Abstract
With almost 30 million individuals predicted to be diagnosed by the year 2025, type 2 diabetes mellitus (T2DM) has become one of the most prevalent diseases in the United States. Because of the progressive dysfunction of the pancreatic β-cells and increasing insulin resistance over time, the need for treatments with different mechanisms or addition of medications to a regimen is becoming commonplace. Because of this, developing new medications to treat T2DM has been the focus of a lot of recent research and drug development. Molecular substrates such as glucagon-like peptide-1 (GLP-1), dipeptidyl peptidase-4 (DPP-4), and the sodium glucose transporter-2 (SGLT2) have all become new therapeutic targets. GLP-1 agonists and DPP-4 inhibitors are 2 of the newest classes of Food and Drug Administration–approved medications for diabetes. By increasing GLP-1 to supraphysiologic levels (GLP-1 agonists) and delaying endogenous GLP-1 degradation (DPP-4 inhibitors), these drugs increase insulin secretion and decrease glucagon production. SGLT2 inhibitors, the newest antihyperglycemic class, promote glycosuria by inhibiting sodium and glucose reabsorption in the proximal tubule of the renal nephron. Other novel agents for the treatment of diabetes include the use of the dopamine agonist bromocriptine, the cholesterol medication colesevelam, and a new form of inhaled insulin.
Keywords
‘[Type 2 diabetes mellitus] requires continuous patient self-management, medical staff support, and an array of medication therapies for treatment.’
Introduction
Type 2 diabetes mellitus (T2DM) is a complex, chronic metabolic illness that is characterized by insulin resistance and decreased pancreatic β-cell function, which may lead to poor glycemic control. This disease requires continuous patient self-management, medical staff support, and an array of medication therapies for treatment. T2DM accounts for 90% to 95% of all DM cases and is currently the seventh leading cause of death in the United States, largely because of complications, which are associated with poor glycemic control. 1
The World Health Organization has recently updated the prevalence of DM. It is estimated that by the year 2025, 30.3 million people in the United States will be diagnosed with DM with a total of 380 million people worldwide. 2 By the year 2050, projections show that 45.6 million Americans will be diagnosed with T2DM and another 17.2 million will go undiagnosed. 3
Type 2 diabetes mellitus is associated with and may be the result of a sedentary lifestyle, obesity, and lack of exercise in an aging population. It is typically diagnosed in patients older than 30 years with a positive family history of the disease and in those who are overweight or obese. However, insulin resistance may begin to develop many years before diagnosis. 4 Because of the increasing rates of adolescent obesity, T2DM is becoming more common in younger populations. Patients are clinically diagnosed with T2DM when glucose levels reach those of a diagnostic criterion (Table 1). These patients are at risk for potential complications, including damage to blood vessels causing microvascular (neuropathy, nephropathy, and retinopathy) and macrovascular (peripheral vascular disease, cerebrovascular disease, and cardiovascular disease) complications. Patients with controlled glycemic levels have shown delays in the progression of microvascular complications, with unclear benefits for macrovascular components.2,5 The United Kingdom Prospective Diabetes Study showed benefits of intensive glycemic control (fasting blood glucose less than 6 mmol/L [108 mg/dL]) in reducing all-cause death, including complications from diabetes.6,7 In addition to these complications, patients with T2DM often have a group of pathologies called metabolic syndrome, which increases risk of heart disease and other serious cardiovascular manifestations, including stroke and myocardial infarction.
Glucose Testing and Diagnostic Criteria of Type 2 Diabetes Mellitus. a
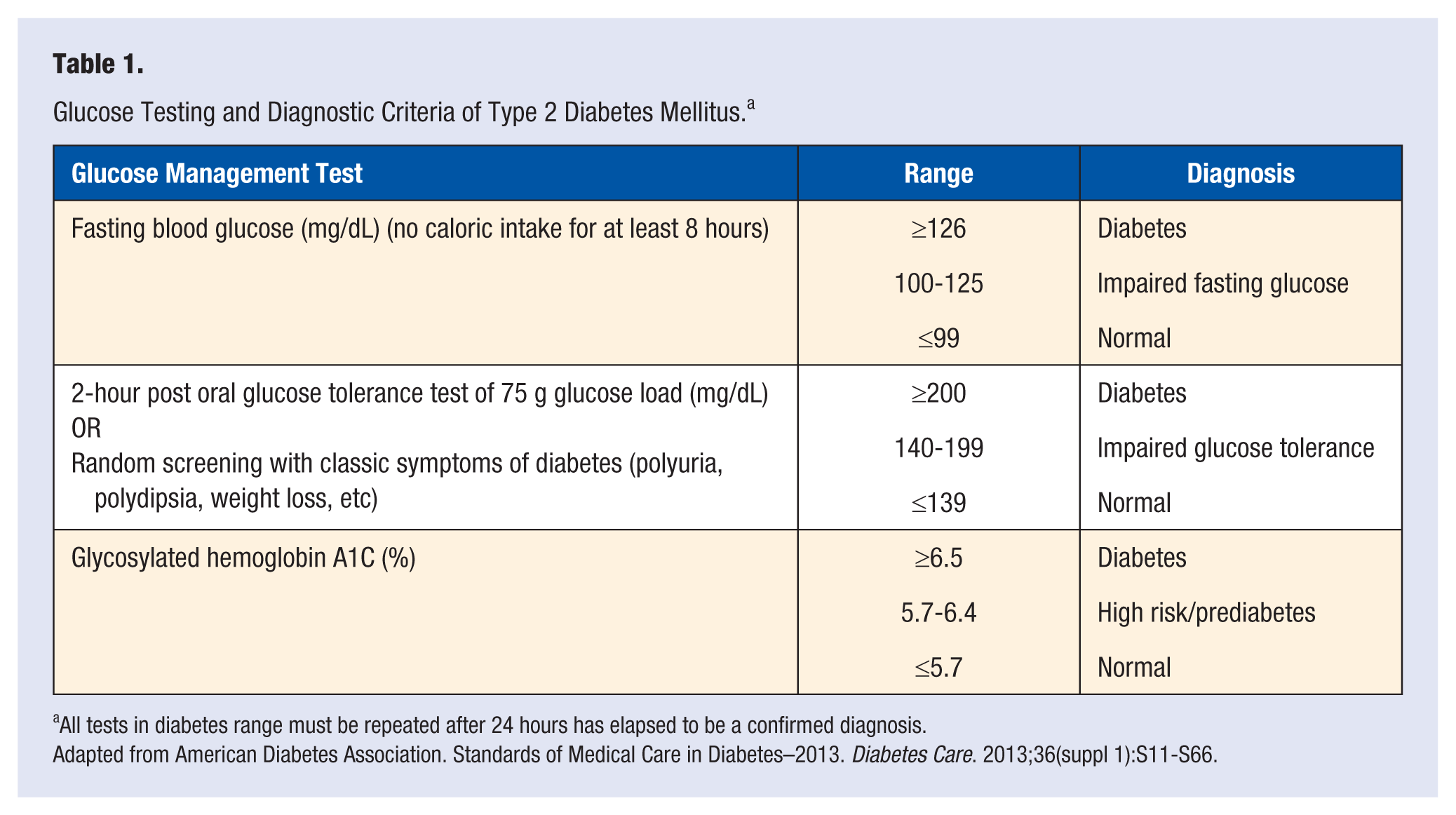
All tests in diabetes range must be repeated after 24 hours has elapsed to be a confirmed diagnosis.
Adapted from American Diabetes Association. Standards of Medical Care in Diabetes–2013. Diabetes Care. 2013;36(suppl 1):S11-S66.
Over the past few years, more pharmacologic therapies have been approved for treatment of T2DM. Currently, there are 12 classes of antihyperglycemic drugs approved for use in the United States. 1 They are insulin analogues, biguanides, sulfonylureas, meglitinides, thiazolidinediones (TZDs), dipeptidyl peptidase-4 (DPP-4) inhibitors, glucagon-like peptide-1 (GLP-1) receptor agonists, α-glucosidase inhibitors, sodium glucose transporter 2 (SGLT2) inhibitors, and amylin analogues. There are also nonconventional agents that improve glycemic control as well. These include Cycloset (bromocriptine) and Welchol (colesevelam). An inhaled form of insulin and multiple drugs from different classes are in the late stages of clinical trials.
The current standard of care for patients newly diagnosed with diabetes is blood glucose lowering medications paired with proper diet and exercise. With proper management of all contributing factors such as glucose, glycosylated hemoglobin A1C (A1C), and lifestyle modifications, disease progression and complications can be effectively reduced. This review discusses current treatment and the role of oral medications in T2DM with a focus on new medications and treatments currently in clinical trials or awaiting approval from the Food and Drug Administration (FDA).
