Abstract
Following physical maturity, all body functions inevitably progressively decline with aging; however, there is a great deal of variability in the rate of decline and the associated development of significant functional impairments. Both genetic and lifestyle factors, including physical activity habits, contribute to this variability in physiological changes, as well as to the associated risk of chronic degenerative diseases. The effect of healthy natural aging on functional capacity and its related cardiovascular and respiratory contributors has primarily been studied by cross-sectional and, less frequently, by longitudinal studies. In these studies, physically active men and women at all ages were observed to have a superior functional capacity, as well as a lower rate of cardiovascular disease morbidity and mortality, as compared with matched sedentary individuals. In addition, a limited number of studies, involving healthy elderly men and women, have demonstrated that moderate-to-vigorous exercise training can substantially improve functional capacity and modify many of the age-related cardiovascular changes. The purpose of this article is to review the effects of natural or so-called primary or eugeric aging on the cardiovascular system and associated functional capacity and the extent of modification of these changes by regular exercise.
Human adults are commonly classified chronologically by age as shown in Table 1 with age 65 years arbitrarily accepted, primarily for sociological reasons, as the demarcation line for the beginning of old age or so-called senescence. However, there is a great deal of variability in the rate of structural and functional changes associated with natural/primary or eugenic aging due to both genetic and lifestyle factors. This variability has given rise to alternative ways to classify relative age based on functional parameters, 1 as well as more recently by cellular DNA dating and vascular aging assessment techniques.
During early adulthood, at the time of sexual and physical maturity, humans generally attain their peak physical and functional capacity, as assessed by maximal oxygen uptake.
Classification of Adults by Chronologic Age
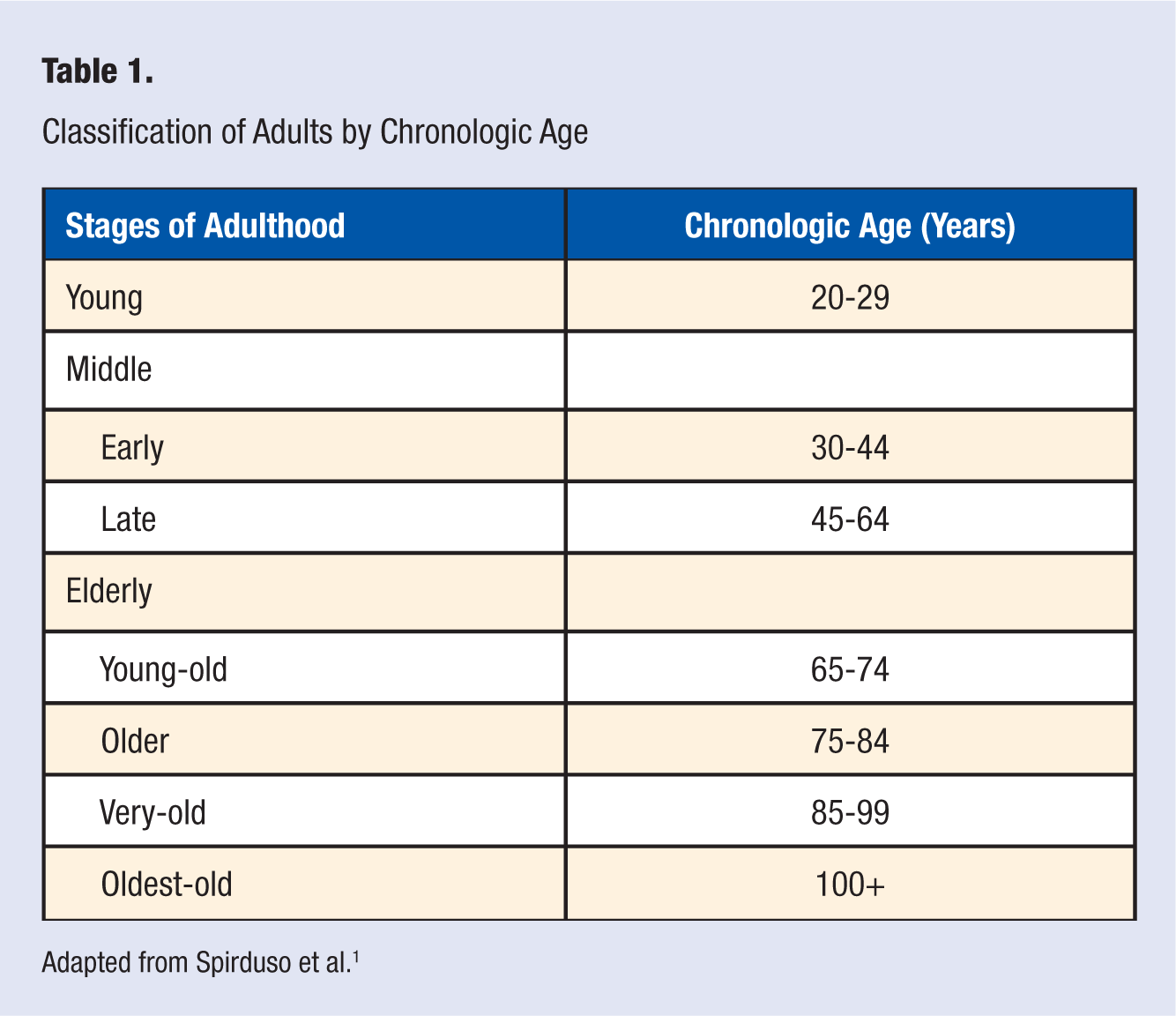
