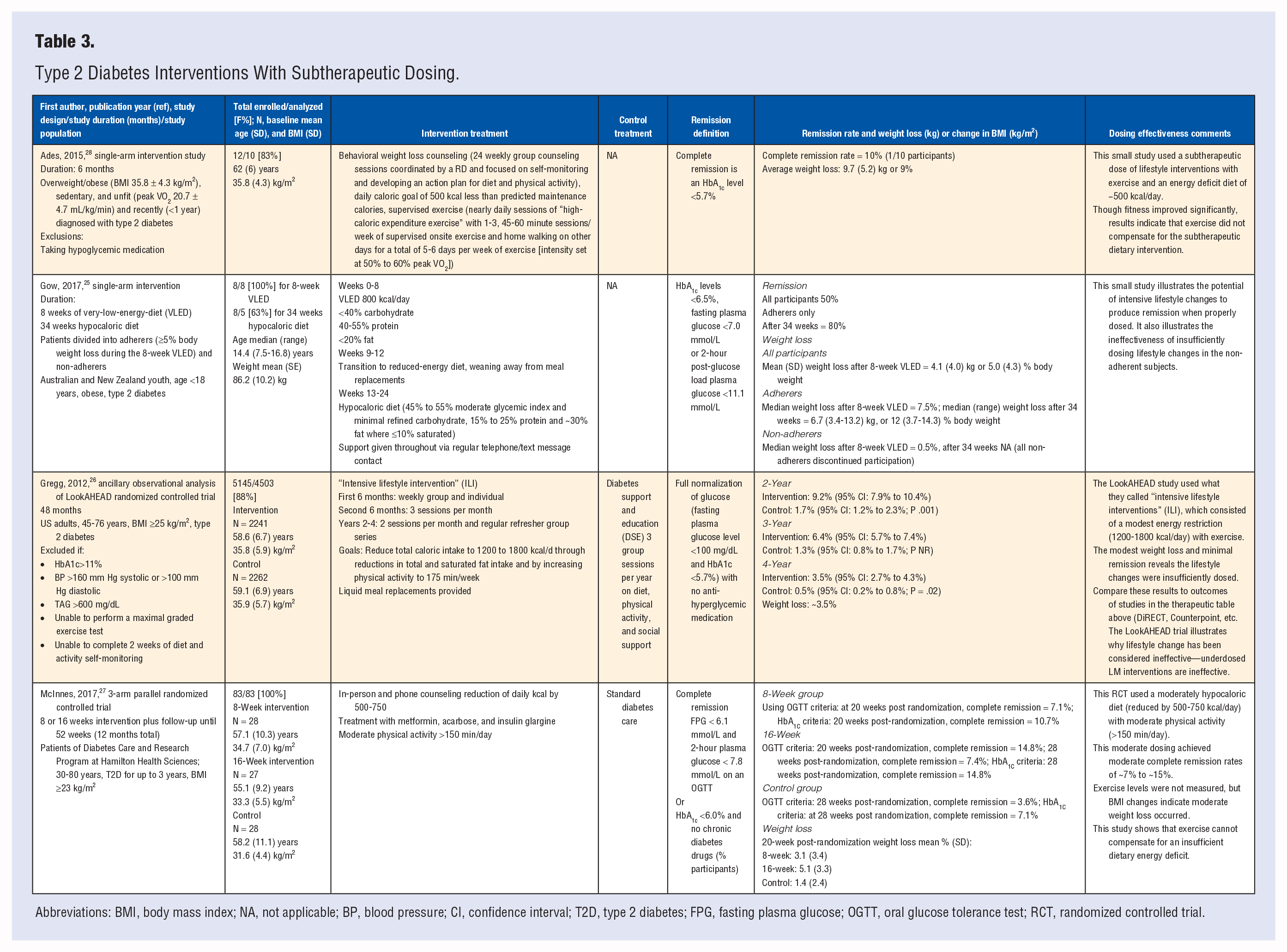
Abbreviations: BMI, body mass index; NA, not applicable; BP, blood pressure; CI, confidence interval; T2D, type 2 diabetes; FPG, fasting plasma glucose; OGTT, oral glucose tolerance test; RCT, randomized controlled trial.
Commentary on the dosing and effectiveness are included in the rightmost columns of Tables 2 and 3.
