Abstract
Prostate cancer is the most common cancer in men in the United States excluding skin cancer. Androgen deprivation therapy is initially effective for most patients with metastatic prostate cancer; however, castration-resistant prostate cancer (CRPC) usually develops when disease progresses despite conventional hormone therapies. There are few treatment options for progressive CRPC. Until recently, only one treatment, docetaxel, had been shown to prolong survival, and no agents had been shown to improve survival in docetaxel-refractory disease. This changed in June 2010 with the U.S. Food and Drug Administration approval of cabazitaxel. Cabazitaxel is a new option for patients with CRPC whose disease progresses during or after docetaxel treatment (docetaxel refractory). Cabazitaxel is a novel taxane that has been shown to have poor affinity for the p-glycoprotein drug efflux pump, a major mechanism of resistance to docetaxel. In combination with prednisone, cabazitaxel was shown to significantly prolong overall survival by 2.4 months when compared with mitoxantrone in a large randomized phase III trial in metastatic CRPC patients previously treated with first-line docetaxel. The most common toxicities with cabazitaxel are neutropenia (88%), anemia (81%), thrombocytopenia (43%), fatigue (27%), nausea (23%), and diarrhea (11%). This review will examine the clinical development, efficacy, adverse effects, and suggested monitoring for this new agent. The combination of cabazitaxel and prednisone is an important new treatment option for men with docetaxel-refractory metastatic CRPC. However, this agent should be administered with close, appropriate monitoring, especially in men at high risk of neutropenia or diarrhea. Future research directions for this new agent will include exploring its role in combination with other agents and comparing with docetaxel for the first-line treatment of metastatic CRPC.