Adapted from Spirduso et al. 1
During early adulthood, at the time of sexual and physical maturity, humans generally attain their peak physical and functional capacity, as assessed by maximal oxygen uptake (V
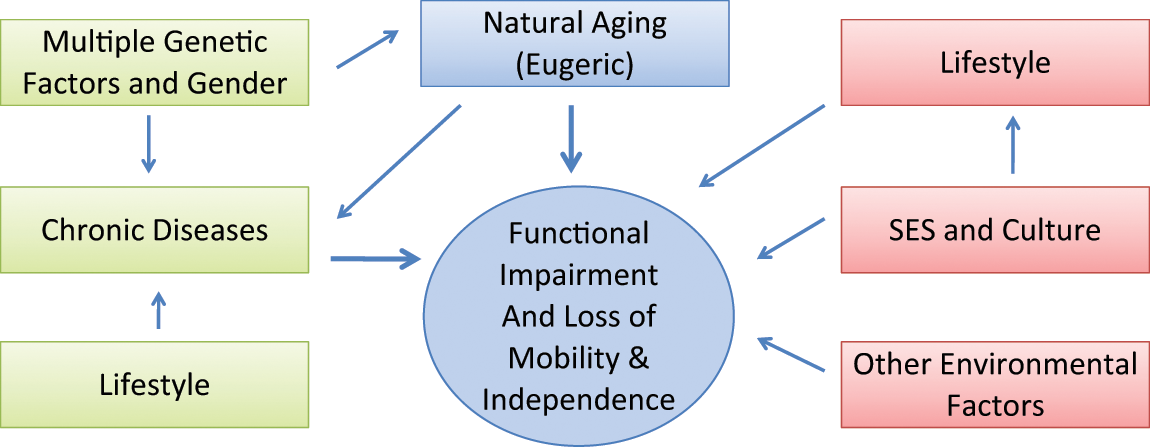
Factors Contributing to the Heterogeneity of the Aging Process and Development of Functional Physical Disability
Because of the combination of aging-related functional decline and the associated increased risk of chronic diseases with aging, a great deal of concern has been expressed by public health, medical, and sociological planners for the progressively increasing prevalence in the population of people age 65 and older in developed countries. According to the US Census Bureau, the prevalence of older Americans increased from about 4% of the population in 1900 to 12% in 2000, and is projected to reach 20% in 2030 with the subgroup age 85 and older showing the largest increase. 5 These concerns are based on stereotyping all older people as having serious physical or mental impairments, limiting their ability to live independently, and thereby requiring a great deal of social services support and health services. However, in a landmark study on so-called successful aging, Rowe and Kahn, 6 on surveying a large sample of Americans age 70 to 100 years, found that only a relatively small percentage classified themselves as sufficiently disabled to require full-time external social support and/or substantial medical attention. This encouraging finding supports the hypothesis that healthy aging principles can result in the so-called rectangularization of the aging curve. 7 This theory postulates that an increasing number of the aging population via good health habits could achieve their disability-free biologically determined maximal chronological age, while compressing major impairments and disease morbidity into a relatively brief period at the very end of life.7-9 The purpose of this article is to review the inevitable negative impact of natural aging on the cardiovascular system and functional capacity of healthy, nonsmoking adults and present evidence of the attenuation of some of these negative aging effects by regular exercise training (ET).
