Abstract
Objectives. A systematic review of randomized clinical trials and meta-analyses evaluating the efficacy, tolerability, and safety of statins in preventing cardiovascular disease (CVD) in women without cardiovascular disease. Background. Several meta-analyses have been performed evaluating statins in CVD primary prevention trials involving women. This review is an update incorporating the results of recent CVD primary prevention trials in women and the recent concerns of statins and new-onset diabetes. Method. PubMed database was searched for primary prevention trials and meta-analyses. The key terms “statins, cardiovascular disease, primary prevention in women” were used. Search was limited to all English publications published up to October 2012. Results. Statin use led to a trend towards reduction in cardiovascular mortality and morbidity in women. No significant increased risk in adverse events was observed. The slight increased incidence of diabetes is outweighed by the greater cardiovascular benefit derived from statin use. Conclusions. The data support the use of statins for primary prevention of CVD in women at higher risk of CVD. The lack of statistical significance in prior randomized controlled trials and meta-analyses is attributable to the lower numbers of women enrolled in these trials and the lower CVD risk of women in the trials resulting in the inadequate powering of these studies. Higher risk women who may benefit from CVD primary prevention with statins may be identified using validated tools such as the Reynolds scoring system, the 2011 American Heart Association risk algorithm for women, and the forthcoming National Heart, Lung, and Blood Institute risk equations.
Keywords
‘Statin therapy to lower low-density lipoprotein cholesterol (LDL-C) has consistently been shown to reduce cardiovascular events in numerous primary and secondary prevention trials’
Introduction
Cardiovascular disease (CVD), primarily manifested as coronary heart disease (CHD) and stroke, is the leading cause of death in women. Data from the Centers for Disease Control and Prevention show that 1 in 3 women die from cardiovascular disease.1,2 In the past 2 decades, the absolute number of women who died from CVD has been greater than men. 3 Differences have been demonstrated in the presentation and outcomes of CVD in women. In women, the onset of clinical coronary artery disease appears to lag by about 10 to 15 years compared with men. 4 However, compared with men of the same age, women age younger than 60 years have increased mortality from myocardial infarction. 5
The reasons for the sex disparities in CVD outcomes are incompletely understood. Unique characteristics have been demonstrated in the pathophysiology of ischemic heart disease (IHD) in women. Compared with men, women have less coronary artery plaque burden6,7 and in up to a third of women with chest pain, no obstructive coronary artery disease is seen on coronary angiography.8,9 Young women with CVD usually do not develop the classical plaque rupture with subsequent thrombus formation, but rather, have plaque erosion with subsequent distal embolization of microemboli.10-12 Microvascular dysfunction has also been demonstrated as a potential etiology of IHD in women. 13
Despite differences in CVD presentation and pathophysiology, the same traditional risk factors predict the risk of CVD for both women and men: age, smoking status, total cholesterol, high-density lipoprotein cholesterol (HDL-C), blood pressure, and antihypertensive treatment. 14 In addition to the traditional risk factors for CVD, female-specific (hormonal) CVD risk factors have also been identified. Premenopausal estrogen deficiency has been hypothesized as a potential etiology for increased risk of atherosclerosis and CVD in young women. 15 Menstrual irregularity is associated with increased adverse cardiovascular events 16 and androgen excess as seen in polycystic ovarian syndrome is associated with increased atherosclerosis and IHD.15,17 Aging in women and menopause leads to unfavorable lipid profiles. 18 Pregnancy and its complications have also been associated with CVD risk. 19
The discovery of these sex-specific risk factors and the differences in CVD presentation in women prompted several trials to evaluate the efficacy of several interventions for the primary and secondary prevention of CVD in women. The Women’s Health Initiative (WHI)20,21 and the Heart and Estrogen/Progestin Replacement Study 22 demonstrated the failure of postmenopausal hormone replacement therapy for primary or secondary prevention of cardiovascular disease. Moreover, the Women’s Health Initiative study demonstrated an increased risk of myocardial infarction (MI), stroke, breast cancer, and pulmonary emboli with hormone replacement therapy. All-cause mortality was not altered.
Statin therapy to lower low-density lipoprotein cholesterol (LDL-C) has consistently been shown to reduce cardiovascular events in numerous primary and secondary prevention trials that included both women and men. 23 As a result, statin therapy to lower LDL-C is recommended for both women and men on the basis of nearer term (10-year) CVD risk.24,25 Most of the early trials evaluating statins in CVD primary prevention enrolled predominantly men.26-32 Sex-specific results were reported in only 31% of primary prevention trials and only 25% of study participants were women. 33 This has raised concerns about the utility and safety of extrapolating findings and recommendations from predominantly male study populations to women. To be deemed an effective primary prevention intervention, the intervention must either reduce potential suffering (morbidity) or prolong life (reduce mortality) and the benefits from the intervention outweigh potential adverse events. To address the benefits of statin therapy in women, we conducted a systematic review of all randomized placebo-controlled trials and meta-analyses of cardiovascular disease primary prevention statin trials involving women and in which clinical outcomes in women were reported. Given the recent concerns of statins and possible diabetes risk, our objective was to provide an updated review of the efficacy, effect on mortality and morbidity, safety, and tolerability of statins for primary prevention of cardiovascular disease in women.
Methods
We performed a systematic review of randomized placebo-controlled trials and meta-analyses published between 1990 and October 2012. Our objective was to determine the efficacy, tolerability, and safety of statins in preventing CVD in women without a prior history of CVD. Studies were included in our review if the trial or meta-analysis met the following criteria: (a) language of publication in English, (b) trial or meta-analysis evaluated statin use for primary prevention, (c) trial design was randomized, and (d) sex-specific results were reported. PubMed database was searched using the terms: “statins, cardiovascular disease, primary prevention in women,” and use the filters “humans and English language.” During the time period specified, 262 articles were identified (Figure 1). All articles were reviewed. Data were collected for sex-specific effects on all-cause mortality, CHD-related mortality, CVD events and outcomes as reported as relative risks (RRs) with corresponding confidence intervals (CIs).
