Abstract
Background. Nutrition and probiotics supplementation are considered as nondrug strategies for the control of coronary artery disease (CAD). The aim of the present study was to investigate the interactive effects of probiotic supplementation and weight loss program on anthropometric and metabolic syndrome (MetS) indices in patients with CAD. Methods. A randomized, double-blind, placebo-controlled trial was performed in 44 overweight CAD patients. All the patients received weight loss program (5% to 10% of initial body weight) throughout the study course. The subjects were randomly assigned into intervention or placebo groups (n = 22, each) and received a probiotic or maltodextrin capsule/day, respectively, for 12 consecutive weeks. Anthropometric and MetS indices were assessed. Results. There was a significant decrease in total cholesterol (−30.7 ± 49.83 vs −5.9 ± 65 mmol/L, P = .043) and low-density lipoprotein cholesterol (−25.64 ± 51.7 vs −5.44 ± 70.1 mg/dL, P = .049) in the probiotic group compared to the placebo group. Other MetS feature indices and blood pressure did not differ significantly within or between groups. Conclusion. Probiotics supplementation improved total cholesterol and low-density lipoprotein cholesterol but had no effect on other lipid profile parameters and anthropometric indices. The weight loss diet plus probiotics supplementations resulted in more favorable decrease of cardiovascular risk factors compared to a weight loss program alone.
‘Weight loss and management of risk factors are the treatment of choice for CAD [coronary artery disease] and other cardiovascular diseases in patients with overweight or obesity.’
Coronary artery disease (CAD), also called coronary heart disease, is a common term for the buildup of plaque in the heart’s arteries that could lead to heart attack. It has been shown that the mortality and morbidity rates of CAD are higher in subjects who are overweight; also obesity is known as an independent risk factor underlying this disorder.1,2 Abdominal obesity may be associated with dyslipidemia, hypertension, diabetes, and elevated levels of inflammatory factors, all of which also increase the risk of CAD. 3 In addition, the association between metabolic syndrome (MetS) features and CAD has been indicated in previous studies. 4 MetS includes several cardiometabolic risk factors and is characterized by 4 chief components including impaired glucose tolerance, dyslipidemia hypertension, and abdominal obesity; all these factors increase the risk of CAD. 5
Subject with MetS are over 5.5 times more likely to have CADs. 6 Therefore, it is important to identify effective strategies to control these cardiometabolic risk factors. Weight loss and management of risk factors are the treatment of choice for CAD and other cardiovascular diseases in patients with overweight or obesity. 7 Although many guidelines propose following a weight loss program to achieve 5% to 10% weight reduction, considerable weight management is not usually achieved. 8 On the other hand, the majority of physicians and dietitians currently emphasize the importance of calorie restriction; however, there is usually poor compliance to these dietary plans. 9 Therefore, albeit the popular demand for effective weight loss methods, it is not surprising that the use of alternative and complementary weight loss strategies has increased. 10
Among the known therapeutic options for CAD treatment, there are wide complementary approaches such as supplements that can be used along with medical nutrition therapy to control this disorder. These methods are becoming popular nowadays and probiotics are one of them. Studies on probiotics have revealed beneficial and promising results in prevention and management of CAD and MetS and weight management. 11 Probiotics are defined as living microorganisms, which when administrated in sufficient amounts, beneficially influence the health of the host by improving the composition of gut microbiota. 12 There is also lot of evidence that obesity is linked to chronic inflammation in the cardiovascular system. By improving gut health, probiotics may reduce systemic inflammation and protect against CAD. 13 In addition to enhancement of intestinal gut health, probiotics are able to impede weight gain and improve cardiovascular risk factors.14,15 Effects of probiotics administration on lowering lipids and cardiovascular disease risk factors have not been conclusive in other studies.16,17 To the best of our knowledge, the effects of Lactobacillus rhamnosus GG (LGG) in combination with a weight loss program on lipid profile and other features of MetS in CAD subjects have not been investigated before.
Therefore, considering the anti-inflammatory and anti-obesity effects of probiotics alone and in combination with weight loss diet, and due to the lack of clinical trials on the interactive therapeutic roles of LGG and weight loss program in patients with CAD, this study was designed to investigate the effects of a 12-week intervention with LGG and weight loss diet on anthropometric indices, lipid profile, and blood pressure in patients with CAD.
Materials and Methods
Study Design
A 2-arm parallel randomized controlled trial was conducted from July 2018 to October 2018 at Shahid Madani Heart Center, affiliated to Tabriz University of Medical Sciences (TBZMED), Tabriz, Iran. The trial was registered at the Iranian Registry of Clinical Trials (IRCT20121028011288N15), after approval of the study protocol by the Medical Ethics Committee of TBZMED (IR.TBZMED.REC.1397.184). 18
Informed consent was taken from all the study subjects. All the patients with CAD admitted to the participating hospitals were considered for possible enrollment into the study, and they were screened by an expert cardiologist for eligibility. The eligibility criteria for this study were (1) agreeing to participate in the study; patients who provided written informed consent were included; (2) being diagnosed as new myocardial infarction (MI) after successful percutaneous coronary intervention (PCI); (3) men and women aged between 30 and 70 years; (4) an echocardiographic left ventricular ejection fraction (EF) ≥30%; and (5) being at the body mass index (BMI) range of 25 to 35 kg/m2 and having no infections and no kidney and liver diseases. Participants were excluded if (1) they were in urgent need for revascularization procedure; (2) were diagnosed with heart failure (function class III and IV), heart valve disease, uncontrolled diabetes, or any other chronic disease including significant renal dysfunction (serum creatinine above 1.5 mg/dL); (3) regularly taking any immunosuppressive drugs; history of supplementation with pre-/pro-/symbiotic or antioxidants during or 2 months prior to the study; and (4) taking weight loss drugs or recent weight reduction. All the participants were requested to report any change in their medications. The subjects in both groups received a weight loss diet considering their dietary habits. They were advised not to modify their physical activity habits. To ensure that these habits were not changed during the study, the participants were trained to record physical activity diaries, which were checked weekly during the visits, by a dietitian.