Prostate cancer is a leading cause of cancer morbidity and mortality and is a major public health issue in the United States and other industrialized nations. It is the most common type of cancer in men, with an incidence of 217 730 new cases in the United States in 2010, and was responsible for 32 050 deaths. 1 Prostate cancer can be diagnosed at early stages, also called localized or locoregional disease. Most men diagnosed with this locoregional disease have an excellent prognosis when treated with local therapy with either surgery, called a radical prostatectomy, or radiation as the primary therapy, with or without androgen deprivation therapy (ADT). ADT can be provided through surgical castration via orchiectomy or hormonal/medical castration therapy to achieve testosterone levels <50 ng/dL (<1.7 nmol/L) and, optimally, <20 ng/dL (<0.7 nmol/L) based on updated castration trials.2,3 Androgen deprivation agents that induce castration levels of testosterone in the blood include luteinizing hormone releasing-hormone (LHRH) agonists such as leuprolide and LHRH antagonists (Table 1). Additionally, antiandrogens, which directly block the effects of androgens such as testosterone, can be used in combination with medical or surgical castration; this method is also referred to as complete androgen blockage (Table 1). 4
Hormonal Therapies in Prostate Cancer 4 .
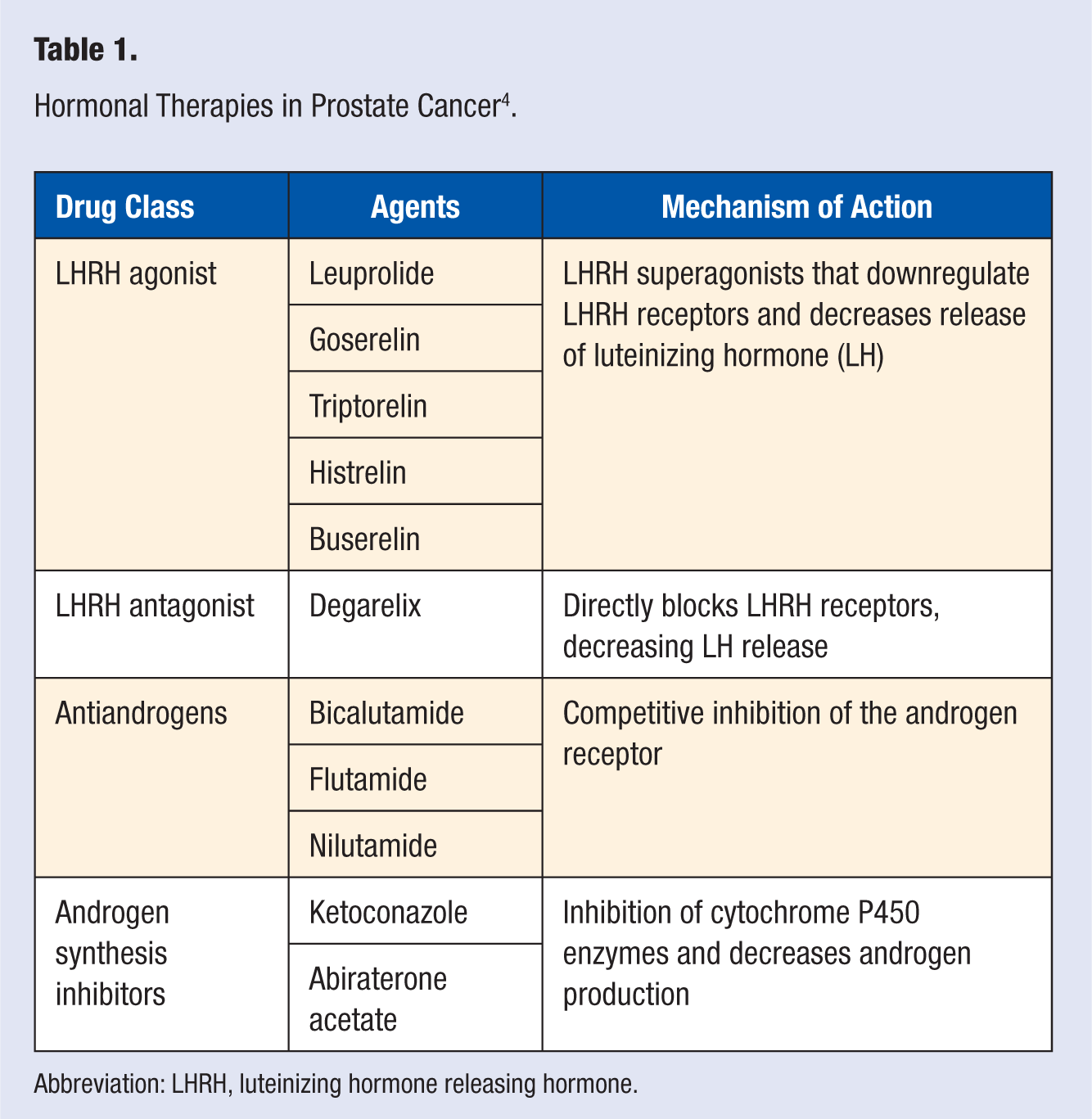
Abbreviation: LHRH, luteinizing hormone releasing hormone.
It [prostate cancer] is the most common type of cancer in men, with an incidence of 217 730 new cases in the United States in 2010, and was responsible for 32 050 deaths.
