Abstract
Evidence indicates that chronic diseases, such as cardiovascular disease, obesity, and asthma are part of a group of conditions linked by inflammatory dysregulation. One explanation for these associations is that the Western diet is obeseogenic and promotes the secretion of inflammatory biochemical signals. Weight gain results in lipid accumulation and adipocyte stress—factors known to disrupt the balance of systemic cell signaling (adipokines and cytokines) that favors inflammation. Markers are used as indicators of the inflammatory milieu and include the measurement of proinflammatory (C-reactive protein, interleukin-6, tumor necrosis factor-a) and anti-inflammatory (adiponectin) signaling molecules. Consumption of a high ratio of omega-6 to omega-3 fatty acids is also an important indicator for a potentially inflammatory profile. This ratio may determine the direction of biological pathways for another family of inflammatory mediators—eicosanoids—which vary according to the omega fatty acid precursor. An improved dietary pattern that emphasizes balanced energy and nutrient intake resulting in a reduced ratio of omega-6 to omega-3 fatty acids plays a particularly important role in inflammation management.
Epidemiological evidence indicates that when asthma and obesity are both present, obesity most often precedes asthma . . .
Obesity is well established as a risk factor for chronic inflammatory diseases 1 such as cardiovascular disease and type-2 diabetes mellitus and is also a newly emerging risk factor for allergic diseases 2 such as asthma.3-5 According to a Center for Disease Control survey, 8.2% of the United States population, or approximately 24.6 million people, are reported to have asthma. 6 In recent decades, allergic disorders and asthma prevalence have risen dramatically and in parallel to the rise in obesity prevalence observed across cultures. Allergic airway disease and obesity are linked by inflammatory dysregulation and often coexist as chronic disorders. 7 Epidemiological evidence indicates that when asthma and obesity are both present, obesity most often precedes asthma, and the strength of this association increases linearly with BMI. 5 A reduction in fat mass associated with dietary intervention has been shown to abrogate asthma-related symptoms. 8 A Western diet, common in industrialized nations, is often described as being an inflammatory stimulus and a risk factor for asthma,9-11 though the mechanisms of that risk are not fully understood.
One explanation is that the Western diet is obeseogenic and promotes secretion of inflammatory biochemical signals. Swinburn et al 12 assert that the present obesity epidemic in the United States can be explained by an increase in the average daily energy intake resulting from the food environment. Kerver et al 13 define a Western dietary pattern as being characterized by high intakes of energy-dense foods, such as processed meats, eggs, red meats, and high-fat dairy products. The Western diet also includes a majority of novel foods that are created or altered by processing. Within these foods are a combination of refined cereals, sugars, and vegetable oils along with added salt and fat. The resulting excess energy intake increases lipid accumulation and adipocyte stress—factors shown to disrupt the balance of systemic cell signaling (adipokines and cytokines) to favor a proinflammatory milieu. 14 The recruitment of macrophages to abdominal fat depots is a significant step in the shift toward an inflammatory profile.15,16
The presence of an inflammatory response can be determined by the measurement of the serum concentration of both proinflammatory and anti-inflammatory biochemical signals. Proinflammatory markers commonly tested include, and are not limited to, C-reactive protein (CRP), interleukin-6 (IL-6), tumor necrosis factor alpha (TNF-α), and interleukin 18 (IL-18).17,18CRP is synthesized in the liver as a first response to an inflammatory stimulus. Obesity-induced adipose tissue inflammation is associated with an increased expression of IL-6, IL-18, and TNF-α, which are synthesized by both macrophages and adipocytes. 19 Adiponectin, an anti-inflammatory adipokine, is released exclusively by adipocytes and regulates insulin sensitivity; however, concentration decreases with obesity. 20
When examining a dietary lifestyle that leads to weight gain, it is also important to consider the contribution of the pattern of foods consumed and the associated nutrient profile. Berg and colleagues 21 conducted a study of men and women and identified food patterns that promote obesity and obesity-related disorders. Both BMI and waist-to-hip ratio were greater in groups that regularly consumed medium- and regular-fat milk, cheese, white bread, sweets, snacks, and alcoholic beverages and infrequently consumed fruits, vegetables, and low-fat products. These food patterns were compared with a reference healthy diet that included more fiber, lower fat, and less sugar. In other research, dietary patterns of Iranian women were examined, and the authors concluded that a Western dietary pattern, when compared with a healthier one (high in fruits, vegetables, tomatoes, poultry, legumes, tea, and whole grains), was associated with higher BMI, waist-to-hip ratio, and waist circumference. In contrast to the healthy pattern, those in the upper quintile of the Western pattern dietary scores were more likely to have general and/or central obesity. 22
Nutrients are highly correlated within a pattern of foods. For instance, observing the ratio of omega-6 to omega-3 fatty acids is perhaps more important than considering one without the other. In the Western diet, the ratio is reported to be in a range as high as 15:1 to 16.7:1. 23 Other estimates conclude that a ratio of 10:1 is more typical and a relatively stable description of the US diet over the past 40 years. 24 Nonetheless, this is a dramatic shift in nutrient balance away from the 1:1 ratio that is theorized to be the dietary profile of early humans. 25 In today’s modern food environment, dietary intake of the omega-6 family of nutrients is largely influenced by linoleic acid found predominantly in grain crops and vegetable oil. Arachidonic acid (AA), a metabolite of linoleic acid, is found in grain-fed animal tissue. Fish flesh and fish oils, high in eicosapentaenoic acid (EPA) and docosahexaeoic acid (DHA), make up the majority of dietary omega-3s. EPA and DHA are intermediary products of linolenic acid (ALA) metabolism. 26
Recognition of the Western diet may be particularly meaningful in health risk evaluation associated with inflammation. Fernandez-Real et al 27 linked fatty acid concentration to inflammation in overweight individuals. In their cross-sectional study of the relationship between dietary fatty acids, insulin resistance, obesity, and inflammatory markers in overweight and lean individuals, the inflammatory marker IL-6 was positively associated with the percentage of saturated fats and omega-6 fatty acids and negatively associated with CRP in the overweight group. These associations were not present for the lean group. These results are both consistent and inconsistent with those of Kalogeropoulos et al 28 who reported an inverse association of plasma omega-6 fatty acids with both CRP and IL-6. The ratio omega-6:omega-3 exhibited the strongest positive correlations with all the markers studied. The authors maintain that this study provides evidence that omega-6 fatty acids are anti-inflammatory. However, they concluded that the proportions of the fatty acids, as measured by the omega-6 to omega-3 ratio, could reflect levels of inflammation, with lower ratios being associated with decreased inflammation.
