Abstract
The importance of optimal bone growth in childhood and adolescence has been recognized as one of the key strategies in osteoporotic fracture prevention. Low birth size, poor childhood growth, and low peak bone mass at the cessation of growth have been linked to the later risk of osteoporosis and hip fracture. Formerly, the focus was merely on maximizing bone mineral accrual because a high peak bone mineral mass may prevent attainment of a critical “fracture threshold” associated with age-related bone loss and osteoporosis. More recently, the focus has shifted away from bone mineral accrual—as measured by dual-energy X-ray absorptiometry (DXA)—toward the optimization of bone strength. This is partly because of the advances in bone imaging that have enabled estimation of bone strength beyond bone mass. In this review, we briefly describe long-bone growth and structural development and our abilities to assess bone properties by medical imaging tools. In addition, we summarize the evidence of factors contributing to skeletal growth, bone fragility, and the development of strong, healthy bones.
‘Encouraging optimal bone growth in childhood and adolescence has been recognized as one of the key strategies in osteoporotic fracture prevention.’
Introduction
Osteoporosis, through its association with age-related fractures, is one of the most common causes of long-standing pain, functional impairment, disability, and death in elderly populations. 1 Encouraging optimal bone growth in childhood and adolescence has been recognized as one of the key strategies in osteoporotic fracture prevention. 2 Low birth size, poor childhood growth and low peak bone mass at the cessation of growth have been linked to the later risk of osteoporosis and hip fracture.2,3 Formerly, the focus was merely on maximizing bone mineral accrual because a high peak bone mineral mass may prevent attainment of a critical “fracture threshold” associated with age-related bone loss and osteoporosis. 4 More recently, the focus has shifted away from bone mineral accrual—as measured by dual-energy X-ray absorptiometry (DXA)—toward the optimization of bone strength. This is partly a result of the advances in bone imaging that have enabled estimation of bone strength beyond bone mass.5,6 New imaging techniques, such as peripheral quantitative computed tomography (pQCT) and high-resolution pQCT (HR-pQCT), can provide information on bone size, geometry, architecture, and tissue densities within the imaged portion of the long bone, and these parameters can be used to estimate bone strength. Moreover, both pQCT and HR-pQCT can distinguish trabecular (spongy bone) and cortical (compact bone) bone properties. 7 It should be noted that peripheral imaging techniques can measure long bones in the upper and lower extremity only, and thus, the obtained information is limited to the assessment of long-bone growth and development. However, this information is important because long-bone ends are susceptible to fractures during growth and later in life.8,9
In this review, we will first briefly describe long-bone growth and structural development in terms of alterations in length and size in addition to mineral mass and density. This is followed by a description of our abilities to assess bone properties by medical imaging tools. These considerations are important for our understanding of factors that contribute to skeletal growth, bone fragility, and the development of strong, healthy bones. Evidence of these factors will be summarized in the following sections.
Bone Growth Processes
There are 2 distinct skeletal growth processes in humans. Flat bones, such as the scapulae, are formed by a process called (intra)membranous ossification. Long bones, such as the femur, develop by a combination of membranous and endochondral bone formation. 10 Because it is the long bones that are most susceptible to fragility fractures, a brief discussion of long-bone growth is warranted. During childhood and adolescence, bone structure is altered by growth in length and size (eg, width or area). 11 Bone growth in length occurs at the growth plate (also called the epiphyseal plate), located just before the long-bone end (epiphysis). At the growth plate, cartilage is replaced by bone, and this process is known as endochondral ossification. A child’s bone lengthens when bone formation proceeds outward from these growth plates. To maintain the structural stability of growing long bone, the bone diameter needs to enlarge along the long-bone shaft.11,12 Growth in diameter occurs by apposition of new bone on the outer (periosteal) bone surface of the cortex. Concurrently, resorption in the inner (endosteal) bone surface excavates the marrow cavity in the tubular long-bone shaft. When periosteal apposition is greater than endosteal resorption, the cortical wall thickens or drifts farther away from the bone center in bone shafts. 13 To preserve long-bone shape (wider ends and narrower shafts), the long bone undergoes metaphyseal inwaisting in the region between bone end and shaft (metaphysis). 13 Metaphyseal inwaisting is achieved by periosteal resorption and endocortical apposition of bone. 11 Overall, growth in length and size are associated with increases in bone mass and density and constant alterations in bone structure.
Changes in skeletal length and size are closely tied to changes in bone mass, with approximately one-quarter of adult total-body bone mineral accrued during the 2 years around the adolescent growth spurt. 14 These structural and material changes are under mechanical regulation and influenced by the hormonal environment.6,10,15 During growth, bones must continually adapt their structure and mass to withstand loads from increases in bone length (ie, stature), muscle mass, and external forces. 11 However, the tempo, timing, and extent of such adaptations are also closely regulated by systemic hormones.16,17 Changes in the bone mass, size, and structure are modified by 3 distinct processes: growth, modeling, and remodeling. 18 During the life span, a single process may dominate at certain times, or the 3 processes may function concurrently at other times. In the immature skeleton, all 3 processes may be active simultaneously, with each having a different function.
Growth is the expression of the genetically programmed, hormone-mediated process of enlargement for the entire skeleton without regard to concurrent changes in shape that may be occurring regionally in response to local loading factors. In humans, skeletal growth is largely completed before the third decade of life.