Although only 4% of men with prostate cancer are initially diagnosed with advanced, metastatic (also called stage 4), prostate cancer that has spread to distant sites outside the prostate, some men may also relapse after local, primary therapy for early stages of prostate cancer. Approximately 20% to 30% of men experience biochemical relapse after local therapy, as evidenced by a prostate-specific antigen (PSA) increase above the normal range of 0.2 ng/mL, and approximately 30% of these men with biochemical relapse develop metastatic disease recurrence within 10 years after local primary therapy. 5
In 1941, Huggins 6 first described an association between the beneficial effects of castration and the androgen (testosterone) dependence of prostate cancer. Hence, ADT, either as monotherapy with an LHRH agonist or complete androgen blockage with the addition of an antiandrogen agent (see Table 1), is the standard initial therapy for metastatic or recurrent progressive prostate cancer to reduce testosterone to castrate levels (<50 ng/mL). This is effective for most patients, as evidenced by PSA, a measure of tumor burden, which decreases to normal range values in about 70% of patients.7,8 Resistance to ADT usually develops in approximately 14 to 20 months after starting this initial hormonal therapy. 9 Two basic mechanisms have been proposed to explain this tumor resistance: the tumor may escape hormonal control by being composed of cells that are hormone independent or the tumor may be stimulated by extratesticular androgens from the adrenal glands. For patients who progress after initially responding to hormonal therapy, a second hormonal manipulation may be indicated. Examples include the addition of an antiandrogen, withdrawal of an antiandrogen in the setting of disease progression after an initial response, high doses of ketoconazole for inhibition of adrenal androgen production, or megestrol acetate. Often, the use of additional hormonal therapy such as ketoconazole is effective in inducing a PSA response with temporary symptom improvement; however, the response is usually of relatively short duration, such as about 2 to 3 months. 10 Invariably, the tumors become castration resistant and progress to what is referred to as castration-resistant prostate cancer (CRPC). Although the nomenclature of CRPC has been debated, CRPC generally refers to patients with progressive prostate cancer despite a castrate testosterone level (<50 ng/dL). 11
For patients with metastatic CRPC in whom multiple hormonal therapy options have failed, the standard of care is cytotoxic chemotherapy with docetaxel (in combination with prednisone). Docetaxel has shown a survival benefit of 2 to 3 months, when compared with mitoxantrone, another cytotoxic chemotherapy agent that has been used in CRPC. Among patients with metastatic CRPC treated with docetaxel, median survival is 18.9 months. 12 Until recently, no other treatment option had demonstrated an improvement in survival for patients with metastatic CRPC. Additionally, no second-line therapies had been approved by the U.S. Food and Drug Administration (FDA) to treat CRPC resistant to docetaxel therapy (docetaxel-refractory disease). On June 17, 2010, the FDA approved the taxane cabazitaxel (Jevtana; Sanofi-Aventis US LLC, Bridgewater, NJ) for the treatment of hormone-refractory CRPC previously treated with a docetaxel-containing regimen.13,14 The purpose of this review is to discuss the mechanism, clinical data that led to FDA approval, administration, adverse effects, monitoring, and future research directions for this newly available second-line chemotherapy option that has shown a survival benefit in metastatic CRPC.