Discussion
The literature review supports the feasibility of T2D remission in diabetic patient populations using lifestyle as a primary means of treatment. Physical activity appeared to have a minor role compared to diet. While most studies increased physical activity,20,22,26-28 some studies achieved remission without an increased exercise protocol.20,24 Nevertheless, physical activity has been shown to improve insulin resistance 30 and beta-cell function. 31
Mechanisms of T2D and Remission
The underlying causes of T2D have long been identified as excess weight,32,33 and insulin resistance. 34 There has been a lack of consensus on whether insulin resistance may be caused by excess weight, or whether excess weight is caused by insulin resistance.35,36 It has been suggested that there is bidirectional relationship. 37 However, one thing these 2 “causes” have in common is that they most often result from excessive caloric intake. This causes rapid insulin resistance due to increased oxidative stress, in as little as 1 to 2 days. 35 This occurs before weight has been gained. Ongoing intake of excess, poorly chosen calories maintains the insulin resistance.
Human physiology preserves and conserves calories, even when consumed to excess as illustrated by morbid obesity. While adipose tissue stores much of the excess calories, as the excess accumulates it is stored in other tissues, notably in the liver, pancreas, and muscle. 38 In these non–adipose tissues caloric excess leads to lipotoxicity, meaning the excess fat (intracellular lipids) stored in these cells inhibits their proper functioning. 39 This disrupts normal glucose and insulin metabolism. Myocellular lipotoxicity inhibits glucose uptake by affecting the Glut-4 transporter. 40 This produces peripheral insulin resistance, requiring greater amounts of insulin to “push” glucose into muscle tissues. This resistance is reduced with exercise as muscle tissues consume energy and become more responsive to insulin. 30
Pancreatic lipotoxicity, on the other hand, inhibits beta-cell insulin production, reducing the amount of insulin available to overcome the insulin resistance in muscle and other energy-consuming tissues. 41 Dramatic caloric restriction has been shown to remove triacylglycerols from the pancreas and liver in a matter of days, restoring beta-cell insulin production. 23 This occurs before significant weight loss happens, indicating it is not the excess weight but excess calories 35 driving insulin resistance and T2D. Over time, this also drives weight gain.
Thus, as caloric intake exceeds energy needs and overwhelms the storage capacity of adipose tissue, energy is stored in non–adipose tissues, causing lipotoxicity. This process produces weight gain as the unused energy is conserved and stored. The lipotoxicity disrupts cellular glucose and insulin metabolism, resulting in insulin resistance and beta-cell dysfunction. As this metabolic state continues and worsens, beta-cell production declines until hyperglycemia occurs. Secretagogues (which increase insulin secretion) and exogenous insulin increase insulin levels to “push” glucose into cells, sustaining the toxic metabolic state and accelerating the dysfunction. Metformin reduces peripheral insulin resistance, 42 but this does not remove the underlying toxic etiology of excess caloric intake. Until the cause is removed, metabolic dysfunction must continue. The dysfunction is in reality a normal physiologic response to the toxic state induced by excess calories.
Some of the newer diabetes medications are designed to cause the kidneys to spill glucose into the urine as a way to lower serum glucose. 43 Normally, the kidneys strictly conserve glucose as the body’s main energy source. When glucose spills into the urine it fuels bacterial growth, which can result in a urinary tract infection, requiring treatment to avoid systemic infection and sepsis. It is therefore common for individuals using these medications to be prescribed antibiotics on an ongoing basis to control the urinary tract infections.8,43
In sum, these treatment complications are the direct result of an attempt to eliminate the excess glucose from overconsumption of calories.
Importance of Dosing
The importance of appropriate dosing in the context of lifestyle modification can hardly be overstated. Dosing is of prime therapeutic importance in a pharmaceutical context; with LM it is no different. Simple aspirin provides an excellent illustration—treatment for headache requires aspirin dosing of 325 to 650 mg once or twice a day. This outcome cannot be achieved using a dose of 81 mg aspirin daily, the dosing typically prescribed as an antithrombotic agent.
