Abstract
Recently reported SEER data revealing an increase in the number of younger women who present with advanced breast cancer at diagnosis has led to much speculation regarding epidemiological factors that play a role in breast cancer risk and that may be modifiable by lifestyle. Molecular genomics has identified biological pathways that affect breast cancer prognosis and inform treatment decisions. Depending on menopausal status and breast cancer subtype, evidence is emerging that lifestyle could be implicated not only in the risk for breast cancer but also outcomes following breast cancer diagnosis. Several biologic mechanisms have been explored explaining the physiologic role of lifestyle and cancer risk and survival. In this report, we explore the interaction between lifestyle, breast cancer phenotype, and outcomes and review potential molecular mechanisms linking lifestyle to risk and prognosis in order to establish a platform for future exploration. The cost of breast cancer treatment and breast cancer morbidity and mortality during a woman’s productive years impose a psychosocial and socioeconomic burden on our nation. Efforts to ease that burden through lifestyle modification should be embraced and supported as we reevaluate and scrutinize the allocation of health care resources in our nation.
Keywords
‘Lifestyle across the lifespan is emerging as a modifiable factor that contributes both to the risk of developing breast cancer as well as to the risk of dying from it.’
A recent report from the US Surveillance, Epidemiology, and End Results (SEER) data earlier this year, indicating a small but significant increase in the proportion of young women presenting with stage IV breast cancer, has led to speculation regarding possible etiologies of this trend. 1 The annual percentage change was seen in 25- to 39-year-old women of all races and ethnicities, and estrogen receptor–positive breast cancer accounted for most of the increase. 1 Several risk factors for breast cancer are widely recognized, some that are potentially modifiable and some that inherently cannot be altered. 2 Lifestyle across the lifespan is emerging as a modifiable factor that contributes both to the risk of developing breast cancer as well as to the risk of dying from it. These observations could aid us in our understanding of the changing epidemiology of breast cancer and provide new strategies for prevention. An epidemiologic review of physical activity, diet, and adiposity showed a relationship between adiposity/physical activity and breast cancer mortality. 3 A new taxonomy of breast cancer classification based on molecular subtypes has provided insights into outcomes of breast cancers by molecular phenotype and has assisted in personalized treatment planning for patients. 4 In this review, we present an update of the literature linking lifestyle to breast cancer risk and breast cancer mortality with a focus on body mass index (BMI), adiposity, and physical activity including possible pathophysiologic mechanisms to explain the interactions and the relationship to breast cancer molecular phenotypes.
Breast Cancer Risk Factors: Accepted and Speculative
Several risk factors have been identified that contribute to increased breast cancer risk. Gender, increasing age, family pedigree, germ line mutation status, a diagnosis of proliferative breast disease, menarche, reproductive and lactation history, use of combination postmenopausal hormone replacement therapy, alcohol use, radiographic breast density, and prior radiation exposure have all been associated with increased lifetime risk of breast cancer. 5 Smoking, dietary consumption, use of low-dose oral contraceptives, and postmenopausal exposure to low-dose estrogen remain somewhat unclear in terms of risk relationship.5-7 Obesity, BMI, and physical activity are gaining recognition as modifiable lifestyle factors that can affect the development of breast cancer and contribute to prognosis after diagnosis.2,8
Breast Cancer Molecular Taxonomy and Phenotype
In the human mammary gland, 2 distinct types of cells have been identified by immunohistochemical staining—basal (also termed myoepithelial) cells and luminal epithelial cells. 9 In addition to the traditional classifications and staging of invasive breast cancer—ductal versus lobular, estrogen and progesterone receptor positive or negative, Her2 neu normal or amplified, grade 1 through 3, and stage I through IV—the new classification includes luminal A (best prognosis), luminal B (intermediate prognosis), basal, and ERBB2+/Her2 neu+ (worst prognosis). 4 For treatment purposes, luminal A cancer is more sensitive to endocrine therapy and less sensitive to chemotherapy (strong estrogen and progesterone receptor expression, low proliferation), and luminal B less sensitive to endocrine therapy (lower estrogen receptor and low to negative progesterone receptor expression, high proliferation or HER-2 overexpression [luminal HER-2]) but with increasing sensitivity to chemotherapy (and trastuzumab for the luminal HER-2). 10 ERB2+ or Her 2 neu amplified breast cancers clearly benefit from chemotherapy plus trastuzumab. 11 Basal-like breast cancers (sometimes referred to as triple negative, although the 2 groups do not entirely overlap) are treated with chemotherapy and are the phenotype commonly seen in patients who carry a germ line mutation in the BRCA1 gene.4,12 Based on the biology of these tumor subtypes, lifestyle factors that modify sex hormones, growth factor ligands, metabolic pathways, and the tumor microenvironment may play a role in tumor development and prognosis.