Aging-Related Cardiovascular Changes
Resting Changes
Aging in healthy adults is associated with minimal changes in indices of cardiac function in the resting state. 10 These generally include no significant change in resting heart rate, in resting cardiac index (liters per minute per meter square of body surface area), stroke volume, or left ventricular (LV) ejection fraction.
Decline in Vo 2max
Most of the age-related decline in cardiovascular functions becomes evident only during heavy physical exertion. The most extensively studied physiological change associated with aging is a decline in peak oxygen uptake or V
It should be noted that the US Social Security Administration recognizes V
The major cardiovascular contributor to the decline in V
Vascular Changes
The earliest detectable vascular manifestation of aging is evident by the third or fourth decade of life.18-21 It consists of a progressive stiffening and loss of distensibility of large elastic conduit arteries, which is considered the “hallmark of vascular aging.” 18 This is caused by fatigue fractures and loss of elastin in the media of these major conduit arteries, an increase in 100-fold less elastic collagen, and the deposition in the media and apventitia of calcium by altered smooth muscle cells (mycro blasts). These vascular changes are attributed to the mechanical stress caused by repetitive pulsatable expansion of these arteries with each heart beat (estimated to occur about 36.8 million times per year for someone with an average heart rate of 70 bpm). 18 Advanced glycation end products of proteins, inflammation and endothelial dysfunction also are postulated to contribute to arterial stiffness.20,21 As a consequence of the loss of elasticity in the aorta, there is an up to 4-fold increase in pulse wave velocity (PWV), associated with blood ejected by the LV, and transported to the peripheral vessels. PWV is commonly accessed by a Doppler technique measuring its transmission from a carotid to a femoral artery, which is considered the best validated, noninvasive procedure to quantify the severity of aortic stiffness. 19
As a consequence of the increase in PWV, there is an accelerated return of the wave to the heart and root of the aorta during late systole, rather than as it normally occurs following the completion of LV ejection. The accelerated rate of return results in an augmentation in systolic blood pressure (BP). As a consequence of increased aortic stiffness and associated increased PWV, the majority of the elderly population (a 60% incidence by age 60) experiences isolated systolic hypertension, a major risk factor for both cerebrovascular and coronary heart disease (CHD). Typically diastolic BP also increases until late middle age, and then it progressively decreases, resulting in a 60 mm Hg or higher pulse pressure in an 80 year old, as compared to a 20 year old. 18
Another consequence of the loss of the cushioning effect of the elastic expansion of the aorta is elevated pulsatable shear stress during systole on the microvasculature. 18 This can cause irreversible damage to these microvessels, resulting in microvascular bleeding and thrombi, especially in the brain and kidneys, 18 contributing to progressive renal dysfunction and increased risk of dementia.
There also is evidence that vascular conductance of blood flow to the lower extremities declines with primary aging, independent of loss of skeletal muscle mass 22 due to an increase in adrenergic-induced vasoconstrictor activity, which contributes to the associated about 1% a year increase in peripheral vascular resistance (PVR). Furthermore, older adults have a reduced ability to augment limb blood flow by vasodilatation in response to increased metabolic demands during exercise, due to vascular endothelial dysfunction, involving both conductance and muscular/resistance arteries.3,20,21
Aging also is associated with an increase in venous stiffness with loss of capacitance. The resulting venous insufficiency, along with an aging-associated disturbance in baroreflex responsiveness due to autonomic system dysregulation, contributes to a high prevalence of orthostatic (postural) hypotension in elderly individuals. 1 The resulting precipitous drop in BP, on assuming the upright posture from a sitting or supine position, can cause synscope, injuries from falling, and even a coronary event or a cerebrovascular accident.
Aging-induced vascular stiffness also adversely affects coronary artery perfusion and even can induce myocardial ischemia, independent of coronary atherosclerotic perfusion impairments.