Randomization and Blinding
Each eligible participant was randomly assigned into intervention or placebo group, according to 1:1 equal proportion rule. The sequence of random allocation was generated using a software of random sequence generator. The criteria for matching were sex and BMI. The random code was kept by an independent investigator not involved in the assessment of the participants or in the data collection and analysis. Another person who was not involved in the intervention enrolled the participants. All participants and investigators were blinded to treatment allocation.
Interventions
Participants in the intervention group received probiotic (containing the 1.6 × 109 colony forming unit (CFU) freeze-dried LGG) or placebo (maltodextrin, 150 mg/day) for 12 consecutive weeks. The appearance and labeling of the supplements were identical (TakGen Co, Tehran, Iran). The subjects were educated to use LGG capsules or placebo capsules with their lunch, daily. All the participants received a moderate calorie restricted dietary plan during the 12-week intervention period. In this study, the program was designed to enable weight loss of 7% to 10% of weight, at a rate of 0.5 to 1 kg/week throughout the intervention. The weight loss program was calculated to introduce a 500- to 1000-kcal energy-deficit based on estimated energy requirements at the baseline of the intervention.
Assessment of Anthropometric and Biochemical Variables
Body weight was measured using a scale (Seca, Hamburg, Germany) without shoes, with 0.5 kg accuracy. A tape with 0.5 cm accuracy was used to measure height. BMI was calculated by dividing weight (kg) by height 2 (m). Waist circumference (WC) was measured using a nonstretchable tape measure, at the midpoint between the end of the rib cage (10th rib) and the iliac crest. 19 All the measurements were performed by trained dietitians. Body fat percentage and body fat mass and fat free mass (FFM) were measured by the bioelectrical impedance method using a scale for body composition (Takara BC-418, Japan).
After 10 to 12 hours of fasting, 10 mL blood was taken from all participants to evaluate biochemical variables. Lipid profile including total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and triglyceride (TG) concentration were determined by enzymatic kits (Pars Azmun, Iran). Friedewald formula was used to calculate low-density lipoprotein cholesterol (LDL-C) values. Serum fasting blood sugar (FBS) level was measured using the glucose oxidase method with a commercial kit (Pars Azmun, Iran).
Morning systolic and diastolic blood pressure (SBP and DBP, respectively) were recorded in seated subjects after a 5-minute rest using an automatic oscillometric device (Omron Healthcare Co, Ltd) before and after intervention.
According to ATP III criteria, 20 MetS was defined as the presence of 3 or more of the following components: (1) WC >102 cm in men and >88 cm in women; (2) serum TG >150 mg/dL; (3) serum HDL-C <40 mg/dL in men and <50 mg/dL in women; (4) FBS level >110 mg/dL; and (5) elevated blood pressure (130/85 mm Hg).
Food intake was assessed using 3-day food record method at baseline, and 12 weeks later. Nutritionist IV software (The Hearst Corp, San Bruno, CA) was used for performing nutrient calculations for the 3-day dietary records.
Physical activity level of the participants was determined using the International Physical Activity Questionnaire (IPAQ).
Statistical Analysis
Statistical analyses were performed using SPSS (version 19; SPSS, Inc, Chicago, IL) software. Normal distribution was examined with the Kolmogorov-Smirnov test. The results are expressed as mean for normally distributed quantitative data and frequency (percentage) for qualitative data. The χ2 test and the independent samples t test were used to compare the 2 groups for background characteristics and baseline measures for qualitative (nominal) and normally distributed quantitative data, respectively. Analysis of covariance (ANCOVA) was used to compare the 2 groups for the measures at the end of the study after adjusting for the baseline measures and covariates. Comparisons between baseline data and final results within each group was made by paired samples t test and Wilcoxon signed-rank test for normally and nonnormally distributed data, respectively. P values of <.05 were considered statistically significant.
Results
Baseline Characteristics of Participants
After screening for inclusion criteria, a total of 44 subjects were recruited. The flow chart of the study is provided in Figure 1.