Cabazitaxel Use in Metastatic Castration-Resistant Prostate Cancer
Mechanism of Action
Cabazitaxel is a semisynthetic taxane analog. It is prepared by semisynthesis with a precursor extracted from yew needles. Taxanes are anti-microtubule agents that stabilize microtubules by direct binding to β-tubulin subunits. This stops division in the mitotic phase of the cell cycle and results in apoptosis or cell death. In other words, cabazitaxel works to bind free tubulin to promote their assembly into stable microtubules but then inhibits the disassembly so that it ultimately freezes the cell within mitosis. 13 Other FDA-approved taxanes include paclitaxel, docetaxel, and albumin-bound paclitaxel. Paclitaxel was the first FDA-approved taxane, and docetaxel is a semisynthetic more potent version of docetaxel. Albumin-bound paclitaxel is a taxane, which is currently the only FDA-approved taxane for metastatic breast cancer, that was most recently developed to avoid the hypersensitivity infusion reactions seen with the original paclitaxel formulation. Although docetaxel is FDA approved for metastatic CRPC, its effectiveness is limited by the high substrate affinity for p-glycoprotein (P-gp), an adenoside triphosphate (ATP)–dependent drug efflux pump that decreases intracellular concentrations of the taxanes paclitaxel and docetaxel. It has been shown that cancer cells such as metastatic CRPC that express P-gp become resistant to docetaxel when they overexpress the multidrug resistance (MDR) gene that encodes for P-gp. This is a commonly implicated mechanism of taxane resistance. 15 Cabazitaxel was selected for development for metastatic CRPC on the basis of its poor affinity for P-gp and higher blood–brain barrier penetration compared with docetaxel and paclitaxel in preclinical models. Additionally, it did show activity in docetaxel-resistant cell lines in in vitro testing. 15
Pharmacologic and Pharmacokinetic Considerations
Cabazitaxel is approximately 89% to 92% bound to human serum proteins, including serum albumin and lipoproteins. After administration of the 25 mg/m2 dose, the volume of distribution is at least 4000 liters at steady state. It is extensively metabolized in the liver, mainly by the CYP3A4/5 (80% to 90%) and to a lesser extent by the CYP2C8 isoenzyme. Cabazitaxel does not appear to inhibit or induce the major CYP isoforms. Cabazitaxel is the main active circulating compound in plasma; however, there are 20 metabolites of cabazitaxel, mainly inactive and excreted in the feces (76%) and 2.3% as unchanged drug in the urine. The elimination half-life of cabazitaxel follows a 3 compartment model with α, β, and γ half-lives at 4 minutes, 2 hours, and 95 hours, respectively.13,15
Although other taxanes such as paclitaxel and docetaxel have a high substrate affinity for the MDR proteins, which can confer both constitutive and acquired resistance, cabazitaxel has a poor affinity for the ATP-dependent drug efflux pump p-glycoprotein. It also appears to have greater penetration of the blood–brain barrier, in animal studies, and this may also be because of its poor affinity for efflux pumps. 15
Although strong CYP3A4 inducers or inhibitors may be expected to affect cabazitaxel pharmacokinetics, no formal drug interaction trials have been conducted. Therefore, the use of strong CYP3A4 inducers or inhibitors, such as ketoconazole, erythromycin, or clarithromycin, should be avoided, if possible. The exception is prednisone at 10 mg daily, which was studied in combination with cabazitaxel in formal clinical trials, and does not appear to affect cabazitaxel pharmacokinetics. As cabazitaxel is primarily metabolized in the liver, hepatic impairment is likely to increase concentrations, although no trials in hepatic impairment have been completed at this time. The use in this population is listed as a precaution in the FDA package labeling.13,15 According to the National Cancer Institute clinical trials database, a study in this population is ongoing. 16
