Abstract
Introduction:
Polycystic ovary syndrome is a common chronic condition characterized by insulin resistance and hyperandrogenism, leading to significant health risks and impaired quality of life. Sodium-glucose transporter type 2 inhibitors have shown promise in improving the metabolic profile of women with polycystic ovary syndrome. However, their impact on hormonal parameters and cycle disorders remains uncertain.
Methods:
This systematic review analyzed randomized clinical trials published up to 1 December 2023, comparing sodium-glucose transporter type 2 inhibitors to metformin, other antidiabetic agents, or placebo in women with polycystic ovary syndrome. The primary outcomes were changes in total testosterone, free androgen index, dehydroepiandrosterone sulfate, delta-4 androstenedione, and cycle disorders.
Results:
Five randomized studies were included, evaluating canagliflozin, dapagliflozin, licogliflozin, or empagliflozin against metformin, exenatide, or placebo, with a total of 214 participants. Improvements in total testosterone and dehydroepiandrosterone sulfatewere observed in some studies, but the effects were inconsistent across drugs and outcomes. Additionally, two studies reported beneficial effects on cycle disorders.
Conclusions:
Sodium-glucose transporter type 2 inhibitors appear to have a potential but variable impact on hormonal parameters in women with polycystic ovary syndrome. However, larger and longer-duration studies are needed to fully elucidate their long-term efficacy in addressing hyperandrogenism and improving overall outcomes in these patients.
Keywords
Introduction
Polycystic ovary syndrome (PCOS), a common endocrinopathy characterized by hyperandrogenism, ovulatory dysfunction, and insulin resistance, remains a major therapeutic challenge.1–3 Although conventional treatments like oral contraceptives and metformin are widely used, long-term efficacy and management of associated comorbidities remain public health issues.3,4
Inhibitors of the sodium-glucose cotransporter type 2 (ISGLT2), a therapeutic drug class with proven hypoglycemic and cardio-renal protective action in diabetic and nondiabetic patients, have recently attracted growing interest in treating PCOS. 2 Their mechanism of action, involving inhibition of renal glucose reabsorption and increased urinary glucose excretion, offers promising therapeutic potential in this complex pathology. 2
Numerous studies have suggested that ISGLT2 may exert beneficial effects beyond their action on blood glucose by modulating several pathophysiological pathways involved in PCOS. 4 These include improving insulin sensitivity, promoting weight leading and overall protective cardiovascular effects. 4 Recent studies have highlighted the role of insulin resistance in the pathophysiology of PCOS, particularly its relationship with hyperandrogenism. Genetic insights, such as the involvement of genes related to insulin receptors and steroidogenesis, have provided a deeper understanding of the mechanisms driving PCOS.5,6 These advances make SGLT-2 inhibitors a promising therapeutic option due to their ability to improve insulin sensitivity and potentially mitigate hyperandrogenism.
However, while the effect of ISGLT2 on improving the cardiometabolic profile of women with PCOS is well identified, 4 the impact of this therapeutic class on the clinical and biological hyperandrogenism that constitutes an important pillar of PCOS pathophysiology remains poorly elucidated.
This systematic review aims to evaluate the efficacy of ISGLT2 on clinical and biological hyperandrogenism and cycle irregularities in women with PCOS.
Methods
Type of study
This is a systematic review of the literature encompassing randomized clinical trials investigating the effects of ISGLT2 on hormonal parameters, clinical hyperandrogenism, and cycle irregularities in women with PCOS. The review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. 7
Search strategy
The search was carried out using the PUBMED, Sciences Directs, and Cochrane databases. The keywords used were “PCOS” and “Sodium-Glucose Transporter 2 Inhibitors,” linked by the Boolean tool “AND.” The search for these words was limited to the title and abstract in the Sciences Direct database. In the PUBMED database, the “Clinical Trial” filter was applied. No restriction by year of publication was applied. The last search date was 1 December 2023.
The first step was the authors’ selection of articles based on their titles, according to the selection criteria detailed below, and eliminating duplicates. The two authors then made a second selection after reading the full text. In the event of discrepancies, the choice was made after consultation.