Body Habitus and the Epidemiology of Breast Cancer
A comprehensive review of adiposity and breast cancer outcome identified mixed results, with an overall association of obesity with a 30% increase in mortality including one compelling study linking type 2 diabetes mellitus to newly diagnosed breast cancer and mortality. 3 Recent data suggest that obesity causes inflammation systemically and in the breast tissue, contributing to the development and progression of breast cancer. 13 The question of dietary content versus adiposity remains unclear, and the data are insufficient to support any one particular dietary habit or nutrient base. It appears that obesity regardless of type of nutrient intake is the prevailing influence on breast cancer risk and mortality.3,7 In a summary of prospective studies and randomized clinical trials investigating the association between lifestyle and breast cancer prognosis, women with weight gain after a diagnosis of breast cancer were found to have a greater risk of recurrence and mortality. 14 In an Italian multicenter case–control study, BMI >30 kg/m2, and weight gain of 5 BMI units or more between age 30 and age at diagnosis were associated with higher breast cancer mortality. 8
In addition to BMI and obesity, skeletal muscle mass may play a role in breast cancer outcome. In a study of 471 breast cancer patients who participated in the Health, Eating, Activity, and Lifestyle (HEAL) study, reduction in skeletal muscle mass 2 standard deviations below the young healthy adult female mean was associated with an increase in breast cancer–specific mortality leading to potential new avenues of study of the effect of sarcopenia (low BMI but little to no muscle mass) and breast cancer events. 15
The impact of body habitus and breast cancer phenotype is less clearly defined. Data from the Women’s Health Initiative were analyzed for BMI, waist–hip ratio, and breast cancer phenotype. 16 Women in the highest BMI quartile had a significant increase in basal-like (triple negative) and estrogen receptor–positive breast cancers, and increased waist and hip circumference were positively associated with endocrine-sensitive breast cancer. 16 A study of BMI at diagnosis and tumor characteristics showed an association between increased BMI and larger tumor size in pre- and postmenopausal women, increase in the risk of lymph node metastases, and evidence of vascular invasion in obese premenopausal women. 17 However, there was no association between BMI and breast cancer phenotype (luminal A, B, Her2 amplified, or basal-like). 17 In a pooled analysis of 34 studies from the Breast Cancer Association Consortium including 35 568 invasive breast cancer patients, BMI > 30 kg/m2 was associated with estrogen receptor–negative and progesterone receptor–negative breast cancer (both triple negative and Her2-neu amplified), whereas in older women, increased BMI was associated with progesterone receptor positivity, an indicator of endocrine sensitive subtype. 18 In 535 postmenopausal women with operable breast cancer, increasing BMI was associated with lower incidence of Her2-neu amplification. 19 It has been implicated that there is biological “cross talk” between estrogen receptor pathways and Her2-neu pathways, and this inverse relationship may be related to the increase in estrogenic effect on tumors arising in postmenopausal obese women. 20 Also, inflamed white adipose tissue within the breast has been associated with elevated levels of pro-inflammatory mediators, enhanced expression of aromatase, and increased estrogen receptor expression. 