Adverse Effects, Safety, Pre-cautions, and Suggested Monitoring
Similar to other taxanes, the notable toxicities of cabazitaxel include myelosuppression, peripheral neuropathy, and hypersensitivity reactions. Additionally, the rates of neutropenia, febrile neutropenia, and diarrhea appeared to be higher than those reported for docetaxel in prostate cancer and were higher than the mitoxantrone comparator arm in the phase III clinical trial. 17 This may have been due to the taxane pretreated population in the phase III randomized clinical trial. Although peripheral neuropathy was higher in the cabazitaxel arm versus the mitoxantrone comparator arm (14% vs 3%), only 1% of patients in each group experienced grade 3 neuropathy in the phase III trial. 17
Neutropenia was the dose-limiting toxicity in clinical trials. Grade 3-4 (severe to life-threatening) neutropenia was reported in 82%, febrile neutropenia in 7%, and 5 patients were reported to have experienced fatal events from neutropenic sepsis in the phase III trial. The mitoxantrone arm, in comparison, reported a rate of grade 3-4 neutropenia and febrile neutropenia of 58% and 1%, respectively. According to the FDA-approved manufacturer’s prescribing information (labeling), primary prophylaxis with a colony stimulating factor (G-CSF), such as filgrastim or pegfilgrastim, should be strongly considered for patients at “higher risk” of experiencing neutropenia, especially those older than 65 years, who were found to be more likely to experience neutropenia and febrile neutropenia in the phase III clinical trial. The definition of “high risk” is not defined in the labeling, and it should be noted that the American Society of Clinical Oncology (ASCO) guidelines for blood cell growth factors do not currently recommend primary prophylaxis with G-CSF such as pegfilgrastim over the alternative option of a dose reduction (in noncurable cancers), because the risk of febrile neutropenia with cabazitaxel is not more than 20%. The ASCO guidelines consider patients at higher risk of experiencing febrile neutropenia to also include those with lower bone marrow reserve because of being previously pretreated with several other cytotoxic chemotherapy regimens and/or experiencing febrile neutropenia with past treatments. 18 According to the manufacturer’s prescribing information, frequent complete blood count monitoring is also recommended, at least weekly for the first 2 cycles, and cabazitaxel is contraindicated with neutrophil counts <1500/mm3. 13
Deaths related to diarrhea and electrolyte imbalance occurred in the randomized phase III clinical trial, and all-grade diarrhea was reported in 47% in the cabazitaxel arm (grade 3 or 4 = 6%). The FDA-approved package labeling includes dose modifications for grade 3-4 neutropenia and/or diarrhea (Table 2).13,17 Severe (grade 3-4) hypersensitivity infusion reactions were reported in 2 patients (<1%) in the phase III clinical trial. Severe hypersensitivity reactions are characterized by rash, pruritis, hypotension, dyspnea, angioedema, and/or bronchospasm. Intravenous premedication with histamine-1 (such as diphenhydramine 25-50 mg) and histamine-2 antagonists (such as ranitidine 50 mg, or equivalent) as well as dexamethasone 8 to 10 mg, 30 minutes before cabazitaxel, is required per the manufacturer’s prescribing information. 