Abstract
This prospective study examined the immediate health benefits associated with participation in a brief intensive behavioral weight and diabetes management program for individuals with type 2 diabetes. Participants were 55 adults with type 2 diabetes and obesity (mean BMI = 44.5) who completed a 4-week residential treatment program. Pretreatment and posttreatment weight, fasting glucose, lipids, hemoglobin A1c (HbA1c), and diabetes medications were evaluated. Treatment effects were also examined by pretreatment HbA1c levels (HbA1c ≤ 7% vs. HbA1c > 7%). Significant reductions in average weight (283 to 267 lb), fasting glucose (143.2 mg/dL to 118.3 mg/dL), lipids, and HbA1c (7.5% to 6.9%) were observed after 4 weeks of treatment. Results were clinically meaningful, evidenced by mean body weight reductions, and HbA1c reductions, and mean posttreatment glucose and lipid levels being in the normal range. Nearly two thirds of participants significantly reduced or eliminated at least 1 diabetes medication. Participants having higher pretreatment HbA1c achieved greater reductions in glucose, HbA1c, and lipids. The 4-week behavioral weight and diabetes management program yielded significant weight and health improvements. This is one of the first studies to report rapid and significant HbA1c changes as a result of following moderate nutrition and physical activity guidelines while receiving diabetes management and weight loss psychoeducation.
Numerous studies suggest that lifestyle modification and weight control are beneficial for successful long-term management of type 2 diabetes.1-6 Weight control is recommended for overweight individuals with diabetes because losing 10% body weight can significantly improve glycemic control, lipid profile, and blood pressure and subsequently decrease cardiovascular risks.6,7 Lifestyle interventions designed to produce weight loss through improved nutrition and physical activity have been shown to yield significant reductions in the incidence of diabetes 2 and cardiovascular risk factors,1,2,6 and some have helped individuals with diabetes to maintain weight losses for a number of years. 6 Furthermore, lifestyle interventions have yielded better outcomes (eg, higher percentage weight loss) than diabetes education, support, or usual care.1,4,6 Some interventions have included cognitive-behavioral components in recognition of the detrimental effects of stress and negative emotions on glycemic control,8,9 and positive results have been reported when techniques to improve stress, 10 mood, 11 and adherence to diabetes self-management 12 were added to standard treatment. These studies suggest that incorporating cognitive-behavioral techniques into traditional diet- and exercise-based interventions may significantly enhance glycemic outcomes.
Lifestyle interventions for diabetes often involve months of participation in sessions or groups; however, a small number of studies show that health outcomes relevant to diabetes (eg, reductions in weight, cholesterol, triglycerides, and fasting glucose) can be achieved through briefer interventions, such as 3- and 4-week intensive residential programs.
Lifestyle interventions for diabetes often involve months of participation in sessions or groups6,13,14; however, a small number of studies show that health outcomes relevant to diabetes (eg, reductions in weight, cholesterol, triglycerides, and fasting glucose) can be achieved through briefer interventions, such as 3- and 4-week intensive residential programs.15,16 In addition to the health indicators mentioned, hemoglobin A1c (HbA1c) and diabetes medication status are also relevant outcomes, yet they are less likely to be evaluated at posttreatment in brief lifestyle interventions. Further research is needed to determine the extent to which these variables can be improved immediately following brief interventions.
The American Diabetes Association recommends a target HbA1c of <7.0% among individuals with diabetes. 17 Education and lifestyle interventions can help individuals achieve recommended HbA1c levels, with studies reporting HbA1c reductions of 0.59% to 0.7%.1,10,13,18 Yet as an indicator of mean glucose control, HbA1c is typically only measured over several months, and it is believed that changes in HbA1c are not evident until at least 3 months following reductions in blood glucose. 13 Recent guidelines recommend the use of HbA1c along with blood glucose to diagnose and monitor diabetes,19,20 and there is support that HbA1c may be more sensitive to the effects of a brief intervention. 21 This study sought to determine whether an intensive lifestyle-based weight loss intervention could produce HbA1c changes in a brief (4-week) period of time.
Pretreatment HbA1c levels may predict treatment outcomes, but studies have been mixed regarding the direction of findings. Some have shown that individuals with higher HbA1c responded less favorably to standard diabetes care. 22 Other studies have reported that individuals with higher HbA1c showed the greatest improvements after a lifestyle intervention 23 or found no significant differences in outcomes based on initial HbA1c levels. 13 It is not known whether pretreatment HbA1c levels predict immediate outcomes of a brief intervention. This study evaluated treatment outcomes for the entire sample as well as for the sample divided by pretreatment HbA1c status.