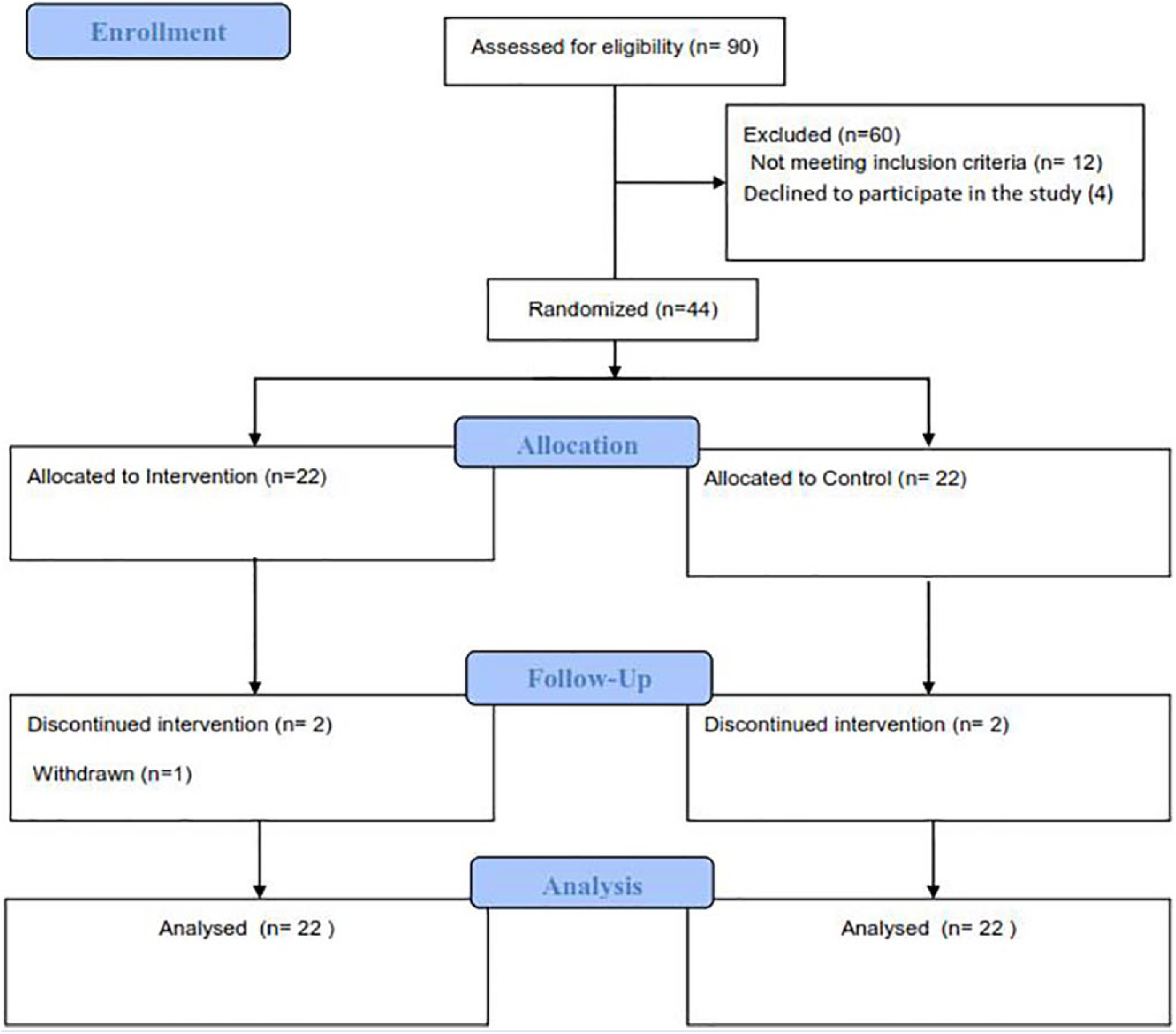
Flowchart of study.
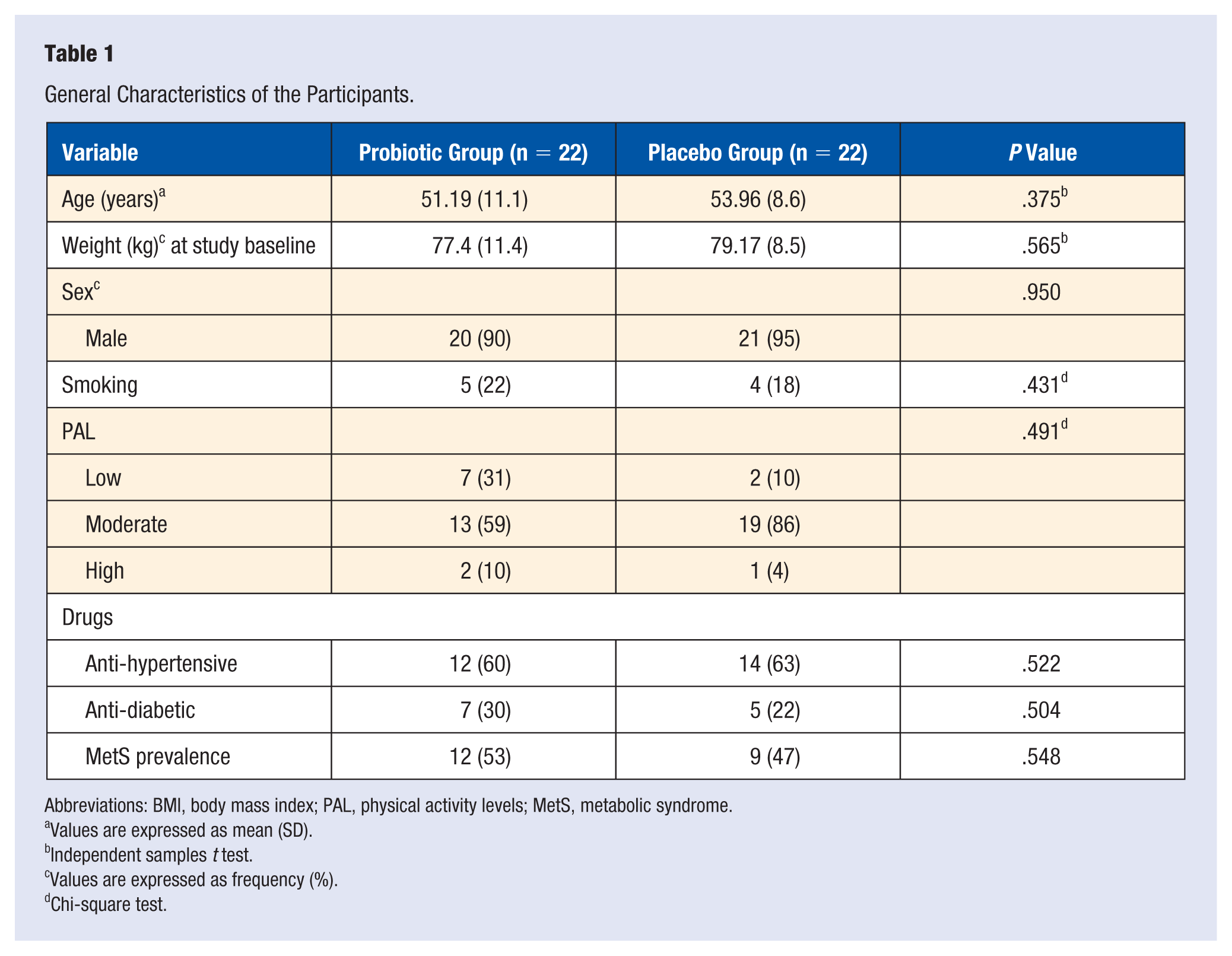
Subjects did not report any adverse side effects throughout the intervention of the study, related to probiotics or placebo consumption. Mean BMI was 27.37 kg/m2 (range 25-32). There were 6 females and 38 males. Of the patients, 90% and 95% were male in intervention and placebo groups, respectively. The baseline characteristics of the patients are presented in Table 1. The mean age of participants was 51.19 (11.1) and 53.96 (8.6) years in the probiotic and placebo groups, respectively, with no statistically significant difference between them (P = .375). Diabetes was present in 30% and 22% of the patients in the probiotic and placebo groups, respectively (P = .504). Despite this, greater decrease of FBS level was observed in the probiotics group than in the placebo group (−17.90 mg/dL vs −8 mg/dL, P = .843), after adjusting for baseline differences.