Incretins
Incretins are molecules secreted in the stomach and intestine in response to oral food intake. About 50% or more of the insulin secretion during meals in healthy individuals results from the secretion of these peptides. 4 The 2 major incretin molecules are glucose-dependent insulinotopic peptide (GIP) and glucagon-like peptide-1 (GLP-1). GIP currently has no therapeutic value for the treatment of T2DM. GLP-1, a 30-amino acid signaling polypeptide, is rapidly secreted from the L-cells of the distal small intestine (ileum) after a meal and is responsible for limiting glucose elevations by stimulating insulin secretion, with better efficacy at higher glucose levels.4,8 In T2DM, the secretion of this peptide does not differ from that of healthy individuals. However, because of the disease process of diabetes, insulin secretion is substantially reduced making these peptides of particular use for treatment of T2DM. 8
Glucagon-like peptide-1 has multiple mechanisms that correspond to its ability to lower glucose levels. This peptide stimulates insulin production from the pancreatic β-cells and increases glucose usage in the adipose and muscle tissue. GLP-1 also suppresses appetite and slows gastric emptying to synergistically reduce food intake. It also decreases glucagon secretion, reduces glucose production, and may reduce apoptosis and promote proliferation of pancreatic β-cells. Several small studies also indicate that GLP-1 can increase cardiac chronotropic and inotropic activity.2,4,8
Glucagon-like peptide-1 induces insulin secretion by binding to the GLP-1 receptor on the pancreatic β-cell. This binding activates secondary messenger systems such as cyclic adenosine monophosphate (cAMP), protein kinase-A, and eventually voltage-gated calcium channels. As a result, an influx of calcium ions into the β-cell stimulates the release of insulin from the cell. 8
Incretins such as GLP-1 are quickly degraded by an enzyme called dipeptidyl peptidase-4 (DPP-4). DPP-4 cleaves active incretins on the N-terminus of their structure, at the alanine residue, resulting in a half-life of only 1 to 2 minutes for endogenous GLP-1. Thus, the clinical use of GLP-1 is based on the ability to develop a peptide that is resistant to the effects of DPP-4.
Glucagon-like peptide-1 analogues that are resistant to the effects of DPP-4 and are approved by the FDA include Byetta (exentatide) and Victoza (liraglutide). Recently, the FDA approved Bydureon LAR (exenatide once-weekly). Another method to increase the effects GLP-1 is to prevent its metabolism by DPP-4. Currently, the FDA has approved 4 DPP-4 inhibitors: Januvia (sitagliptin), Onglyza (saxagliptin), Tradjenta (linagliptin), and the most recent, Nesina (alogliptin).
Glucagon-Like Peptide-1 Agonists
Byetta (Twice-Daily Exenatide)
Amino acid substitutions in GLP-1 made synthetically can stabilize the molecule from metabolism by DPP-4. The first molecule found to be similar to that of endogenous human GLP-1 was exendin-4, a peptide from the saliva of the Gila monster (Heloderma suspectum). The first GLP-1 agonist approved by the FDA in the United States is a synthetic version of exendin-4, called exenatide. 8 Exenatide, a complete agonist to the GLP-1 receptor, was approved in 2005 for the treatment of refractory T2DM. Exenatide binds to and activates the GLP-1 receptor on pancreatic β-cells leading to an increased secretion of insulin and glucose-dependent synthesis of insulin. 8 It also suppresses the action of glucagon, suppresses appetite, and slows gastric emptying.
Exenatide is given subcutaneously twice a day, usually within 60 minutes of the morning and evening meals for optimal efficacy. It is initiated at 5 µg/dose during the first 4 weeks of usage to reduce gastrointestinal side effects, including nausea and vomiting. 9 Nausea occurred in about 44% of patients taking any dose of exenatide with metformin and/or a sulfonylurea compared with 18% on placebo in a 30-week clinical trial. 9 Forty percent of patients on TZDs with or without metformin using exenatide also experienced nausea. 9 If the patient can tolerate side effects of exenatide, the dose may be increased to 10 µg/dose after the 28-day period. Because nausea, vomiting, and other side effects are dose-dependent, patients may experience these more frequently while on the 10 µg dose. Other side effects of note for exenatide include injection site reactions such as pruritus and uticaria, rash, abdominal pain, diarrhea, constipation, dysgeusia, weight loss, altered renal function, and increased serum creatinine. Hypoglycemia with exenatide is unlikely unless the patient is on concurrent medications that are known to cause hypoglycemia, such as sulfonylureas.
A beneficial effect in patients treated with exenatide is the increased weight loss seen when on this medication. This can be a very important factor in choosing a regimen for a patient if controlling weight is a concern. A meta-analysis of 18 different studies reported an average weight loss of about 3 kg with losses ranging from 2.3 to 3.4 kg. When comparing weight differences for those treated with insulin instead of exenatide, the difference between weight gained on insulin and weight lost using exenatide was around 5 kg.10,11
Based on postmarketing reports, patients using exenatide may be at risk for pancreatitis that can potentially be fatal.9,12 Despite the warning, several meta-analysis have yet to discover a significant correlation between exenatide use and acute pancreatitis.13,14 This is extremely rare but exenatide should be stopped immediately if patients experience an extended episode of severe abdominal pain. If pancreatitis is confirmed, exenatide should not be rechallenged. 9 Another contraindication to exenatide is patients with a history of gastroparesis. Since exenatide slows gastric emptying, patients with gastroparesis may experience an exacerbation of symptoms, and thus exenatide should be avoided.
Exenatide is eliminated primarily through the kidneys with some small metabolism via proteolytic degradation. 9 Therefore, exenatide is not recommended in patients with renal insufficiency (creatinine clearance, CrCl <30 mL/min) or end-stage renal disease. 9 There is no dosage adjustment required for patients with moderate renal disease (CrCl 30-50 mL/min); however, caution should be used for increasing the dose from 5 to 10 µg. 9 The rapid elimination of exenatide gives a measurable concentration for about 10 hours in most patients with adequate renal function, requiring the medication to be administered twice a day.
Exenatide is clinically effective in lowering postprandial glucose, fasting glucose, and hemoglobin A1C values. Most clinical trials have found an average reduction in A1C to be between 0.4% and 0.9%. 15 These reductions depend on the concurrent medications and baseline A1C values. Patients being treated with metformin and/or sulfonylurea tend to be closer to 1% reduction in A1C, which is seen in most studies.10,15,16 Fasting plasma glucose reduction ranged from 17 to 24 mg/dL across multiple clinical trials. 9
Approximately 40% to 50% of patients exposed to exenatide developed anti-exenatide antibodies, which for a few, resulted in failure of therapy or required dosage and/or regimen adjustment. 15 For patients who have low antibody titers, dosage adjustment may not be necessary as patients with low titers have the same glycemic response as those with no antibody production. Thus, attenuated response to exenatide occurs in patients with high titers of antibodies, or roughly 3% to 4% of all patients treated with exenatide. 9
The cardiovascular side effect profile of exenatide also appears beneficial to patients with T2DM. The effect of weight loss may have some connection with improved outcomes in patients treated with exenatide as compared with non-incretin glucose control medications. Different mechanisms for the beneficial cardiovascular profile has been proposed, but to date, no trial has been conducted that showed improvements in cardiovascular events. 17 Meta-analyses conducted show patients treated with exenatide are less likely to have a cardiovascular event than non-exenatide patients (hazard ratio [HR] = 0.81; P = .001) and also have lower rates of cardiovascular-related hospitalizations (HR = 0.88; P = .02) and all-cause hospitalizations (HR = 0.94; P < .001). 18 A meta-analysis examined decreases in systolic blood pressure associated with exenatide use and found a significant decrease of 2.8 mm Hg (P = .0002) versus placebo. 19 There was no evidence of change in diastolic blood pressure. 19 Heart rate was also affected slightly, ranging from an increase of 2 to 4 beats per minute. Confirmation studies are ongoing to explain this increase in heat rate, but it is thought that GLP-1 receptors in heart tissue have a role. It seems that there are other potential benefits on lipids as well. In an analysis of the clinical trials associated with exenatide versus insulin glargine, it was noted that patients treated with exenatide had significant reductions in postprandial triglycerides, free fatty acids, and high-density lipoprotein. There was no significant difference between the 2 groups for low-density lipoprotein (LDL), total cholesterol, Apo-A1, and other cholesterol components. 17
Bydureon (Once-Weekly Exenatide)
Recently, researchers have developed a new formulation of exenatide to improve patient compliance and ultimately improve glycemic control. Bydureon (exenatide once-weekly; exenatide LAR), approved by the FDA in January 2012, is a long-acting formulation of exenatide that allows for once weekly dosing. To allow for extended interval dosing, researchers formulated exenatide encapsulated in a 0.06 mm diameter microsphere (roughly the diameter of a human hair) composed of poly-(
The slow approach to steady state may improve tolerability of the drug. Nausea and vomiting are less frequent with once weekly dosing as compared to twice a day dosing of exenatide, as seen in Table 2.22-29 However, there is a more frequent mild inflammatory response to the microspheres, which may result in small nodules after injection, seen in up to about 80% of patients who used once weekly exenatide. 20 Other side effects that are seen with once-weekly exenatide are similar to those seen in the twice-daily formulation and include headache, diarrhea, and possible hypoglycemia, especially if taken concomitantly with a sulfonylurea.
Summary of the Diabetes Therapy Utilization: Researching Changes in A1C, Weight and Other Factors Through Intervention With Exenatide Once Weekly (DURATION) Studies.