Modeling is the main process altering the shape and mass of the skeleton. 10 Modeling represents a regional response to specific loading conditions, whereby new bone is formed without prior resorption, resulting in a change in shape and size of bone. Because young bone has a greater potential for adaptation to loading than aging bone, 19 the modeling process occurs primarily during growth and results in a net gain in bone over time. 20
Remodeling, although present in young individuals, is the predominant process modifying bone shape and mass in adults. It allows for the replacement of fatigue-damaged bone with new bone. 21 In addition to serving a preservation function, it provides a mechanism for the maintenance of calcium homeostasis. The remodeling cycle begins with an activation phase, followed in sequence by a resorption phase and a formation phase, which are coupled. 10 In mature bone, bone remodeling results in a net loss of bone over time because newly formed bone never completely replaces the bone that has been resorbed, resulting in a decrease in bone mineral mass (ie, bone mineral content, BMC) and alteration in bone structure. 22 In contrast, growing skeleton is able to gain bone mineral from modeling or remodeling. 18 Finally, bone remodeling is an integral part of fracture healing in both the growing and mature skeleton. 23
Measuring Bone Mass and Strength in Growing Skeleton
Medical imaging tools enable noninvasive assessment of bone properties, which can be used to estimate bone strength development during growth. Bone strength can be defined as the bone’s ability to resist fracture at a given loading condition. The strength of a long bone depends on the amount of bone mass, the spatial distribution of the bone mass (eg, bone size, geometry, and architecture), and the bone’s material properties.6,24
The clinical assessment of bone strength in pediatric studies previously relied on DXA-derived BMC (g) or areal bone mineral density (aBMD, g/cm2). 7 Because of a high correlation between actual skeletal mass and total-body BMC, the term bone mass is often used synonymously with BMC. 25 Although DXA-derived bone mass and aBMD predicts experimentally derived bone strength (eg, failure load),7,26,27 aBMD is a poor indicator of fracture risk.28,29 Fractures unrelated to aBMD could be a result of deterioration in bone size, structure, and strength, which 2-dimensional (2D) DXA measures cannot detect. Because of this 2D limitation, DXA-derived aBMD is influenced by bone size, and individuals with larger bones have (artificially) higher aBMD. Therefore, bone mass (BMC) rather than aBMD should be reported in pediatric DXA studies. 30 Limitations aside, a low effective radiation dose (eg, about 10 µSV for a whole-body scan 31 ) from DXA scanning makes DXA suitable for monitoring bone mass changes in growing skeleton, especially during the pubertal growth spurt when bone mineral is accrued rapidly.14,32 Figure 1 illustrates a total-body BMC change from 8 to 14 years of age in a healthy girl. During 6 years of growth, her total body BMC more than doubled, from 937 to 2098 g.
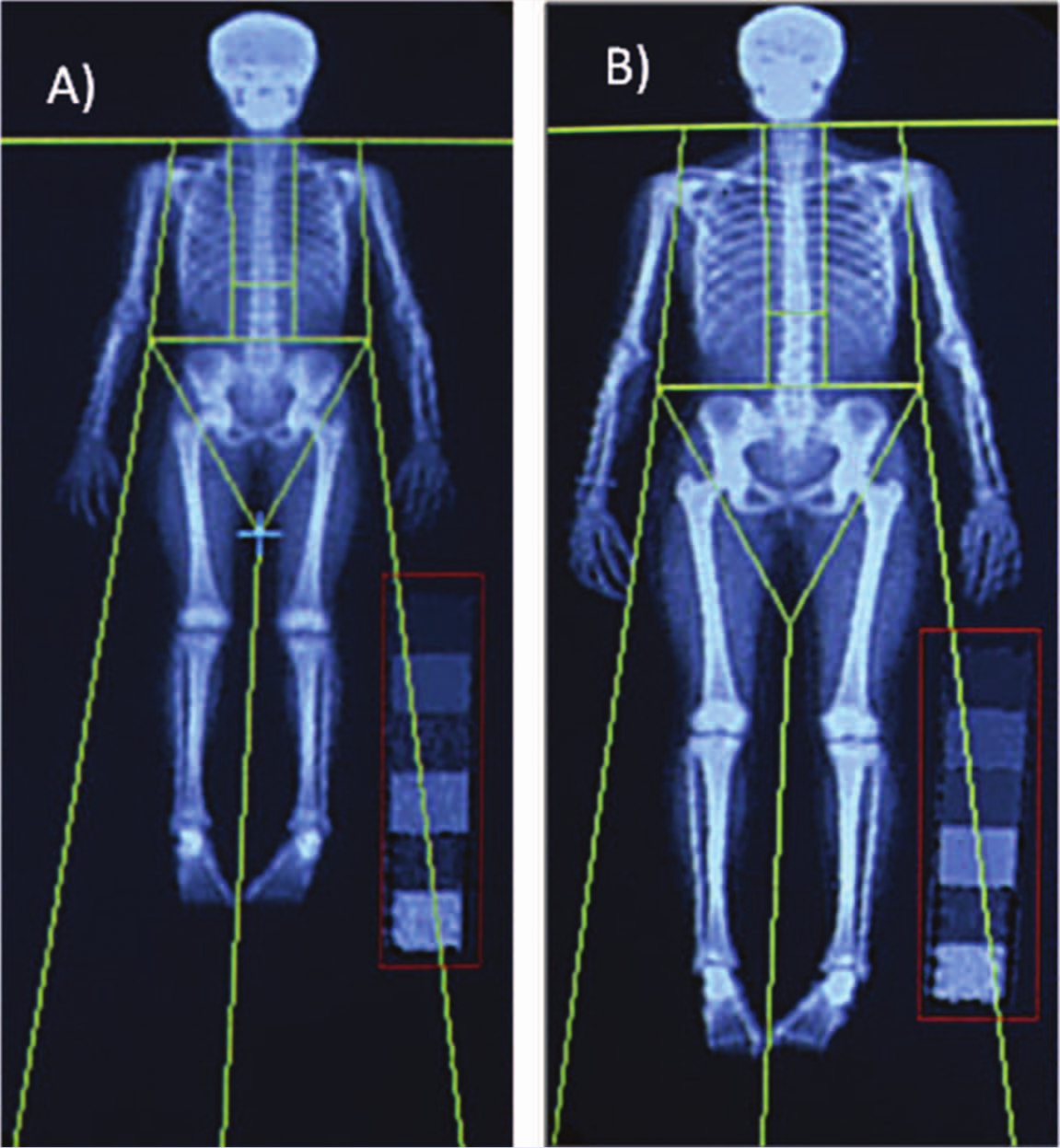
Whole-Body DXA Scans From a Female PBMAS Participant Illustrates 6 Years of Skeletal Growth From (A) 8 Years to (B) 14 Years of Age.