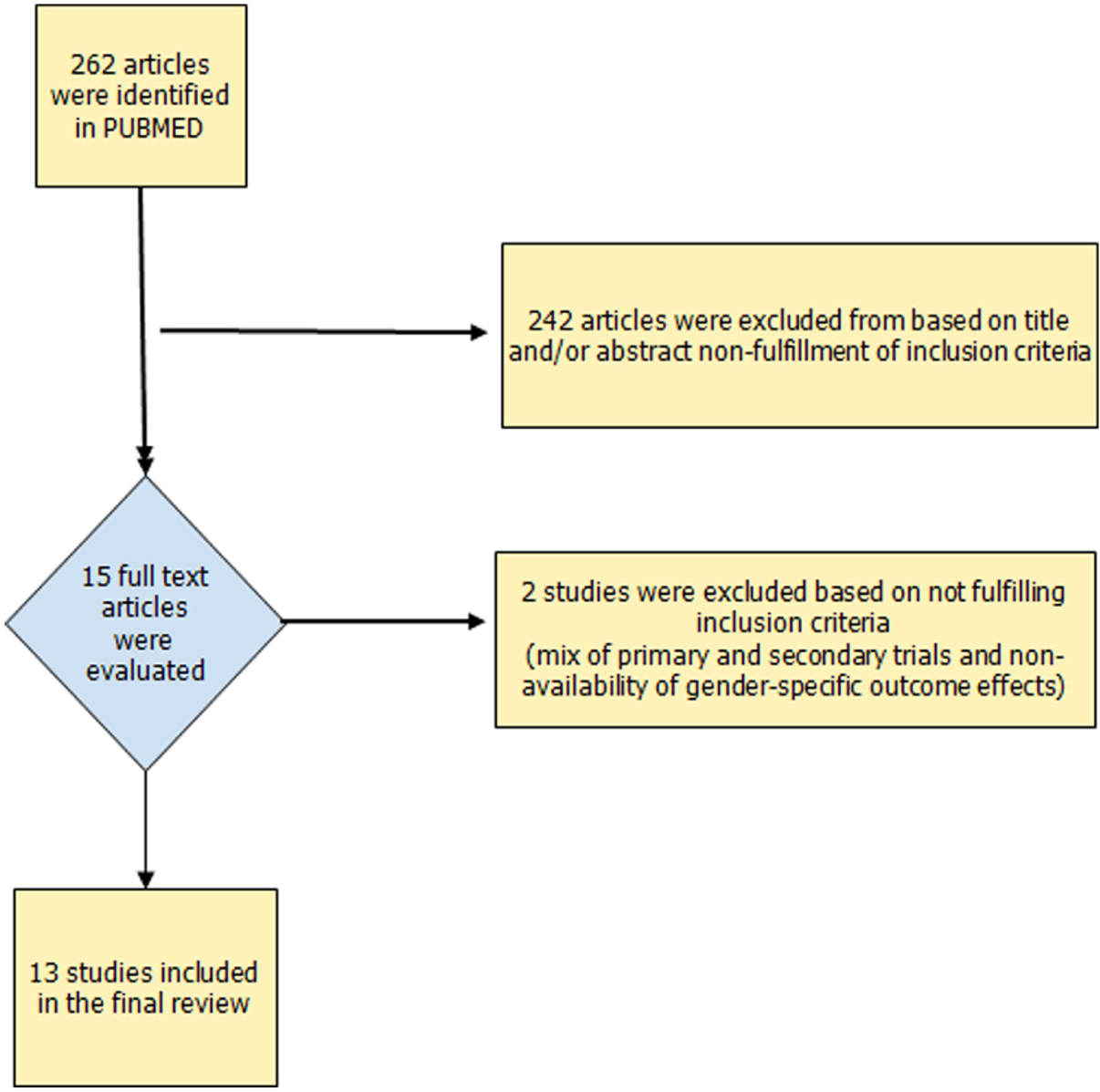
Flow diagram of selection of studies for inclusion in the review article.
Results
Six primary prevention trials that included women or reported outcomes in women29,30,32,34-36 were identified (see Table 1 for study characteristics). The Heart Protection Study 37 and the Pravastatin in elderly individuals at risk of vascular disease (PROSPER) 28 studies were excluded as they were a mix of primary and secondary prevention populations and the primary prevention gender-specific outcome effects were not available. Four meta-analyses of statin use in primary prevention in both men and women were also identified38-41 as well as an additional 3 meta-analyses specifically designed to evaluate outcomes in women.36,42,43 One of these meta-analyses evaluated both primary and secondary prevention of CVD 42 (see Table 2 for meta-analyses characteristics).
List of Primary Prevention Trials in Women.
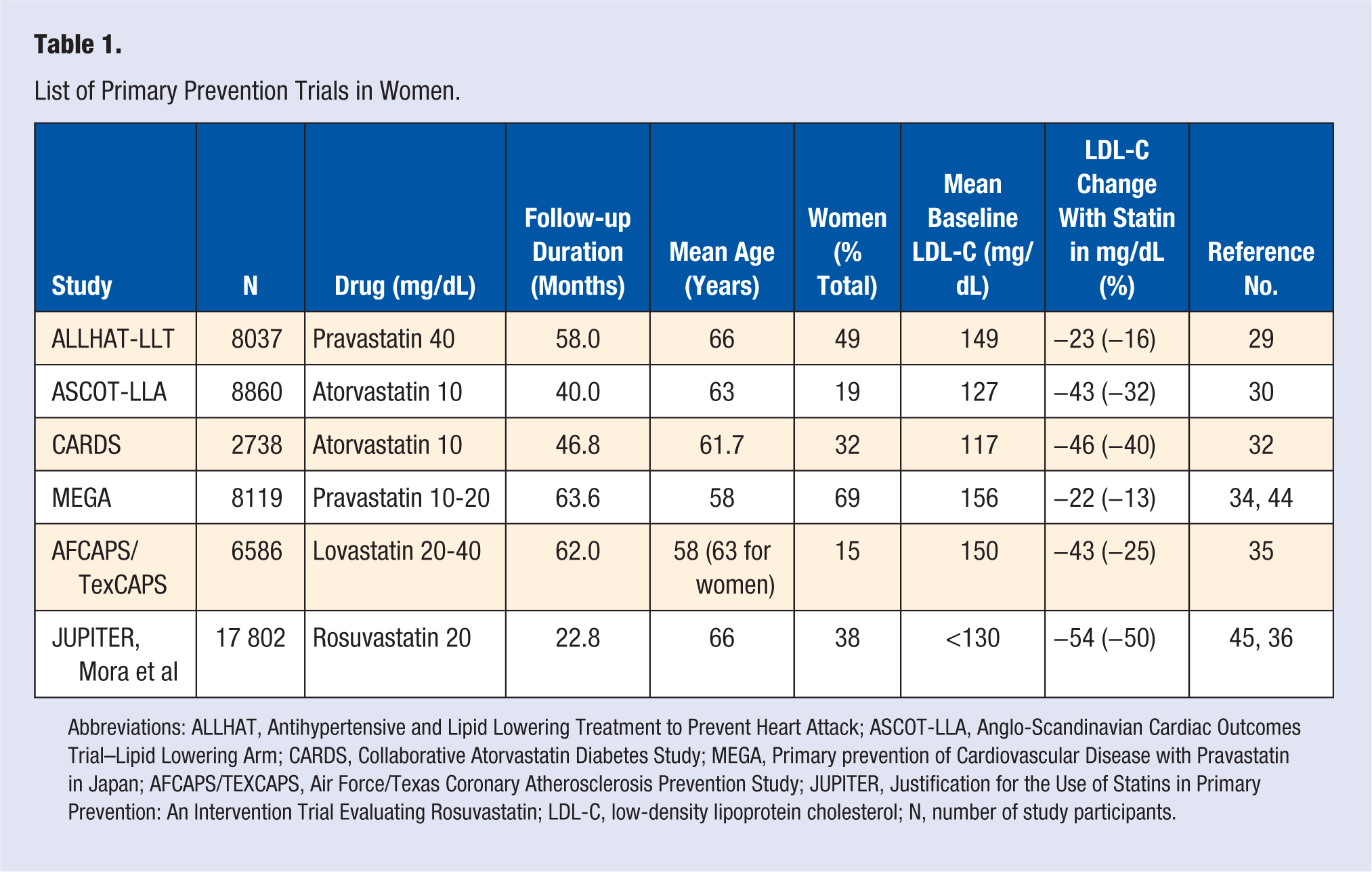
Abbreviations: ALLHAT, Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack; ASCOT-LLA, Anglo-Scandinavian Cardiac Outcomes Trial–Lipid Lowering Arm; CARDS, Collaborative Atorvastatin Diabetes Study; MEGA, Primary prevention of Cardiovascular Disease with Pravastatin in Japan; AFCAPS/TEXCAPS, Air Force/Texas Coronary Atherosclerosis Prevention Study; JUPITER, Justification for the Use of Statins in Primary Prevention: An Intervention Trial Evaluating Rosuvastatin; LDL-C, low-density lipoprotein cholesterol; N, number of study participants.