Inclusion criteria
This systematic review included randomized clinical trials:
– Double-blind or not
– Investigating hormonal parameters (total testosterone (TT) or free testosterone (FT), SDHEA, sex hormone-binding globulin (SHBG), free androgen index (FAI) estradiol, gonadotrophins), clinical signs of hyperandrogenism (hirsutism) and/or cycle disorders in women with PCOS
– Using: (i) ISGLT2 alone versus placebo (ii) ISGLT2 versus metformin (iii) ISGLT2 combined with metformin versus metformin alone (iv) ISGLT2 versus another antidiabetic agent
– Which included patients aged over 18, diagnosed with PCOS according to the Rotterdam 2003 or National Institutes of Health criteria. 8
Noninclusion criteria
Studies were not considered for inclusion if they met any of the following broad criteria:
– Nonrandomized controlled trials (RCTs), such as observational studies, systematic reviews, meta-analyses, case reports, and letters to the editor.
– Studies that did not focus specifically on women diagnosed with PCOS.
– Studies that did not use SGLT-2 inhibitors as the primary intervention, whether alone or in combination with other treatments.
– Studies that lacked evaluation of hormonal parameters (e.g., TT, SHBG) or clinical manifestations of hyperandrogenism (e.g., hirsutism) and/or menstrual irregularities.
Exclusion criteria
We have excluded:
– Studies involving participants with hyperandrogenism due to conditions other than PCOS.
– Inaccessible full text: Studies for which the full text was unavailable and thus could not be fully assessed.
– Studies with significant methodological concerns, such as high risk of bias, lack of a control group, or insufficient reporting on hormonal and menstrual outcomes.
– Studies that did not provide adequate data on key hormonal parameters or menstrual irregularities as required for this systematic review’s analysis
Assessment of study quality
The assessment of studies for bias was carried out by both authors independently using the “Revised Cochrane risk of bias tool for randomized trials (RoB 2)” in its 22 August 2019 version. 9 This tool explores five domains and six subdomains, assigning each subdomain one of the following risk-of-bias grades: Low, Some concern, High. 9 The evaluation was carried out independently by two authors, and any discrepancies were addressed after consultation.
Data extraction and representation
Data extraction was carried out independently by two authors, using preestablished formula. The data studied were the baseline characteristics of the study population (inclusion criteria, age, presence of overweight or obesity, total number of subjects), the course of the study (duration of study, randomization, number of subjects in the different groups, molecules, and doses used in the intervention and control groups, clinical and biological parameters assessed) and the results of the interventions. No sensitivity, subgroup, or statistical analysis was conducted to account for heterogeneity or compare outcomes across studies. Results were synthesized into tables and figures by two independent authors.
Results
Study selection and study populations
A total of five RCTs were included, with a combined total of 214 participants (Figure 1).10–14 The interventions evaluated in these studies included various SGLT-2 inhibitors, specifically canagliflozin (100 mg/day), dapagliflozin (10 mg/day), licogliflozin (100 mg/day), and empagliflozin (25 mg/day). The control groups in the studies were treated with either metformin (doses from 1500 to 2000 mg/day), exenatide (2 mg/week), or placebo. Study durations ranged from 2 to 24 weeks. Four of the studies used the Rotterdam 2003 criteria to diagnose PCOS, while one study employed the NIH 1990 criteria. 11

Flow diagram of included studies according to PRISMA 2020 guidelines for new systematic reviews.
The study populations were somewhat heterogeneous, with differences in BMI, age, and comorbidities. These factors likely contributed to the variability observed in the outcomes. Some studies included patients who were overweight or obese, while others included women with a wider range of body compositions. The mean age of participants across the studies ranged from 23 to 35 years, and comorbidities such as insulin resistance and dyslipidemia were frequently reported (Table 1).
Summary of the methodology and characteristics of study population of the included studies.

D-4A: delta 4 androstenedione; DHEA: dehydroepiandrosterone; FAI: free androgen index; FSG: Ferriman Gallaway score; FSH: follicle stimulating hormone; FT: free testosterone; LH: luteinizing hormone; N/A: not applicable; PCOS: polycystic ovary syndrome; SDHEA: dehydroepiandrosterone sulfate; SHBG: sex hormone binding globulin; TT: total testosterone.