Finally, medication changes represent a clinically meaningful outcome for diabetes interventions, yet the extent to which brief lifestyle interventions can lead to diabetes medication reduction is uncertain. To our knowledge, only 1 other published study has evaluated diabetes medication changes following a brief intensive program. 16 Longer duration lifestyle interventions for diabetes, such as the Look AHEAD and ICAN trials, have reported reductions in the number of individuals taking glucose-lowering medications at follow-up.1,6,13 Thus, a final aim of the current study was to conduct an analysis of medication changes to better understand the type of changes that can be achieved through brief lifestyle interventions.
In summary, the primary aim of this study was to evaluate the extent to which HbA1c levels and diabetes medications improved following a brief intensive lifestyle intervention that incorporated diet and physical activity modification as well as cognitive-behavioral techniques for stress and mood management. A secondary aim was to determine how pretreatment HbA1c levels were related to health outcomes at posttreatment. Predictions included the following: (1) participants would experience significant reductions in HbA1c from pretreatment to posttreatment; (2) reductions in the number and dosage of diabetes medications would occur by posttreatment; and (3) participants with poorer pretreatment diabetes control (HbA1c > 7.0%) would show greater health benefits (ie, greater reductions in fasting glucose, HbA1c, and triglycerides) following the brief lifestyle intervention.
Research Design and Methods
Study Participants
Participants were drawn from a group of 157 individuals with type 2 diabetes and obesity who completed a 4-week residential weight and diabetes management program between 2005 and 2008. Of these, 117 provided consent to participate in the prospective outcome evaluation study prior to treatment, and 55 (38 females, 17 males) met study inclusion criteria—completing 4 weeks of the program and having no missing data in the variables included in the analyses. Ages ranged from 29 to 77 years (mean = 56.6 years), and 91% of participants were white, non-Hispanic. BMI at the start of treatment ranged from 29 to 67 kg/m2 (mean = 44.5 kg/m2). At baseline, 85% of participants were prescribed at least 1 oral diabetes medication, and 32.7% were taking insulin.
Treatment Description
Participants were enrolled in the residential program at Structure House Center for Weight Control and Lifestyle Change. The program is multidisciplinary and emphasizes cognitive-behavioral principles of weight control, including but not limited to daily self-monitoring of food, weight, and physical activity as well as behavioral practice in exercise, menu planning, mindful eating, grocery shopping, and restaurant eating. The program is based on the philosophy of Structured Eating®, which instructs participants to preplan 3 nutritious, portion- and calorie-controlled meals per day and eliminate snacking. Participants consume meals prepared on-site from standardized menus with options for individualizing based on preferences and specific caloric needs. Menus are based on nutrition guidelines from the American Heart Association (AHA),
24
and daily calorie levels were determined using the Mifflin-St Joeur formula,
25
which takes into account an individual’s height, weight, gender, age, and expected activity level and is intended to produce a modest weight reduction of 1% loss of fat per week. The daily calorie assignments for study participants ranged from 1000 to 1800 kcal/d (mean = 1287 kcal/d). Given the importance of adequate protein when following a hypocaloric nutrition plan,26,27 the macronutrient composition of meals included 20% fat, 51% carbohydrate, and 29% protein. Participants were encouraged to meet the current physical activity recommendations
28
of 150 minutes per week of exercise at their assessed fitness level, and individualized fitness consultations provided specific exercise plans to facilitate adherence to exercise recommendations for weight loss. Participants received a standardized schedule of programming to attend weekdays between 8
To improve diabetes management, participants also received diabetes-specific classes and individual appointments with an endocrinologist, health psychologist, exercise physiologist, and certified diabetes educators (registered nurse and registered dietitian). Daily glucose and medications were monitored, and participants received diabetes education, information about psychological factors (eg, stress and negative emotions) that affect self-management and glycemic control, and instruction in cognitive-behavioral techniques that improve diabetes control (eg, progressive relaxation and cognitive restructuring).