3D Imaging techniques, such as pQCT and HR-pQCT, are common research tools. Peripheral QCTs provide (volumetric) information of bone structural and densitometric properties from total, trabecular, and cortical compartments7,33 (Figure 2). Effective radiation doses are low for pQCT (about 0.3 µSV per scan) and HR-pQCT (about 3 µSV per scan), making these techniques suitable for pediatric studies.34,35 Volumetric density is independent of bone size and thus can be assessed in pediatric studies. 36 However, the reliability of cortical density assessment depends on the cortical thickness and resolution of the obtained images.37,38 Cortical density can be measured at long-bone-shaft sites using pQCT (resolution commonly 400-600 µm), whereas HR-pQCT (resolution 82 µm) is needed to assess cortical bone properties from thin cortex at the wrist (distal radius) and ankle (distal tibia).35-37 High-resolution imaging also enables assessment of other microarchitectural characteristics of bone, such as cortical porosity, trabecular number, and trabecular thickness at the distal radius and tibia.39,40 HR-pQCT can also be used in conjunction with engineering-based computer simulation, known as finite-element (FE) modeling, to estimate bone strength. This method evaluates how a structure with a complex shape and varying tissue properties behaves when subjected to loading. Its basic premise is to divide a complicated object into a finite number of small manageable pieces (elements), whereby the behavior of each element can be described mathematically and evaluated computationally. 41 FE modeling of imaged bone provides estimates of bone failure load and whole bone stiffness (flexibility of bone), 27 which previously were only attainable via destructive mechanical testing of cadaveric bone samples. The FE method also provides unique information impossible to measure experimentally, such as stresses and strains inside whole bones. A specific advantage of FE modeling is that bones can be analyzed multiple times with different loading directions and bone properties to simulate how whole bone will behave under different conditions and disease states.
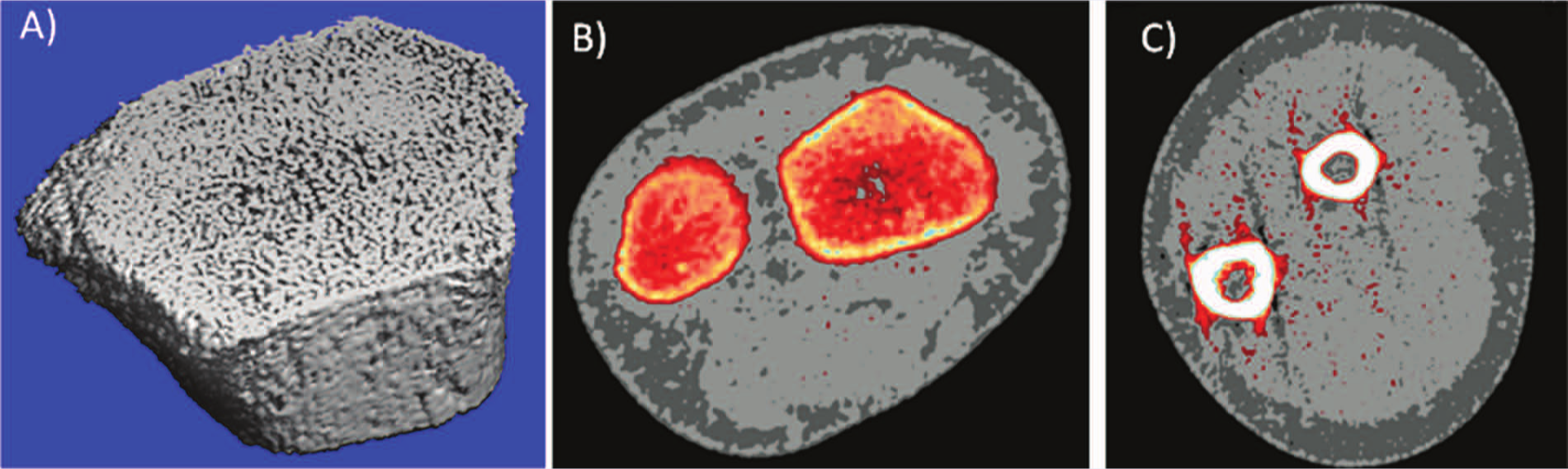
A. High-Resolution Peripheral Quantitative Computed Tomography (HR-pQCT) Image Illustrating Trabecular Microarchitecture and Thin Cortical Shell at the Distal Radius (Metaphysis) From a 9-Year-Old Girl. B. Cross-sectional pQCT Image of the Wrist Demonstrating Ulna (Left) and Radius Metaphyses and Surrounding Soft Tissue. C. pQCT Image of the Forearm Illustrating Cortices of Ulna (Left) and Radius Shafts (Diaphyses) and Surrounding Muscle (Light Gray) and Subcutaneous Fat (Darker Gray) Tissues From the Same Girl Measured 10 Months Prior to HR-pQCT Scanning. Red Streaks in the Forearm Image Are Minor Movement Artifacts That Can Be Excluded From the Image During the Scan Analysis Process.