The same dosing principles apply to LM; the lifestyle modifications required to produce T2D remission are significantly more intensive than those sufficient to prevent T2D. Clinicians whose patients achieve remission do so by making sufficiently dosed lifestyle changes (eg, diet, exercise, etc). Remission has not been reported with inadequately dosed lifestyle changes, such as eating more salad, or simply reducing meat consumption. Research now shows that remission is achievable for a majority of short duration (<8 years) T2D patients, and many with longer duration, with sufficiently intense LM interventions.22,23 Most failures are due to inadequate dosing of LM, and not to any inherent deficiency of LM as treatment. Patients find intensive LM treatment acceptable and desirable when treatment produces remission. 1 A personal demonstration of remission can reset the lifestyle goals patients set for themselves.
At the same time, degree of dosing is also influenced by patient willingness to change 44 and the inherent challenges in making healthy changes 45 without adequate support and guidance. Adherence to conventional diet and lifestyle guidelines for diabetes management has been identified as problematic for T2D patients specifically. 46 Key barriers to adherence include lack of communication and support from health care providers, as well as inadequate education. 47 We argue that this underscores the importance of working with specialists trained in LM 48 to provide the education, support, and modeling necessary to successfully and realistically achieve a sufficiently strong intervention dose and accomplish the goal of remission.
Therapeutic Dosing
Research shows that sufficiently intensive dosing of lifestyle interventions will induce remission in half or more of all subjects with T2D. Studies also reveal that not all lifestyle interventions produce remission, and that remission rates are lower for longer-duration T2D. Adherence and other individual factors also affect remission rates. The highest remission rates are similar to those with bariatric surgery.22-24,49
The case-control study by Lim et al from 2011 provides a strong example of appropriate, therapeutic dosing. 23 This study achieved immediate remission (1-4 weeks) with a very intensive LM intervention, prior to significant weight loss. The Counterbalance study by Steven et al from 2015 is another example of therapeutic dosing. 24 These researchers chose to use liquid meal replacements with good effect. Rehackova et al 50 conducted semistructured interviews of participants before and after completion of the 8-week Counterbalance study and found high acceptability for the VLED (very low energy diet) meal replacement. Steven et al published self-reported remission in subjects inspired by the Lim paper to make intensive lifestyle changes on their own, 1 indicating the presence not only of high acceptance but also high motivation for diets that induce remission. Few people are willing to make dramatic changes in behavior unless they can perceive meaningful benefit.
Not all attempts at T2D remission are equally successful. Not all lifestyle interventions are of equal strength or intensity. The LookAHEAD study compared standard DSE (sDSE) to what researchers called an “intensive lifestyle intervention” (ILI), achieving a remission rate ~4 to 6 times higher with ILI than with the sDSE. 26 This sounds excellent until we learn that the sDSE rate was ~2% while the ILI rate was ~6% to 12%. When compared to adequately dosed LM such as that used in the DiRECT and Counterpoint trials, which achieved remission rates of 46% to 86%, and the 6% to 12% is subtherapeutic.
Similarly, in the pilot study by Ades et al, 28 partial remission was achieved in 70% of completers (7 of 10) but complete remission in only 10% (1 of 10). The subjects had recently been diagnosed with T2D (mean 93 days) and would be expected to achieve remission more readily than those with long-duration disease. The dosing was insufficient to produce the remission that more intensive dosing achieves. The diet was only 500 kcal less than required for weight maintenance. This dosing is more suited for prevention than remission. While more effective than the LookAHEAD trial, compared the therapeutic dosing rates of 46% to 86% in the DiRECT and Counterpoint trials, it is subtherapeutic.