13 Patients must be monitored closely during the first 2 infusions for signs and symptoms of hypersensitivity reactions. 13 Polysorbate 80 (Tween 80) is a water-soluble emulsifier used as a diluent for both cabazitaxel and docetaxel. Polysorbate 80 may cause hypersensitivity reactions, although the risk is decreased when patients are premedicated as detailed above, as significant reactions occurred in less than 1% of patients in the phase III trial. However, if the patient has previously experienced a severe reaction to docetaxel, it may be that the patient reacted to the polysorbate 80, which is a diluent for both docetaxel and cabazitaxel, and therefore, the patient should not receive cabazitaxel, as per the manufacturer’s prescribing information. Another taxane, paclitaxel, has a higher rate of hypersensitivity reactions than both docetaxel and cabazitaxel, which is related to its diluent, Cremophor EL, a castor oil–based chemical commonly implicated in causing anaphylactic, severe hypersensitivity infusion reactions.
Cabazitaxel Dosage Adjustments Based on Toxicities 13 .
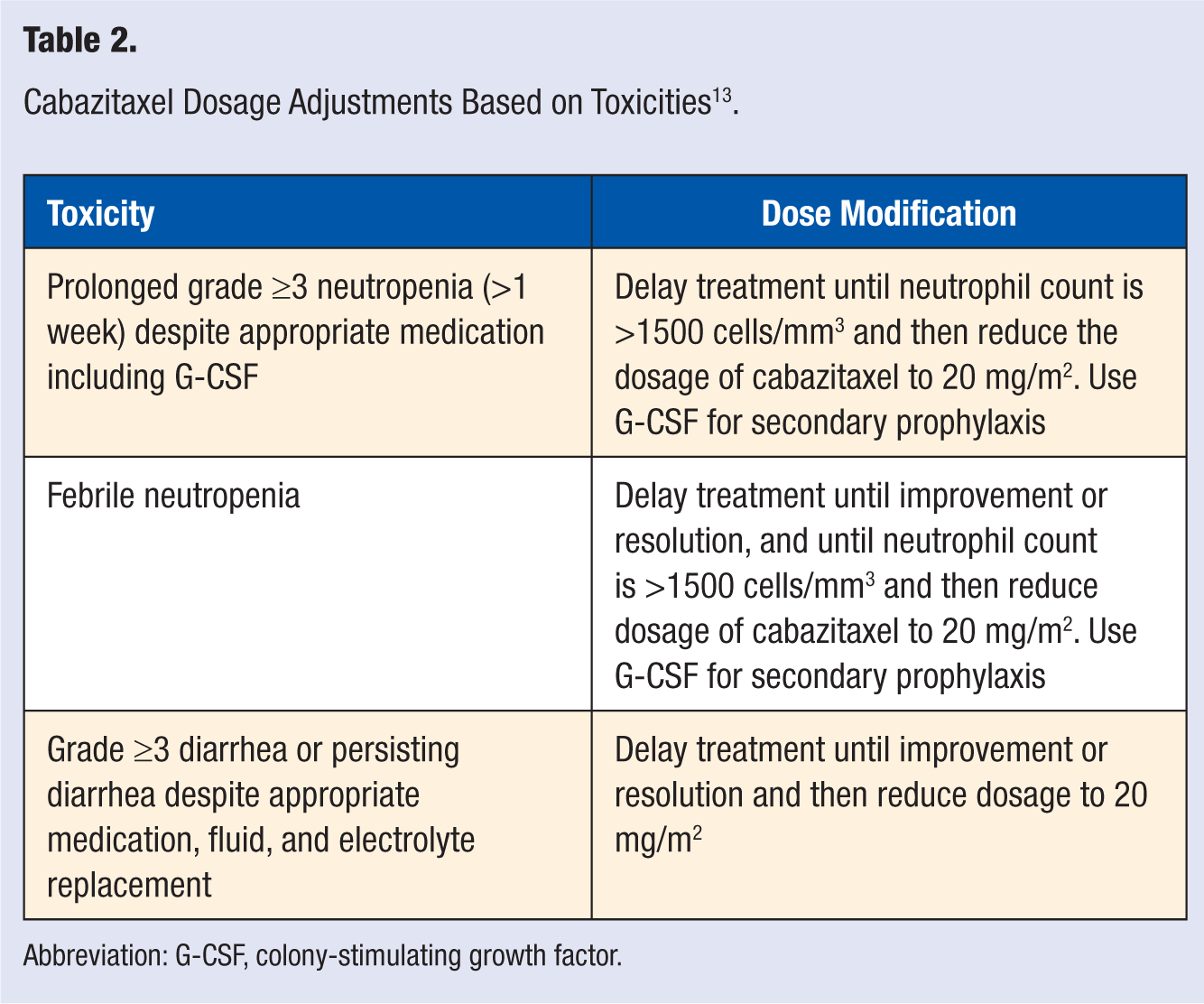
Abbreviation: G-CSF, colony-stimulating growth factor.
The most frequently reported drug-related adverse effects of cabazitaxel in the phase III clinical trial included anemia, neutropenia, diarrhea, fatigue, nausea, anorexia, peripheral neuropathy (any grade neuropathy 13%, grade 3-4 ≤1%), dysguesia, arthralgia, pyrexia, and alopecia. Drug toxicity led to study discontinuation in 18% of patients who received cabazitaxel (the most common reason was neutropenia) as compared with 8% of patients who received mitoxantrone.
Patient monitoring and teaching related to hypersensitivity reactions should include close and frequent monitoring of vital signs during the first and second doses of cabazitaxel. Other objective signs to monitor for include flushing, sweating, and/or edema. The patient should be counseled to promptly report any symptoms such as itching, dizziness, sweating, chills, nausea, abdominal cramps, anxiety, or confusion. The infusion should be discontinued during any signs/symptoms and should be permanently discontinued for severe hypersensitivity reactions, according to the package labeling. 13
Patient monitoring and teaching for the risks of myelosuppression, especially neutropenia, should include complete blood count monitoring as detailed above and prior to any chemotherapy dose. The patient should be taught the signs and symptoms of infection, how to use a thermometer, and to report any symptoms or fever immediately. Additionally, patients should be taught self-care tactics to reduce risk of infection such as hand hygiene, avoidance of persons with active infections, avoidance of undercooked fish/meats, and of pet feces.
Patient monitoring and teaching for nausea and diarrhea should include self-administration of antiemetics for any nausea and self-administration of over-the-counter loperamide during episodes of diarrhea. Additionally, increased fluids during episodes of diarrhea and bland foods such as bananas, rice, and crackers should be emphasized, as well as the importance of prompt physician or emergency department visits for diarrhea lasting longer than 24 hours to assess fluid and electrolyte status and to receive intravenous fluids and electrolytes as needed.
Signs and symptoms of peripheral neuropathy not only include sensory symptoms, such as numbness and tingling in the fingers and toes, but also motor function symptoms, such as altered gait and balance. Patients should be closely monitored and questioned about these effects on activities of daily living and quality of life before any dose is administered.