Skeletal Growth and Bone Fragility
Prospective follow-ups show that bone mineral mass more than doubles between 8 and 15 years at total-body and different skeletal sites.14,42 The peak gains in BMC at the lumbar spine, femoral neck, and total body occur at about age 13.0 for girls and 14.4 years for boys, which is explained by the earlier onset of sexual maturation in girls. 14 Longitudinal DXA data from the Saskatchewan Pediatric Bone Mineral Accrual Study (PBMAS) indicated that in the 3 years around peak height velocity (ie, growth spurt), about 30% of BMC in the total body and lumbar spine was laid down and about 20% of femoral neck BMC was accumulated.14,43 These data are consistent with other studies showing that bone mineral accrual increases progressively in early childhood44,45 and then accelerates during adolescence.42,45 In a PBMAS cohort, both girls and boys achieved about 90% of their final adult height at the time of the peak height velocity but had only achieved about 57% of their estimated adult total-body and lumbar spine BMC and about 70% of the estimated femoral neck BMC values. 46 For all sites, peak velocity in BMC occurred over a year later than the growth spurt in both girls and boys. 46 This lag time between mineralization and rapid growth in bone length and size has important clinical implications because this dissociation may partly explain increased fracture incidence during adolescence.38,47-49
During the adolescent growth spurt, the most common fracture site is the distal forearm.47,50,51 It is poorly understood why fractures are common during the adolescent growth spurt. 52 DXA-derived whole-body BMC continues to increase during puberty and thus cannot explain this temporary increase in bone fragility. 42 Cross-sectional pQCT evidence indicates that total bone area at the distal radius increases across puberty but not always enough to offset the external load from a fall.38,53 It is assumed that the rapid metaphyseal inwaisting results in a thin, weak cortex predisposing to fracture if a child falls.11,38 To support this assumption, evidence from HR-pQCT comparison indicated lower cortical thickness and bone volume fraction and higher cortical porosity in boys and girls around the peak incidence of forearm fractures.53,54
Growth during infancy, childhood, and adolescence is important for bone health later in life. Infant and early childhood growth predicts adult bone mass independently of adult lifestyle. 2 Both low birth size and poor childhood growth are directly linked to the later risk of hip fracture. 55 The evidence of mineral accrual during adolescence indicates that the population variance in bone mass and in structural traits is probably largely established by the end of the second decade.56-58 Pubertal timing seems to determine some variance in bone development.
Earlier maturation may be beneficial for bone development and protective for fracture risk in adolescence and young adulthood. Evidence from the PBMAS cohort, followed for 15 years from adolescence to young adulthood, showed that late-maturing girls gained less mineral in their total body than their early or average maturing peers. 59 However, maturation timing was not associated with bone mass accrual in boys or development in the estimated structure and strength at the hip (proximal femur) in either sex in the PBMAS cohort.56,59 In contrast, maturational timing predicted bone mass negatively at the age of 16 in both boys and girls in a shorter prospective follow-up. 60 Cross-sectional evidence from a large cohort of young adult men also suggested that maturational age is a negative predictor of both cortical and trabecular bone size and volumetric BMD. 61 It is important to note that later maturational age was linked to fractures in this cohort. 61 Recent evidence from studies of young women lends credence to this finding, showing later pubertal timing in women who had suffered one or more fractures during childhood and adolescence when compared with their fracture-free peers. 62 Association between fractures and later maturity might be related to the observed deficits in trabecular density, thickness, and estimated strength (stiffness, failure load, and apparent modulus) in the distal radius or tibia in the women experiencing fractures when compared with their peers who did not.62,63 Based on this evidence, the earlier maturing girl may have an advantage in her bone mass and strength development over the late-maturing girl, and this advantage may be protective against fractures. However, site- and sex-specificity in the role maturational timing plays in adult bone structure and strength needs to be addressed in future follow-ups.
Optimizing Bone Mass and Strength Development During Growth
To optimize bone strength development, we need to identify those modifiable factors that can enhance or retard the attainment of bone mass, structure, and strength during growth. Bones adapt their strength according to the mechanical forces they experience. Studies with healthy children and adolescents highlight the potential of modifiable factors such as physical activity, lean mass, or muscle cross-sectional area in determining bone mass accrual, bone size, tissue density, and estimated strength. Key factors that determine and influence the development of bone mass, structure, and strength are listed below.