Assessment of study quality:
Overall, the studies were of good quality with no high risk of bias (Table 2).
Evaluation of bias risk of the included studies.

Efficacy of ISGLT-2 on clinical or biological hyperandrogenism and cycle disorders in women with PCOS
Table 3 summarizes the results of the various studies included. The efficacy of ISGLT-2 on hormone levels was inconsistent across studies (Table 4).
Summary of the results of different interventions in the included studies.

D-4A: delta 4 androstenedione; DHEA: dehydroepiandrosterone; FAI: free androgen index; FSG: Ferriman Gallaway score; FSH: follicle stimulating hormone; FT: free testosterone; LH: luteinizing hormone; N/A: not applicable; PCOS: polycystic ovary syndrome; SDHEA: dehydroepiandrosterone sulfate; SHBG: sex hormone binding globulin; TT: total testosterone.
Summary of the effect of varying SLGT-2 inhibitors on hormonal parameters, menstruations, and clinical hyperandrogenism in patients with PCOS.

D-4A: delta 4 androstenedione; DHEA: dehydroepiandrosterone; FAI: free androgen index; FSG: Ferriman Gallaway score; FSH: follicle stimulating hormone; FT: free testosterone; HA: clinical hyperandrogenism; LH: luteinizing hormone; PCOS: polycystic ovary syndrome; SDHEA: dehydroepiandrostrone sulfate; SGLT-2: sodium-glucose cotransporter; SHBG: sex hormone-binding globulin; TT: total testosterone; ↘: significant decrease; ↗: significant increase; NS: nonsignificant effect; —: not studied.
The primary hormonal parameters evaluated across the studies were TT, FAI, sex hormone-binding globulin (SHBG), and serum dehydroepiandrosterone sulfate (S-DHEA). The results regarding these hormonal outcomes were inconsistent across the trials:
•
•
•
•
•
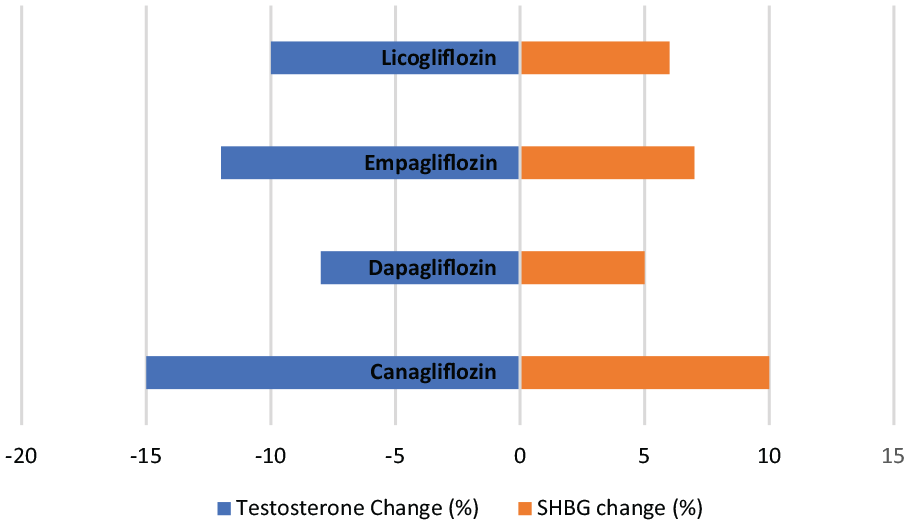
The effect of different studied SGLT-2 inhibitors on testosterone and SHBG levels expressed in percentage of baseline levels.
Menstrual cycle disorders
The impact of SGLT-2 inhibitors on menstrual cycle regularity was assessed in two studies. Both studies, which evaluated canagliflozin, reported improvements in menstrual cycle regularity. However, the improvements were not significantly different from the control groups, which were treated with metformin or exenatide. While SGLT-2 inhibitors appeared to restore more regular menstrual cycles in some participants, the variability in study populations and differences in baseline menstrual irregularities made it difficult to draw definitive conclusions about their efficacy in this regard.