General Characteristics of the Participants.
Abbreviations: BMI, body mass index; PAL, physical activity levels; MetS, metabolic syndrome.
Values are expressed as mean (SD).
Independent samples t test.
Values are expressed as frequency (%).
Chi-square test.
No significant difference was seen in other baseline parameters including weight, sex, drug history, smoking, and physical activity level between the 2 groups (P > .05). No significant differences were found between the 3 group in term of MetS prevalence (53%:47%, P = .548).
Dietary Intakes
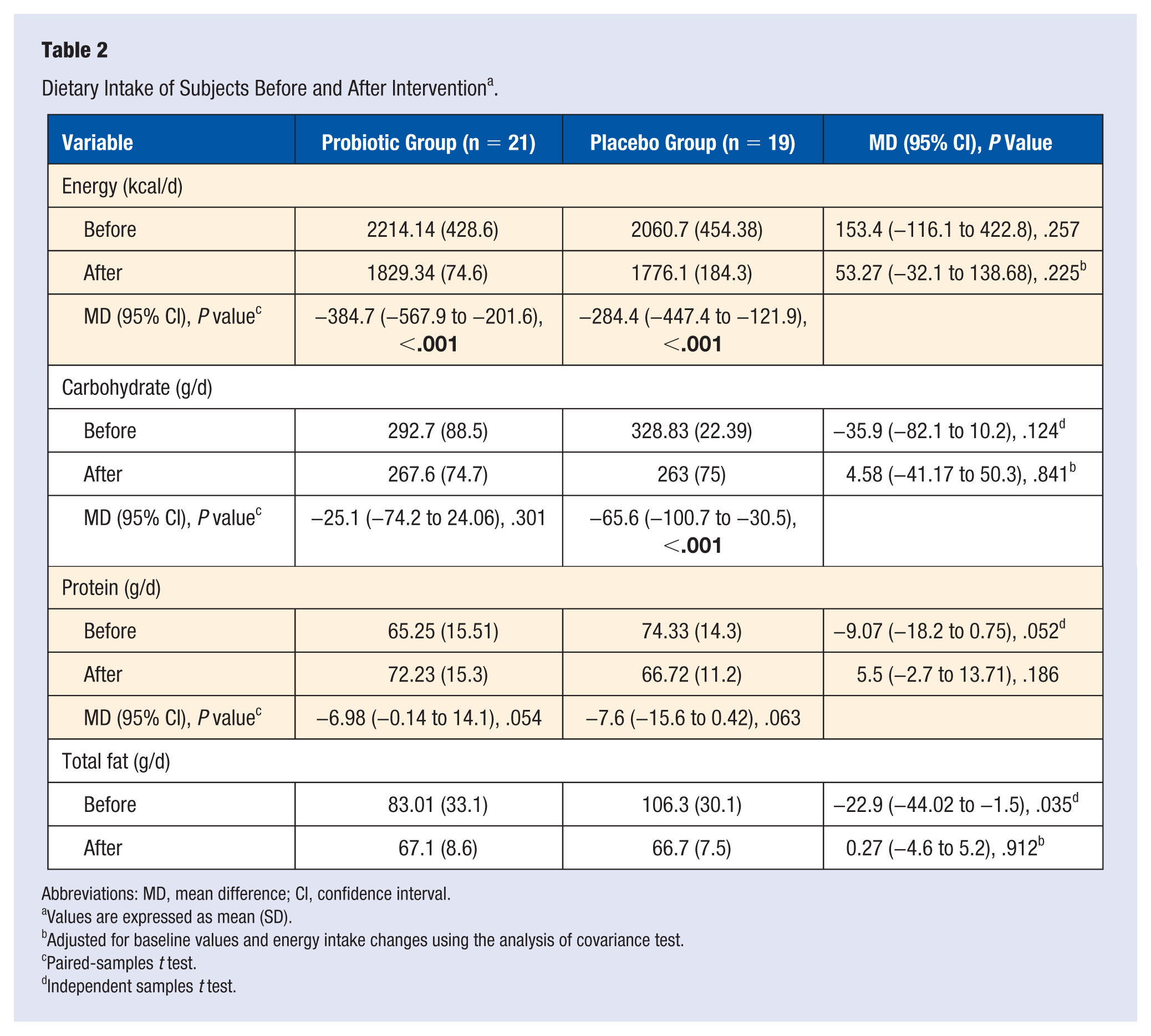
At baseline, no significant differences were observed in dietary intake of the 2 groups, based on the 3-day food record (P > .05). After 12 weeks of weight loss diet, energy intake decreased significantly in the both groups, whereas no significant differences in percentage of macronutrient intake and types of dietary fatty acid intake were observed between the groups; only the percentage of fat increased insignificantly in the probiotics group. At the end of the intervention, no significant differences were observed in dietary intake between the 2 groups. Energy and total fat intakes significantly decreased by the end of intervention, compared to the baseline (P > .05; Table 2).