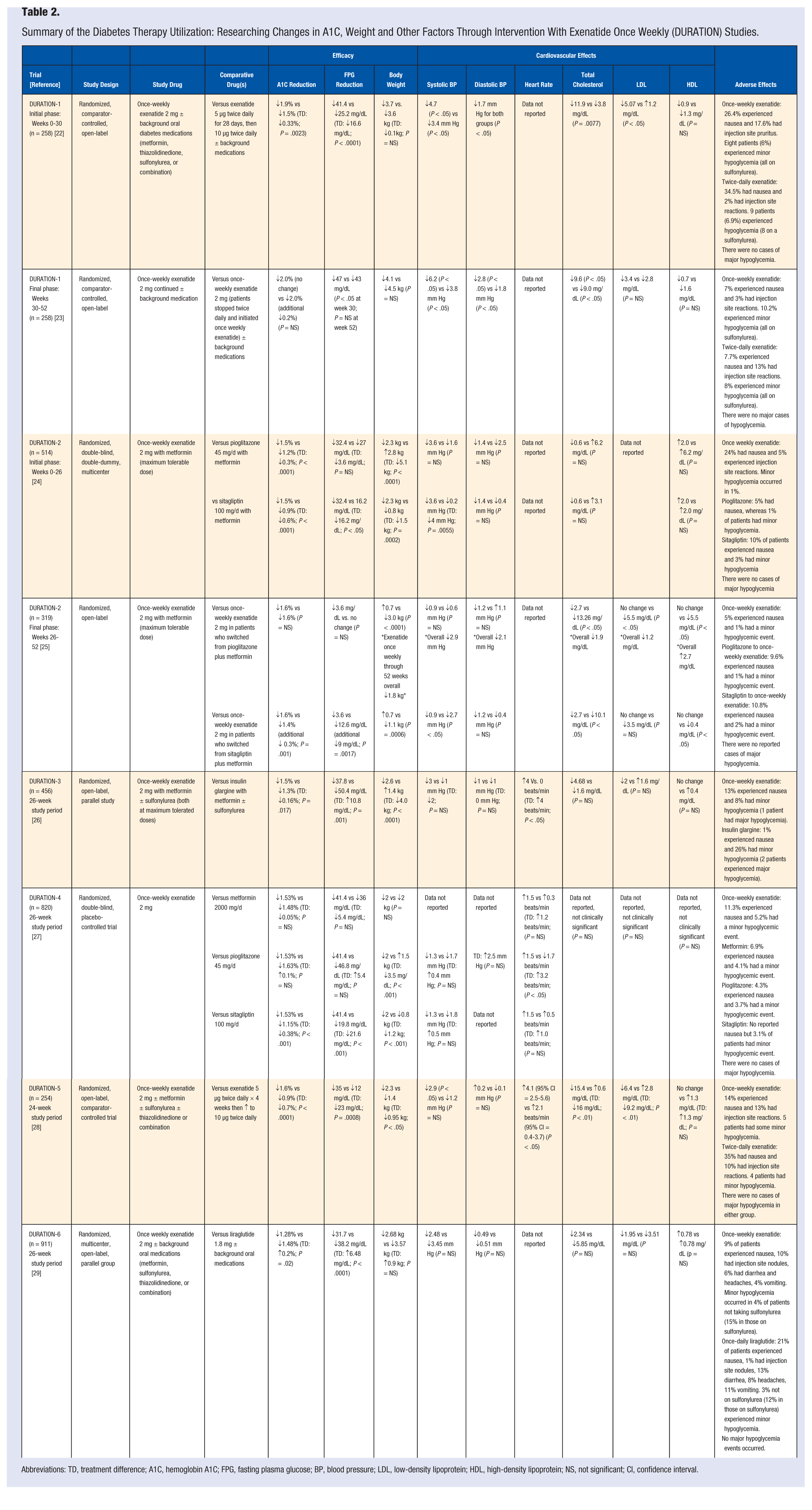
Abbreviations: TD, treatment difference; A1C, hemoglobin A1C; FPG, fasting plasma glucose; BP, blood pressure; LDL, low-density lipoprotein; HDL, high-density lipoprotein; NS, not significant; CI, confidence interval.
Once-weekly exenatide is also associated with reports of pancreatitis and the use of it is not recommended in patients with a history of pancreatitis, although this is rare. Patients who experience severe abdominal pain lasting several days should stop using once-weekly exenatide. Although not studied in once-weekly exenatide, gastroparesis can be exacerbated and thus it should not be used in patients with a history of gastroparesis. Recently, there has been a concern of thyroid cancer risk based on rat models that have not been extrapolated in humans. GLP-1 activates GLP-1 receptors on thyroid chief cells (C-cells) in mice, causing release of serum calcitonin. In rat models, this caused proliferation into C-cell adenomas and C-cell carcinomas. Until the risk has been clarified, once-weekly exenatide should not be used in patients with medullary thyroid carcinoma or hereditary C-cell hyperplasia or in individuals with first-degree family members who have these diseases because of the hereditary risk associated with them.8,30 For this reason, the FDA has issued a black box warning for exenatide once-weekly warning against thyroid C-cell tumors.
Table 2 summarizes the Diabetes Therapy Utilization: Researching Changes in A1C, Weight and Other Factors Through Intervention with Exenatide Once Weekly (DURATION) clinical trials, which showed Bydureon is an effective agent for lowering A1C and blood glucose levels. Bydureon was superior to twice-daily exenatide and DPP-4 inhibitors (specifically sitagliptin), noninferior to metformin and sulfonylureas, and inferior to once-daily liraglutide in regards to A1C-lowering ability.22-25,27-29 Bydureon was slightly more effective in reduction of A1C when compared to insulin glargine. With an intensive insulin protocol in place to achieve fasting blood glucose levels of less than 100 mg/dL, Bydureon lowered A1C 0.2% more (P = .017). 26 However, insulin protocols were not strictly monitored and poor patient adherence to this regimen could affect outcomes.
Once-weekly exenatide can reduce A1C 1.3% to 1.9%, depending on baseline A1C values and concomitant medications. In addition to reduction of A1C, once-weekly exenatide reduces fasting plasma glucose by 30 to 47 mg/dL while also decreasing body weight by an average of 2 to 4 kg. There are minor cardiovascular effects associated with the use of once-weekly exenatide. This includes minor lowering of the systolic blood pressure, although statistical significance is not associated with this effect in some trials, and improvement in fasting cholesterol levels. Significant reductions in total cholesterol and LDL are associated with exenatide; however, long-term data associated with these findings are not available.22,25 The Exenatide Study of Cardiovascular Event Lowering Trial (EXSCEL) is examining the benefit of once-weekly exenatide on major cardiovascular events. Specifically, the researchers will investigate the benefit of adding once weekly exenatide to a treatment regimen versus placebo on their primary outcome of cardiovascular-related death. Secondary outcomes will include nonfatal myocardial infarction and nonfatal stroke. However, with an estimated completion date of March 2017, results will not be known for some time.
Since exenatide is an exogenous product that is injected into the body, antibodies did develop in some patients during clinical trials. Antibody titers were higher with once weekly than in twice-daily exenatide but levels decreased over time in most patients. Patients with higher titers were associated with smaller reductions in A1C, and also had more injection site reactions such as itching and redness. 23 Thus, many patients who are not seeing clinical improvement in A1C and blood glucose values may have formed high titers to exenatide, and will require an alternate regimen for their diabetes treatment.
Overall, low withdrawal rates, a favorable tolerability profile with less nausea and vomiting, and rare events of hypoglycemia allow Bydureon to be an effective agent for the treatment of T2DM. This drug is a good choice for patients looking for easier administration compared to daily injections and for those individuals who have problems with compliance. A trial of twice daily exenatide is not needed prior to initiating once-weekly exenatide, which allows this medication to be used primarily as an add-on agent for combination therapy treatment of T2DM.
Victoza (Liraglutide)
Victoza (Liraglutide), approved in January 2010, was the second GLP-1 agonist approved for use in the United States. Liraglutide contains a fatty acid chain that facilitates its binding to albumin, leading to a half-life of approximately 13 hours, with active drug still in circulation up to 24 hours after injection. 31 The long half-life allows liraglutide to be injected subcutaneously once a day and because of the slow release from albumin, the maximum plasma concentration occurs 9 to 14 hours after injection. 8
Liraglutide is initiated at a dose of 0.6 mg injected subcutaneously once daily into the abdomen, thigh, or upper arm. Liraglutide can be given at any time of day, independent of meals. As a starting dose, 0.6 mg of liraglutide a day is not clinically effective in lowering blood glucose and this dose is used to decrease the occurrence of gastrointestinal side effects of nausea and vomiting before administering a higher dose. The dose is then titrated after 1 week to 1.2 mg daily. Additional titration to 1.8 mg daily should be at the discretion of the clinician. A1C should be rechecked in 3 months to determine if the 1.2 mg dose is sufficient or if the patient requires a higher dose of 1.8 mg daily to achieve their goal. However, some patients may not tolerate the higher 1.8 mg dose. Doses of 1.2 mg a day are clinically effective in A1C and blood glucose reductions, and thus this is an alternative for patients who become intolerant to the higher dose. 32 Liraglutide does not require dose adjustment in renal dysfunction, but caution is advised in patients with severe renal impairment (CrCl < 30 mL/min). 32
Liraglutide, as shown in the Liraglutide Effect and Action in Diabetes (LEAD) trials (Table 3),33-39 is superior to exenatide at reducing A1C, fasting blood glucose, and body weight. Not only can it be more efficacious versus a comparable GLP-1 agonist, it is also significantly more effective than some oral antihyperglycemic medications, such as sitagliptin. Liraglutide can usually decrease A1C 1% with many patients seeing a reduction of up to 1.5%, a percentage that few other medications can reach in optimal circumstances. To determine the potential beneficial long-term effects liraglutide could provide, clinical trials with a long follow-up period need to occur. Currently, a postmarketing, long-term (5 years), randomized, placebo-controlled clinical trial is evaluating liraglutide’s effects on cardiovascular outcomes. The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results trial is monitoring the time from which a patient is enrolled in the trial and randomized until the patient experiences death due to a cardiovascular event or nonfatal events such as myocardial infarction and stroke. This is currently the only ongoing trial that assess long-term safety and cardiovascular benefit of liraglutide. 8
Summary of the Liraglutide Effect and Action in Diabetes (LEAD) Trials Plus Head-to-Head With DPP-4.