• One study noted that the participants who experienced the greatest improvement in menstrual regularity had higher baseline testosterone levels and were more insulin-resistant, suggesting that SGLT-2 inhibitors may be particularly effective in a subset of women with more severe metabolic disturbances.
Comparison with metformin
In three of the studies, metformin was used as a control treatment. When compared to metformin, SGLT-2 inhibitors showed similar benefits in terms of improving insulin sensitivity and reducing body weight. However, metformin demonstrated more consistent improvements in both menstrual regularity and hyperandrogenic symptoms such as hirsutism.
•
The results suggest that while SGLT-2 inhibitors may offer comparable metabolic benefits to metformin, their effects on hormonal regulation, particularly in the context of hyperandrogenism and menstrual irregularities, are less consistent
Discussion
PCOS is the most common endocrinopathy in women of childbearing age.1–3,15 Depending on the diagnostic criteria, its prevalence varies from 4% to 20%.1,2 Its pathophysiology is complex and not completely understood. It includes genetic (genes involved in steroidogenesis, gonadotropin genes and their receptors, AMH genes, insulin genes and their receptors, etc.), epigenetic, prenatal, and postnatal factors (intrauterine exposure to androgens), environmental (endocrine disruptors such as bisphenol A), dietary (high-fat hypercaloric diet, Vitamin D deficiency), social (chronic stress), as well as the involvement of the gut microbiota, mitochondrial dysfunction, oxidative stress, and chronic low-grade inflammation.1,15–19 The main pathophysiological elements of PCOS are insulin resistance and hyperinsulinism, and hyperandrogenism, both of which are interrelated and responsible for the clinical phenotype of patients followed for PCOS. Insulin resistance is a cornerstone mechanism in both the development and persistence of PCOS. 6 Insulin resistance in PCOS patients is due to an insulin receptor defect resulting from excessive serine phosphorylation and decreased tyrosine phosphorylation, leading to reduced activation of the phosphatidylinositol-3-kinase signaling pathway by insulin, resulting in increased blood glucose levels. 6 Patients with PCOS are exposed to an increased prevalence of cardiovascular risk factors, such as hypertension (through activation of the sympathetic and/or renin-angiotensin systems), obesity, dyslipidemia (with an atherogenic profile), and chronic kidney disease, responsible for increased morbidity and mortality. 2 In addition, women with PCOS are at greater risk of impaired quality of life, anxiety, and depression. 20 Currently, PCOS is usually managed by changes in lifestyle, oral contraceptives, and metformin, prescribed to restore regular menstruation, lower insulin resistance, and maintain adequate body weight.3,6,18 In cases of infertility, letrozole, and clomiphene citrate are frequently prescribed to induce ovulation.3,16,18,20 Focusing on treating symptoms and the complexity of the physiopathological mechanisms in PCOS prevents the optimal efficacy of different treatment regimens. 15 With ISGLT2 proving their beneficial metabolic effects in women with PCOS, 4 this systematic review aimed to investigate the efficacy of ISGLT2 on clinical and biological hyperandrogenism and cycle disorders.
The diagnostic criteria for PCOS used in these studies were the Rotterdam 2003 criteria in four studies and the National Institutes of Health (NIH) 1990 criteria in the study by Elkind-Hirsch et al. 11 PCOS, a diagnosis of exclusion, can be characterized using three different sets of diagnostic criteria: the 1990 NIH criteria, the 2003 Rotterdam criteria, and the 2006 Androgen Excess-PCOS Society (AE-PCOS) criteria. The Rotterdam criteria include (i) oligo-ovulation or anovulation, (ii) clinical or biochemical signs of hyperandrogenism, or (iii) polycystic ovarian morphology evidenced by intravaginal pelvic ultrasound or AMH elevation, 3 and require the presence of two out of three to confirm the diagnosis of PCOS. 1 Based on the Rotterdam criteria, PCOS cases are distinguished into four different phenotypes (A, B, C,D ) according to the presence or the absence of the three diagnostic criteria. 8 The NIH and AE-PCOS criteria consider hyperandrogenism to be a necessary condition for diagnosis, 8 corresponding to phenotypes A and B of the Rotterdam criteria. 1 In the five randomized studies we included, the effect of ISGLT2 use was inconsistent on hormone levels.