Plant-Based Diets Versus Meal Replacements
Other T2D and weight loss researchers have used unprocessed plant-based diets with good effects,51-53 indicating that liquid meal replacements may be unnecessary to achieve the very low energy diets needed for remission. A study of 5 diets by Turner-McGrievy et al from 2015 found a totally plant-based diet (vegan) was more effective in reducing energy intake than other diets tested, leading to significant weight loss. 54 The fasting mimicking diet studied by Cheng et al found a periodic fast restored beta-cell function and normoglycemia in T1D and T2D mouse models, with similar in vitro effects in human pancreatic tissue. 55 Intermittent fasting has been found effective in reducing hyperglycemia and stimulating beta-cell neogenesis.18,56
Very low energy diets and fasting, whether using liquid meal replacements, fasting mimicking diet, or simply intermittent fasting, can be quite effective. This is consistent with other evidence indicating that reduction or avoidance of unhealthy foods may yield a greater benefit than inclusion of healthier foods. Schwingshackl et al found consumption of risk-decreasing foods (whole grains, dairy, fruits, and vegetables) resulted in a 42% risk reduction of T2D, and consumption of risk-increasing foods (red and processed meats, sugar-sweetened beverages, and eggs) was associated with a threefold increased risk, compared to nonconsumption. 57
Current Best Evidence and ACLM’s Position Statement on T2D
The numerous studies in Table 2 show that sufficiently intensive lifestyle interventions successfully produce remission of T2D and do so with a success rate comparable to other modalities, including bariatric surgery. This fact has not been recognized and has not appropriately informed or shaped the present approach to lifestyle interventions in the treatment and management of T2D. It is inconsistent with the evidence to favor higher risk, invasive treatments over lifestyle change. It is ACLM’s position that this perspective on lifestyle interventions needs to change and that LM should be a primary component of every patient’s treatment plan for T2D.
The studies in Table 3 demonstrate that inadequately dosed lifestyle interventions do not successfully produce remission of T2D. The present diminution of lifestyle interventions in the treatment and management of T2D is undoubtedly due in part to the failure to distinguish between therapeutically dosed interventions and subtherapeutically dosed interventions. For example, the LookAHEAD study concluded from their subtherapeutic “intensive lifestyle interventions” that lifestyle change was not sufficiently effective to be a viable alternative to bariatric surgery. 26 This is actually an incorrect conclusion because it was based on what appears to be a subtherapeutic intervention. Other investigators who have produced remission have arrived at a different conclusion (Table 2).
Lifestyle recommendations have not historically focused on the remission of T2D, rather focusing on glycemic improvements and disease management. Several organizations, including the American Association of Clinical Endocrinologists and the American College of Endocrinology, 58 acknowledge the benefit of a plant-based diet as the optimal nutrition plan for the management of T2D. 58 In fact, the International Diabetes Federation 3 plainly states that lifestyle behavior, including poor diet, is the most influential factor in the development of T2D.
In 2019, the ABCD and the PCDS published a position statement 12 on T2D remission stating that effective methods of diabetes remission included either (1) bariatric surgery or (2) a short-term dramatic caloric reduction with total dietary replacement followed by a stepped food re-introduction. Overall, there is movement in the professional medical community toward emphasizing lifestyle modifications for T2D management, though an emphasis on remission as a goal is still emerging.
The Historical Context
Although the concept of using diet to achieve remission seems new in the context of conventional treatment, it is not a new idea. Over the course of the 20th century, diet recommendations for diabetes prevention and management varied widely, 59 alternately supporting either low-carbohydrate/high-protein60,61 or high-carbohydrate62-65 eating patterns. However, instead of remission as a goal, research typically focused on glycemic improvement and disease management.8,66,67
During that time, the use of medication emerged as the primary means of treatment. 8 The discovery of insulin in 1921 was hailed as a breakthrough therapy for severe diabetes, 68 later known as type 1 diabetes. While this discovery of insulin was life-saving for patients with type 1 diabetes, it also became the standard of care for patients with type 2 diabetes. This diverted attention from the approach of seeking to reverse the condition by addressing the underlying causes. Unfortunately, the medication-oriented approach has progressed to the point that some practitioners now advocate for starting insulin immediately after diagnosis. 69 This approach may prolong beta-cell function but does not lead to remission.
Thus, a gap developed in lifestyle research and treatment, causing a lack of results around clinically significant, enduring T2D remission. Because of these failures, more dramatic interventions were sought. Bariatric surgery, despite the expense and risk of complications, started as a treatment for weight loss circa 1987 70 and became commonplace as a treatment for T2D by 2010. 71 While the 30-day mortality is between 0.1% and 1.1% for bariatric surgery, the risk of developing acute and long-term complications is high (17%). 72 The list of possible complications from bariatric surgery is wide-ranging including anemia, reflux esophagitis, marginal ulcer, nutritional deficiencies, gastrointestinal bleeding, intraabdominal abscess, intraperitoneal bleeding or minor bleeding, urinary tract infection, and wound infection, while the rate of reoperation after surgery is 7%.