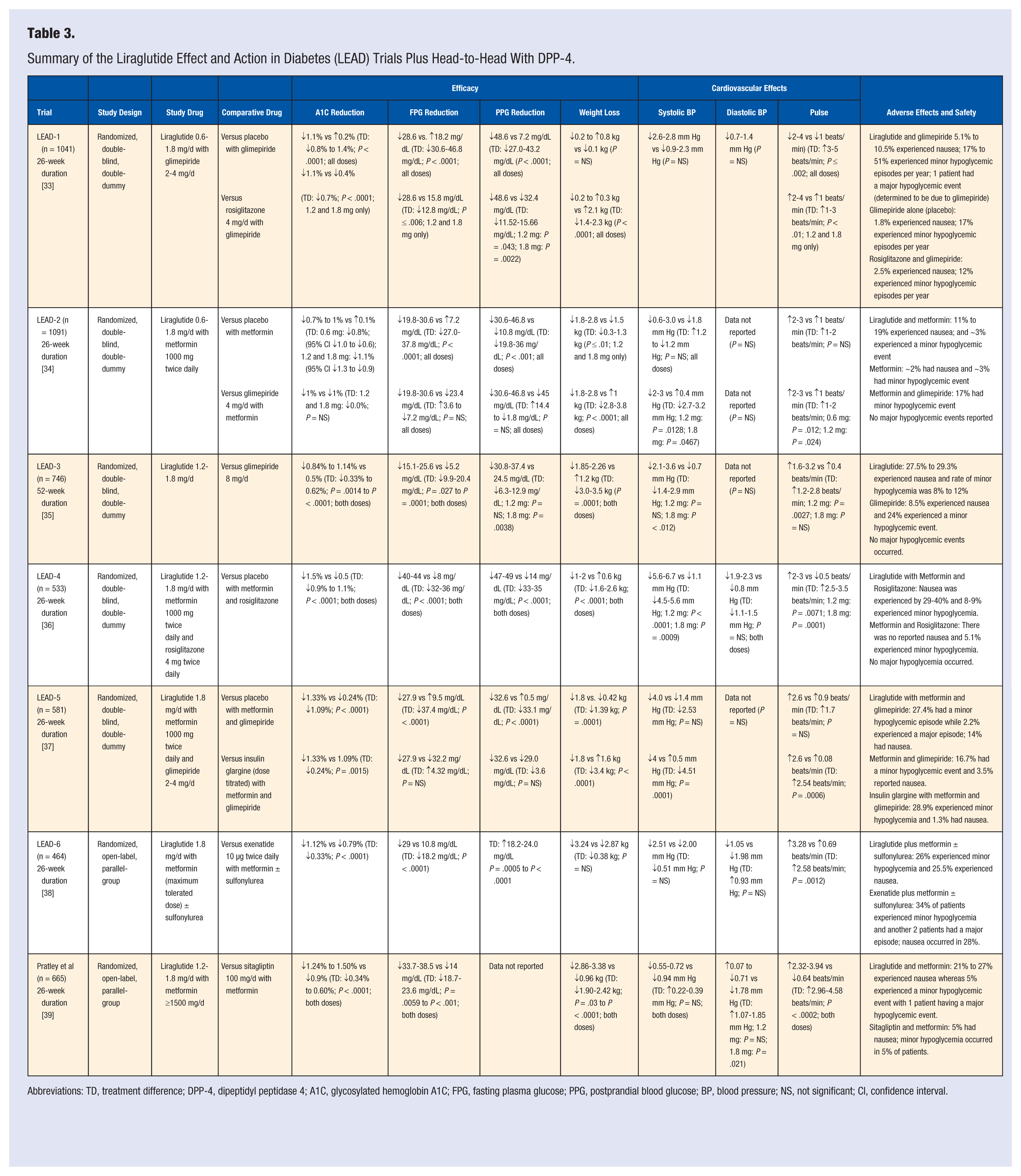
Abbreviations: TD, treatment difference; DPP-4, dipeptidyl peptidase 4; A1C, glycosylated hemoglobin A1C; FPG, fasting plasma glucose; PPG, postprandial blood glucose; BP, blood pressure; NS, not significant; CI, confidence interval.
Nausea was the most common adverse effect in patients treated with liraglutide, with roughly 6% to 41% of patients reporting it, as seen in Table 3. This was most frequently noted within the first 3 days of treatment and was transient, or dissipating over time. Other side effects reported included headache, vomiting, diarrhea, and constipation. Acute pancreatitis was also reported (<0.2%) and concordant to the exenatide recommendation, liraglutide should not be used in patients with a history of pancreatitis or at high risk for pancreatitis. 31 The risk of pancreatitis associated with liraglutide use is extremely low and patients should not be rechallenged with liraglutide if there is confirmed pancreatitis. Liraglutide should be immediately stopped after an extended period of time consisting of severe stomach and abdominal pain. As with exenatide, hypoglycemia with liraglutide is rare, unless combined with other medications, such as a sulfonylurea, which are prone to this effect. Liraglutide also has a black box warning for risk of medullary thyroid carcinoma and hereditary C-cell hyperplasia. Although it has not been shown in humans, patients with a history of these thyroid conditions or have a first-degree family member who has these diseases should not be started on liraglutide. Liraglutide should also be avoided in gastroparesis, as exacerbation of symptoms could occur.
Antibody titers of anti-liraglutide antibodies were reported in 8.6% to 13% of patients.8,33,37 When compared with exenatide, 61% of patients in the exenatide comparator arm had anti-exenatide antibodies after 26 weeks. 38 After the 26-week trial, patients on exenatide were switched to liraglutide, and reported an additional 0.4 % reduction in A1C. 40 This reduction shows that anti-exenatide antibodies do not affect A1C response to liraglutide, and thus patients who have inadequate response to exenatide may have a better response with liraglutide. Overall, because liraglutide is 97% homologous to endogenous GLP-1, it is less immunogenic compared with exenatide (53% homologous), and patients are at lower risk for treatment failure because of their immune system.
Summary
As a class, GLP-1 agonists are beneficial agents to treat type 2 diabetes because of their A1C, FPG, and PPG lowering effect. Currently, only exenatide twice daily is approved for monotherapy but all 3 are approved for combination therapy in refractory T2DM. A common side effect is nausea in up to half of patients who are started on GLP-1 agonists; however, this and other gastrointestinal side effects seem to be transient. Gradual titration to therapeutic doses is recommended to help decrease incidence of adverse effects and help to improve tolerability. Use of GLP-1 agonists in patients with history of pancreatitis and gastroparesis should be avoided. Bydureon and Victoza have labeling reporting risk of thyroid C-cell tumors and thus they should be avoided in patients with a history of thyroid C-cell cancers or patients with first-degree relatives with a positive history. Byetta currently does not have labeling on this risk, but it should be used with caution in these patients.
On average, these medications can reduce A1C anywhere from 1.0% to 1.5%, with liraglutide showing superiority over both exenatide formulations. They also have beneficial effects on decreasing weight, decreasing blood pressure, positive effects on lipid levels, and possible effects in decreasing β-cell apoptosis, which has yet to be proven with these agents. Because of the different pharmacokinetic profiles of these 3 medications, therapy should be tailored to specific patient needs. Patients in need of additional fasting blood glucose reductions may see more benefit with Bydureon and Victoza. On the other hand, patients needing postprandial blood glucose control may see better effects with Byetta. These effects should be weighed against the approximate cost of treatment, which is shown in Table 4.
Cost Comparison of Glucagon-Like Peptide-1 Agonists.