Mechanisms of action on androgen levels
In PCOS, hyperandrogenism and insulin resistance appear to be interrelated. Insulin and insulin-like growth factors act synergistically with LH to increase androgen synthesis in ovarian theca cells. 21 Insulin also decreases SHBG by reducing its hepatic synthesis, resulting in increased FT, 21 capable of binding and activating the androgen receptor. In peripheral tissues, testosterone can be reduced to 5α- dihydrotestosterone (DHT). 22 DHT being the most potent agonist of the androgen receptor is responsible for androgenic features like hirsutism and acne. 22 Additionally, on the other hand, short-term androgen administration in women has been shown to decrease insulin sensitivity, as demonstrated both in hyperinsulinemic hyperglycemic and euglycemic clamps 23 and in mouse models.24,25 The effects of ISGLT-2 on androgen levels may be explained by their beneficial effect on patients’ metabolic profiles. These molecules improve insulin sensitivity even in nondiabetic patients,5,26 thus reducing insulin levels and its pro-androgen effect. By improving insulin sensitivity and reducing hyperinsulinemia, SGLT-2 inhibitors may indirectly decrease androgen production and increase SHBG levels, which reduce free androgen bioavailability.
Despite these promising findings, the effects of SGLT-2 inhibitors on androgen levels were not consistent across all trials. This variability could be attributed to differences in baseline insulin resistance, BMI, and the duration of treatment. For example, participants with higher baseline insulin resistance may have experienced more pronounced improvements in androgen levels due to greater reductions in insulin levels. Additionally, studies that used higher doses or longer treatment durations may have had more time to observe significant changes in hormonal parameters.
Their beneficial effect on anthropometric and metabolic parameters in PCOS women has also been demonstrated. 4 These effects include an increased insulin sensitivity, thus lowering total insulin levels. 4 Lowering insulin levels can explain the potentially beneficial effect of ISLGT2 on the androgen levels shown in this systematic review. ISGLT2 offers cardiovascular protection in patients with or without T2DM, through its action on the renin–angiotensin system and/or the sympathetic nervous system, and improvement of mitochondrial function; all are important pathophysiological pathways in PCOS.
Potential role in menstrual cycle regulation
Restoration of menstrual regularity is a key therapeutic goal in managing PCOS, as menstrual irregularities are often the first sign of endocrine disruption in these patients. Two of the included studies reported improvements in menstrual regularity following treatment with canagliflozin. These findings suggest that by reducing hyperinsulinemia and improving insulin sensitivity, SGLT-2 inhibitors may restore the balance between LH and follicle-stimulating hormone (FSH), leading to improved ovulatory function. Dapagliflozin, an SGLT-2 inhibitor, has been shown to partially restore reproductive function in obese female mice by improving LH profiles and increasing ovulation rates. 27 In high-fat diet-fed mice, dapagliflozin ameliorated ovulation disorders by attenuating microglia-mediated hypothalamic inflammation and improving insulin and leptin sensitivity. 28 SGLT-2 inhibitors have demonstrated neuroprotective properties and the ability to suppress microglia-induced neuroinflammation in the central nervous system. 29
However, the improvements in menstrual regularity observed with SGLT-2 inhibitors were not significantly different from those seen with metformin. Metformin, a well-established treatment for PCOS, also improves insulin sensitivity and has been shown to regulate menstrual cycles in insulin-resistant women. 30 The similarity in outcomes suggests that while SGLT-2 inhibitors offer a promising alternative, their effects on menstrual cycle regulation may not be superior to traditional treatments such as metformin.
Further research is needed to explore the mechanisms by which SGLT-2 inhibitors influence menstrual cycle regulation. Animal studies have shown that improved insulin sensitivity can restore normal hypothalamic–pituitary–ovarian axis function by modulating gonadotropin-releasing hormone (GnRH) pulsatility and reducing androgen production. It is possible that SGLT-2 inhibitors exert similar effects, but the lack of long-term data limits our understanding of their full potential.