List of Meta-Analyses of Primary Prevention Trials in Women.
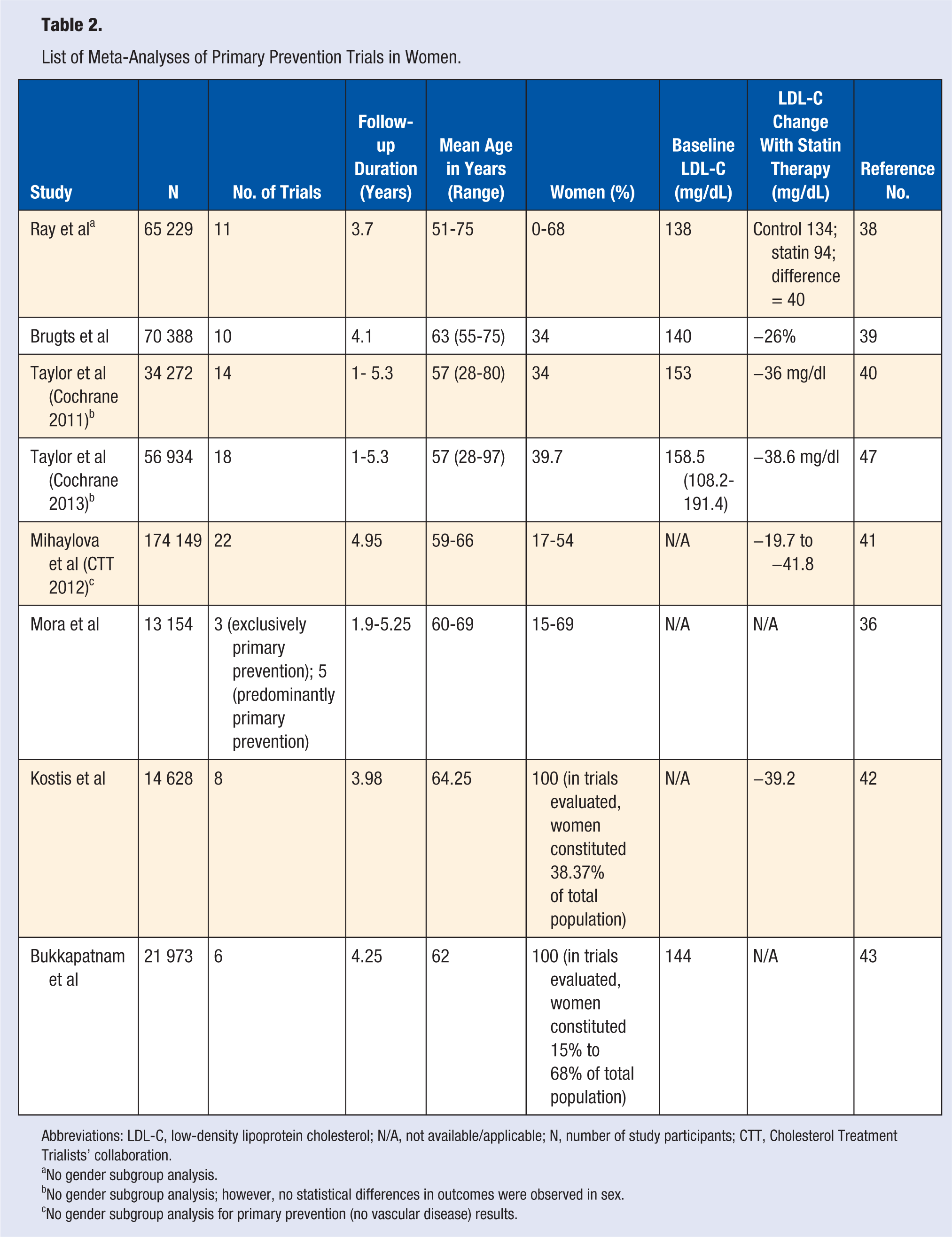
Abbreviations: LDL-C, low-density lipoprotein cholesterol; N/A, not available/applicable; N, number of study participants; CTT, Cholesterol Treatment Trialists’ collaboration.
No gender subgroup analysis.
No gender subgroup analysis; however, no statistical differences in outcomes were observed in sex.
No gender subgroup analysis for primary prevention (no vascular disease) results.
Effects on Morbidity
Five of the trials evaluated low- to moderate-intensity statin therapy (LDL-C reductions of <35% from pravastatin 10-40 mg, lovastatin 20-40 mg, and atorvastatin 10 mg29,30,32,34,35; one trial evaluated a high-intensity statin therapy (LDL-C reduction 50% from rosuvastatin 20 mg). 36 Three of the 4 moderate-intensity statin trials demonstrated a nonsignificant reduction of CVD events in women, although the magnitudes of effect were similar to the effects in men, with no evidence of treatment heterogeneity by sex observed.
The Management of Elevated Cholesterol in the Primary Prevention Groups of adult Japanese (MEGA) study, 34 evaluated the efficacy of a low-intensity statin intervention (pravastatin 10-20 mg) in a primary prevention population of Japanese individuals with hypercholesterolemia (total cholesterol 220-270 mg/dL). MEGA enrolled a total of 7832 patients (40-70 years old), of which 68% were women. The mean follow-up period was 5.3 years. The study showed a lower event rate for CHD in the diet plus pravastatin group (66 events) compared with the diet-alone group (101 events); hazard ratio [HR] = 0.67, 95% CI = 0.49-0.91, P = .01). A subgroup analysis was performed to evaluate the effect of pravastatin for primary prevention in women. 44 The MEGA study showed low-dose statin use in women (pravastatin 10-20 mg) led to a nonsignificant reduction in outcomes compared to diet only. There was a 25% lower incidence of CHD compared with the control group (HR = 0.75, 95% CI = 0.45-1.25, P = .27), a 37% reduced incidence of stroke (HR = 0.63, 95% CI = 0.36-1.10, P = .10), a 28% reduced incidence of all cardiovascular events (HR = 0.72 95% CI = 0.50-1.02, P = .07), a 23% reduced incidence of cerebral infarction and transient ischemic attack (HR = 0.77, 95% CI = 0.41-1.44, P = .42), and a 26% reduced incidence of CHD and cerebral infarction (HR = 0.74, 95% CI = 0.50-1.12, P = .15). The number needed to treat (NNT) to prevent 1 CHD or CHD plus cerebral infarction event in women ≥55 years was 176 and 109, respectively, and 106 and 61, respectively, for women ≥60 years (see Table 3 for event rates in women). Statin use led to a significant reduction in revascularization/unstable angina in women (HR = 0.24, 95% CI = 0.11-0.51), which was significantly greater than the reduction observed in men (HR = 0.63, 95% CI 0.46-0.85, P for heterogeneity = .01).
Primary Prevention Trials Event Outcomes in Women.