Because the subject population of the latter study was community based and not an obese sample, the differing results of the 2 studies in regard to IL-6 might be explained by the presence or absence of obesity. Indeed, De Lorenzo et al 29 found that plasma proinflammatory cytokine concentrations were elevated in normal-weight, obese and preobese-obese women and that circulating concentrations were correlated with percentage fat mass. Esposito and colleagues 30 conducted a randomized trial of otherwise healthy premenopausal obese women whose baseline diets contained large amounts of saturated fat, cholesterol, and refined carbohydrates plus small amounts of fiber and omega-3 fatty acids. Baseline values of IL-6, IL-18, and CRP were higher than those reported in nonobese women. After a weight reduction diet, the intervention group that consumed more complex carbohydrates and monounsaturated fat had a lower ratio of omega-6 to omega-3 fatty acids and less dietary energy, saturated fat, and cholesterol than controls. Serum concentrations of IL-6, IL-18, and CRP were significantly reduced in the intervention group compared with controls, whereas adiponectin levels were significantly increased. 30
Is the ratio of omega-6 to omega-3 of greater significance in an inflammatory profile than singular fatty acids? A study of healthy men and women examined the effect of 10 weeks of diet modification to decrease the omega-6:omega-3 ratio on cardiovascular risk factors and resting energy expenditure. The dietary intervention was associated with significant reductions in TNF-α and increased adiponectin. Guebre-Egziabher and colleagues 31 concluded that the reduced omega-6:omega-3 polyunsaturated fat ratio achieved with simple dietary counseling is potentially protective for multiple metabolic disorders and low-grade inflammation.
A disproportionate ratio of omega-6 to omega-3 essential fatty acids may form the foundation through which the nutrient-inflammation theories are most strongly linked. Omega-6 and omega-3 fatty acids complete the pool of biologically available fatty acids used by the human body. 23 AA, EPA, and DHA are partitioned to the phospholipid bilayer of all cell membranes in a concentration reflective of dietary intake. For inflammatory cells like the macrophage, AA, EPA, and DHA are competitive substrates for intracellular COX (cyclo-oxygenase) enzymes, the gateway for eicosanoid metabolism. Eicosanoids are potent inflammatory mediators that have differing effects among inflammatory cells and target tissues. 32 The biological properties of eicosanoids vary according to the omega fatty acid precursor; therefore, modifying the availability of these substrates through the diet would limit the inflammatory potential of the eicosanoid pool when the ratio of omega-6 to omega-3 is lower (Figure 1).
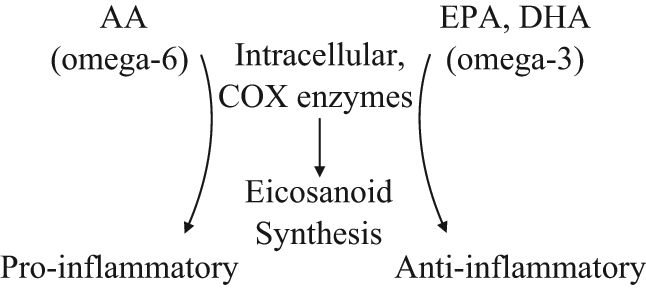
The inflammatory signaling milieu is influenced by the concentration of dietary substrates AA (arachidonic acid), EPA (ecosapentaneoic acid, and DHA (docosahexanoic acid) in association with COX (cyclooxygenase) synthesis of eicosanoid.
Evidence suggests that an inflammatory state may be activated or exacerbated by a Western dietary pattern, which promotes obesity and the increased production of inflammatory cytokines and decreased production of anti-inflammatory cytokines. This unbalanced environment favors a proinflammatory condition and predisposes individuals to increased incidence of inflammation-associated disorders. A review by Giugliano et al 33 on the effects of diet related to inflammation provided general conclusions and foundational practices for dietary modification to prevent inflammatory conditions:
Increase consumption of omega-3 fatty acids from fish or plant sources.
Reduce saturated and trans fats by substituting nonhydrogenated unsaturated fats.
Consume a diet that is high in fruits, vegetables, nuts, and whole grains in addition to low amounts of refined grains and sugars.
In conclusion, the association between dietary pattern, an increase in fat mass, and the onset of a chronic disorder, such as allergic asthma, may be partially explained by the low-grade inflammation common to all 3 conditions. Creating a healthy eating pattern is a long-established practice associated with lifestyle changes that are important to weight management. An improved dietary pattern that emphasizes balanced energy and nutrient intake resulting in a reduced ratio of omega-6 to omega-3 fatty acids plays a particular role in inflammation management (Figure 2).
A healthy dietary pattern, relative to energy and nutrient balance, is inclusive of a lifestyle plan for reducing systemic inflammation and risk of chronic disease.