Data Collection
All study procedures were approved by the Duke University Health Systems Institutional Review Board. Participants weighed themselves daily wearing light indoor clothing on a computerized scale with a maximum weight of 1000 lb and a precision of 0.1 lb. Height (in inches) was measured during the health evaluation on day 1 and used with body weight data to calculate BMI. On day 2, participants provided an overnight (8-10 h) fasting plasma sample to evaluate blood chemistries. All blood samples were refrigerated approximately 30 hours prior to delivery to a local laboratory for standardized analyses. The present study focused on changes in glucose (mg/dL), cholesterol (mg/dL), triglycerides (mg/dL), and HbA1c (%) from the sample drawn on day 2 compared with a posttreatment sample drawn on day 25. HbA1c (%) was analyzed in a laboratory certified by the National Glycohemoglobin Standardization program (NGSP) using a COBAS INTEGRA Tina-quant Hemoglobin A1c Gen.2 whole blood application on Roche clinical chemistry analyzers, standardized to the International Federation of Clinical Chemistry (IFCC) and Laboratory Medicine (for HbA1c mmol/mol) and transferable to Diabetes Control and Complications Trial (DCCT) and NGSP (HbA1c %).
Pretreatment diabetes medications were assessed and recorded on day 1 by a registered nurse. Any changes made during weekly appointments with the endocrinologist were recorded. Information collected included medication name, class, daily dosage, and dosage frequency.
Data Analysis