Dietary Intake of Subjects Before and After Intervention a .
Abbreviations: MD, mean difference; CI, confidence interval.
Values are expressed as mean (SD).
Adjusted for baseline values and energy intake changes using the analysis of covariance test.
Paired-samples t test.
Independent samples t test.
Effect of Probiotic Supplementation on Anthropometric Indices and Body Composition
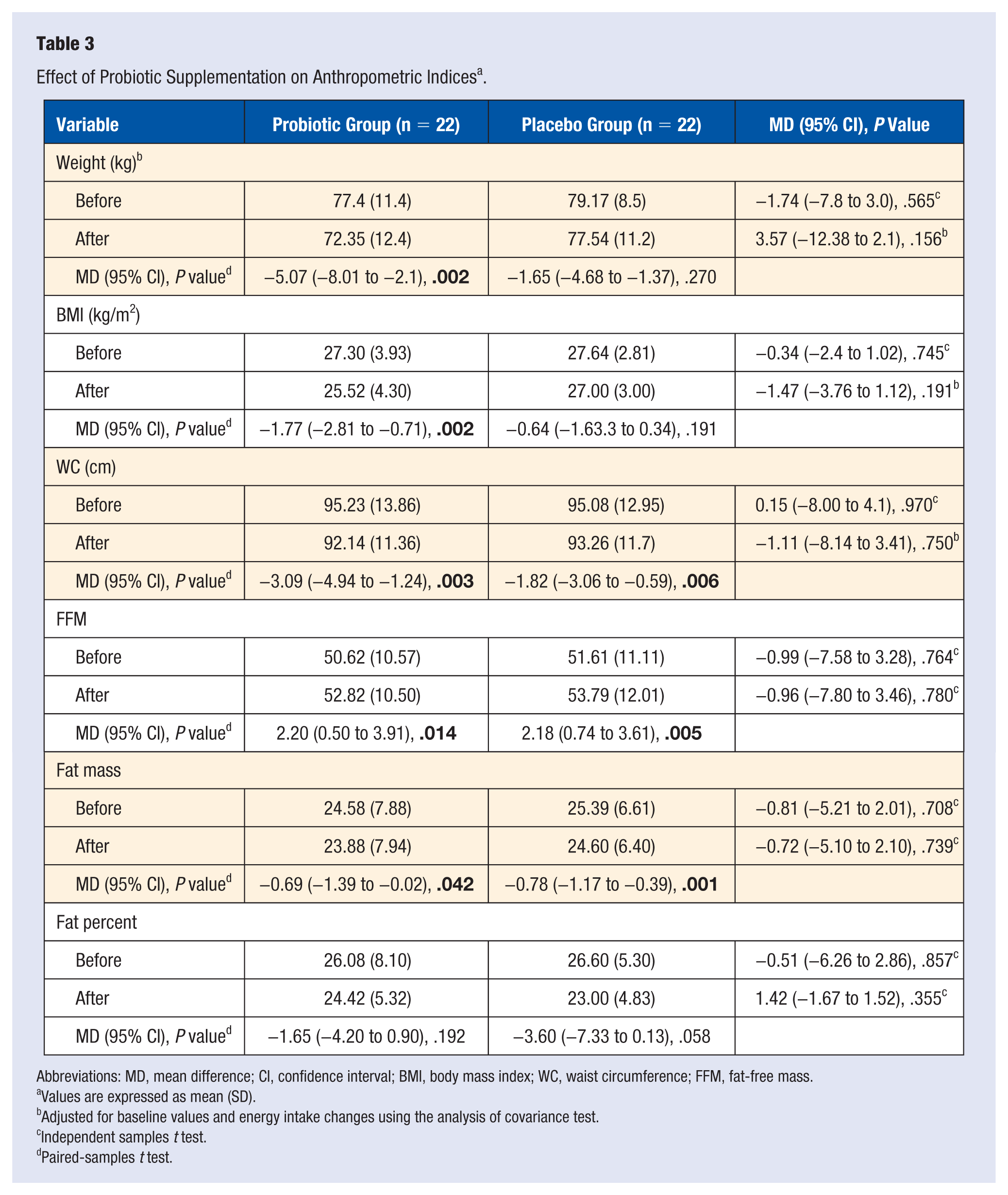
As shown in Table 3, weight, BMI, WC, FFM, and fat mass decreased significantly by the end of study in both probiotics and placebo groups, although no statistically significant difference was observed between the 2 groups at baseline and after intervention (P > .05). In the adjusted model, after controlling for confounding variables including energy and baseline values, there were no significant differences in anthropometric indices between the groups. Within-group analyses indicated that anthropometric indices significantly decreased in response to adherence to low-calorie diet in both groups; greater decrease was seen in the probiotic-treated group.
Effect of Probiotic Supplementation on Anthropometric Indices a .
Abbreviations: MD, mean difference; CI, confidence interval; BMI, body mass index; WC, waist circumference; FFM, fat-free mass.
Values are expressed as mean (SD).
Adjusted for baseline values and energy intake changes using the analysis of covariance test.
Independent samples t test.
Paired-samples t test.
