Abstract
Objectives
Ischemic/reperfusion (I/R) injury seriously affects human health. Renal tubular epithelial cell injury and inflammation are important pathogenic mechanisms involved in I/R injury. Recent studies have shown that quercetin (QU) is one of the most effective natural products against inflammatory responses and is mainly used as an adjuvant treatment for chronic diseases in clinical practice. However, the effect of QU on renal I/R injury remains unclear.
Methods
The therapeutic effect of QU was evaluated in a mouse model of renal I/R injury in vivo, and its anti-inflammatory mechanisms were preliminarily investigated in Normal Rat Kidney-52E (NRK-52E) cells. Subsequently, STING knockout mice were used to verify the anti-inflammatory mechanism.
Results
Our results showed that QU alleviates renal injury and inflammation subsequent to I/R. In vitro, QU alleviated inflammatory responses induced by H2O2. Mechanistically, QU alleviated inflammation by inhibiting the STING/NF-κB pathway.
Conclusion
These results demonstrated that QU protected against renal I/R injury by suppressing inflammation via the STING-NF-κB axis, highlighting its therapeutic potential.

Introduction
Renal ischemic/reperfusion (I/R) injury is a prevalent injury that increases the morbidity and mortality of renal diseases, such as acute renal failure and acute kidney injury. 1 Renal I/R injury is commonly observed in surgical departments and is prevalently detected after kidney transplantation, partial nephrectomy, and nephrolithotomy. Furthermore, various infectious diseases and hypovolemic shock can also induce renal I/R injury, but kidney transplantation remains the most common etiology. 2 Previous studies have reported that the incidence of acute kidney injury ranges from 6.9% to 11.6% among hospitalized patients in China. To address I/R injury in patients, numerous studies have developed treatment and prevention methods by targeting various mechanisms, including anti-inflammatory and antioxidant therapies. Despite extensive research endeavors, there is still a lack of therapeutic drugs and strategies to protect against renal I/R injury in clinical practice. Therefore, actively exploring the mechanisms behind renal I/R injury is essential to identify effective therapeutic targets and provide a theoretical basis for clinical application.
Pathologically, renal I/R injury is characterized by damage to renal tubular epithelial cells. The causes of damage to renal tubular epithelial cells include apoptosis, necrosis, abnormal ATP metabolism, and inflammatory responses. 3 Currently, it is believed that inflammatory responses play a crucial role in the pathophysiology of renal I/R injury. 4 It has been reported that nuclear factor-κB (NF-κB)-mediated inflammatory response aggravates renal I/R injury. 5 Stimulator of interferon genes (STING), as an important intracellular inflammatory regulatory molecule, can recognize second messengers and activate interferon regulatory factor 3 (IRF3) and NF-κB. This process can rapidly and strongly produce type I interferons and inflammatory factors, thereby causing inflammatory responses.6-8 Recent studies have demonstrated that the cyclic GMP-AMP synthase (cGAS)-STING pathway serves as a critical driver of inflammatory injury in renal I/R injury. Upon I/R injury, damaged mitochondria release mitochondrial DNA (mtDNA) into the cytosol, which can activate cGAS and subsequently stimulate STING-dependent NF-κB signaling, thereby exacerbating tissue inflammation and renal dysfunction. 9 Notably, pharmacological inhibition of STING has been shown to confer robust protection against renal ischemic injury, establishing the cGAS-STING-NF-κB axis as a promising therapeutic target. 9
Quercetin (QU) is a natural flavonoid found in plants. It is known for its diverse biological properties, including antioxidant, anti-inflammatory, and cardioprotective properties. 10 Notably, QU alleviated I/R injury in the brain and heart, 11 improved neuroinflammation via the nuclear factor erythroid 2-related factor 2 (Nrf2)/heme oxygenase-1(HO-1) pathway, 12 and downregulated inflammatory mediators by inhibiting the Toll-like receptor 4 (TLR-4)/nuclear factor-kappa B (NF-κB) axis. 13 Crucially, emerging evidence indicates that natural flavonoids can modulate the cGAS/STING pathway. For instance, epimedium flavonoids improve cGAS/STING-mediated inflammatory diseases by disrupting STING-IRF3 interactions and preventing the activation of the downstream molecule NF-κB. 14 More recently, QU has been indicated to specifically inhibit the cGAS/STING pathway, thereby significantly downregulating STING expression and NF-κB-mediated inflammatory responses.15,16 However, the effect of QU on renal I/R injury remains unclear.