Nonmodifiable Factors
Genetics and Sex
Evidence from familial resemblance and twin studies indicates that genes control about 50% to 85% of the variance in bone mass or aBMD 64 but a much smaller (<20%) proportion of the variance in rate of bone loss 65 or fractures.66-69 Genetic influences on the skeleton are thought to contribute mainly to the acquisition of bone mass during childhood and adolescence,70,71 possibly by interacting with environmental factors such as physical activity and nutrition. The trait variance in the population is largely the result of diversity in genetic makeup rather than lifestyle.64,72 However, at the individual level, environmental effects such as muscle paralysis 73 or exercise 19 have profound effects on bone structure and estimated strength.
As the heritability of bone mass is stronger than aBMD, some of the familial resemblance in aBMD may simply reflect the heritability of body size. 74 Only 1 study has reported the genetic influences on bone size, showing a heritability estimate of 27% for the cross-sectional area at the distal radius in young adults. 74 Heritability estimates for trabecular and cortical bone density has been shown to be higher: 40% to 60% depending on the bone site. 75 These results are in accordance with a meta-analysis of genome-wide scans, which concluded that genetic regulation of bone mass is sex and site specific. 76 Part of the collinearity between heritability of body and bone size (or mass) may also be related to lean mass and muscle size and their development.77-80 The important aspect regarding bone fragility prevention is that these traits can be enhanced by a physically active lifestyle and healthy diet.
Fractures of the wrist and hip have strikingly different age- and sex-specific incidence patterns. 9 To optimize preventive efforts, we need to understand how bone strength develops and declines for both sexes. Small or nonexistent sex differences in BMC, density, and structure at axial or appendicular skeletal sites have been reported in childhood and adolescence.12,46,81-89 Observations appear to be specific to the measured site and technology used and how body size and composition—especially lean mass or muscle size—were controlled for in the analysis. The most comprehensive evidence of sex differences in bone development during pubertal growth comes from longitudinal PBMAS data. After adjusting for differences in maturation, height, and body composition, BMC development in the total body and femoral neck were noted to be slightly greater in boys than girls across puberty. 46 Structural analysis of hip bone scans from the same cohort indicated a greater estimated strength (section modulus) in femoral neck across the pubertal growth in boys when compared with girls but a smaller increase in the femoral neck area after the growth spurt. 85 This evidence of a greater bone mineral gain (relative to muscle) in the female skeleton occurring later in the pubertal growth period is supported by cross-sectional comparisons between male and female participants in studies.77,84,90-92 It has been suggested that this “extra” bone mineral provides a calcium reservoir for reproductive purposes.22,91-94 Observations of similar cortical thickness between sexes have indicated that girls have either endosteal apposition 95 or diminished resorption at the endosteal (inner) bone surface in puberty. 12 Because women may gain more endosteal than periosteal bone, their bones strengthen less than in men, who tend to have more periosteal bone formation and thus greater increase in overall bone size during the growth. Absolute sex differences in adult BMC and bone size may also reflect greater overall body growth because of the longer pubertal growth period in boys than girls. In women, earlier completion of longitudinal growth with epiphyseal fusion and earlier inhibition of periosteal apposition likely results in smaller bone size and generally weaker bone when compared with men.12,95
Modifiable Factors
Physical Activity
It is important to note that modeling and remodeling described earlier in the article are essentially under the control of the mechanical strain environment (such as strain magnitude, rate, and frequency). This functional model of bone development is based on the mechanostat model.22,96,97 In this model, bone cell action is coordinated by the mechanical requirements of the bone.96,97 If bone is not exposed to mechanical loading, the lack of stimulus (strain) results in increased remodeling, with subsequent loss of bone mineral and likely alterations in bone structure. Mechanical loading that is familiar to bone will be sufficient to maintain bone mass and structure, thereby maintaining remodeling and modeling in a steady state. Loading that produces an unusual strain environment to bone tissue can initiate a remodeling and/or modeling response. In the mechanostat model, the bone response to an altered strain environment is based on physiological set points, which act as thresholds for the initiation or inhibition of bone modeling and remodeling. Increases in stature, body, and muscle mass during growth will, in part, increase the forces placed on bone, leading to greater bone strain and adaptation. 