Finally, as per the manufacture’s labeling and other occupational exposure guidelines for handling hazardous agents such as antineoplastic agents, this agent should only be administered by those with experience in administering hazardous agents, using appropriate protection and precautions for handling and disposal, as well as access to emergency resuscitation medical equipment and medications due to the risk of allergic hypersensitivity infusion reactions. 13
Dosing and Administration
The recommended dosage of cabazitaxel is 25 mg/m2, which is administered intravenously every 3 weeks as a 1-hour infusion in combination with a daily administration of oral prednisone 10 mg. Patients should be premedicated with histamine-1 and histamine-2 antagonists and dexamethasone 8 to 10 mg intravenously at least 30 minutes prior to the infusion. The daily oral prednisone was not held on the day of cabazitaxel administration in the phase III clinical trial. Cabazitaxel is contraindicated in patients with a history of severe hypersensitivity reactions to cabazitaxel, or other drugs formulated with polysorbate 80, such as docetaxel. Antiemetic prophylaxis with a serotonin antagonist is recommended to be added prior to subsequent administrations of cabazitaxel if a patient experiences nausea with a previous dose, as per the manufacturer’s prescribing information. 13 Like other taxanes, the emetogenic potential appears to be moderate rather than high (as compared with cisplatin for example). Dosage adjustments may be required during treatment for adverse events as described in Table 2. As with paclitaxel and docetaxel, use of an in-line filter is required during administration, and cabazitaxel should be diluted in polyvinyl chloride–free containers. This is recommended to minimize patient exposure to the plasticizer DEHP—di-(2-ethylhexyl)phthalate—which may be leached from polyvinyl chloride infusion bags or sets by agents usng the diluents polysorbate 80 (cabazitaxel and docetaxel) or Cremophor EL (paclitaxel).
Although cabazitaxel is extensively metabolized in the liver, no trials have been completed in patients with hepatic impairment, although one is ongoing, according the NCI clinical trials database. 16 Therefore, there are no specific dosage adjustments for hepatic dysfunction at this time, and the package labeling suggests that cabazitaxel not be given to patients with hepatic impairment (total bilirubin > upper limit of normal (ULN), or aspartate aminotransferase and/or alanine aminotransferase >1.5 × ULN). 13
Efficacy
Based on promising activity of cabazitaxel in the metastatic breast and prostate cancer populations in a phase I trial of advanced solid tumors, a phase II trial involving 71 patients with taxane-resistant metastatic breast cancer was conducted and demonstrated an objective response rate of 14%, with 8 partial and 2 complete responses seen. Although no phase II study in advanced prostate cancer was ever conducted, the dosing for the phase III trial to evaluate the efficacy and safety of cabazitaxel in metastatic CRPC was based on the efficacy demonstrated in the phase II trial of metastatic breast cancer.15,19
Phase III results
Results from the pivotal phase III clinical trial was reported at the 2010 Annual Meeting of the American Society of Clinical Oncology (ASCO) by de Bono et al. 17 The TROPIC study was a phase III, open-label trial of 755 patients, which compared cabazitaxel in combination with prednisone versus mitoxantrone with prednisone in men with metastatic CRPC progressing during or after docetaxel, with performance status between 0 and 2 and renal and hepatic function within normal limits. 17 Patients were randomly assigned to receive either cabazitaxel 25 mg/m2 every 3 weeks or mitoxantrone 12 mg/m2 every 3 weeks, both in combination with a daily dose of oral prednisone 10 mg. Both treatments were administered for a maximum of 10 cycles, or until development of unacceptable toxicity. The primary endpoint was overall survival, and secondary endpoints were progression-free survival and tumor response rates. An update of the final outcomes, with a median follow-up of 12.8 months, was presented at the ASCO 2010 annual meeting. 20 The authors reported that men randomized to the cabazitaxel group had a median overall survival of 15.1 months compared with 12.7 months for men in the mitoxantrone group, and this difference of 2.4 months was statistically significant (hazard ratio = 0.72; 95% confidence interval = 0.61-0.84; P < .0001). Investigator-assessed tumor response, which included complete and partial responses, was 14.4% in the cabazitaxel arm versus 4.4% in the mitoxantrone arm, P = .0005, although no complete responses were reported.13,17,20
Cabazitaxel was FDA approved in June 2010, under the agency’s priority review program, which expedites approval of agents that offer a treatment for a disease state where none or few had existed previously.