Long-term clinical implications
The use of SGLT-2 inhibitors in nondiabetic women with PCOS is a novel therapeutic approach, and the long-term clinical implications of this treatment are still unclear. While SGLT-2 inhibitors have been shown to provide cardiometabolic benefits in diabetic populations, their safety and efficacy in nondiabetic women with PCOS have not been thoroughly evaluated. This is especially important given the increased cardiovascular risk associated with PCOS, which includes a higher prevalence of hypertension, dyslipidemia, and obesity.
The potential cardiovascular protective effects of SGLT-2 inhibitors, such as reductions in blood pressure, body weight, and visceral fat, could offer additional benefits for women with PCOS. However, the long-term use of these drugs raises concerns regarding potential adverse effects, such as an increased risk of urinary tract infections and euglycemic diabetic ketoacidosis. These risks may be more relevant for nondiabetic women, and thus larger, long-term studies are needed to assess the risk-benefit profile of SGLT-2 inhibitors in this population
Strengths and limitations
This study analyzed five RCTs, selected through a rigorous literature search and the use of strict inclusion and exclusion criteria to identify high-quality literature for analysis. We also carried out a careful assessment of bias in these studies. This included population across all studies with PCOS patients with clinical or biological hyperandrogenism (Phenotype A and/or B, NIH criteria) and insulin-resistant phenotype. However, a key limitation of this review is the lack of statistical analysis to account for heterogeneity or compare outcomes across studies. Future studies should address this limitation to enhance the comparability and robustness of findings. In addition, the hormonal parameters measured across the five studies were heterogeneous and the effects on clinical hyperandrogenism and menstruation were measured in one and two studies, respectively.
Clinical implications
While SGLT-2 inhibitors present a promising alternative for managing insulin resistance in PCOS, further research is essential to assess their long-term safety and efficacy, especially in nondiabetic populations. The potential risks of prolonged SGLT-2 inhibitor use, such as urinary tract infections, warrant careful consideration, particularly in women with PCOS who may not have diabetes.
Recommendations for future research
Future studies should focus on direct comparisons between SGLT-2 inhibitors and established treatments like metformin and oral contraceptives to better evaluate their relative efficacy in managing hyperandrogenism and menstrual irregularities. Additionally, studies should investigate which specific subgroups of women with PCOS (e.g., those with higher baseline insulin resistance) may benefit most from SGLT-2 inhibitors. Larger and longer-duration trials are also needed to establish a clearer understanding of the long-term effects and safety of SGLT-2 inhibitors for reproductive and metabolic outcomes in women with PCOS.
Conclusion
This systematic review highlights the potential role of SGLT-2 inhibitors in managing PCOS, particularly through their effects on insulin sensitivity, androgen levels, and menstrual regularity. The findings suggest that SGLT-2 inhibitors may offer benefits comparable to metformin for metabolic improvements, with the added advantage of possible cardioprotective effects. However, the impact of SGLT-2 inhibitors on hyperandrogenism and menstrual regulation remains inconsistent across studies. This variability may be influenced by the differences in drug type, dosage, duration, and patient characteristics, such as baseline insulin resistance and BMI. In conclusion, SGLT-2 inhibitors represent a valuable addition to the therapeutic options available for PCOS management, but further research is required to fully understand their role and establish clear guidelines for their use in this patient population.
Footnotes
Acknowledgements
During the preparation of this work, the authors used Google Gemini to proofread the text. After using this tool, the authors reviewed and edited the content as needed and took full responsibility for the content of the publication. The authors acknowledge Dr Rihene Melki for providing a complementary manuscript revision.
Authors contributions
HE Mrabet: Conceptualization, Data curation, Investigation, Formal analysis, Methodology, Supervision, Validation, Writing – original draft, Writing – review and editing. H Ben Salem: Data curation, Formal analysis, Investigation, Methodology, Supervision, Validation, Writing – original draft. T Ach: Conceptualization, Supervision Validation. A Ben Abdelkarim: Supervision, Validation. W Alaya: Writing – review and editing, Supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Not applicable.
Informed consent
Not applicable.