13 A retrospective analysis of 2311 women with stage I to III basal-like (triple negative) breast cancer showed a poor prognosis for this phenotype regardless of BMI. 21 In contrast, a quantitative analysis of 11 published articles evaluating obesity as a risk factor for triple negative breast cancer showed a statistically significant association in premenopausal women. 22 This finding was corroborated by data from the Nurses’ Health Study demonstrating a direct association between waist–hip ratio and risk of estrogen receptor–negative breast cancer in premenopausal women. 23 Furthermore, in a case–control analysis of African-American and white women from the Carolina Breast Study, elevated waist–hip ratio was linked to luminal A breast cancer in postmenopausal women and basal-like breast cancer in both pre- and postmenopausal women with the association in premenopausal African-American women being the strongest. 24 Patients who are carriers of germ line mutations in BRCA1 or BRCA2 (hereditary breast and ovarian cancer or HBOC) have a significantly higher risk of breast and ovarian cancer than the average population and an increase in the development of breast and ovarian cancer at a younger age of onset. 25 Guidelines for management of HBOC include increased surveillance and consideration of risk reduction strategies including prophylactic mastectomies and prophylactic removal of the ovaries and fallopian tubes prior to menopause. 26 In women carrying a deleterious BRCA1 or BRCA2 mutation, a case–control study demonstrated a link between loss of >10 pounds between age 18 and 30 and reduced risk of breast cancer between age 30 and 49. 27 In this study, a weight gain during that time frame was not associated with increased overall risk except in the population of patients with BRCA1 mutation who had at least 2 children. 27
Physical Activity, Breast Cancer Risk, and Survival
In a review of observational studies evaluating the association of physical activity and breast cancer risk as well as survival after diagnosis, a non–statistically significant trend was found between physical activity and approximately 30% reduced breast cancer mortality. 3 In the Iowa Women’s Health Study with 554 819 person-years of follow-up and 2548 cases of breast cancer, physical activity appeared to reduce the incidence of postmenopausal breast cancer, especially the luminal B subtype. 28 In a study of women in Shanghai, China, exercise in adolescence as well as exercise patterns in the previous 10 years from diagnosis appeared to reduce hormone receptor–positive and hormone receptor–negative breast cancer in pre- and postmenopausal patients, with sweating during exercise also being associated with risk reduction in postmenopausal women. 29 Breast cancer incidence and phenotype distribution differ in Asian women compared to Western populations, suggesting that hormonal and nonhormonal mechanisms associated with physical activity may play a role in risk reduction. 29 The National Institutes of Health–AARP Diet and Health Study evaluated nonrecreational physical activity and breast cancer risk in 97 039 postmenopausal women and found that routine activity during the day including heavy lifting or carrying and avoidance of sedentary behavior led to a statistically significant reduction in the diagnosis of invasive breast cancer. 30 In the Nurses’ Health Study, a subset analysis of 2987 women with stage I to III breast cancer demonstrated improved survival associated with physical activity especially in women with endocrine-sensitive phenotypes who walked an average of 3 to 5 hours per week. 14 Likewise, data from the Women’s Health Initiative demonstrated a reduced risk of estrogen receptor–positive and triple negative breast cancer in postmenopausal women in the highest activity tertile. 16