A pivotal finding from bariatric surgery for morbid obesity was that it could produce T2D remission, something that had previously been considered impossible, or at least impractical. 8 While the search continues for a complete understanding of how bariatric surgery rapidly reverses T2D, some very informative research reveals that these results may not depend on the surgery itself.49,73 Lingvay et al evaluated the contribution of caloric restriction versus surgically induced changes in glucose homeostasis in the immediate postoperative period among N = 10 patients in a prospective, 2-period study in which patients served as their own controls. 49 The study periods lasted for 10 days of inpatient stay, and periods were separated by a minimum of 6 weeks of a wash-out period. While no significant improvements were observed in the days immediately following the surgery, the presurgery diet intervention period produced a significant improvement in fasting glucose, maximum poststimulation glucose, and glucose area under the curve (AUC) during a mixed-meal challenge test. 49 These results support reduced calorie ingestion as the causal mechanism initiating marked improvement in glycemic control observed after Roux-en-Y gastric bypass surgery. Studies like the Counterpoint 23 and DiRECT 22 trials reveal that sufficiently intense caloric restriction can indeed induce remission in a majority of T2D cases in a matter of weeks.
Glycemic Index and Blood Glucose
One view of dietary control of blood sugar revolves around glycemic index; 74 thus, the topic requires clarification in the context of diabetes remission versus management. Glycemic index (GI) calibrates the postprandial effect a food or meal has on serum glucose levels (SG) compared to ingestion of 70 g of glucose. 75 High GI values indicate the food produces a relatively greater rise in SG, whereas low glycemic index values indicate the food produces a relatively smaller rise in SG. In general, simple sugars and highly processed carbohydrate foods have relatively higher GI values, while complex and unprocessed carbohydrates and non-carbohydrate foods (protein, fat) have relatively lower GI values.
Not all low GI foods are equivalent in their effects on metabolism, insulin resistance, and T2D, and remission is not directly related to GI. 76 Remission depends on reversing insulin resistance and restoring insulin sensitivity. 23 Thus, Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) is a better measure of the effect a food has on insulin resistance and T2D remission than GI. 77
Remission requires an energy deficit to remove liver and pancreatic triacylglycerols and stop their lipotoxic effects. 23 Thus, consuming low GI foods alone cannot produce remission without an energy deficit. Some low GI dietary patterns that help maintain a low SG do not reverse insulin resistance and do not produce T2D remission. One such pattern is the so-called “ketogenic diet,” which emphasizes a low-carbohydrate, high-fat, and high-protein eating pattern. 78
A VLED produces ketosis, but does it with an energy deficit, whereas the ketogenic diet does it by forcing the body to metabolize fats instead of carbohydrates. The diet is not energy deficient and does not reverse liver and pancreatic triacylglycerols; thus, it helps control SG but not insulin resistance.78,79 Ketogenic diets only appear to eliminate T2D by consistently failing to challenge insulin receptivity rather than by improving or restoring it.
Low-Carbohydrate Diets
The present swing toward a high-protein, low-carbohydrate dietary pattern for improving hyperglycemia is problematic. As summarized by Uusitupa and Schwab in their editorial on diet and T2D,
80
. . . there are no real long-term intervention trials on high protein diets in people with diabetes.
81
. . . the safety of high protein diets has been documented for one year only.
81
. . . the quality or quantity of dietary protein does not seem to play any role in the pathogenesis of type 1 diabetes, whereas regarding type 2 diabetes, higher intakes of red meat and processed meat products have been consistently associated with the increased risk of type 2 diabetes
82
and other chronic non-communicable diseases.
83
This should arouse concern when considering any dietary guidelines.
Long-term prevention trials of T2D have applied diets emphasizing healthy dietary patterns with typical protein intake. 84 The primary goal of the prevention of T2D is permanent weight reduction that can be achieved by healthy dietary patterns and increased physical activity.