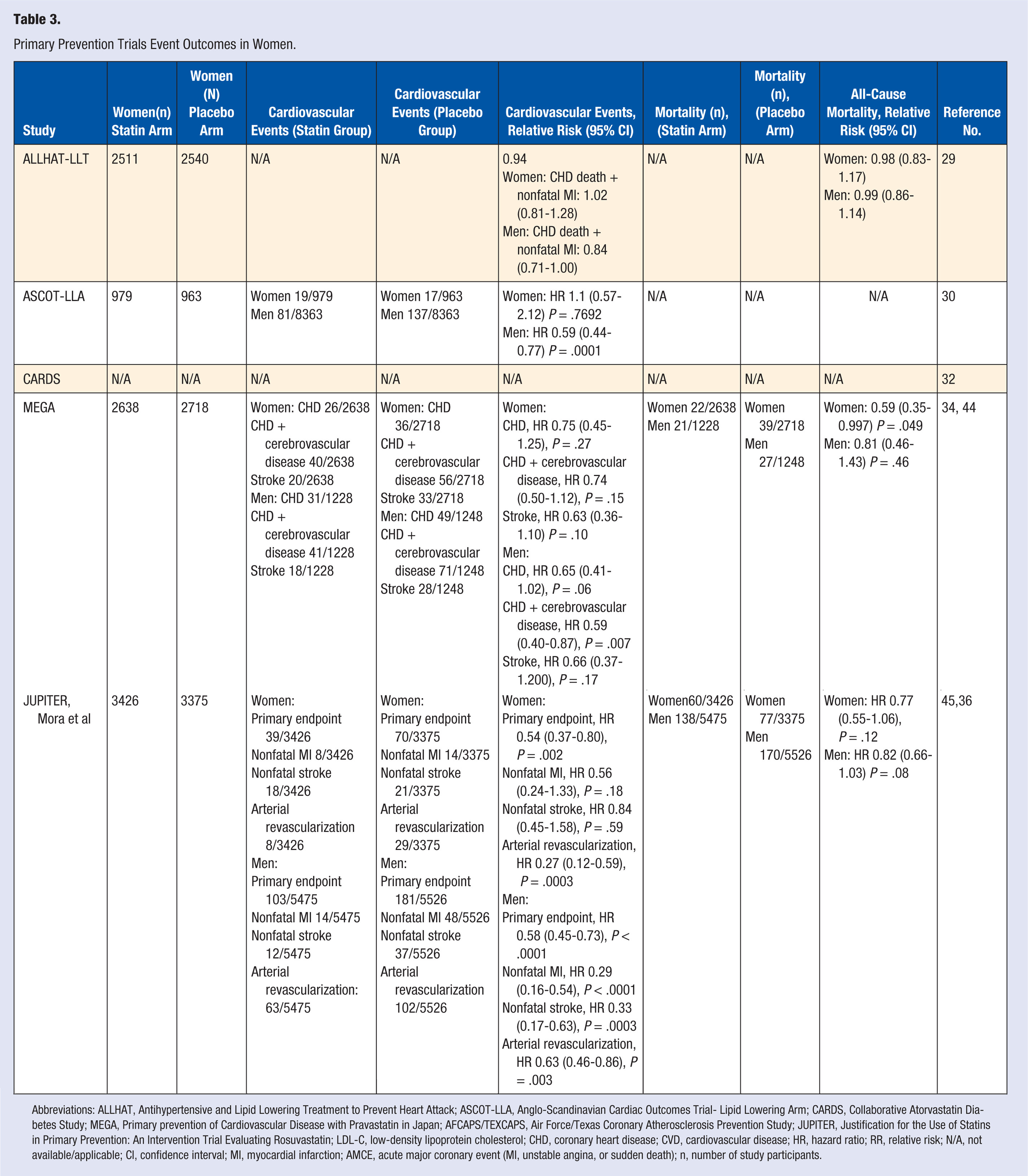
Abbreviations: ALLHAT, Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack; ASCOT-LLA, Anglo-Scandinavian Cardiac Outcomes Trial- Lipid Lowering Arm; CARDS, Collaborative Atorvastatin Diabetes Study; MEGA, Primary prevention of Cardiovascular Disease with Pravastatin in Japan; AFCAPS/TEXCAPS, Air Force/Texas Coronary Atherosclerosis Prevention Study; JUPITER, Justification for the Use of Statins in Primary Prevention: An Intervention Trial Evaluating Rosuvastatin; LDL-C, low-density lipoprotein cholesterol; CHD, coronary heart disease; CVD, cardiovascular disease; HR, hazard ratio; RR, relative risk; N/A, not available/applicable; CI, confidence interval; MI, myocardial infarction; AMCE, acute major coronary event (MI, unstable angina, or sudden death); n, number of study participants.
The Antihypertensive and Lipid-Lowering treatment to prevent Heart Attack Trial (ALLHAT-LLT), 29 evaluated the efficacy of low- to moderate-intensity statin use (pravastatin 10-40 mg) compared with usual care in reducing mortality in older individuals without known CHD, but with hypertension, LDL-C 120 to 189 mg/dL, and an additional CHD risk factor. The ALLHAT-LLT trial enrolled 10 355 participants, of which 49% were women. The mean follow-up period was 4.8 years. In the pravastatin group, there were 380 CHD events compared with 421 CHD events in the usual care group. For both sexes, pravastatin use led to a nonsignificant reduction in the secondary endpoint of fatal CHD and nonfatal MI (RR = 0.91, 95% CI = 0.79-1.04, P = .16), and stroke (RR = 0.91, 95% CI = 0.75-1.09, P = .31). No significant heterogeneity in outcome was reported for sex. There was no benefit with pravastatin use in women on the secondary endpoints of fatal CHD and nonfatal MI (RR = 1.02, 95% CI = 0.81-1.28).
The Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS) study 35 enrolled a total of 6605 participants, of which 15% were postmenopausal women. This study evaluated the efficacy of low- to moderate-intensity statin (lovastatin 20-40 mg) compared with placebo for prevention of the first event of major coronary events in men and women with average cholesterol levels (LDL-C 130-190 mg/dL) and below average HDL-C levels. The mean follow-up period was 5.2 years. With lovastatin, the incidence of first acute major coronary event was significantly reduced (event rate 116 with lovastatin, 183 with placebo; RR = 0.63, 95% CI = 0.50-0.79, P < .001). No statistical difference was reported for treatment (statin) benefit between sexes (P = .859). The study showed that in women, statin use led to a nonsignificant reductions in acute major coronary event (RR = 0.54, 95% CI = 0.22-1.36, P = .183), revascularization (RR = 0.89, 95% CI = 0.32-2.44, P = .814), unstable angina (RR = 0.34, 95% CI = 0.09-1.24, P = .085), MI (RR = 0.67, 95% CI = 0.19-2.37, P = .532), cardiovascular events (RR = 0.67 95% CI = 0.34-1.31, P = .236), and coronary events (RR = 0.56, 95% CI = 0.25-1.28, P = .164). 35 The NNT for 5 years to prevent the first acute major coronary event in women was 88.
The Anglo-Scandinavian Cardiac Outcomes Trial–Lipid Lowering Arm trial (ASCOT-LLA) study 30 did not find any difference in the primary endpoint (nonfatal MI and fatal CHD) with atorvastatin use compared with placebo in women. The ASCOT-LLA 30 evaluated the efficacy of a moderate-intensity statin (atorvastatin 10 mg) in primary prevention of CHD in hypertensive individuals with mildly elevated cholesterol levels. ASCOT-LLA enrolled 10 305 hypertensive individuals with total cholesterol <251.3 mg/dL. Of these participants, 19% were women. The median follow-up period was 3.3 years. Atorvastatin significantly reduced the primary endpoint by 36% (HR = 0.64, 95% CI = 0.50-0.83, P = .0005). The event rate was 100 and 154 for atorvastatin and placebo, respectively. In women, the unadjusted HR with atorvastatin was 1.1 (95% CI = 0.57-2.12, P = .77). The event rate was 19 (atorvastatin) and 17 (placebo). In contrast, in men, a highly significant benefit was noted with the use of statins in reduction of the primary endpoint (unadjusted HR = 0.59, 95% CI = 0.44-0.77, P = .0001). The primary endpoint event rates in men were 81 (atorvastatin) and 137 (placebo). No significant heterogeneity between sex was reported (see Table 3 for the outcome event rates in women).