18
The associated increase in PWV results in prolongation of systole, delaying the onset of diastole at which time about 80% coronary flow ordinarily occurs. In addition, myocardial oxygen (M.V
Myocardial Changes
Between age 20 and 80 years, there generally is an accumulative loss of about 30% of cardiomyocytes. 24 Multiple mechanisms are postulated to be responsible for cardiomyocyte senescence and increased apoptosis (ie, programmed cell death).4,20,21,24,25 These include cumulative reactive oxygen species (ROS) damage to mitochondrial DNA, adversely affecting oxidative metabolism; a progressive loss during cellular division of telomeres, the protective nongenetic strands of DNA at the end of chromosomes; and a decline in cellular activity of the enzyme, telomerase that catalyzes DNA synthesis to maintain telomere length. These aging-related changes result in cellular inability to undergo further division resulting in their senescence and apoptosis. In addition, there is a progressive loss of cardiac progenitor satelite stem cells required for a limited rate of myocyte regeneration.20,21 However, hypertrophy of remaining cardiomyocytes commonly contributes to an increase in LV mass with aging.1,20,21,24,25 The myocardium of older adults also is slower to relax during diastole, and has a reduced contractile reserve, as compared with younger adults, due to biochemical and physiologic alterations of cardiomyocytes, including reduced mitochondrial oxidative enzyme activity and reduced b-adrenergic ionotropic responsiveness.3,26 Protein glycation and cross-linkage (including of myosin) also may contribute to reduced contractility. In addition, there is an increase with aging in collagen and fat deposits in the myocardium. These alterations result in increased myocardial stiffness, contributing to the reduced peak LV ejection during systole, 26 and increased risk of chronic heart failure (CHF) with aging.
Aging also is associated with enlargement of the left atrium and an increased contribution of the force of its contraction to LV diastolic filling. 25 Aging-induced atrial enlargement increases risk of atrial fibrillation, the rate of which doubles with each decade of adult life.
Effects of Exercise Training on the Cardiovascular System
Although the cardiovascular effects of aerobic ET have been extensively explored in young adults, there are limited data available on its effects in older adults. Most of the available data on cardiovascular changes with aging are from cross-sectional rather than from longitudinal observational and ET studies. In addition, it is suspected that some of the earlier observational studies included subjects with underlying clinically evident or occult/silent cardiovascular disease. In both cross-sectional and longitudinal observational studies, physically active men and women, as previously mentioned, had higher V
The effects of moderate (50% to 75% V
As little as 16 weeks of moderate-intensity aerobic ET has been found to increase V
Based on animal research findings, regular exercise is postulated to mitigate age-related attrition of cardiomyocytes by improving antioxidative protection of the myocardium against ROS and by increasing cellular telomerse activity.20,21 However, an endurance ET program initiated in elderly, previously sedentary individuals apparently does not reduce aging-related cardiac stiffness. 27 This is in contrast to the findings in observational studies of the absence of myocardial stiffness in elderly master athletes. In these master endurance athletes, absence of cardiac stiffness probably resulted from them being physically active most of their adult lives. However, ET in sedentary older individuals has been consistently shown to significantly enhance compliance/elasticity of the aorta and other large arteries, as well as to improve endothelial-induced vasodilitation by increasing endothelial NO synthase activity.31,32 An exercise-induced increase in circulating endothelial progenetor cell number and function also is postulated to contribute to repair of damaged endothelium and improvement in vascular endothelial function.20,21 These vascular adaptations in conduit arteries probably play a significant role, along with a reduction in PVR, for the improvement with ET in peak blood flow to the lower extremities of elderly individuals. 31
Reduced Risk of CHD
Regular moderate-intensity physical activity is associated with a reduced risk of fatal and nonfatal CHD as well as with a reduction in all-cause mortality. 1 Multiple mechanisms are postulated to be responsible for these protective effects against CHD. These mechanisms have been classified as atherosclerotic, anti-ischemic, antiarrhythmic, and thrombotic effects. 23 Reduced progression or partial regression of coronary atherosclerosis with ET has been demonstrated in monkeys on an atherosclerotic diet, 33 and in CHD patients, expending 1500 kcal or more a week of aerobic exercise, while on a heart-healthy, reduced saturated fat diet.34,35 Possible mechanisms for these anti-atherosclerotic effects of ET include a reduction in all of the risk factor components of the metabolic syndrome as well as independent anti-inflammatory 36 and antioxidant effects, 37 improving coronary endothelial function and reducing lipid infiltration of the intima.