Based on the critical role of STING/NF-κB signaling in renal I/R injury and the inhibitory effects of QU on this pathway, we hypothesized that QU alleviates renal I/R injury by targeting the STING-NF-κB axis and attenuating inflammatory responses. To investigate this hypothesis, we initially assessed it using a mouse model of renal I/R injury. We found that QU alleviated I/R-induced pathological changes in the kidneys. Furthermore, QU mitigated the inflammatory response induced by I/R through the STING/NF-κB pathway. Therefore, QU holds promise as a potential therapeutic option for renal I/R injury.
Methods and Materials
Study Duration
All experiments were conducted from January 2022 to December 2025.
Animal Experiments
8-week-old male C57BL/6J mice weighing 18-22g were purchased from GemPharmatech (Nanjing, China). STING gene knockout mice with a C57BL/6J background were purchased from the Jackson Laboratory and bred to expand the population and identify the genotypes of the offspring for subsequent experiments. All mice were housed in an independent ventilation cage system with automatic control of temperature, humidity, and air exchange rate. The animals were provided with a 12-hour light and 12-hour dark cycle, as well as free access to food and water.
Experiment 1
After adaptation to their environment preoperatively, C57BL/6J mice were randomly divided into three groups. In the sham operation group (Ctrl), 3% pentobarbital sodium (40 mg/kg) was intraperitoneally injected. After the mice became stable, a layer-by-layer incision was made 1 cm beside the spinal cord and 1 cm below the lower edge of the ribs on the back. The renal artery and vein were carefully dissected and then occluded by sutures. The mice were conventionally raised. I/R group (I/R): The anesthesia and dissection of the renal vessels were the same as those in the sham operation group. Bilateral arteries and veins were rapidly clamped using hemostatic forceps, and ischemia lasted for 45 minutes. Previous studies reported that 45-minute bilateral renal ischemia induces reproducible moderate-to-severe renal injury in C57BL/6J mice, characterized by significant tubular necrosis and inflammatory cell infiltration.17,18 Then, the hemostatic forceps were released to restore blood flow to the kidneys, and the recovery of the kidneys was observed. QU administration group (I/R + QU): One week before the operation, QU (dissolved in 0.5% CMC-Na) was administered by gavage at a dose of 100 mg/kg/day. The anesthesia and surgical methods were the same as those in the I/R group. The sham operation group and the model group received the same volume of solvent (0.5% CMC-Na), and the sham operation group did not undergo ischemia. Other procedures were the same. All animals were sacrificed two days after the surgery. All mice were perfused with physiological saline through the heart, and the kidneys were harvested, frozen at -80°C, and immersed in a solution containing 4% formaldehyde. The 7-day pretreatment regimen (100 mg/kg/day) was adopted based on its common use in preclinical studies. 19 This dosing schedule achieves measurable plasma concentrations in vivo, 19 supporting its application for pharmacological studies.
Experiment 2
After adaptation to their environment before the surgery, the mice were divided into the I/R group (I/R+WT), the STING gene knockout group (I/R+KO), and the STING knockout with QU administration group (I/R+QU+KO).