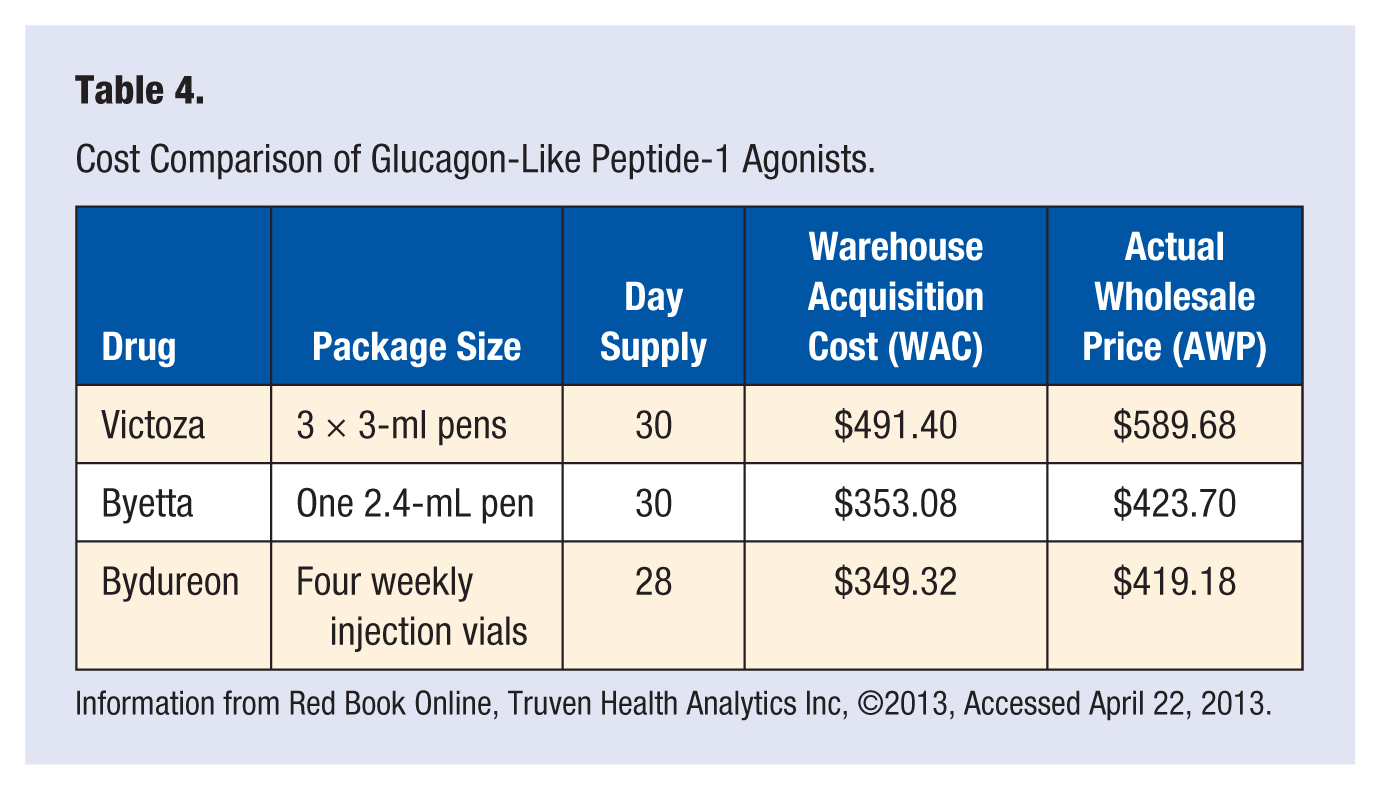
Information from Red Book Online, Truven Health Analytics Inc, ©2013, Accessed April 22, 2013.
Currently, there are three other GLP-1 agonist agents being developed. Albiglutide is a dimer of GLP-1 and human albumin, dulaglutide is a GLP-1 peptide, which is combined to immunoglobulin G4 Fc fragment, and similar to liraglutide, CJC-1134-PC is exendin-4 covalently inked to recombinant human albumin. 20
Dipeptidyl Peptidase-4 Inhibitors
While focusing on the possibilities the incretin effect has in type 2 diabetes with specific GLP-1 agonists, an alternative mechanism has appeared: blocking the degradation of GLP-1 via the DPP-4 enzyme. As previously discussed, DPP-4 is an enzyme in the body that rapidly degrades active GLP-1 into inactive metabolites, thus making it ineffective in stimulating insulin secretion and suppressing glucagon secretion. Unlike GLP-1 agonists, the DPP-4 inhibitors primary mechanism of action is stimulating insulin secretion and suppression the action of glucagon. They have little effect on satiety and gastric emptying. Since 2006, the FDA has approved 4 different DPP-4 inhibitors for the treatment of type 2 diabetes. The first was Januvia (sitagliptin), followed by Onglyza (saxagliptin), Tradjenta (linagliptin), and most recently, Nesina (alogliptin).
Dipeptidyl peptidase-4 inhibitors are usually well tolerated with a minimal adverse effect profile. The most common adverse events reported in clinical trials was nasopharyngitis (5.2% to 6.3%), upper respiratory tract infection (4.5% to 6.2%), headache (1.1% to 5.9%), and rare cases of hypoglycemia. 41 Hypoglycemia alone while on a DPP-4 inhibitor is very rare, unless the patient is on a sulfonylurea as well. DPP-4 inhibitors also carry the same pancreatitis risk associated with GLP-1 agonists, and thus should be used cautiously in patients who have suspected pancreatitis, and not at all in confirmed cases. DPP-4 inhibitors are given orally, an aspect that could potentially provide increased compliance over the injectable GLP-1 agonists. Unlike the GLP-1 agonists, DPP-4 inhibitors are generally weight neutral. However, it is not uncommon to see weight loss or gain in certain patients.
Januvia (Sitagliptin)
Dipeptidyl peptidase-4 inhibitors have moderate efficacy in reducing A1C, depending on concurrent therapies and length of treatments. Sitagliptin, the oldest DPP-4 inhibitor on the market in the United States, has the potential to reduce A1C 0.6% to 1.0% when added to metformin (P < .001). 42 When sitagliptin and metformin therapy were initiated at the same time, A1C reductions peaked at over 2.0% and allowed 66% of patients to reach their A1C goal of <7% versus 44% on placebo with metformin (P < .001). 43 Metformin alone decreased A1C 1.3% meaning sitagliptin contributed to anther 0.7% reduction in A1C (P < .001). 43 When started on triple combination therapy with metformin and glimepiride, sitagliptin can decrease A1C an additional 0.89% versus placebo (P < .001). 44 When compared with a sulfonylurea (glipizide) as an add-on agent for patients being treated with metformin, both groups had A1C reductions of about 0.7% (P = not significant) confirming noninferiority of the 2 agents. 45 Where these 2 groups did differ was in secondary characteristics. Sitagliptin had a similar fasting plasma glucose reduction (10.0 vs 7.5 mg/dL; P = not significant) compared with glipizide, but weight differences of 2.5 kg between the sitagliptin and glipizide groups (−1.5 vs +1.1 kg; P < .001) 45 were significant. The risk of hypoglycemia rises when patients are also taking a sulfonylurea with sitagliptin (vs placebo; 12.2% vs 1.8%; P < .001) 44 and insulin therapy with sitagliptin versus insulin and placebo (15.5% vs 7.8%). 41 Hypoglycemia with glipizide reached 32% in one study versus 5% for those on sitagliptin (P < .001). 45
Sitagliptin is primarily excreted via the kidney as both active metabolite (79%) and inactive metabolites. This medication requires dosage adjustment for patients with renal insufficiency. The normal dosage of 100 mg, taken once daily without regard to meals, applies for individuals with a CrCl >50 mL/min. 41 Patients with moderate renal disease who have a CrCl of 30 to 50 mL/min should receive a dose of 50 mg once daily. 41 Patients with CrCl <30 mL/min or end-stage renal disease should receive 25 mg once daily. 41 Studies have shown that despite renal dysfunction, sitagliptin is equally effective compared to individuals with normal renal function if dosed properly. 46
Onglyza (Saxagliptin)
Saxagliptin, the second approved DPP-4 inhibitor, shares common characteristics with sitagliptin. Saxagliptin, when added to metformin monotherapy, has A1C reductions averaging 0.7% (P < .0001). 47 The same 0.7% A1C reduction was seen when saxagliptin was added onto sulfonylurea monotherapy (P < .0001). 48 When added to a TZD, saxagliptin reduced A1C an additional 0.6% versus placebo (P < .0001). 49 Other trials report A1C reductions totaling 2.3% when saxagliptin and metformin therapy were initiated at the same time, to which saxagliptin added 0.5% to 1.5% versus placebo (P < .0001). 50 Saxagliptin reduced fasting plasma glucose by 10 to 23 mg/dL (P < .05).48,49 Postprandial glucose reductions can be even larger, ranging from 40 to 50 mg/dL (P < .05).47,49 Saxagliptin was not associated with any significant changes in weight.
Available in 2 dosage strengths, 2.5 mg and 5 mg, saxagliptin is taken once a day without regard to meals. Because of extensive renal elimination of both the unchanged drug and active metabolite, saxagliptin should be dose reduced to 2.5 mg in patients with moderate to end-stage renal dysfunction (CrCl <50 mL/min). 51 Since saxagliptin undergoes metabolism through CYP450 3A4/5 to an active metabolite, patients who are on concomitant medications that inhibit CYP450 3A4, such as ketoconazole, should receive a dose of 2.5 mg daily. 51
Tradjenta (Linagliptin)
Linagliptin is the third DPP-4 inhibitor approved for use by the FDA. Unlike the previous 2, linagliptin is not extensively eliminated by the kidneys, and thus does not require any renal dose adjustment. The dose of linagliptin is 5 mg taken once daily without regard to meals. Linagliptin may have some minor metabolism through CYP450 3A4, and thus combination with a strong 3A4 inhibitor, such as rifampin, should be avoided. 52
Linagliptin was shown to be effective in reducing A1C, postprandial plasma glucose, and some minor effects in lowering fasting blood glucose. When added to metformin, linagliptin reduced A1C an additional 0.5% (P < .0001). 53 This same 0.5% reduction in A1C was seen when added onto a sulfonylurea (P < .0001) 54 and when used in combination with a TZD (P < .0001). 55 In patients already on metformin and a sulfonylurea, addition of linagliptin significantly reduced A1C an additional 0.62% (P < .0001). 56 Postprandial glucose was significantly decreased in multiple trials, with average results ranging in reductions from 58 to 67 mg/dL (P < .0001) depending on concomitant medication use.52,53 Minor but significant reductions in fasting plasma glucose were observed. Average reductions of fasting plasma glucose ranged from 14 to 20 mg/dL (P < .001).53,55 Body weight did not differ significantly between linagliptin and placebo groups.