Mechanisms Linking Physical Activity, Obesity, and Body Habitus to Breast Cancer Risk and Mortality
Obesity has emerged as a significant epidemiologic phenomenon of Western civilization, and its implications for diseases including cardiovascular disease, hypertension, diabetes, and metabolic syndrome are well recognized. Growing attention is now focused on the carcinogenic impact of obesity and malignancy with potential pathophysiologic mechanisms of the interaction coming to light. The association of elevated leptin and decreased adiponectin with hyperinsulinemia and activation of insulin-like growth factor signaling via the mTOR pathway has gained significant scientific support as a mediator of cancer pathogenesis. 31 The impact of adiponectin on reduction of mediators of inflammation may also play a role in the neoplastic process. 31 The observation of a significant reduction in cancer diagnosis in Scottish patients with type 2 diabetes who were new users of the insulin sensitizing drug metformin has led to exploration of the role of metabolism and mediators of cellular metabolism as potential targets for cancer reduction and has aided us in our understanding of the lifestyle/cancer link. 32 Furthermore, patients treated with long-acting insulin analogs have been shown to have an increased risk of cancer versus those taking metformin. 33 One intracellular pathway that sits at the crossroads of several signaling networks is the phosphatidylinositol 3-kinase signaling pathway (PI3K). 34 This kinase resides downstream of the insulin-like growth factor receptor and Her2-neu receptor cascades and affects estrogen receptor and progesterone receptor activity. 34 For this reason, it plays a key role in tumor biology and is being studied as a prime target for antineoplastic therapy. It also appears to be modifiable by physical activity and metabolic factors. PI3K plays a role in each phenotypic subclass of breast cancer, luminal A, B, Her2 amplified, and triple negative. 34 Amplification of copies of PI3K is associated with poorer prognosis for each subtype of breast cancer. 35 To further understand the connection of PI3K, physical activity, and obesity with the neoplastic process, the AMP activated protein kinase (AMPK) plays a central role in the physiologic process. 36 AMPK is an evolutionarily conserved protein kinase found from single celled eukaryotes to mammalian species and plays a pivotal role in the regulation of energy balance. 36 Exercise increases AMPK leading to a reduction in mTOR and PI3K activity, lowers leptin activity, and increases insulin sensitivity—all of which can reduce neoplasia. 36 Insulin resistance is a demonstrated risk factor for breast cancer in postmenopausal women. 37 Moderate intensity exercise has been shown to decrease in insulin-like growth factor in postmenopausal breast cancer survivors.38,39 The data on intracellular metabolic pathways and association with physical activity and body habitus are illuminating of the relationship between lifestyle and breast cancer.