96 We have illustrated this concept of functional bone adaptation in Figure 3. 98 Similarly, the mechanostat model could be used to explain bone adaptation to increased bone loading and related increase in bone strain (tissue deflection) resulting from physical activity. For example, if a girl unaccustomed to gymnastics practices handsprings, her bones at the wrist are exposed to a new loading environment with increased strains. In response, these bones will model and adapt to the new loading environment by increasing bone mass, bone size, and modeling bone structure. After adapting bone structure and mass, the bone will be better able to resist applied loads, and strains will return to a steady state (Figure 3). Progressively increased training stimulus is needed for continuous bone adaptation, thereby avoiding acute and chronic injuries related to intensive training. 99 Bone adaptation can be measured noninvasively by assessing changes in bone properties using earlier described imaging techniques (Figure 3). Bone strain characteristics cannot be measured noninvasively or with these imaging techniques. However, there are some promising biomechanical modeling techniques that will likely enhance our ability to use noninvasive imaged data in modeling simulations to estimate bone strain in vivo. 100
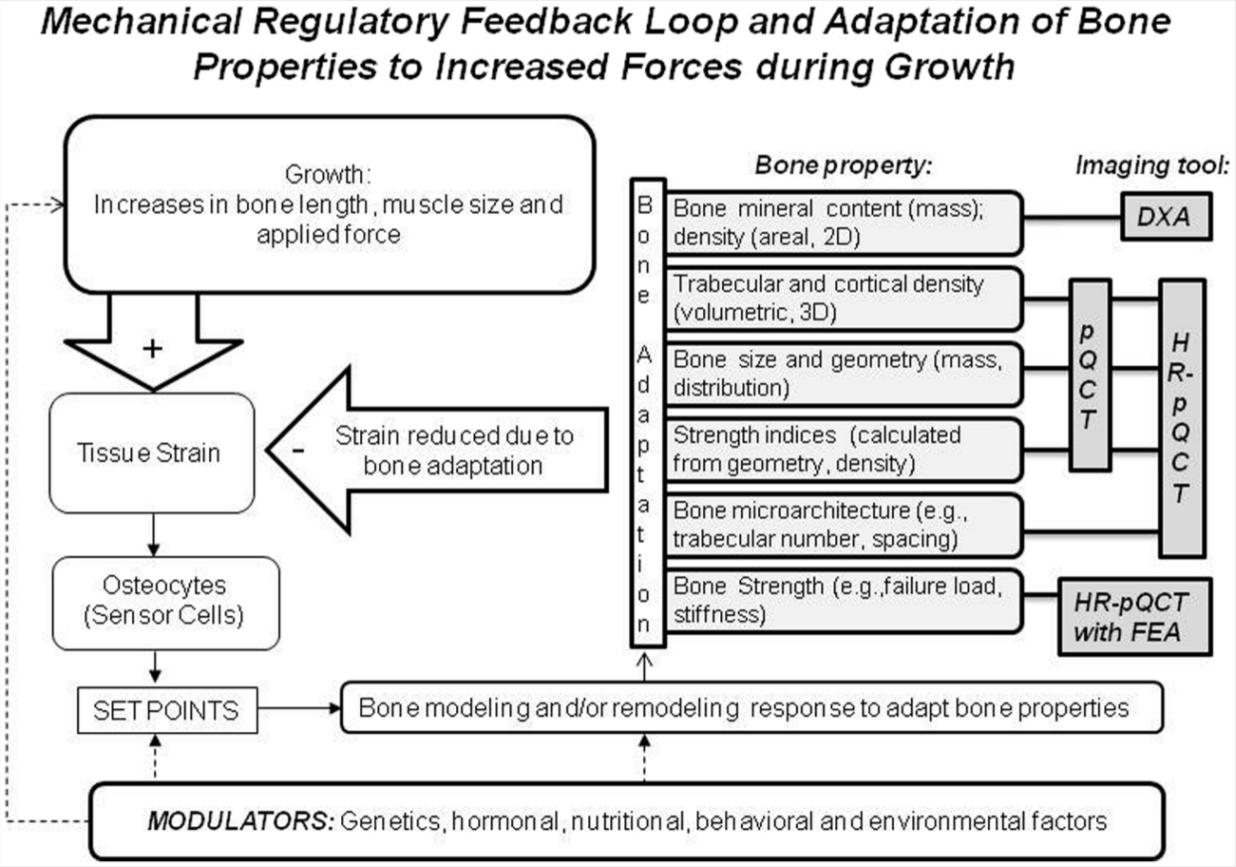
A Functional Model Illustrating the Regulation of Bone Strength Development During Growth. The Model Is Based on the Mechanostat Theory (Modified From References 6 and 98).
Observational studies have provided evidence of greater bone mineral mass, not only in child and adolescent athletes involved in gymnastics or other high-impact activities but also in children and adolescents who simply had a more physically active lifestyle.14,101-103 Exercise trials have supported this evidence by showing a 3% to 6% greater bone mass accrual in children randomized to exercise intervention when compared with the bone accrual in the control group.104-109 Weight bearing and high-impact activities such as jumping exercises and step aerobics have shown bone mass benefits at the loaded bone sites of the lower extremities and lumbar spine. 110 However, what constitutes the optimal exercise program for bone adaptation remains unclear. 110
Evidence remains inconclusive if the bone mass benefit acquired during the growing years persists into adulthood. To date, studies examining the effects of physical activity during childhood and adolescence on adult bone mass have been limited to retrospective assessments of physical activity111-113 or earlier measured youth physical activity114-117 to adult bone mineral status. Follow-up data of retired athletes have provided evidence of both sustained116-119 and partially lost 120 bone benefit after reduced or ceased activity, whereas exercise-induced bone gain has been reported to persist 1 to 8 years after exercise intervention in prepubertal or early pubertal children.121-123
Prospective observations from the Saskatchewan PBMAS cohort showed a 9% to 17% greater bone mineral accrual in physically active children when compared with their less-active peers. 14 These studies have lent credence to the concept that physical activity in prepuberty and early puberty provides a unique opportunity to enhance bone accrual and peak bone mass. However, even if exercise during the growing years affects bone accrual, the importance of these effects from a clinical perspective depends on their permanence. The long-term implications of these benefits on adult bone mass can only be established with prospective longitudinal studies that follow individuals from childhood to adulthood. The follow-up data of these same individuals in young adulthood suggest that these benefits in bone mass persist into early adult life. 124
The effect of physical activities and loading on growing bone structure and strength relies mainly on observational evidence. 125 Unilateral tennis studies that compared side-to-side differences between players who begin their career prior to or after the growth spurt have provided the strongest observational evidence that increased BMC is a result of enlarged bone size, which leads to greater estimated strength in the long bone. 19 Cross-sectional comparisons in young and retired gymnasts supported these findings.126-132 Comparisons of the side-to-side differences in bone mass, structure, and strength between unilaterally loaded tennis players and their controls also pinpointed that the bone benefit is greater if physical activity was initiated prior to skeletal maturation.19,133 Specifically, the benefit in estimated bone strength was more than twice as much (ie, 26%) in women who had begun playing prior to menarche when compared with the 11% strength benefit in the loaded site of the women who started the unilateral activity at a later age. 19 There are no randomized controlled trials that have assessed the effect of exercise on bone structure and strength in the upper extremities. 