Nesina (Alogliptin)
Although alogliptin was the first DPP-4 inhibitor to submit a new drug application to the FDA, it is the fourth of its kind to be approved for use in the United States. Expected to be available in the summer of 2013, alogliptin provides another choice to a rapidly expanding class of medications. Alogliptin is a potent, highly selective DPP-4 inhibitor, with greater than 10 000-fold selectivity for the DPP-4 isozyme over the other DPP isozymes. 57
Alogliptin has been studied extensively in multiple large, randomized, placebo-controlled trials supporting its efficacy for treatment of T2DM. Alogliptin was evaluated as monotherapy in patient’s naïve to treatment, which resulted in a 0.6% A1C reduction compared with placebo (P < .001). 58 Fasting blood sugar was also reduced 22 to 28 mg/dL compared with placebo (P < .001). 58 When added onto a sulfonylurea, alogliptin provided an additional 0.4% to 0.5% reduction in A1C (P < .001). 59 This reduction was thought to be because of postprandial glucose improvements since there was no significant change in fasting plasma glucose. When combined with a stable metformin regimen of ≥1500 mg, alogliptin contributed further reduction of A1C by 0.6% (P < .001). 60 Fasting blood glucose was also reduced 17 mg/dL (P < .001). 60 The largest reduction seen with alogliptin was when it is used with pioglitazone. Initial treatment with combination pioglitazone and alogliptin resulted in an A1C reduction of 1.7%, compared with pioglitazone monotherapy, 1.15%, which resulted in a reduction of 0.56% due to alogliptin (P < .05). 61 However, when added onto therapy in patients already receiving pioglitazone with or without metformin and sulfonylurea, alogliptin reduced A1C by an additional 0.7% to 0.8% (P < .001). 62 Alogliptin combined to patients with insulin alone or with metformin resulted in an additional 0.5% to 0.6% reduction in A1C (P < .001). 63
During clinical trials, similar adverse events seen were compared to other DPP-4 inhibitors. However, skin-related adverse events were noted to be higher in the alogliptin-treated groups (<5%). 57 These reactions not only included primarily pruritus but also included dry skin, rash, and eczema. Patients treated with alogliptin saw increased aminotransferase levels, and there have been postmarketing reports of fatal and nonfatal hepatic failure. 64 However, direct connection and cause related to alogliptin use has not been established. It is recommended to obtain liver function tests prior to starting alogliptin. In patients with abnormal results, alogliptin should be started with caution. 64 There is currently a long-term cardiovascular safety study occurring with alogliptin. The EXAMINE trial is looking at long-term cardiovascular safety from time of randomization to event, defined as cardiovascular related death, nonfatal myocardial infarction, or nonfatal stroke.
Alogliptin is dosed at 25 mg taken once daily without regard to meals. Since alogliptin is excreted primarily through the kidneys, patients with moderate renal impairment (30 to <60 mL/min) should take 12.5 mg daily. 64 Patients with severe renal impairment (<30 mL/min) or in end-stage renal disease should receive 6.25 mg daily. 64 Administration of alogliptin can happen without regard to dialysis schedule. 64
Summary
Overall, all 4 of the DPP-4 inhibitors seem to have similar reductions in A1C. These agents can also reduce fasting plasma glucose by 10.0 to 28.0 mg/dL (P < .05).48,49,53,58 Postprandial glucose reductions can be even larger, from 40 to 50 mg/dL (P < .05).47,49 Weight fluctuation between these 4 agents was minimal, with most trials reporting no weight changes; however, some reported weight loss up to 1.5 kg. Hypoglycemia between these agents was rare, with only a few reports occurring with concomitant treatment of a sulfonylurea, an agent known to cause hypoglycemia.
Differences in selection should be based on other parameters such as renal and liver function. Sitagliptin, saxagliptin, and alogliptin must be dose reduced with moderate to severe renal dysfunction. Studies have shown that despite renal dysfunction and the dosage adjustment, sitagliptin is as effective as described if dosed correctly. 46 Linagliptin is different than these three in the aspect that it undergoes minor metabolism through the liver and is primarily excreted through the enterohepatic system. Therefore, no dose reduction is needed in patients with renal dysfunction. Cost of these medications is shown in Table 5, as many times cost may also play a role in choosing a DPP-4 inhibitor.
Cost Comparison of Dipeptidyl Peptidase-4 Inhibitors. a
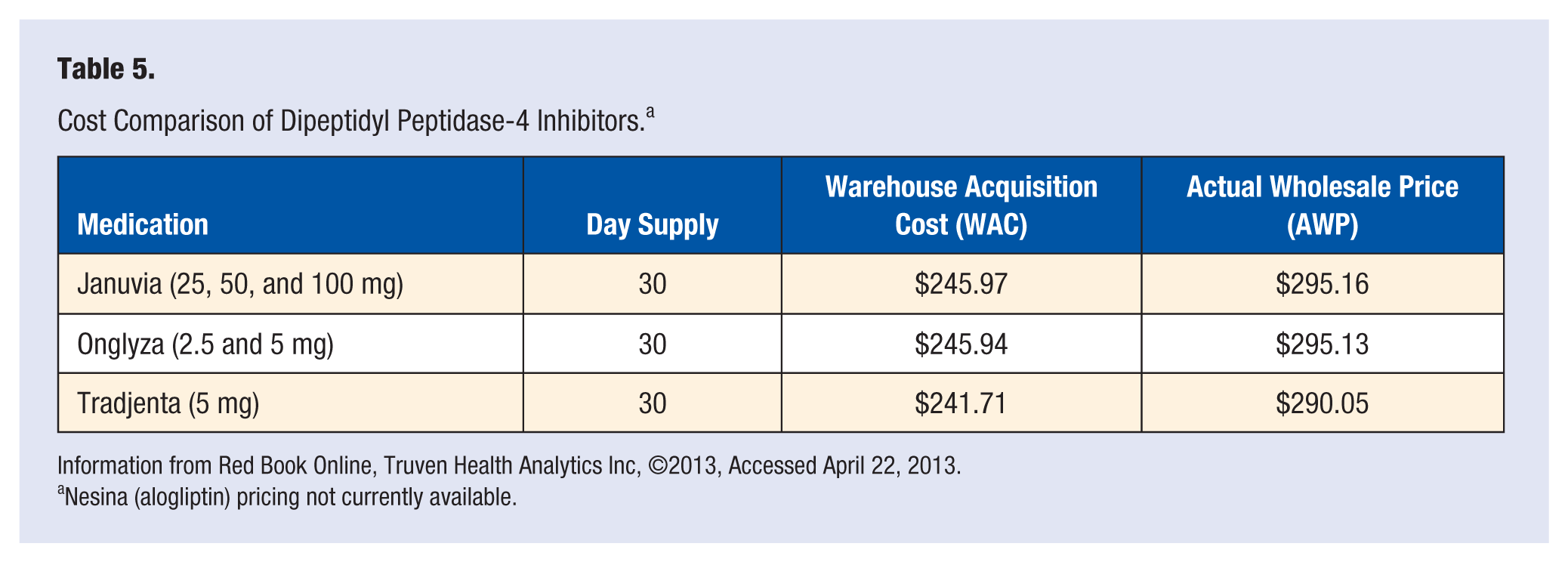
Information from Red Book Online, Truven Health Analytics Inc, ©2013, Accessed April 22, 2013.
Nesina (alogliptin) pricing not currently available.
Dipeptidyl peptidase-4 inhibitors provide another therapeutic agent to treat progressive T2DM. Although having advantages over other similar agents, such as GLP-1 agonists in regards to ease of administration, their A1C reduction is not as great. Advantages for DPP-4 inhibitors include easy oral dosing with a defined dosing schematic, low risk of hypoglycemia, minimal gastrointestinal adverse effects, and unlike other comparable agents such as the sulfonylureas, DPP-4 inhibitors are mostly weight neutral.
Sodium Glucose Transporter-2 Inhibitors
Diabetes often affects multiple organ systems and tissues in the body, including the pancreas, liver, muscle, adipose tissue, and cardiovascular system. Recently, the kidneys have emerged as a new target for diabetes therapy. Patients with high blood glucose levels usually experience glycosuria and nocturia, which are associated with poor glucose control. This glucose load excreted by the body is what is left after glucose has passed through the renal nephron. Most of the glucose is reabsorbed into the blood vessels via lumen wall transporter proteins called sodium glucose transporters (SGLTs). Antagonizing these transporters may provide a therapeutic effect for T2DM.