Effect of Weigh Loss Program on Fasting Blood Sugar, Lipid Profile, and Blood Pressure
Patients were classified into 2 groups based on at least 5% of weight loss: adequate weight loss or unsuccessful weight loss. Although no significant difference was observed in terms of the study variable, the results showed that the MetS features were nonsignificantly lower in those with adequate weight loss than those who experienced unsuccessful weight loss (Table 4).
Effect of Weight Loss on FBS, Lipid Profile, and Blood Pressure a .
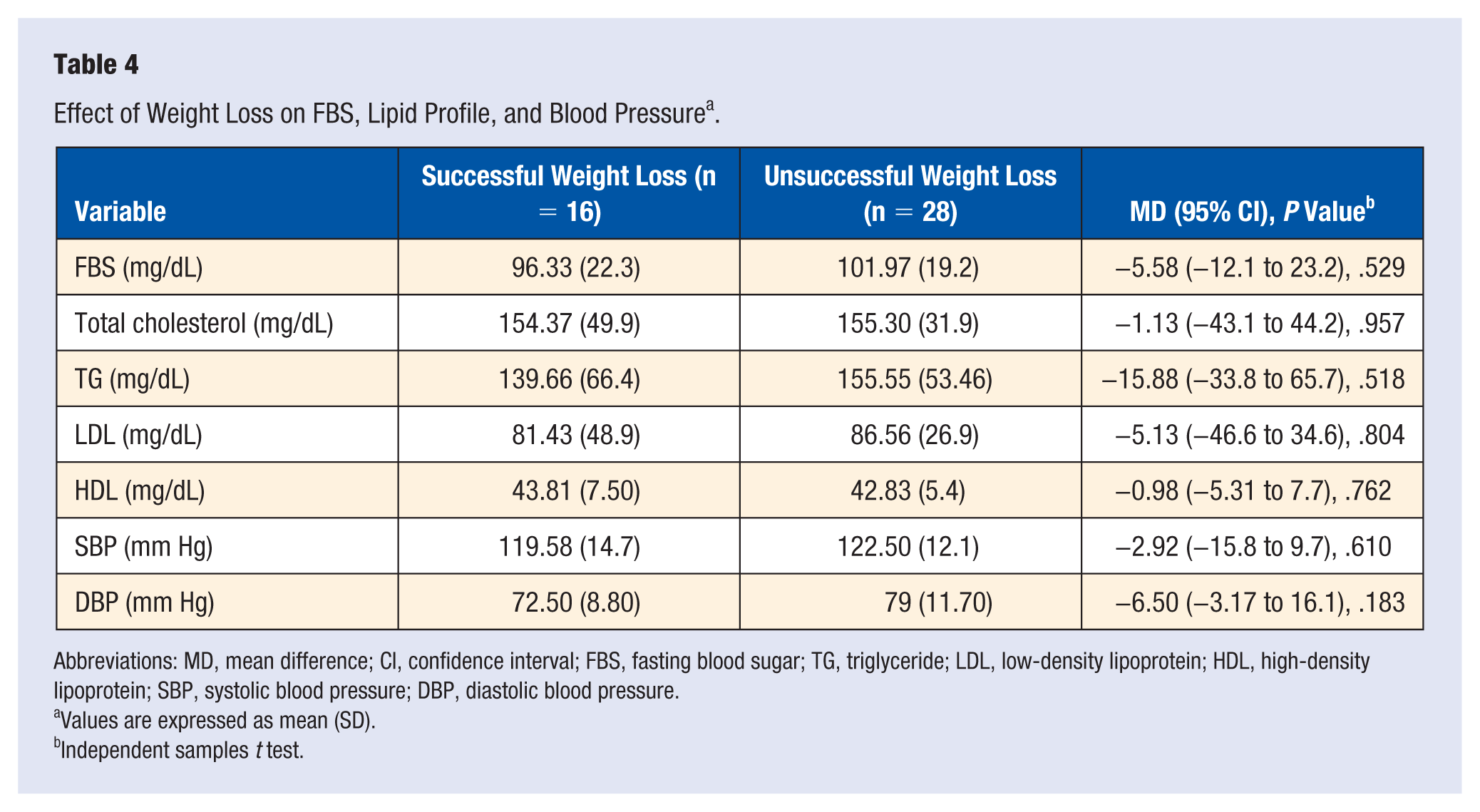
Abbreviations: MD, mean difference; CI, confidence interval; FBS, fasting blood sugar; TG, triglyceride; LDL, low-density lipoprotein; HDL, high-density lipoprotein; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Values are expressed as mean (SD).
Independent samples t test.
Fasting Blood Sugar, Lipid Profile, and Blood Pressure After Probiotic Supplementation
After 12 weeks of probiotics supplementation, changes in total cholesterol, LDL-C, and SBP were found to be significant within groups. No significant change in other metabolic features (FBS, TG, HDL-C, and DBP) were observed within groups; between-group differences were also not significant at baseline, and after supplementation when analyses adjusted for confounding factors. Overall, a significant decrease in serum TC (−30.7 ± 49.83 vs −5.9 ± 65 mmol/L, P = .043), LDL-C concentrations (−25.64 ± 51.7 vs −5.44 ± 70.1 mg/L, P = .049), and a nonsignificant decrease in SBP levels (−7.76 ± 13.88 vs −2.43+16.8 mm/Hg, P = .765) were detected following the supplementation with probiotic, compared to the placebo. Thus, taking probiotics resulted in a significant decrease in metabolic indices in comparison with the placebo (Table 5).
Effect of Probiotics Supplementation on FBS, Lipid Profile, and Blood Pressure a .