Whole Food, Plant-Based Diets
Importantly, control of SG can also be accomplished, without negative side effects from medication, by reducing consumption of excess calories. This can be done in 1 of 2 ways: either by reducing the volume of food (holding energy density constant) or by reducing the energy density (holding the volume of food constant). 85 In order of effect size, the 3 factors of decreasing fat, increasing fruits and vegetables, and increasing water content have been shown to decrease caloric content in ad libitum eating conditions. 86 Consumption of food with lower energy density has been found to produce satiety without feelings of hunger or deprivation, 85 and portion size, independent of greater energy density, has been shown to affect satiety. 87 The dietary pattern that, when followed, offers the greatest reduction in fat content and the greatest increase in fruits and vegetables and water content is a WFPB diet.88,89 In observational studies this dietary pattern largely overlaps with a vegan diet (eliminating all animal products) and is associated with lower BMI and prevalence of T2D.90,91 This is also the diet which the authors use with the greatest success and which the surveyed ACLM member practitioners report as being most important in achieving T2D remission outcomes, when the dietary change represents an appropriately intensive dose.
Summary
Adequate research exists demonstrating that T2D remission is achievable without pharmacological interventions or surgery. The best evidence indicates that a substantial energy deficit is necessary until an adequate reduction in intracellular hepatic and pancreatic fat content is achieved to induce insulin sensitivity and glucose control. The wide variability in results among the interventions discussed here highlight the importance of appropriate dosing in lifestyle interventions, just as with other types of medical treatment.
The dietary intervention used by the authors indicates that a therapeutically dosed WFPB is the preferred intervention. This is consistent with internal ACLM survey results (as-yet unpublished data) that reveal that a large proportion of ACLM members are using diet (predominantly a WFPB diet) as the primary modality in treating and managing T2D, and that the majority have seen improvements in T2D including reduced need for medications and improved glycemia. While these data do not quantitatively capture remission outcomes, it shows support for and need of evidence-based lifestyle interventions to treat and reverse T2D, the conclusion of this position statement. A WFPB diet has multiple benefits of minimizing harmful foods, emphasizing healthy foods, reducing energy density through lower levels of fat and higher amounts of fruits, vegetables, and water. It also facilitates weight loss without feelings of deprivation or hunger, and seems well-tolerated by patients. 92
Regardless of the exact dietary method used to restore beta-cell function and insulin sensitivity, we recommend that T2D remission by intensive lifestyle change be the primary modality for T2D care. Published research reveals that sufficiently intensive lifestyle interventions consistently produce partial or complete remission in a significant proportion of patients. We also recommend that existing pharmaceutical therapies not be abandoned prematurely, but continue to be utilized to achieve and maintain optimal blood sugar control, with the goal of reducing and eliminating these measures as soon as possible, as the recommended lifestyle diet and exercise modalities are applied by the patient and weight loss and restoration of normal endogenous insulin levels are restored. As more experts in the field support a nonpharmacologic approach to T2D treatment, 16 rigorous research to quantify the effects of a WFPB diet as compared to other dietary interventions such as liquid meal replacements or severe caloric restriction is needed.
Limitations
There are inherent limitations present in this review. The lifestyle intervention studies of T2D remission are few in number with wide variability of design as well as being generally limited to short-term outcomes. A majority of the therapeutic studies examined would not meet the definition of remission put forth by Buse et al, 11 though there is a lack of consensus on duration needed to assess remission among clinicians and researchers. 12 Most studies of remission have examined T2D of short duration, and success rates are higher the earlier treatment is initiated after diagnosis. This is not a systematic review; the list of studies (Tables 1-3) is not exhaustive but rather chosen to be representative of the position statement put forth.
Conclusion
As incidence and prevalence of T2D continue to rise, the current best evidence from multiple intervention studies supports that remission achieved with intensive lifestyle modifications should become the preferred treatment and standard of care. To achieve remission, appropriate, therapeutic dosing of lifestyle modifications is necessary, including a hypocaloric diet. Future research should focus on high-quality intervention trials to further quantify the potential of different lifestyle modifications, including consumption of a low-fat, WFPB diet to achieve remission of T2D in comparison to standard pharmacologic treatment and surgical interventions.
Footnotes
Acknowledgements
The authors would like to thank research assistant Kelly Cara for her help in performing data extraction, Jean Tips and Samantha Gallion for editing the manuscript, Rich Rosenfeld as ACLM Expert Panel reviewer, members of the ACLM Type 2 Diabetes Task Force, and the ACLM members who responded to the Lifestyle Medicine Practice Survey.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American College of Lifestyle Medicine.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