Within-subjects Students t tests were performed (SPSS v.15.0; SPSS Inc, Chicago, Illinois) to determine whether weight and health outcomes changed significantly during treatment. The sample was also divided into 2 subgroups based on initial HbA1c, and independent t tests were conducted to compare outcomes based on degree of pretreatment diabetes control. Descriptive analyses (means, standard deviations, percentage of participants, percentage change) were used to provide additional information regarding the changes observed within and between subjects.
Results
Pretreatment values for weight and health indices are shown in Figures 1A to 1E, and pretreatment medication information is provided in Table 1.
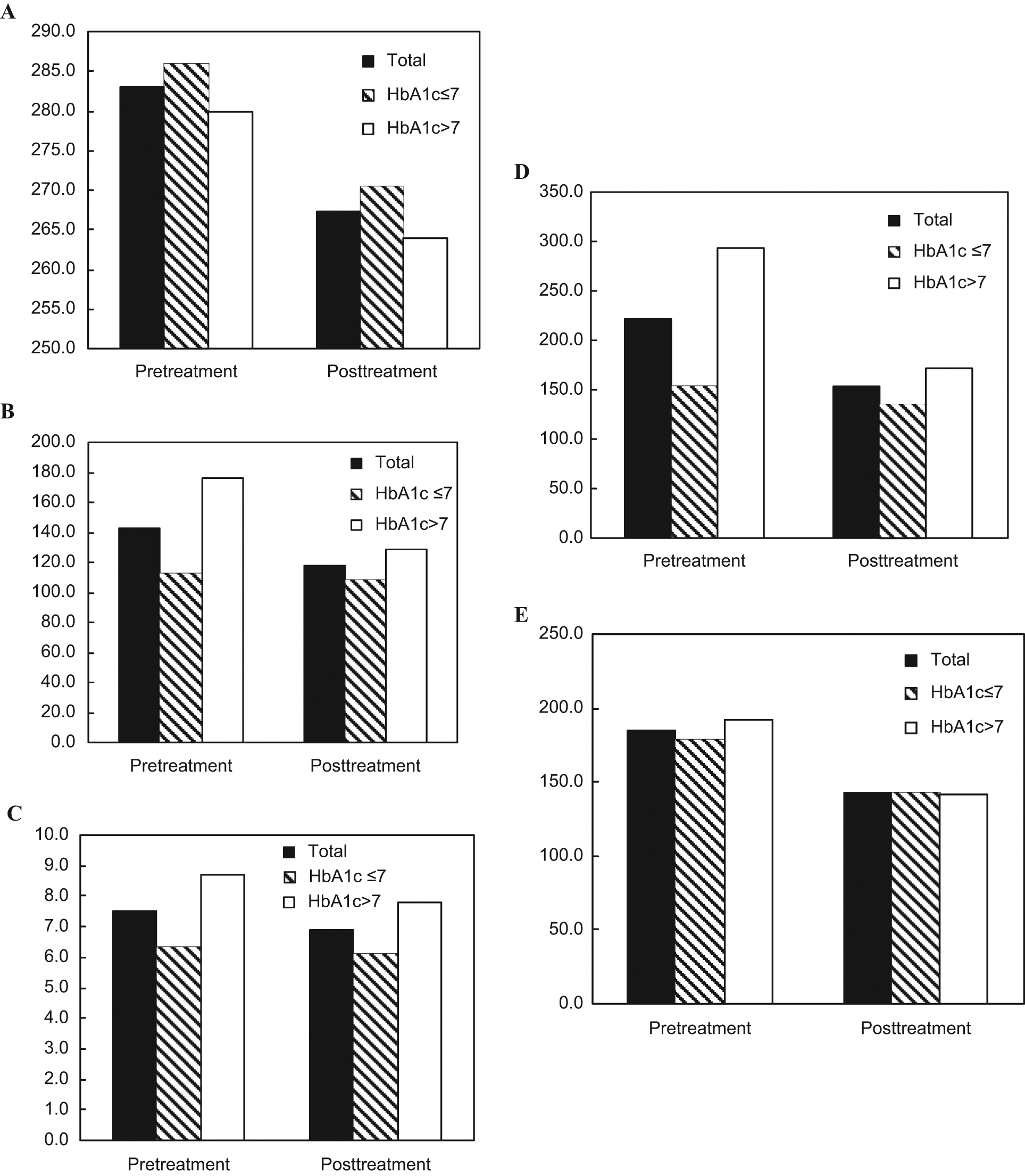
Weight and health changes following brief lifestyle program for diabetes: A. Mean weight (lb). B. Mean fasting plasma glucose (mg/dL). C. Mean hemoglobin A1c (%). D. Mean triglycerides (mg/dL). E. Mean total cholesterol (mg/dL).
Medication Changes by Drug Class for Total Sample and HbA1c Subgroups
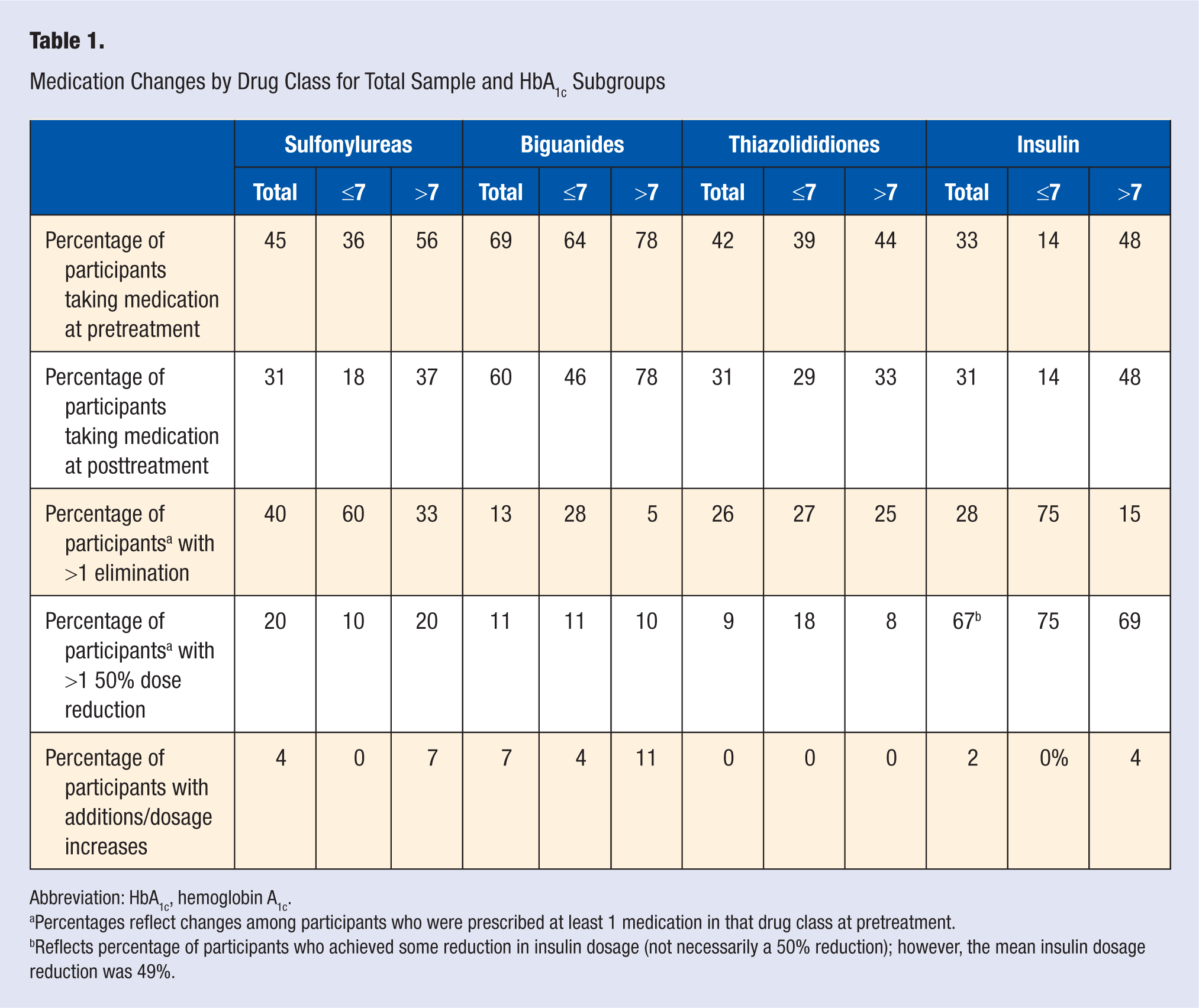
Abbreviation: HbA1c, hemoglobin A1c.
Percentages reflect changes among participants who were prescribed at least 1 medication in that drug class at pretreatment.
Reflects percentage of participants who achieved some reduction in insulin dosage (not necessarily a 50% reduction); however, the mean insulin dosage reduction was 49%.
Weight and Health Outcomes
Results revealed the following statistically significant pretreatment to posttreatment mean (standard deviation) improvements (Ps < .001): Weight decreased from 283.0 lb (9.2 lb) to 267.2 lb (8.7 lb), t(54) = 17.0; waist circumference decreased from 51.0 inches (1.1 inches) to 48.0 inches (1.1 inches), t(50) = 15.8; glucose decreased from 143.2 mg/dL (6.7 mg/dL) to 118.3 mg/dL (3.4 mg/dL), t(53) = 4.4; total cholesterol decreased from 185.3 mg/dL (5.0 mg/dL) to 142.3 mg/dL (3.3 mg/dL), t(54) = 9.8; and triglycerides decreased from 222.3 mg/dL (21.7 mg/dL) to 153.5 mg/dL (10.9 mg/dL), t(54) = 4.8.