Another study evaluated primary prevention of cardiovascular disease with a moderate-intensity statin (atorvastatin10 mg) in individuals with diabetes, the Collaborative Atorvastatin Diabetes Study (CARDS). 32 This study evaluated the efficacy of atorvastatin 10 mg, compared with placebo, for primary prevention of CVD in individuals with diabetes and LDL-C <160 mg/dL. CARDS enrolled 2838 participants (32% women). The median follow-up was 3.9 years. The event rate for major cardiovascular events was lower in the atorvastatin group (83) compared with the placebo group (127). Atorvastatin 10 mg led to a 37% significant reduction (HR = 0.63, 95% CI = 0.48-0.83, P = .001) in major cardiovascular events: acute coronary heart syndrome, stroke, or coronary revascularization procedure, from statin use in all participants (male and female). No significant heterogeneity in outcomes was reported for sex (P = .59).
The only trial that reported significant risk reduction in primary prevention of coronary events in women compared a high-intensity statin (rosuvastatin 20 mg) to placebo. 45 The Justification for the use of statins in prevention: An intervention trial evaluating rosuvastatin (JUPITER) trial 45 enrolled 6801 women and 11 001 men with elevated high-sensitive C-reactive protein (hs-CRP >2 mg/L), LDL-C < 130 mg/dL, and without CVD. The trial showed that rosuvastatin significantly reduced the primary endpoint, which was a composite of MI, stroke, arterial revascularization, hospitalization for unstable angina or cardiovascular death (HR = 0.56, 95% CI = 0.46-0.69, P < .00001). The event rates for the primary endpoint were 0.77 and 1.36 per 100 person-years for rosuvastatin and placebo, respectively. In women, JUPITER 36 showed that high-intensity statin (rosuvastatin 20 mg) significantly reduced the primary endpoint (HR = 0.54, 95% CI = 0.37-0.80, P = .002). No significant sex interaction for the primary outcome was noted (P for heterogeneity = .80). The primary endpoint event rate in women per 100 person-years was 39 in the rosuvastatin group compared with 70 in the placebo group. In women, the 5-year NNT to prevent the primary endpoint (MI, stroke, hospitalization for unstable angina, arterial revascularization, or cardiovascular death) was 36. 46 The 5-year NNT to prevent MI, stroke, or any death in women was 52.
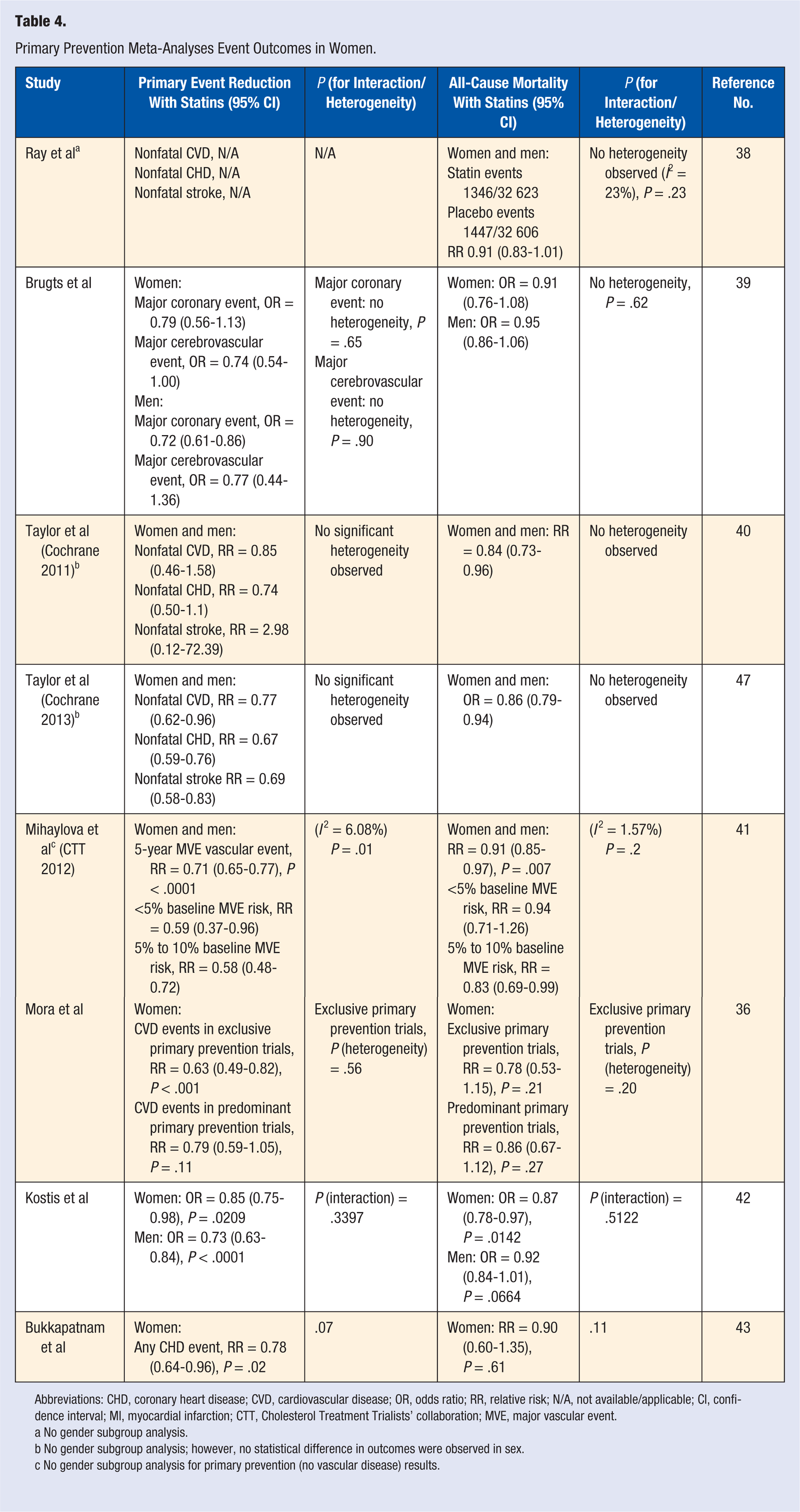
Most meta-analyses evaluating primary prevention with statins were not designed to report gender-specific outcomes.38-41 These non-gender-specific statin primary prevention meta-analyses showed a significant reduction in coronary events with statins. One of these meta-analyses, 39 evaluated 10 randomized controlled trials (RCTs), involving 70 388 men and women. About 6% participants had known CVD. This analysis showed that statin use for primary prevention led to a significant reduction in major coronary events (odds ratio [OR] = 0.70, 95% CI = 0.61-0.81), nonfatal MI (OR = 0.56, 95% CI = 0.41-0.76), major cerebrovascular events (OR = 0.81, 95% CI = 0.71-0.93), and revascularization (OR = 0.67, 95% CI = 0.59-0.76). The 2011 Cochrane meta-analysis 40 evaluated 14 RCTs involving 34 272 subjects with <10% of participants with CVD and a follow-up period of 1.0 to 5.3 years. This 2011 Cochrane analysis 40 showed 30% reduction in combined fatal and nonfatal CVD (RR = 0.70, 95% CI = 0.61-0.79) and 34% reduction in revascularization (RR = 0.66, 95% CI = 0.53-0.83) with statin use. The Cholesterol Treatment Trialists’ (CTT) collaboration performed a meta-analysis of 27 trials evaluating the effects of statin therapy in low-risk patients without vascular disease. 41 The CTT collaboration meta-analysis in patients without vascular disease 41 showed that statin use significantly reduced 5-year major vascular events (RR = 0.71, 95% CI = 0.65-0.77, P < .0001). In participants without prior history of vascular disease and with low 5-year baseline risk for major vascular events (CTT risk categories <5% and 5% to 10%), statin use also reduced incidence of major coronary events (RR = 0.59 [95% CI = 0.37-0.96] and RR = 0.58 [95% CI = 0.48-0.72], respectively). These results were also replicated in the most recent 2013 Cochrane meta-analysis, 47 which evaluated 18 primary prevention trials involving 56 934 participants with <10% of participants having CVD at baseline. Women comprised 39.7% of participants. The analysis showed statin use led to a 25% reduction in combined fatal and nonfatal CVD (RR = 0.75, 95% CI = 0.70-0.81) and 38% reduction in revascularization rates (RR = 0.62, 95% CI = 0.54-0.72). This 2013 Cochrane meta-analysis was published after we had conducted a systematic review of randomized placebo-controlled trials and meta-analyses published between 1990 and October 2012 in the PubMed database. Because of the significance of the results of this 2013 Cochrane meta-analysis, the results have been presented in our systematic review article.