125 To date, evidence from randomized controlled trials assessing bone structural and strength changes is limited to investigating bone adaptation in the lower limb. 134 In this 16-month trial, an additional 15-minute physical activity resulted in an increase in bone strength at the distal tibia (measured by pQCT) in prepubertal boys. 134 This intervention included a novel bone loading component of 3 short (3-minute) jumping sessions during the school day. 134 When bone strength adaptation was estimated in the proximal femur from DXA images, intervention benefit was noted in prepubertal and early pubertal girls only. 135 Further experimental evidence is needed to clarify site, maturity, and sex specificity in bone structural and strength adaptation to exercise during growth.
Muscle and Fat Tissue
Pediatric bone studies have been in the forefront to provide evidence of the importance of lean mass136-139 and muscle cross-sectional area140-142 on bone mass accrual and strength development. Lean mass and muscle size are commonly used as surrogate measures for forces from mechanical loading, and these traits have been associated with enlarged bone mass, size, and strength.143,144 In contrast, higher fat mass has been associated with smaller and less-dense cortical and trabecular bone and lower bone strength in young girls.145,146 In terms of absolute bone strength, overweight children seem to be at an advantage, but this is because of adaptation to their larger muscle size, not excess body fat. 147 These findings are in accordance with the mechanostat model, which postulates that the growing skeleton adapts its mass, structure, and strength to keep loading-induced strains within an acceptable range79,141,148,149 (Figure 3). The age of peak lean tissue velocity precedes the peaks in the estimated bone size and strength at the proximal femur. 150 These observations were derived from DXA images, using hip structure analysis, 150 and they support the theory that muscle development is an important factor in bone strength development. However, there is a lack of comprehensive longitudinal data from both sexes linking bone structural and strength development in growing years to bone strength in adulthood.
Nutrition
In addition to physical activity, diet during the growing years has been associated with bone mineral accrual in childhood and adolescence.42,114,151-153 Two important nutritional aspects related to bone mineral have been noted in the past years: (1) a shift in emphasis from calcium to vitamin D (especially D3 form) influences on bone154,155 and (2) an interest in dietary patterns, in particular the importance of fruit and vegetable consumption on bone health.156,157 The average calcium accretion is 92 to 210 mg/d in 9- to 18-year-old boys and girls, 158 and bone calcium accretion can peak at 300 to 400 mg/d. 42 The current recommendation of 1300 mg calcium per day for 9- to 18-year-olds meets the needs for skeletal growth in practically all adolescents. 159 Calcium supplementations produce a small (1% to 2%) effect in the upper-limb aBMD. 160 This small increase is probably not likely to result in a clinically significant decrease in fracture risk in the growing skeleton; thus, current evidence does not support the use of calcium supplementation in healthy children as a public health intervention. 160 However, according to the Institute of Medicine 2011 guidelines, adolescent girls continue to be a group at risk for low calcium intakes from food sources. 159
The recommended intake of vitamin D is 600 International Units (IU) per day for children and adolescents in North America. 159 Recommendations for vitamin D intakes from dietary sources are complicated because vitamin D is also synthesized in the skin through sunlight exposure, which varies from person to person. Minimal sun exposure was assumed when the current vitamin D recommendations were established. 159 Inconsistent vitamin D tests used in studies and variance in the description of normal and abnormal ranges for vitamin D (25OHD) levels in children further challenges vitamin D recommendations. 161 In northern latitudes without assurance of adequate sun exposure, requirements and recommendations for vitamin D should probably be higher than those currently in place.154,162 Estimated mean daily vitamin D intakes in PBMAS participants have been relatively constant: 200 to 240 IU for girls and 280 to 320 IU for boys. These are comparable to Canadian youth intakes 158 and lower than the recommended dietary allowance for vitamin D (600 IU/d). 159 The evidence of dose, time, and form of vitamin D for bone strength development and maintenance requires further evidence, especially from children and adolescents with limited exposure to sunlight (eg, northern latitude) and poor nutrition as a result of socioeconomic factors.