Sodium glucose transporters are found in the intestine, central nervous system, and most important for diabetes therapy, the renal nephrons. SGLT1 and SLGT2 both facilitate glucose transport and reabsorption but are expressed differently in different areas of the body. For example, SGLT1 is found primarily in the intestine, but only minimally in the nephron. SGLT2 on the other hand, is primarily found in the proximal tubule of the nephron and accounts for approximately 90% of the renal reabsorption of glucose. 65
Sodium glucose transporter-2 is more active and has greater expression in individuals with T2DM. Even though it is upregulated in patients with diabetes, glycosuria is still seen because the amount of glucose being filtered surpasses the reabsorption threshold of SGLT2s, which is approximately 375 mg/min of glucose, resulting in glucose in the urine.4,65 Because blocking this transporter does not depend on the action of insulin and because of the additional SGLT1 transporters further on in the nephron, the risk of hypoglycemia is low with these agents. 4
Invokana (Canagliflozin)
Food and Drug Administration–approved on March 29, 2013, canagliflozin became the first SGLT2 inhibitor approved for use in the United States. This medication was approved with the caveat that the manufacturer completes 5 different postmarketing studies on safety and efficacy of the medication. Currently, one of these studies is already in progress. The Canagliflozin Cardiovascular Assessment Study, originally started to meet the 2008 FDA guidance on cardiovascular safety in diabetes medications, will be looking at cardiovascular risks, including cardiovascular-related death, nonfatal myocardial infarction, and nonfatal stroke, in patients treated with canagliflozin. Follow-up will be for up to 9 years or time to an event. Despite its approval, there are still some concerns. FDA analysis of data submitted found increased risk of stroke; however, this risk seems to be reduced after the first month of use.
Canagliflozin is initiated at 100 mg once daily taken before the first meal of the day. Patients who require additional glycemic control may be increased to 300 mg if their estimated glomerular filtration rate (eGFR) is greater than or equal to 60 mL/min/1.73 m2.66 Patients with a eGFR 45 to <60 mL/min/1.73 m2 should receive a maximum dose of 100 mg per day. 66 Clinicians are not recommended to use canagliflozin when eGFR is <45 mL/min/1.73 m2 and use is contraindicated with eGFR <30 mL/min/1.73 m2. 66 Canagliflozin is metabolized by O-glucuronidation primarily through uridine diphosphate-glucuronosyl transferase and thus use with uridine diphosphate-glucuronosyl transferase inducers, such as rifampin and phenytoin, is not recommended. 66
The most common side effects associated with canagliflozin use include increased occurrence of female and male genital mycotic infections, increased urination, urinary tract infections, hyperkalemia, and weight loss. Mycotic infections associated with canagliflozin use were mild to moderate in nature and responded to treatment with antifungals. 67 Weight loss associated with canagliflozin was statistically significant and ranged from 2.4 to 4.7 kg (P < .001).66,67 Study investigators hypothesized that since canagliflozin increases urinary glucose excretion, patients with T2DM could be urinating up to 400 kcal/d since 1 g of glucose attributes approximately 4 kcal, resulting in significant weight loss. 67 Other associated side effects include orthostatic hypotension, increased thirst, and hypotension. 66 These side effects could possibly be attributed to the osmotic diuresis effect caused by canagliflozin. 67
Currently only 2 trials from the Canagliflozin Treatment and Trial Analysis (CANTATA) are published. The CANTATA studies are determining the safety and efficacy of canagliflozin as a monotherapy option (CANTATA-M), as an adjunct agent for patients already on metformin and a sulfonylurea (CANTATA-MSU) or pioglitazone (CANTATA-MP), and versus sitagliptin (CANTATA-D and CANTATA-D2) or a sulfonylurea (CANTATA-SU). The results from other trials are expected to be published in the near future.
Canagliflozin has shown to be an effective medication in reduction of A1C, fasting plasma glucose, and postprandial blood glucose. The CANTATA-D2 trial was a 52-week randomized trial, which looked at the effectiveness of canagliflozin 300 mg, compared with sitagliptin 100 mg as add-on therapies to metformin and a sulfonylurea in patients with poorly controlled T2DM. Average reduction in A1C was 1.03% in patients treated with canagliflozin compared to a reduction of 0.66% for sitagliptin (treatment difference [TD]: −0.37%; 95% confidence interval = −0.50 to −0.25). 67 This same A1C reduction of 1.03% was seen in the CANTATA-M monotherapy study (TD: −1.16%; P < .001). 68 Canagliflozin had significant effects on fasting plasma glucose and post-prandial blood glucose as well. Fasting plasma glucose can be reduced anywhere from 27 to 34 mg/dL compared with sitagliptin and placebo.67,68 There was also dramatic reduction in post-prandial glucose, with average reduction around 60 mg/dL.67,68
Canagliflozin was also associated with significant reductions in systolic blood pressure when compared to sitagliptin. 67 This reduction is thought to be because of the diuretic affect associated with canagliflozin. Heart rate was not significantly different. 67 The study investigators also saw significant improvements in pancreatic beta cell function. A proposed mechanism is with glucose wasting effects that do not require insulin production, the beta cells are spared and given time to rest and become more effective. 67
Forxiga (Dapagliflozin)
Dapagliflozin was the first SGLT2 inhibitor to be researched. A new drug application was submitted to the FDA but despite positive results in controlling glucose and A1C, concerns relating to possible risk of breast cancer, bladder cancer, and liver dysfunction forced the FDA to reject the application. This has since put the drug on hold as more trials are completed to show exactly what risk is associated with dapagliflozin and if the benefits are worth approval.
Efficacy of dapagliflozin was assessed in 4 phase-3 clinical trials, which looked at more than 2500 patients. The first trial investigated the role of dapagliflozin as monotherapy compared with placebo. A1C reductions at 24 weeks ranged from 0.58% to 0.89% (5 and 10 mg dose: P < .0001). 69 Fasting blood glucose values decreased 24.1 to 28.8 mg/dL (5 and 10 mg dose, respectively) versus a placebo effect of 4.1 mg/dL (P < .0001). 69 There was no significant weight reduction as dapagliflozin was associated with a 0.46 to 1.54 kg weight loss versus placebo. 69 When added on to a sulfonylurea, significant weight reductions of 1.56 to 2.26 kg versus placebo in 5 and 10 mg doses were seen (P = .009; P < .0001). 70 There were nonsignificant systolic and diastolic blood pressure decreases because the higher osmolarity of urine from the diuretic effect. 65 When compared with placebo as an adjunctive therapy to metformin and to a sulfonylurea, A1C reductions were similar, ranging from 0.67% to 0.84% (P < .0001) 71 and 0.58% to 0.82% (P < .001), 72 respectively. Higher A1C values resulted in greater reductions with dapagliflozin, as seen in an exploratory cohort of patients with higher A1C values. Patients with A1C >10% had 2.8% reduction versus placebo 69 ; however, Bailey et al 71 showed a smaller reduction was seen with A1C >9% (−1.35%; P < .029). 71 When dapagliflozin was compared with glipizide as add-on to metformin therapy, the result showed neither was superior, with both medications reducing A1C 0.52% (p = not significant). 70 However, greater reduction in body weight was observed in the dapagliflozin group, −3.22 kg versus +1.44 kg in patients treated with sulfonylurea (P < .0001). 70
Summary
Sodium glucose transporter-2 inhibitors provide a novel mechanism for reducing blood glucose in patients with T2DM. The approval of canagliflozin allows providers more options in treating therapy resistant T2DM. Dapagliflozin, however, may be associated with an increased risk of breast cancer, bladder cancer, and liver dysfunction, and thus will require more research before it will receive approval from the FDA. SGLT2 inhibitors reduce A1C approximately 1% and have significant reductions in fasting and postprandial plasma glucose, averaging 30 and 60 mg/dL, respectively. Possible side effects include increased risk of urinary or genital infection, headache, hypotension, and increased thirst.65,67 With low rates of hypoglycemia and significant weight loss associated with beneficial antihyperglycemic effects, this class provides a good alternative for patients seeking different treatment options.
Nonconventional Therapies
Cycloset (Bromocriptine)
In May of 2009, the FDA approved a unique therapy for the treatment of T2DM. Cycloset (bromocriptine), was shown to be beneficial in lowering blood glucose and A1C levels in patients already treated with 1 or 2 hyperglycemic agents. Bromocriptine, an ergot alkaloid derivative that acts as a dopamine-2 agonist, is approved for the treatment of type 2 diabetes and Parkinson’s disease. It is not well known why bromocriptine has beneficial effects in patients with diabetes; however, there have been some proposed mechanisms. One mechanism is that increased dopamine receptor activation inhibits prolactin release, and thus prolactin may raise glucose levels. 73 Another idea is that neuroendocrine peptides play a major role in body fat stores and insulin sensitivity. The effects of serotonin and dopamine may cause the blood glucose lowering effects and explain a mechanism for bromocriptine. 74
Bromocriptine is given once a day within 2 hours of waking up in the morning and should be given with food to reduce the gastrointestinal adverse effects that are possible such as nausea and vomiting, which seemed to be transient. The most common side effects include nausea (32.5%), dizziness (12.5%), hypoesthesia (18.9%), and headache (16.8%). 75 Other possible side effects include hypotension, syncope, and exacerbation of psychiatric disorders. Doses are initiated at 0.8 mg per day and titrated weekly by 0.8 mg increments to a maximum of 4.8 mg (6 tablets) until goal A1C is achieved or maximum A1C reduction has been met. 75 Study patients typically required 4 to 6 tablets a day to achieve clinical response. Hypoglycemia was rare with this agent, unless combined with a medication known to cause hypoglycemia. When compared with placebo in trials, there was no difference in weight changes. 76
Clinical trials show that bromocriptine rapid release is effective in reducing A1C by 0.5% to 0.6% (P = .009) when added to other medications such as metformin, or a sulfonylurea.74,75 In one study, fasting glucose was also decreased 18 mg/dL on average (P = .02). 74 Patients had a 42% relative risk reduction of cardiovascular endpoint for time to the first myocardial infarction, stroke, hospitalization for angina or congestive heart failure, or revascularization. 76 Only 1.8% of patients taking bromocriptine had a cardiovascular endpoint compared to 3.2% of patients taking placebo (95% confidence interval = 0.35-0.96). 76 One concern noted was 24% of patients discontinued the therapy because of adverse effects compared to 11% in the placebo arm. 76
Bromocriptine is an effective agent in the treatment of refractory T2DM. Despite an unclear mechanism in T2DM, it can potentially reduce A1C 0.6%. Its use, however, should be kept to refractory cases or in cases where it could have dual indications, such as in Parkinson’s disease or hyperprolactinemia.