Anti-ischemic contributions of endurance ET in the presence of advanced coronary artery disease, include both a reduction in myocardial oxygen (M.V
Coronary blood flow is enhanced by the ET-induced improvement of endothelial-mediated vasodilatation and reduced coronary stiffness. 38 In addition, coronary artery remodeling (arteriogenesis) increases the cross-sectional area of the lumen of major coronary arteries.23,33 Furthermore, animal studies in several different species have consistently demonstrated that endurance ET promotes myocardial capillary angiogenesis (analogous to the training-induced increase in capillarization of skeletal muscles of humans). This substantially increases the total cross-sectional area of the coronary bed.23,39-41 Exercise-induced mobilization of endothelial progenitor cells from the bone marrow in response to increase vascular endothelial NO synthase activity is postulated to be involved in promoting myocardial angiogenesis. 32 Coronary blood flow also is improved by reductions in PWV and training-induced heart rate, which result in an increase in the duration of diastole, during which coronary blood flow is at its peak.
A major mechanism postulated to be involved in the apparent protective effect of aerobic ET against fatal ventricular arrhythmias is improved electrical stability of the heart, primarily by increased heart vagal activity, as evidenced by increased resting heart rate variability. 23 Mechanisms for the postulated antithrombotic effects include improved vascular endothelial function, resulting in decreased platelet aggregation and improved blood clot fibrinolysis. Furthermore, ET results in a reduction in circulating blood level of fibrinogen, which ordinarily increases with aging. 23
Cardiovascular Effects of Resistance/Strength Training
The importance of skeletal muscle mass, strength, and metabolic functions for health and performance of physical exertion and activities of daily living (ADL) are well recognized. 42 Thus, progressive muscle loss with aging in sedentary people via sarcopenia is an important contributor to reduced capabilities to perform ADL, essential for living independently. In addition, sarcopenia is an important contributor to loss of bone density and strength, increasing the risk of fragility fractures. 42 Furthermore, sarcopenia is associated with increased risk of type 2 diabetes. 43 The evidence is overwhelming that resistance training (RT) can reduce development of sarcopenia, as well as increase skeletal muscle mass, strength, and endurance at all stages of life. 43 RT in elderly individuals thereby can significantly improve vigor, the ability to perform ADL, the quality of life, and the ability to live longer independently. However, there are contradictory reports on the effects of RT on artery stiffness. Some studies have found that RT reduces conduit artery stiffness, while others reported increased large artery stiffness and an associated increase in PWV in older men and women, who were highly trained strength athletes. 43
Exercise Recommendations
A consensus now exists that a comprehensive exercise program can promote cardiovascular and general health and improve functional capacity, and reduce mobility limitations, and thereby improve the quality of life for people of all ages, including the elderly population. 43 Ideally, an exercise program for elderly individuals should include both aerobic ET and RT, as well as flexibility and balance exercises. Guidelines for such programs for older adults are available, including medical screening procedures to rule out contraindications to exercise and for preparation of individualized exercise prescription.44,45 Such pretraining screening procedures are especially important for elderly individuals, who are extremely deconditioned, functionally impaired, or have existing health problems. Exercise programs should be initiated at a low intensity and volume and the rate of progression should be individualized and tailored to the elderly individual’s tolerance and preference.