In the 3 meta-analyses specifically designed to evaluate outcome effects in women, similar significant results were obtained. Kostis et al, 42 in a meta-analysis of primary prevention trials in women, showed significant reduction in cardiovascular events in women (OR = 0.85, 95% CI = 0.75-0.98, P = .0209). Bukkapatnam et al 43 performed a meta-analysis with data from 6 primary prevention RCTs involving 21 963 women (Table 4). One of the trials included in the meta-analysis was both a primary and secondary study: the Heart Protection Study. 37 The meta-analysis showed that primary prevention with statins significantly reduced CHD in women (RR = 0.78, 95% CI = 0.64-0.96, P = .02). Mora et al 36 performed a meta-analysis with data from the 3 exclusive primary prevention trials in women (MEGA, JUPITER, and AFCAPS/TexCAPS). The meta-analysis of the 3 exclusive primary prevention trials in women showed that statin use significantly reduced CVD in women by one third (RR = 0.63, 95% CI = 0.49-0.82, P < .001; P for heterogeneity = .56). When studies that were predominantly primary prevention studies (ALLHAT-LLT and ASCOT-LLA) were added to the analysis, the reduction in CVD remained but as a nonsignificant reduction (RR = 0.79, 95% CI = 0.59-1.05, P = .11; see Table 2 for meta-analyses characteristics).
Primary Prevention Meta-Analyses Event Outcomes in Women.
Abbreviations: CHD, coronary heart disease; CVD, cardiovascular disease; OR, odds ratio; RR, relative risk; N/A, not available/applicable; CI, confidence interval; MI, myocardial infarction; CTT, Cholesterol Treatment Trialists’ collaboration; MVE, major vascular event.
No gender subgroup analysis.
No gender subgroup analysis; however, no statistical difference in outcomes were observed in sex.
No gender subgroup analysis for primary prevention (no vascular disease) results.
Effects on Mortality
Most of the individual clinical trials30,32,34,36 showed a trend toward reduction in all-cause mortality. Two studies29,35 found no evidence of a reduction in all-cause mortality in women. In the MEGA study, 34 the 5-year all-cause mortality was significantly reduced with use of low-dose pravastatin (HR = 0.68, 95% CI = 0.46-1.00, P = .048). There were 66 deaths in the diet-alone group compared with 43 deaths in the pravastatin group. Gender data analysis showed that all-cause mortality in women was reduced by 41% in the diet plus pravastatin group compared with diet-alone group (HR = 0.59, 95% CI = 0.35-0.997, P = .046). 44 For women, the total mortality event rate was 39 in the diet group versus 22 in the diet plus pravastatin group. The reduction in mortality was primarily via reduced cancer mortality in the statin group. There was no difference between randomized groups in cardiovascular mortality. There was no significant heterogeneity between men and women (P = .43). In the ALLHAT study, 29 the mortality event rate was similar for both groups: 631 deaths in the pravastatin group versus 641 deaths in the usual care group. In women, pravastatin 40 mg use, compared with usual care, did not reduce all-cause mortality (RR = 0.98, 95% CI = 0.83-1.17) or a composite of CHD death and nonfatal MI (RR = 1.02, 95% CI = 0.81-1.28). The RR for mortality in both sexes was 0.99 (95% CI = 0.89-1.11). No significant heterogeneity was reported for sex. In the ASCOT-LLA trial, 30 all-cause mortality and cardiovascular mortality in men and women showed a nonsignificant reduction in the atorvastatin group (HR = 0.87, 95% CI = 0.71-1.06 P = .16) compared with placebo. In the atorvastatin group, there were 185 deaths compared with 212 deaths in the placebo group. No significant heterogeneity for sex was noted. In the CARDS study, 32 the all-cause mortality event rate was lower in the atorvastatin group (61) compared with the placebo group (82). Atorvastatin reduced all-cause mortality by 27% (HR = 0.73, 95% CI = 0.52-1.01, P = .059) overall. There was no significant heterogeneity of effect for sex (P = .59). In the JUPITER study, 45 all-cause mortality was significantly reduced with rosuvastatin (HR = 0.80, 95% CI = 0.67-0.97, P = .02). The event rates were 1.00 and 1.25 per 100 person-years of follow-up for rosuvastatin and placebo, respectively. JUPITER showed a trend toward reduction in all-cause mortality in women (HR = 0.77; 95% CI = 0.55-1.06, P = .12) with rosuvastatin 20 mg compared with placebo. 36 Similar results were obtained in men (P for heterogeneity = .74). These trials contrast with the results obtained from the AFCAPS/TexCAPS study, 35 which showed no reduction in the total mortality rate in women with lovastatin 20 to 40 mg compared with placebo (see Table 3 for the mortality event rates in women).
The meta-analyses demonstrated a reduction in mortality from statin use when women and men were analyzed together. One meta-analysis 39 showed that on exclusion of trials that involved subjects with known CVD, statin use led to a significant decrease in all-cause mortality (OR = 0.87, 95% CI = 0.78-0.97) over a mean follow-up of 4.1 years. The CTT collaboration meta-analysis evaluated the effects of statin therapy in low-risk patients without vascular disease. 41 This meta-analysis showed that statin use significantly reduced all-cause mortality (RR = 0.91, 95% CI = 0.85-0.97, P = .007) and vascular mortality (RR = 0.85, 95% CI = 0.77-0.95, P = .007) in all participants. All-cause mortality was also reduced in participants with 5% to <10% risk for major vascular events (RR = 0.83, 95% CI = 0.69-0.99). The most recent 2013 Cochrane meta-analysis 47 also conclusively demonstrated a reduction in total mortality in an analysis of 18 primary prevention trials. Statins reduced all-cause mortality by 14% (OR = 0.86, 95% CI = 0.79-0.94). The earlier Cochrane meta-analysis 40 evaluated 14 RCTs and found that statins led to a 17% reduction in all-cause mortality (RR = 0.83, 95% CI = 0.73-0.95) over a follow-up period of 1.0 to 5.3 years. In contrast, the meta-analysis by Ray et al 38 showed a nonsignificant reduction in total mortality, although the magnitude of the relative reduction in the risk of overall mortality was similar to that found in the other meta-analyses. This meta-analysis involved 11 RCTs comprising 65 229 men and women without known CVD. This study found that statin use led to a 7% to 9% nonsignificant risk reduction in all-cause mortality (RR = 0.91, 95% CI = 0.83-1.01) over an average of 3.7 years of therapy. No strong evidence of statistical heterogeneity in effect estimate across the studies was seen (I2 = 23%, 95% CI = 0% to 61%, P = .23).