Welchol (Colesevelam)
Another alternative agent for the management of type 2 diabetes is Welchol (colesevelam). Colesevelam, a bile acid sequestrant, is normally used for LDL reduction in patients with high LDL levels despite primary treatment with a statin or in patients intolerant to statins. Bile acid sequestrants work in the intestinal tract and are not absorbed. In the gastrointestinal tract, sequestrants bind to bile acid and prevent its reabsorption, leading to less cholesterol in circulation as the body uses cholesterol as a precursor to produce more bile. A proposed mechanism of action for colesevelam in T2DM is that it affects glucose absorption or changes the kinetics of absorption, thus lowering blood glucose. Another possible mechanism is some bile acids are ligands to a family of receptors called farnesoid X receptor. Farnesoid X receptors are thought to have a role in maintaining blood glucose, bile acid and cholesterol metabolism through downstream regulation of transcription and growth factors. When bile acid binds, it activates a pathway that may improve glycemic control. 77
Recently, studies have shown that colesevelam can also reduce blood glucose and A1C values. Currently, 75% of patients diagnosed with T2DM have LDL values above the current goal of 100 mg/dL. 77 These patients are also at higher risk for experiencing a coronary artery disease (CAD) event, such as myocardial infarction or stroke, and thus diabetes has been classified as CAD risk equivalent. This drug works to potentially reduce CAD risk by 3 ways; reducing LDL, total cholesterol, and blood glucose.
Colesevelam has modest efficacy reducing A1C and blood glucose. When added to metformin, sulfonylurea, and/or insulin, colesevelam reduced A1C an additional 0.41% to 0.54% (P < .001), and reduced LDL from 12.8% to 16.7% (P < .001).77-80 Additionally, colesevelam reduced fasting plasma glucose by an average range of 13.9 to 15.1 mg/dL (P = not significant to P = .01).77-79 However, triglyceride levels did increase 15% (P < .001) with colesevelam treatment. 79
The most common side effect noted with colesevelam was constipation occurring in around 7% of patients. 80 Other side effects noted include dyspepsia, rare hypoglycemia, flatulence, and nausea. There are no significant weight changes associated with adding colesevelam therapy. Colesevelam is dosed in 3 tablets (1.875 g) taken twice daily or 6 tablets (3.75 g) taken once daily with a full glass of liquid.
Overall, colesevelam is a viable option for patients near their A1C goal level who also require additional LDL lowering. It is considered an effective agent, lowering A1C roughly 0.5% when added on to other antihyperglycemic medications. Side effects are reported but not severe and are mostly gastrointestinal in nature. Although colesevelam has positive effects in reduction of CAD risk factors, such as LDL and blood glucose, its beneficial effects on reducing overall CAD are yet to be established.
Future Agents
Inhaled Insulin
A new delivery method for rapid acting insulin is currently in development. Afrezza is a new, rapid insulin therapy for type 1 and type 2 diabetes and is currently in the late stages of clinical research. Afrezza is used as mealtime insulin and helps lower postprandial blood glucose. It is formulated as a dry powder containing rapid-acting human insulin molecules. Researchers believe that it could potentially change the art of insulin administration.
Afrezza is administered using Technosphere insulin technology, a drug carrying moiety that allows for pulmonary absorption and administration of drugs. The main excipient in the Technosphere administration system is fumaryl diketopiperazine microparticles. These particles deliver the drug to the deepest part of the lung and are not only being developed for insulin delivery, but also for GLP-1 administration. Fumaryl diketopiperazine particles are not metabolized and are excreted unchanged via the kidneys. 81
In head-to-head studies of Afrezza and subcutaneous regular insulin, peak insulin concentrations occurred at 12 to 17 minutes for Afrezza compared with 134 minutes for regular subcutaneous insulin. 82 This quick peak in insulin concentrations also allows quicker excretion than regular insulin, which results in a shorter duration of action. The bioavailability of inhaled insulin may be dose dependent and varies from 21% to 25%, with lower dosages being better absorbed. 82 Technosphere insulin reduced A1C by 0.4% to 0.72%, compared with placebo Technosphere powder (P < .05 to P = .003).83,84 Postprandial glucose levels were also lowered significantly, with reductions up to 56% compared with baseline (P < .0001). 83 The most common adverse events associated with use were mild hypoglycemia and a mild, but transient nonproductive cough.
Pulmonary administration does not seem to be significantly affected in those with chronic obstructive pulmonary disease (COPD) or in individuals who smoke cigarettes. In a study comparing pharmacokinetic parameters in individuals with COPD and without, mean peak insulin was roughly the same, 34.7 µU/ml versus 39.5 µU/mL (P = .29). 85 Time till maximum concentration occurred 3 minutes later in the COPD group, 15 versus 12 minutes in the non-COPD arm (P = .24). 85 Also total insulin exposure time was not statically significant, indicating that absorption and clearance was not affected. 85
Many practitioners may feel uncertain about the clinical use of Afrezza, especially after the first inhaled insulin, Exubera, was eventually pulled from the market by the manufacturer. Because of erratic dosing schedules, limited options for dosages, nonlinear dosing with higher doses and inconsistent lowering of blood glucose, it was not an option used often. Especially for patients with type 2 diabetes who require higher amounts of insulin to lower their blood glucose, multiple inhalations of Exubera was an inconvenience and resulted in stopping the treatment. Afrezza, however, seems to have addressed certain issues that allow it to be a reliable insulin source. It has larger dose cartridges to allow higher dosages to be used, while still keeping a linear kinetic profile. Another concern was the large size of Exubera. Afrezza is available as a compact, pocket-sized inhalation device. As patients breathe in, medicine will be drawn into the lungs. They do not have to line up their breaths as with Exubera. The pharmacokinetics of Afrezza also match up to that of endogenous insulin, something that was not associated with the insulin in Exubera. With clinical studies still ongoing, it is too early to tell how efficacious this product is, but it looks to be an effective way to administer insulin.
For future insulin therapy in both T1DM and T2DM, the inhaled insulin Afrezza may change the way insulin is administered. Technosphere insulin is absorbed faster, has a shorter duration of action and preliminary data show similar efficacy to subcutaneous regular insulin. This form of insulin therapy has not been associated with any weight changes yet, unlike that of subcutaneous insulin delivery. 84 Afrezza may also increase patient satisfaction and compliance by the ease of insulin therapy.
Conclusion
Because of the progressive nature of T2DM, new agents are needed to help combat this potentially deadly disease. Recent years have seen the FDA approve efficacious new drugs for use in patients with refractory disease. The GLP-1 agonists, which can dramatically reduce A1C up to roughly 1.5% while at the same time decreasing body weight and potentially improving the function and number of pancreatic β-cells, are becoming more common in practice. Other agents such as DPP-4 inhibitors are good alternative agents because of easy, daily, oral administration. DPP-4 inhibitors act mostly on postprandial glucose and are weight neutral. Although their effect on A1C is not as great as GLP-1 agonists, they are comparable to the sulfonylureas, without the risk of hypoglycemia and weight gain.
The newest agent approved by the FDA is the novel SGLT2 inhibitor. Blocking the reabsorption of glucose in the renal nephron results in markedly increased glycosuria, resulting in reduced blood glucose concentrations. These agents are associated with about a 1% reduction in A1C. They also cause significant weight loss patients. The patient is essentially urinating calories in the form of glucose, resulting in fat being used as an alternative source for energy. The adverse event profile is complicated by an increased occurrence of genital mycotic infections, urinary tract infections, and hypotension. However, severe side effects such as hypoglycemia are rare since SGLT2 inhibitors do not affect insulin secretion.
Third-line agents for individuals who need additional control to achieve their goal include bromocriptine and colesevelam. Although these agents have only a modest effect in reducing A1C, about 0.4% to 0.6%, they can be good choices for patients who have other conditions. Colesevelam may be a good choice for patients with high LDL levels as it also reduces LDL values by approximately 12%, whereas bromocriptine can be used in patients with Parkinson’s disease or hyperprolactinemia.
There are some effective drugs in the pipeline, which may have important clinical effects if they come to the market and are approved by the FDA. This includes Afrezza, a new form of inhaled insulin that is showing modest efficacy as a rapid-acting insulin delivery system. Patient compliance and satisfaction with this form of insulin medication may be at an all-time high because of its convenient and pain-free administration and its small, convenient size.
In conclusion, there is a continued need for more agents to treat T2DM. As patients progress with the disease, medications may become ineffective, and others may be contraindicated in use. In many situations, practitioners are left with only a few options to treat the disease. Thus, treatment regimens should be individualized and combination treatment should be used to target multiple aspects of type 2 diabetes. Despite lacking long-term outcome data, many new treatment options are available to help improve glycemic control while at the same time reducing risk of hypoglycemia, decreasing weight in a growing obese population, and improving patient compliance with easier dosing regimens, including once-daily and once-weekly options.