Conclusions
Major cardiovascular anatomical and physiological changes associated with aging are summarized in Table 2. Based on this review, it is concluded that there is consistent strong and growing evidence that a significant contributor to these cardiovascular changes is a sedentary lifestyle and an associated reduction in cardiorespiratory fitness. Initiation of moderate-intensity aerobic ET by elderly individuals can ameliorate all the aging-related vascular changes listed in Table 2, as well as attenuate many of the cardiac changes. Favorable ET-induced changes include an increase in V
Summary of Aging-Related Cardiovascular Structural and Physiological Changes
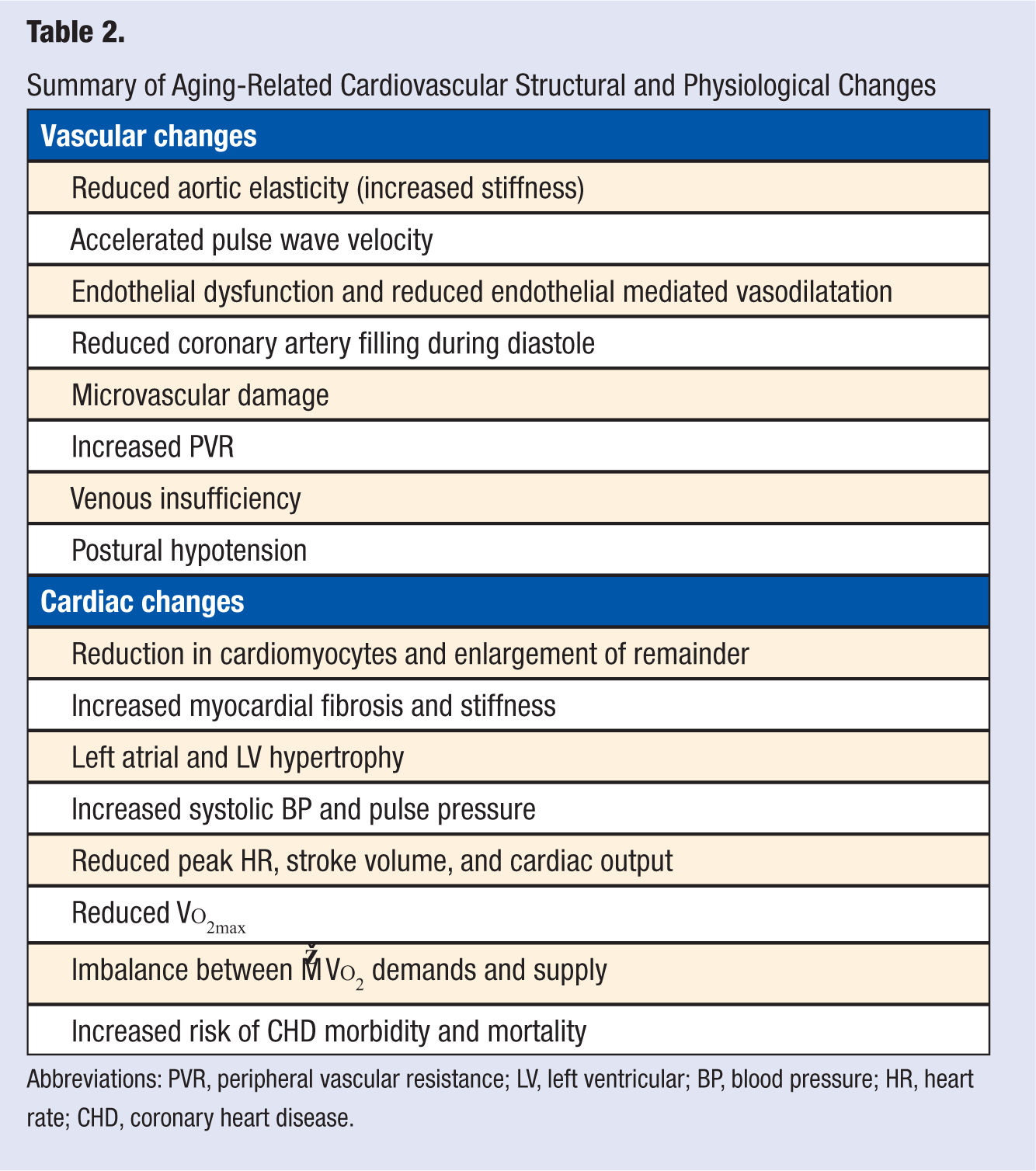
Abbreviations: PVR, peripheral vascular resistance; LV, left ventricular; BP, blood pressure; HR, heart rate; CHD, coronary heart disease.
In addition, endurance ET reduces risk of CHD by multiple mechanisms. Cardioprotective effects include decreasing the rate of progression or partial regression of coronary atherosclerotic plaques. M.V
Footnotes
Acknowledgements
This article was presented in part by Dr Leon by invitation at the 21st International Puijo Symposium: Physical Exercise, Aging, and Disabilities: Current Evidence; Kuopio, Finland; June 29 to July 2, 2011.
The author wishes to extend his appreciation to Ms Linda Estrem for preparation of this article and Scott Brown, MS, for preparation of the figure. Appreciation also is expressed to Professor Li Li Ji for his review and critique of this article. Dr Leon is partially supported by the Henry L. Taylor professorship in Exercise Science and Health Enhancement and by an NIH-supported grant (Exercise Training to Reduce Claudication [EXERT] Study).