Place in Therapy and Future Directions
Cabazitaxel is indicated in combination with prednisone for treatment of patients with metastatic CRPC previously treated with a docetaxel-containing treatment regimen (docetaxel-refractory disease). Until recently, there were no other FDA-approved second-line treatment options that have shown an overall survival advantage for CRPC. In 2010, treatment with cabazitaxel and prednisone was the first therapy approved by the FDA that offers a survival benefit to men with metastatic CRPC whose disease has progressed during or after docetaxel treatment. However, 2 other treatments were FDA approved for metastatic CRPC in 2010 and will affect future research directions for cabazitaxel therapy, either in sequence or in combination with these agents. Sipuleucel-T (Provenge, Dendreon Corporation, Seattle, WA) was the first immunotherapy treatment approved by FDA for any cancer; this agent, an autologous cellular immune therapy, involves training the patient’s T cells to attack prostate cancer cells. This agent did show an overall survival advantage in a phase III placebo-controlled trial in metastatic CRPC; however, this population differed from the cabazitaxel study population in that the majority of patients were chemotherapy naive and asymptomatic. Presently it is only FDA approved for use in the asymptomatic or minimally symptomatic metastatic CRPC population (per the manufacturer’s package labeling). In addition, in October 2010, overall survival results were presented from a phase III placebo-controlled clinical trial of abiraterone acetate—an oral CYP17 inhibitor that impairs extragonadal androgen synthesis—demonstrating an overall survival advantage in men with metastatic docetaxel-pretreated CRPC randomized to the abiraterone versus the placebo, and recently this agent was FDA approved for this population. 21 These new agents have greatly expanded the available treatment options for metastatic CRPC.
Future trials will further elucidate the role of cabazitaxel, alone and in combination with other agents, in both treatment naive and refractory CRPC. At present, cabazitaxel is only indicated for second-line therapy after docetaxel; however, this may change in the near future as trials directly comparing docetaxel to cabazitaxel are planned. Additionally, trials evaluating the possible synergy between the cabazitaxel and the new agents abiraterone acetate and/or sipuleucel-T are highly anticipated. Currently, tumor biomarkers that identify who will respond the best to any of these new agents is yet to be found, and this will need to be an important goal of future clinical trials. Until then, clinicians will have to continue to weigh the benefits in terms of prolongation of life versus the risks for each particular patient with respect to each agent’s adverse effect profile. Because of the modest survival benefit with cabazitaxel of 2.4 months, the risks of neutropenia and diarrhea from this agent should encourage careful patient selection in this risk–benefit decision process for each patient. In addition, close monitoring is suggested for safe prescribing. Future cost-effectiveness analyses comparing the available agents for metastatic CRPC will be required to more fully define the treatment algorithms for CRPC. Finally, future trials will explore the feasibility and dosing of cabazitaxel use in liver impairment and also evaluate lower dosing or alternative dosing schedules to reduce the rates of neutropenia seen in the pivotal phase III TROPIC study.16,22
Conclusion
Treatment options for metastatic CRPC are limited, and therefore research into this unmet need is paramount. Until recently, only one agent—docetaxel—was FDA approved for the treatment of metastatic CRPC, and options for CRPC were suboptimal, particularly for patients progressing on docetaxel. In 2010, cabazitaxel was approved by the FDA as the first chemotherapy agent that offers a survival benefit in men with metastatic CRPC whose disease has progressed during or after docetaxel. Challenges and questions remain for the immediate future, such as defining optimal sequencing and combinations with other new or existing agents for metastatic CRPC. The comparative effectiveness with docetaxel in the first-line metastatic CRPC setting remains to be defined. Future pharmacoeconomic analyses will hopefully further define this agent’s place in therapy in comparison with docetaxel, or with other new options for CRPC, such as the orally available abiraterone acetate. Ongoing and future clinical trials should help answer these questions and further elucidate the role of all the newly approved agents in the treatment of prostate cancer.16,22 Although additional treatment options for the common disease state of prostate cancer are needed, the approval of cabazitaxel has added a promising and much needed treatment option for metastatic CRPC that has progressed on first-line chemotherapy with docetaxel.