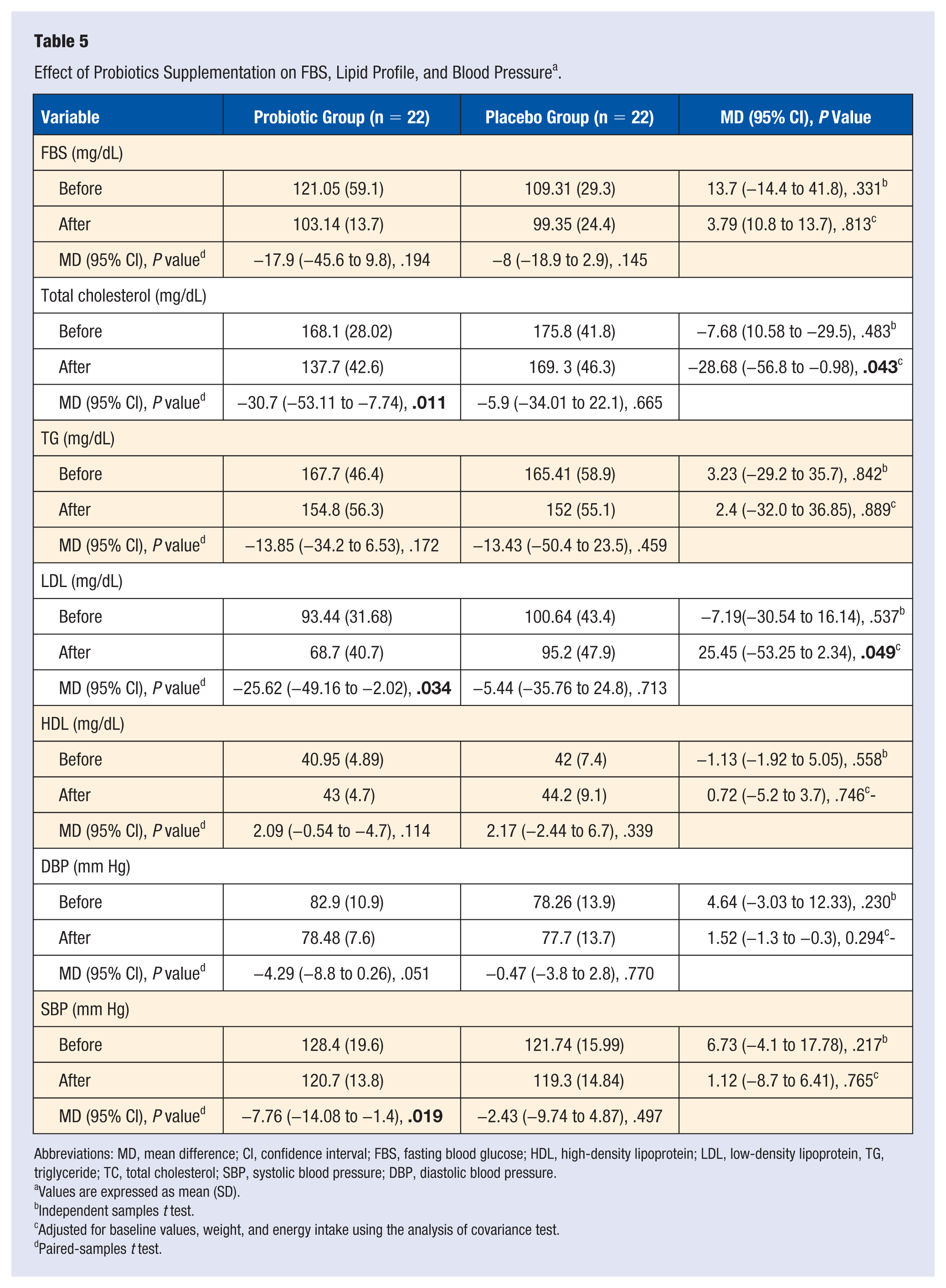
Abbreviations: MD, mean difference; CI, confidence interval; FBS, fasting blood glucose; HDL, high-density lipoprotein; LDL, low-density lipoprotein, TG, triglyceride; TC, total cholesterol; SBP, systolic blood pressure; DBP, diastolic blood pressure.
Values are expressed as mean (SD).
Independent samples t test.
Adjusted for baseline values, weight, and energy intake using the analysis of covariance test.
Paired-samples t test.
Discussion
To our knowledge, this is the first study to compare weight and metabolic features in patients with CAD receiving weight loss diet on either a probiotics supplementation or a conventional diet. Our results indicated that 12 weeks of supplementation with LGG had a significant effect on lipid profile in patients with CAD. We observed that the LGG supplementation leads to changes in TC and LDL. We found no significant difference in overall weight loss between the 2 groups. The data presented here proposes that weight loss diet will provide beneficial effect on cardiovascular risk factors, but accompanying the diet with probiotics may better promote MetS features. In summary, the weight loss diet plus probiotics supplementation could significantly enhance MetS features.
There are many studies with controversial results on the effects of probiotics administration on anthropometric indices. In the current study, reduction in anthropometric indices (weight, BMI, and WC) in the probiotic group was more than in the placebo group, but this did not reach to significant levels compared to the placebo group. The findings are in line with some previous studies. In one study conducted on patients with type 2 diabetes, receiving multistrain probiotic capsules for 12 weeks did not influence anthropometric indices including body mass and waist-to-hip ratio. 21 Sadrzadeh-Yeganeh et al, who supplied participants with 300 g/day probiotic yoghurt containing Lactobacillus acidophilus and Bifidobacterium lactis, failed to observe any anti-obesogenic effect of probiotics in healthy females. 22 However, Kadooka et al reported that 200 g/day of fermented milk containing Lactobacillus gasseri for 12 weeks had powerful effects on abdominal adiposity, body weight, and other measures in adults with obesity. 23 Temporarily, there are many studies on the beneficial effects of probiotics on metabolic disorders and body weight regulation; these effects are usually attributed to inhibition of lipid absorption. In addition, probiotics have an important role in body weight regulation by influencing energy metabolism and the inflammatory status of the body. 24 In the present study, we did observe statistically significant changes in nutrient intakes during the intervention; there was a descending trend in the total energy intake in both groups, because we provided the patients with a weight loss diet. Although weight loss was similar between the 2 groups, the effects on cardiovascular risk factors were still more favorable after probiotic supplementation when the results were adjusted for differences in weight loss. Taken together, we observed that weight loss diet in combination with the probiotics supplementation led to a significant decrease in weight, BMI, and WC; diet alone had no significant effect on these variables.