Three meta-analyses were specifically designed to address the effects of primary prevention with statins in women.36,42,43 Mixed results were obtained, with 1 meta-analysis demonstrating significant reduction in all-cause mortality and the other 2 meta-analyses demonstrating nonsignificant reduction in mortality, although the magnitude of the effects was similar to that observed in the first meta-analysis. . The meta-analysis by Kostis et al 42 showed that statins significantly reduced all-cause mortality in women without CVD (OR = 0.87, 95% CI = 0.78-0.97, P = .0142). Mora et al 36 analyzed data from the 3 exclusive primary prevention trials in women (MEGA, JUPITER, and AFCAPS/TexCAPS). This meta-analysis showed that statin use nonsignificantly reduced total mortality in women (RR = 0.78, 95% CI = 0.53-1.15, P < .21). When studies that were predominantly primary prevention studies (ALLHAT-LLT and ASCOT-LLA) were added to the analysis, the nonsignificant reduction in CVD persisted (RR = 0.86, 95% CI = 0.67-1.12, P = .27). The third meta-analysis 43 evaluated 6 primary prevention RCTs involving 21 963 women. One of the trials included in the meta-analysis was both a primary and secondary study: the Heart Protection Study. 37 This meta-analysis showed that statin use did not significantly improve all-cause mortality (RR = 0.90, 95% CI = 0.60-1.35, P = .61) over a mean follow-up of 4.25 years.
Tolerability/Adverse Events
Incidence of severe adverse events, including cancer, was similar in the statin and nonstatin groups.30,32,35,36,44 Several meta-analyses of trials involving men and women showed no increase in cancer incidence or mortality with statin use.41,47-49 One randomized study 50 showed that a 6-month use of statins (simvastatin 20 mg or pravastatin 40 mg) for primary prevention in women led to reduced energy and increase in exertional fatigue compared with placebo (P = .01). This study enrolled 1016 participants (692 men and 324 women) without CVD or diabetes and with LDL-C levels of 115 to 190 mg/dL. However, it is important to note that exertional fatigue and change in energy levels were evaluated using the EnergyFatigEx score, which has not been previously validated. An association between statin use and development of diabetes mellitus (HR = 1.49, 95% CI = 1.11-2.01, P = .008) has been observed in both women and men. 36
Discussion
The reduction in CVD events with statins is directly proportional to the magnitude of LDL-C lowering, with each 38 mg/dL (1 mmol/L) reduction in LDL-C associated with a 22% reduction in CVD events. 48 Greater LDL-C reduction (32% to 50%) was obtained in trials that used moderate- and high-intensity statin compared with trials that used low- to moderate-intensity statins (see Table 1 for the LDL-C reduction in the different trials). The inability of ALLHAT-LLT 29 to demonstrate a significant reduction in CVD events and total mortality largely arose from the smaller than expected difference in total cholesterol reduction between the treatment arms (17.2% pravastatin vs 7.6% usual care total cholesterol reduction), as well as the nonblinded study design and high crossover rate between treatment arms. This smaller-than-expected cholesterol differential may have contributed to the ALLHAT-LLT study being underpowered to distinguish expected mortality reduction and the null hypothesis with statin use. The magnitude of LDL-C reduction may also explain the greater statistical significance in cardiovascular event reduction in the primary prevention trial JUPITER (47% reduction to mean LDL-C 55 mg/dL) compared with MEGA (20% reduction to LDL-C 123 mg/dL) and AFCAPS/TexCAPS trial (25% reduction to median LDL-C 116 mg/dL). 51
In AFCAPS/TexCAPS, 35 ASCOT-LLA, 30 MEGA study, 44 and JUPITER, 36 the incidence of coronary events was lower in women compared with that in men. It has been proposed that this low incidence of events in women may have contributed to the non–statistically significant results obtained in some of the clinical trials. However, the relative reduction in CVD events was similar for women and men in these studies. The MEGA study 44 showed that despite this low incidence of cardiovascular events, the risk for CHD was reduced by 25% in women in the statin group compared with the nonstatin group and was comparable to the 35% reduction seen in the men who received statins. The low incidence of events and the comparatively fewer number of women enrolled in these studies compared with the JUPITER may explain why these studies showed non-significant trends toward cardiovascular event reduction. When these trials were considered together in meta-analysis, 42 statins for primary prevention of CVD showed significant benefit in both mortality and morbidity in women. In future trials, it will be important to enroll sufficient numbers of higher risk women to detect a treatment effect, and ensure that women do not continue to be underrepresented in clinical research. 52
The results from the MEGA study 44 show that the efficacy of statins in primary prevention in women increases with patients’ age. Indeed, one explanation offered for the significant cardiovascular event benefit seen with women in JUPITER is that older women were enrolled (median age 68 years in JUPITER, mean age 62.5 years in AFCAPS/TexCAPS, and mean age 60 years in MEGA). 51 In ALLHAT-LLT, 29 results suggest that a trend of benefit was seen in patients with elevated LDL-C levels. However, the JUPITER trial showed benefit of statin use for primary prevention in women with LDL-C levels <130 mg/dL and elevated hs-CRP. Of note, in JUPITER, the mean Framingham score for women was <5%, a population that using the National Cholesterol Education Panel Adult Treatment Panel III (NCEP ATP III) guidelines would not merit statin therapy.
These studies suggest the importance of considering the risk conferred by all risk factors and not just LDL-C levels alone, to identify the right subgroup of patients to benefit from primary prevention with statins. Studies in women have shown that an increase in the number of traditional cardiac risk factors such as hypertension, diabetes, obesity and dyslipidemia, increases the mortality rate of CVD. 53 CARDS 32 results indicate that patients with diabetes, an additional CVD risk factor and baseline LDL-C levels greater than 70 mg/dL, also benefit from CVD primary prevention with statins. The metabolic syndrome has also been associated with increased risk for CVD in women. 54
Familial hypercholesterolemia (FH) refers to a group of inherited genetic defects resulting in severely elevated LDL-C. Studies have shown that all patients with familial hypercholesterolemia should be regarded as high risk for atherosclerosis and premature CHD. In women with FH, the mean age of onset of a cardiovascular event is the early 50s. 55 FH is associated with a >30% risk of premature CHD by age 60 in women.55,56 Smoking57,58 and total cholesterol/HDL-C level 57 are associated with premature CAD in patients with FH. One study 59 involving 2146 patients with FH and without clinical CVD showed that treatment with statins reduced CHD risk by about 76% over a mean 8.5 years. Another prospective study 60 involving 3382 patients with heterozygous FH showed that statin use in patients without CHD led to a 48% reduction in CHD mortality from 2-fold baseline CVD excess to none. Greater reduction in coronary mortality was noted in women compared to men. Current recommendations are that patients with FH should receive first-line therapy with high-potency statins to achieve a 50% reduction of baseline LDL-C levels.55,61,62 The use of the Framingham risk score has been discouraged in FH patients because of their inherent high risk for CAD.