Some investigations have indicated a positive relationship between fruit and vegetable consumption and bone mineral accrual.156,157 This evidence has provided some support to the theory that a low-fruit and low-vegetable diet may lead to chronic, low-grade metabolic acidosis, which may have detrimental effects on long-term bone health.163-165 On the other hand, high fruit and vegetable consumption may reflect overall healthy dietary patterns. In addition, protein represents another key nutrient for bone health. Prospective studies have suggested that high protein consumption is associated with greater bone mass and lower incidence of osteoporotic fracture. 163 Furthermore, dietary protein intake of ~2 g protein/kg bodyweight was positively associated with cortical bone size and estimated strength in the radial shaft of healthy children aged 6 to 18 years. 166 Milk and milk products provide not only protein but also calcium and vitamin D, all of which are important for the developing skeleton. 163 However, North American adolescents have shifted their beverage intake away from milk to sugary, carbonated beverages. 167 High intakes of these beverages may reduce bone mass accrual in adolescents. 167
Smoking
A few studies have shown a negative association between bone mineral and smoking during growth and adulthood in young men114,168 and women, 169 whereas others have not demonstrated such a relationship. 79 Smoking during adolescence was negatively associated with aBMD only in men but not in women 114 ; therefore, this relationship may be gender dependent or related to the amount of cigarette consumption. 168 In elderly smokers (both men and women) in whom the effect of smoking has accumulated over several years, aBMD is reduced, bone resorption markers are increased, and fracture risk is elevated. There is limited evidence of an association between smoking and bone size, structure, and volumetric density. In a cohort of young Swedish men, daily smoking was associated with lower cortical thickness, 170 whereas no association was reported in older Spanish women. 171
Recommendations
There are still many questions remaining about the complicated mechanisms controlling bone mineral accretion and structural development during the growing years. Prospective studies and well-designed trials are needed to assess adaptation in bone structure and strength, the role of nutrition in this process, and whether bone adaptation related to physical activity during growth is maintained into later years irrespective of adult activity levels.
From a public health perspective, perhaps the greatest questions have to do with the development of strategies directed at relating a condition that appears in elderly people to a largely disinterested teenage population that will not be at risk for 40 or 50 years. The ultimate target population for the prevention of osteoporosis may be the young and not the elderly population. 172 Taking into account the fact that our knowledge of the complicated mechanisms controlling bone mass and structure is still incomplete, on the basis of what we do know, it is possible to offer some sound and prudent lifestyle advice to young people regarding how to optimize bone strength development in order to reduce the risk of bone fragility and fractures later in life.
For overall health benefits, WHO and several national guidelines recommend at least 60 minutes of moderate- to vigorous-intensity physical activity daily for children and adolescents.173-176 In the US and Canada, this recommendation includes activities that strengthen muscle and bone at least 3 days per week. Examples of bone-strengthening activities include the following: running, jumping rope, basketball, tennis, soccer, playing tag, hiking, and hopscotch. 176 It has to be noted, however, that this recommendation relies on evidence from interventions limited to bone mass (or areal density) assessment and are supported by observational data linking greater bone mass with activities that generate relatively high ground-reaction forces on the lower extremities.177,178 With further evidence from exercise trials assessing bone structural and strength adaptations in both sexes during skeletal growth, we anticipate that future recommendations will be based on observed dose relationships and include site-specific exercises to optimize bone strength development at fracture-prone sites, including the wrist and forearm. Finally, the accumulating evidence on the negative consequences of sedentary lifestyles on health will likely provide sufficient knowledge to formulate a basis of recommendations on physical inactivity in the near future. 173
In regard to the current knowledge of physical activities beneficial for bone development in the young, the following fundamentals should be kept in mind: (1) the skeletal response to exercise is greatest at the loaded site and (2) to cause an adaptive response, the training stimulus must be greater than that habitually encountered. On the basis of these fundamentals and the review of the pertinent literature, the following recommendations can safely be offered to the public at large.
An individual should make a lifelong commitment to physical activity at an early age. Growing bones respond to mechanical loading by the addition of new bone and/or a strengthened bone structure. The growing skeleton has better ability to adapt to mechanical loading than the mature skeleton.
Weight-bearing activities that provide impact loading like skipping, aerobics, squash, and basketball are beneficial to bone mass and strength development—especially at the lower extremity bones—than weight-supporting activities such as swimming or cycling.
A variety of physical activities (such as gymnastics) that can be short in duration and include diverse, impact-type loading stimuli to several skeletal sites, including non-weight-bearing upper extremities are recommended.
Progressive increased training allows musculoskeletal adaptation and prevents overloading and related injuries.
Activities that increase muscle size and strength should be encouraged because these can be osteogenic.
Inactivity and immobility should be avoided. When this is not possible because of sickness or injury, even brief periods of physical activity will likely help conserve bone mineral.
The responsiveness of the growing skeleton to physical activity is dependent on the sensitivity of bone to circulating hormone levels and nutritional adequacy. This has important implications for exercise prescription in adolescents, which leads to the following important recommendations.
Children should have a diet of nutritious foods that will meet the recommended dietary intake for calcium and vitamin D, provide adequate but not excessive protein, and limit the intake of sodium and caffeinated beverages (soft drinks and coffee).
A well-balanced diet that is sufficient to meet the energy demands of growth and physical activity should be encouraged.
Cigarettes should be avoided: they may interfere with the attainment of an optimum level of bone mineral and structure following skeletal maturation.
Footnotes
Acknowledgements
Dr Kontulainen acknowledges financial support from the Canadian Institute of Health Research and Saskatchewan Health Research Foundation New Investigator award.