In addition to the effect on intracellular metabolic pathways, other mechanisms may explain the effect of exercise and obesity on breast cancer epidemiology. When lean and obese rodents with mammary tumors underwent surgical oophorectomy and were subsequently overfed, nutrient tracing studies demonstrated preferential deposition of nutrients into tumor cells in the obese animals whereas the lean animals showed preferential deposition in peripheral tissues and nonmalignant mammary tissue. 38 In the animal model, treatment with metformin led to tumor regression and downregulation of progesterone receptors. 40 Overweight postmenopausal female breast cancer survivors who were placed on a low-fat/high-fiber diet and who attended daily exercise classes for 2 weeks were shown to have a significant reduction in estrogen levels and insulin/IGF-1. 41 An in vitro corollary of this study exposed breast cancer cell lines to serum obtained from patients before and after the intervention, and the cell lines demonstrated a decrease in cell growth and increase in apoptosis after exposure to the postintervention patient serum. 41 In the Alberta physical activity and breast cancer prevention trial, a statistically significant reduction in serum estradiol was seen in the exercise group versus control group after a yearlong intervention. 42
Modulators of inflammation and immunologic responses may also play a role in exercise and breast cancer risk and one of the pathophysiologic mechanisms could involve the tumor microenvironment and tumor-associated macrophages. 43 Epigenetic changes could also be implicated in the link between physical activity and breast cancer outcome potentially via reduction in methylation of tumor suppressor genes (see Table 1). 44
Exercise and Body Habitus: Impact on Cellular Mechanisms.
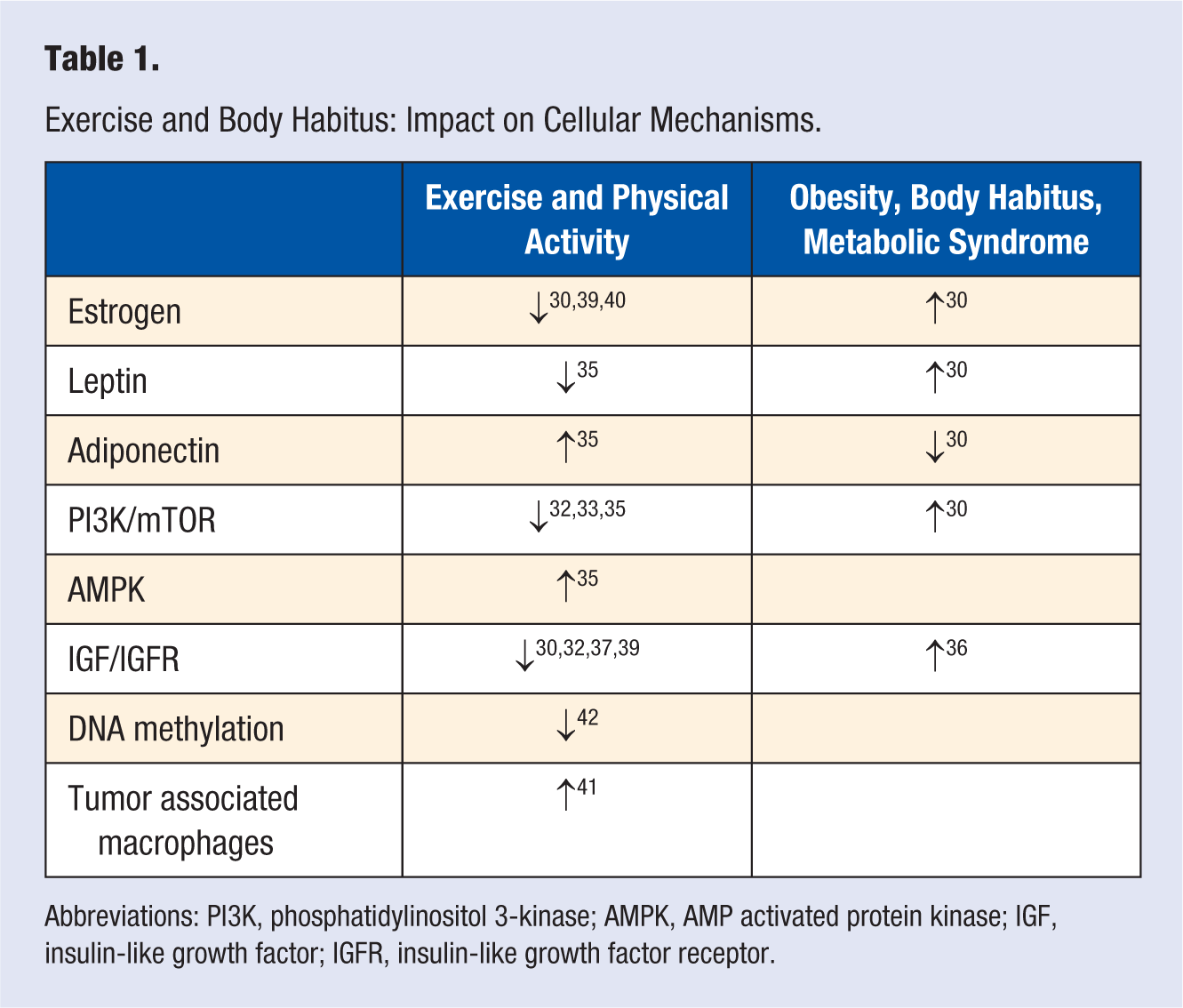
Abbreviations: PI3K, phosphatidylinositol 3-kinase; AMPK, AMP activated protein kinase; IGF, insulin-like growth factor; IGFR, insulin-like growth factor receptor.
Conclusions and Future Directions
Increasing physical activity and improving body habitus are 2 lifestyle strategies that can address the epidemics of obesity and breast cancer in Western culture, and also diabetes mellitus and hypertension. The links between lifestyle modification and cancer epidemiology provide modifiable factors that can reduce cancer risk or decrease its recurrence. Emerging science has illuminated several possible mechanisms of the interaction between lifestyle and neoplasia. Factors that affect cellular metabolism and intracellular signaling networks may provide targets for future intervention. Guidelines have been created for obesity and cancer prevention.3,31 The greatest challenge we face is in developing strategies for patient motivation and compliance to alter the epidemiology of breast cancer.
A study of breast cancer care in California in 2001 revealed that $279 million was spent on direct cost of care. In addition, premature loss of life was estimated to cost the state $1.1 billion in lost productivity and premature death. 45 As our nation embraces health care reform, the increased importance of the role of exercise physiology and nutrition research should be recognized and supported. Furthermore, insurance coverage for wellness programs, personal training, and enrollment in lifestyle enhancement and weight reduction would be a wise use of dollars under the Affordable Care Act.
Footnotes
Acknowledgements
The authors acknowledge Clarence Brown, MD, for the original concept and Dr Eleftherios Mamounas for his assistance in review of the article.