Scoring systems for predicting the nearer term risk of CVD have been developed to identify the ideal treatment population. The current NCEP ATP III guidelines state that patients without CHD, but with diabetes, hypertension, and dyslipidemia, are at increased risk for CHD. 63 The Framingham risk score 64 is traditionally used to classify a person’s 10-year risk for developing CAD and to guide the ideal LDL-C level based on the risk strata. According to NCEP ATP III, drug therapy should be initiated for primary prevention in women with a Framingham 10-year risk of 10% to 20% and ≥2 CHD risk factors and LDL-C ≥130 mg/dL. Updated risk prediction equations are anticipated as part of the National Heart, Lung, and Blood Institute’s Cardiovascular Risk Reduction initiative.
Newer risk markers such as hs-CRP may add prognostic value to traditional risk factors for detecting cardiovascular events in women. 65 New risk factor scoring models have been developed. The Reynolds Risk Score 66 is a validated tool and includes hs-CRP, hemoglobin A1c, and family history of CAD in the scoring model. The Reynolds Risk Score has been shown to be a better risk predictor of CVD in women compared with the Framingham scoring system in some studies. 67 In 2007, the American Heart Association (AHA) provided a validated algorithm to stratify CVD risk in women. 68 This algorithm classified women as “at high risk (≥1 high-risk condition such as diabetes, CHD, stroke, or chronic kidney disease), at risk (≥1 major CVD risk factor such as hypertension, family history of premature CVD, metabolic syndrome, obesity, history of preeclampsia, or gestational diabetes) or at optimal risk (Framingham risk <10%, healthy lifestyle and no CVD risk factors)” for CVD. This classification was based on the high lifetime risk for CVD in almost all women, the limitations inherent in traditional risk scoring models and the fact that most clinical trials included women at high risk with known CVD or healthy women with some degree of risk. The panel recognized that while a high Framingham risk score (>20%) identified women at high risk for developing CVD, a low score did not effectively predict women at low lifetime CVD risk. The algorithm includes family history of CVD, medical history, and lifestyle history in the risk classification. This algorithm has been evaluated and shown to be valid and appropriately predict event rates for the different risk groups. 69 However, one shortcoming identified was the inability of this AHA algorithm to classify all women (13% uncategorized). Also, although the 2007 AHA algorithm CHD event risk prediction was similar for the Framingham categories of <10% and 10% to 20%, the AHA algorithm was less accurate than the modified Framingham categories of <5%, 5% to 20% and >20% (P < .0001). In 2011, this algorithm was updated 70 and is more reflective on an effective-based approach for management of CVD, rather than an evidence-based approach. The current AHA 2011 guideline reclassifies CVD risk in women as “high risk, at risk, or ideal cardiovascular health.” It should be noted the 2012 meta-analysis by the CTT study found that statin therapy substantially reduced CVD risk in individuals with ≥5% risk of a major CVD event (MI, stroke, CVD, death, or coronary revascularization). 41 Women with a family history of premature CVD may accrue greater benefit from primary prevention with statins (HR = 0.20, 95% CI = 0.06-0.69) than those without premature CVD family history (HR = 0.63; 95% CI = 0.41-0.96), P for interaction = .07. 36
Statins should never be used in pregnant women or breast-feeding women and are considered pregnancy category X. With regard to safety, no increased incidence of malignancy or memory loss has been demonstrated. Rates of serious muscle adverse effects (myopathy or rhabdomyolysis) and hemorrhagic stroke are very low (0.5 excess cases per 1000 individuals treated for 5 years for each or NNT to harm 2000). 41 However, concerns have been raised recently regarding an associated increased risk of diabetes with statin use. 71 A recent meta-analysis involving 13 statin trials, most of which were moderate intensity statin therapy showed that the excess risk of new diabetes was 5 per 1000 statin-treated persons over 5 years. 72 The 5-year number needed to harm (NNH) was 200. In a reevaluation of the JUPITER trial, which involved the use of rosuvastatin 20 mg, the excess risk of new diabetes was 15 per 1000 statin-treated persons over 5 years and 54 per 8901. 73 All cases of diabetes occurred in patients with impaired fasting glucose levels at baseline. The 5-year NNH was 66. The diagnosis of diabetes was on average 6 weeks earlier in the rosuvastatin group. The predictors for developing diabetes appear to be influenced by high baseline fasting glucose levels, metabolic syndrome profile 74 and older age. 72 This has led to the hypothesis that statin use may merely unmask patients who were likely to have developed diabetes irrespective of statin therapy.75,76 Despite concerns that use of statins are associated with an increased risk of diabetes, the risk–benefit ratio of statin use outweighs the possible risk of diabetes when the right population group for statin therapy is identified. For a given patient, the net benefit from statin therapy can be estimated by calculating the predicted 10-year absolute risk of CVD, applying the relative risk reduction from the intensity of statin therapy (about 30% for moderate-intensity statin therapy and 45% for high-intensity statin therapy), and then comparing the estimated reduction in absolute risk with the risk for the threshold for adverse events.
The evidence from the 2013 Cochrane meta-analysis 47 suggest that statins for primary prevention of CVD is both cost-effective and may improve quality of life without increased risk for adverse outcomes. Given the data from 2012 CTT study, 41 the 2013 Cochrane authors emphasize that their previously expressed caution regarding primary prevention with statins in low risk individuals especially women, was no longer valid and the evidence supports statin use in low-risk individuals. 47
Healthy lifestyle habits such as regular exercise, smoking cessation, healthy weight attainment, and diet rich in vegetables, fruit, and low salt should be emphasized as the recommended first-line primary prevention therapy for all women. 70 One barrier to adopting healthy lifestyle habits is the lack of awareness among health care providers and the general population that heart disease is the leading cause of death in women.77,78 The AHA has adopted the Go Red for Women campaign annually to increase awareness regarding heart disease in women. This campaign has had much success in improving awareness in women. However, a recent survey showed that the awareness that CVD is the leading cause of death in women is still needed among racial and ethnic minorities (African American, Hispanic, and Asian women).79,80 This knowledge gap should continue to be addressed at all levels in the community. We emphasize that given the limited evidence supporting primary prevention with statin in women at moderate to high risk, health care providers should not neglect the proven importance of patient and caregiver education regarding heart disease and lifestyle modifications.
Conclusion
In higher risk women and all women ≥ 20 years old with familial hypercholesterolemia, the evidence supports the use of statins for primary prevention. Higher risk women include those with diabetes and those with multiple risk factors. Risk can be estimated by the Reynolds Risk Score, the updated Framingham CVD risk profile, or the updated/modified AHA algorithm for risk assessment in women. Recent data suggest that statin therapy may benefit women with 5-year risk of major CVD (including coronary revascularization), which translates into about a 5% 10-year risk of CHD, stroke, and CVD death; however, the net benefit of statin therapy and patient preferences need to be considered. A combination of multiple risk factor scoring, genetic family history, age, and hs-CRP may be useful to consider when deciding to initiate statin therapy for primary prevention of CVD in women. Nonpharmacologic therapies such as heart-healthy diet, regular exercise, weight management, and lifestyle changes, including smoking cessation/avoidance remain the first line of therapy for primary prevention of cardiovascular disease in all women (AHA class I recommendation). 70 Clinical trials need to report sex-specific results and more women should be encouraged to enroll in clinical trials, to improve our knowledge of gender-specific outcomes in cardiovascular disease prevention.