In 4 weeks, 64% of the participants achieved ≥5% weight loss. Although nearly half of the participants (46%) had pretreatment fasting glucose levels above the recommended value of 130 mg/dL for type 2 diabetes, 17 at posttreatment less than one third (29%) had glucose above 130 mg/dL. At pretreatment, 35% of participants had total cholesterol greater than 200 mg/dL; at posttreatment, 100% of participants had total cholesterol values less than 200 mg/dL. Regarding triglycerides, less than half (46%) had levels within the recommended (<150 mg/dL) range at baseline, 16% had levels in the borderline high (150-199 mg/dL) range, and 38% had levels in the high (>199 mg/dL) range. At posttreatment, 58% fell within the normal range, 22% in the borderline high range, and 20% in the high range.
Hemoglobin A1c
Overall, 51% of participants had at least a 0.5% reduction in HbA1c levels. A paired sample t test showed a statistically significant reduction in HbA1c from 7.5% (±0.21) to 6.9% (±0.21); t(54) = 7.4; P < .001. At pretreatment, approximately half of the participants (49%) had an HbA1c level of >7%. At posttreatment, 64% of the participants completed treatment with an HbA1c level ≤7%.
With regard to the weight and health variables examined, men and women did not differ at pretreatment nor in degree of change at posttreatment, with the exception of weight change. Men showed a greater percentage of weight loss than women (6.63% vs 5.0%; t(53) = 3.1; P < .01) and greater decrease in waist circumference (3.56 inches vs 2.69 inches; t(49) = 2.3; P < .05).