The evidence about the effect of probiotic on blood pressure has shown conflicting results.25,26 In the present study, we found a statistically significant change in systolic and diastolic blood pressure in probiotic group, but no significant effect of LGG on blood pressure compared to placebo was observed. In a study by Ivey et al, no significant changes of blood pressure was seen with daily consumption of probiotic yoghurt or supplements containing 2 strains of L acidophilus and Bifidobacterium animalis subsp for 6 weeks. 25 On the contrary, in a trial conducted on prediabetic patients, 500 mg probiotic capsules (which contained 7 × 109 CFU Lactobacillus casei, 2 × 109 CFU Lactobacillus acidophilus, and 7 × 109 CFU Bifidobacterium) for 8 weeks did significantly affect SBP, with no effect on DBP. 26 Several confounding factors including the type of subjects, range of blood pressure, different probiotic strains and species, and dietary changes during intervention have been identified and might have played a role in obtaining diverse results from various studies.
The main mechanisms by which probiotic administration might lower blood pressure are not well understood. Probiotics may act directly on the renin-angiotensin system through the production of angiotensin-converting enzyme inhibitory peptide. Aside from the vasodilatory effect of probiotics on vascular tone, their effects on a reducing plasma glucose and the onset of inflammatory process may also explain the effect of probiotics on BP.27,28
We found that LGG supplementation had no statistically significant effect on FBS. However, the amount of improvement in FBS levels in the probiotic group might have clinically significant effect on micro- and microvascular complications. 29 The finding is supported by some results of other clinical trials that supplemented patients with different probiotic strains. Mazloom et al 21 indicated that actobacillus probiotics including L acidophilus and bifidum for 6 weeks did not result in change of FBS. However, in one study among adults with type 2 diabetes, consumption of 300 g/day probiotic yogurt containing L acidophilus and B lactis for 8 weeks significantly influenced HbA1c and FBS levels. 30
In the current study, we found a significant reduction in serum TC and LDL-C, but not in other lipids, suggestive of probable cardioprotective properties of LGG. Previous studies reported diverse effects of probiotics administration on lipid profile. Based on Mahboobi et al, 500 mg probiotic capsules did not contribute to significant changes in lipid profile, in prediabetic patients after 8 weeks. 31 Moroti et al revealed that 4 weeks of receiving a symbiotic product can decrease serum TG and TC levels and increase HDL-C concentration significantly. 32 It has been confirmed that some species of Bifidobacterium, Lactobacillus, and Streptococcus bacteria are able to improve blood lipids. 33 In the present study, we used capsules containing LGG that did beneficially affect some markers of lipid profile. Fermentation products of Lactobacillus bacteria, mainly short chain fatty acids, may decrease cholesterol absorption by improving the binding of cholesterol to the intestinal lumen.34,35 They may also inhibit binding of cholesterol to cellular surface, and lead excretion of cholesterol with de-conjugated bile in stool. 36 Furthermore, these bacteria can inhibit the enzymatic synthesis of cholesterol. Moreover, in the present study, we administered weight loss diet, thereby the improvement in lipid profile found in patients supplemented with probiotic could be somewhat described by the induction of weight loss in these subjects. In our study, high lipid levels were not considered as the eligibility criteria for the participants. The lack of a significant effect of LGG on some lipids may be partly due to the low levels of these outcomes among participants at baseline; also 70% of subjects were under statin therapy.
There were some limitations and strengths in the current study. First, the other known risk factors of CAD such as Apo A, Apo B, and inflammatory markers were not measured. Moreover, the potential effects of probiotics on endothelial markers such as intercellular adhesion molecule 1 were not examined. Second, the intervention period was relatively short and sample size was relatively limited. One of the strengths of the current study was planning and monitoring the patients’ diets during the study. Also, physical activity and dietary intakes of the subjects were checked before and after the intervention. The novelty of current study is related to the use of LGG in CAD subjects, which had not been assessed before.
Conclusion
In summary, we observed similar weight loss in subjects randomly assigned to a weight loss diet plus probiotics, or the weight loss diet plus placebo. Despite modest overall weight loss in both groups, assignment to the probiotics group had a direct and more favorable effect on TC and LDL in patients with CAD; therefore, it is expected that probiotics might significantly reduce the overall CVD risk factors. These results give further evidence that weight loss program in combination with probiotics supplementation may have favorable effects on some metabolic feature. Most important, upcoming studies will need to assess whether this combination has more favorable effects on cardiovascular risk factors compared to probiotics alone.
Footnotes
Acknowledgements
We would like to thank all participants of the present study for their ideas, suggestions, participation, and support. Moreover, the authors wish to thank Tabriz University of Medical Sciences for financial support.
Authors’ Note
Data will be available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported financially by Tabriz University of Medical Sciences as a thesis proposal for the PhD degree of the first author.
Ethical Approval
The study protocol by the Medical Ethics Committee of Tabriz University of Medical Sciences (IR.TBZMED.REC.1397.184).
Informed Consent
Informed consent was taken from all the study subjects.
Trial Registration
The clinical trial was registered in the WHO | Iranian Registry of Clinical Trials (IRCT) (https://www.who.int/ictrp/network/irct/en/) with identifier IRCT20121028011288N15 and is available at ![]() .
.