Medication Outcomes
Analysis of medication data revealed that many who completed the 4-week program were able to modify their pretreatment medication regimen. All except 1 participant were taking diabetes medications at pretreatment, and the mean number of medications prescribed per participant was 2.2 (standard deviation = 1.2). At posttreatment, the mean number of diabetes medications prescribed per participant was 1.7 (1.1); t(54) = 4.4, P < .001. There was also a significant pretreatment to posttreatment reduction in the number of participants taking 3 or more medications (38% vs 22%; t = 2.89, P < .01). Evaluating all medication classes together, 42% of participants were able to eliminate at least 1 medication in 4 weeks. In addition to eliminations, 31% were able to decrease dosage or frequency of at least 1 medication, and the mean dosage reduction was 50%. Together, nearly two thirds of participants (64%) either eliminated or significantly reduced at least 1 medication (and 9% had both a medication elimination and dosage reduction). Even more dramatic reductions were achieved by some participants, including elimination of all diabetes medications among 9% of participants and a 50% reduction in number of prescribed medications by 15% of participants.
Medication changes by drug class
Medication outcomes were further evaluated by drug class. Table 1 presents pretreatment and posttreatment medication data. Participants were able to eliminate and decrease dosages for each medication class evaluated, with the most substantial modifications among sulfonylureas and thiazolididiones.
Outcomes by pretreatment HbA1c status
To determine whether the intervention yielded differential effects on participants based on pretreatment diabetes control status, we divided the sample into 2 subgroups—HbA1c ≤ 7.0% (n = 28) and HbA1c > 7.0% (n = 27)—and conducted between-group univariate analyses.
As expected, between-group comparisons revealed pretreatment differences in fasting glucose [F(53) = 18.7] and triglycerides [F(53) = 16.3], with Ps < .001, such that the HbA1c > 7.0% group began treatment with higher values for each of these risk factors. There were no pretreatment differences in age, weight, waist circumference, or total cholesterol. At posttreatment, weight change (% weight loss) did not differ based on HbA1c status. As predicted, participants with HbA1c > 7.0% at pretreatment achieved significantly greater improvements in HbA1c [F(53) = 45.5], fasting glucose [F(52) = 20.4], and triglycerides [F(53) = 16.2], with Ps < .001, and marginally greater improvements in total cholesterol [F(53) = 2.8; P = .098].
With regard to medication changes by HbA1c subgroup, Table 1 shows the overall pattern: higher percentages of participants with HbA1c > 7.0% are prescribed each class of diabetes medication at pretreatment and posttreatment, and they are less likely to be taken off medications compared with the HbA1c ≤ 7.0% group, with the exception of thiazolididiones, which were eliminated similarly across groups. Across most drug classes, the HbA1c > 7.0% group achieved rates of dosage reductions that were similar to those for the HbA1c ≤ 7.0% group.
Discussion and Conclusions
This study examined the effects of a brief intensive lifestyle intervention for obese individuals with type 2 diabetes. The intervention produced a weight loss of ≥5% in the majority of participants, with significant reductions in fasting glucose, HbA1c, total cholesterol, and triglycerides. Findings are consistent with previously published short-term behavioral weight loss outcomes, 15 including significant health improvements occurring with 5% to 10% weight loss. 29
Hemoglobin A1c
The mean 0.6% reduction in HbA1c was significantly greater than the minimal changes found during an earlier brief dietary intervention, 29 slightly greater than the 0.5% reduction reported 12 months following a stress management intervention, 10 slightly less than the 0.9% reduction obtained after an 8-week, 850 cal/d liquid diet, 21 and less than the 0.7% reduction obtained 3 months following a self-management education program. 18 Reductions in HbA1c ≥0.5% are associated with positive health effects and regarded as a desirable target. This appears to be the first study to report significant reductions in HbA1c in such a short period of time using moderate lifestyle changes. The nutrition component of the program was based on recommendations for weight loss from the AHA 24 and National Institutes of Health, 30 and daily calorie levels were intended to produce no more than 1% of fat loss per week. Participants were never assigned <1000 cal/d, and the majority (57%) were assigned 1200 to 1400 cal/d (23% consumed 1000-1150 cal/d, and 20% consumed 1400-1800 cal/d). Compared with programs that prescribe greater fat restriction (eg, <10%) 16 or greater caloric deficit, 31 the diet consisted of approximately 20% of calories from fat, 51% from carbohydrates, and 29% from protein. The HbA1c results are encouraging because they were obtained after only 4 weeks of health behavior change, and they were obtained using moderate and maintainable methods of weight control based on national health and nutrition guidelines.
As predicted, and similar to some previous research, 14 higher pretreatment HbA1c was associated with greater improvements in HbA1c, fasting glucose, and triglycerides at posttreatment. The lifestyle intervention yielded a stronger effect among individuals who began treatment with poorer glycemic control. Those with higher HbA1c levels may have been practicing the fewest diabetes self-management behaviors prior to treatment. Initiating these behaviors during treatment may have yielded a stronger effect on these individuals compared with those who may already have been engaging in more healthy behaviors. Given that past research has shown that individuals with higher HbA1c levels may be poor responders to standard diabetes care, 21 it is possible that intensive, comprehensive, yet still brief interventions may be a particularly valuable treatment modality for individuals with a history of low adherence, poor self-management behaviors, or difficult-to-control glucose levels.
Medication
Patients with diabetes often express a strong desire to take less medication. At posttreatment, we found a significant reduction in the mean number of medications prescribed per patient, and 42% of individuals, under the care of an endocrinologist, were able to eliminate at least 1 diabetes medication. Achieving desired medication reductions so quickly after initiating behavior changes may provide much-needed reinforcement for individuals to continue with healthy lifestyle behaviors. An important question for future research is whether early medication reductions contribute to improved longer term adherence to diabetes self-management behaviors.
Analysis of medication changes by drug class was particularly interesting when considering changes based on the initial HbA1c subgroup (≤7.0% or >7.0%). Although individuals with less-controlled glycemic levels were less able to eliminate medications in 4 weeks, many achieved significant dosage reductions in a brief period of time. This supports the few published studies that demonstrate that some individuals may be able to reduce diabetes medications rapidly following initiation of healthy lifestyle behaviors 16 and adds to reports that improving glycemic control through lifestyle change is associated with diminished need for diabetes medications.13,31 An important contribution of the present study is that it offers a more in-depth analysis of the specific types of medication changes that occurred after 4 weeks as well as a comparison of medication changes attained by individuals based on pretreatment glycemic control.
Limitations and Future Directions
Because the study was conducted in an applied clinical setting and no control group was available, changes that occurred were not compared with wait-list, education-only, or usual care groups, nor were participants randomized into treatment conditions. However, research generated from applied settings offers important opportunities to disseminate information about “real world” interventions that have broad inclusion criteria and imperfect experimental conditions, such as participants with multiple comorbidities. A review of randomized controlled studies suggests that the weight, HbA1c, and medication changes observed in the present study are greater than control group changes reported in other studies and similar in magnitude to changes seen in the Look AHEAD intervention groups.1,2,6
Another limitation involves the absence of follow-up data beyond treatment, so the degree to which early treatment improvements were sustained is unknown. Efforts are under way to collect 6- and 12-month posttreatment data from current participants with diabetes for follow-up analyses. An advantage of focusing on the immediate effects is the ability to demonstrate that brief interventions can offer rapid stabilization and control of metabolic and cardiovascular risk factors using a moderate, safe, and realistic approach to diet and physical activity. There is evidence from previous studies that early positive response to a weight management program is associated with longer term success.23,32 Results offer providers and patients evidence that modest, sustainable lifestyle changes can produce meaningful weight loss and reductions in HbA1c, triglycerides, fasting glucose, total cholesterol, and diabetes medications within a month of consistent behavior change. Seeing significant improvements early in the process of making lifestyle changes can provide a sense of accomplishment to individuals and direct evidence that their efforts are worthwhile, potentially increasing the likelihood for adherence.
Finally, the private-pay residential setting in which the intervention was delivered poses a limitation to the extent to which results can be generalized to individuals who initiate healthy lifestyle changes using less-intensive interventions. Though the dietary, physical activity, and behavior modification strategies were all designed to translate to a home setting, the provision of meals, ready access to fitness facilities, peer and professional support, and availability of educational and therapeutic services are unique to this setting and undeniably contribute to the success of the intervention. Given that residential treatment is not widely available and affordable, important future tasks include identifying the types of individuals who may be most in need of intensive weight and diabetes treatment programs and developing more accessible methods of delivery for the effective treatment components (eg, Web- or mobile-based interventions).
Footnotes
Jennifer Pells, Anna Stout, Chandra Quigley, and Gerard Musante are employed by Structure House, LLC, a private facility owned by CRC Health Group. Michelle Rodriguez-Diaz was formerly a research assistant employed by Structure House, LLC. Findings reported in this article are derived from data collected from individuals participating in the Structure House program who provided informed consent to participate in outcomes evaluation research.