Genotyping
The alkaline lysis method was employed to extract DNA from the mice. The toes or tails of 7-10-day-old neonatal mice were placed in tubes. Next, 100 μL of 50 mM NaOH was added to the samples and heated at 95°C in a metal bath for 30 minutes. Then, 30 μL of pH 7, 1 M Tris-HCl was added and mixed by vortexing for neutralization. Finally, the mixture was centrifuged at 15,000 rpm for 2 minutes at 4°C, and the supernatant was collected as the DNA solution. Subsequently, polymerase chain reaction (PCR) was conducted to amplify the sequence, and PCR amplification products were detected using agarose gel electrophoresis.
Pathological Staining
The selected tissue sections were placed in an oven at 60°C for 1 hour, followed by dewaxing and rehydration. Subsequently, hematoxylin-eosin staining and Masson’s trichrome staining were performed according to the instructions. Finally, the sections were dehydrated and mounted. Images were obtained under the bright field of a microscope.
Immunohistochemical Staining
The sectioned tissues were selected and placed in an oven at 60°C for 1 hour. Then, the subsequent steps were followed for dewaxing and rehydration. Antigen retrieval was conducted using sodium citrate buffer (0.01M; pH=6.0). The sections were rinsed with PBS three times for 5 minutes each. An appropriate amount of endogenous peroxidase blocker was added, and the samples were incubated at room temperature for 10 minutes. The sections were rinsed with PBS for 3 minutes, and then blocked with 5% donkey serum at room temperature for 30 minutes. After removing the blocking solution, the primary antibody was diluted with the blocking solution at a ratio of 1:200 and incubated at 4°C overnight. The next day, the sections were rinsed with PBS three times for 5 minutes each. Thereafter,100 μL or an appropriate amount of biotin-labeled goat anti-rabbit IgG polymer was added and incubated at room temperature for 10-15 minutes. The sections were rinsed with PBS three times for 3 minutes each. Subsequently,100 μL or an appropriate amount of horseradish peroxidase-labeled streptavidin working solution was added and incubated at room temperature for 10-15 minutes. The sections were rinsed with PBS three times for 3 minutes each. An appropriate amount of freshly prepared DAB solution was added and incubated at room temperature. The samples were rinsed with water and incubated with hematoxylin staining solution. The images were obtained under the bright field of a microscope.
Cell Culture
Rat renal tubular epithelial cells NRK-52E were purchased from Procell. The cells were cultured in DMEM medium containing 10% fetal bovine serum and 1% penicillin-streptomycin.
H2O2 Stimulation
Based on the established protocols described in the literature, a 100 µM concentration of H2O2 and a treatment duration of 30 minutes were selected for NRK-52E cell stimulation. Zhao et al reported that treating NRK-52E cells with 100 µM H2O2 for 0.5 h can effectively induce oxidative stress, evidenced by increased levels of reactive oxygen species and reduced cell viability, without excessive cell death. 20 This time point captures the acute phase of H2O2-induced signaling activation, including NF-κB phosphorylation and inhibitor of nuclear factor-κB α(IκB-α) degradation, which are the early events in the inflammatory cascade. 21 To assess inflammatory cytokine expression, the cells were treated with 100 µM H2O2 for 6 h, a time point previously shown to induce interleukin-1β (IL-1β) and tumor necrosis factor-α(TNF-α) expression in NRK-52E cells. 22 Therefore, for experiments evaluating inflammatory responses, NRK-52E cells were pretreated with QU (at indicated concentrations) for 1 h, followed by stimulation with 100 µM H2O2 for 30 min. The cells were then harvested for subsequent analyses.
Real-Time Quantitative PCR
Primers for qRT-PCR
Western Blotting
The cells were fully lysed using cell lysis buffer to extract proteins. After denaturation, the proteins were separated by SDS gel electrophoresis. Then, the proteins were transferred to an NC membrane through membrane transfer. After transferring, the membrane was immersed in a 5% skim milk solution prepared in TBST in an incubation box. The membrane was incubated at room temperature with gentle shaking for 1 hour to achieve blockade. The primary antibody (1:1000) was diluted in a 5% skim milk solution prepared in TBST (dilution ratio and dilution buffer were determined based on the instructions of the kits) and incubated overnight at 4°C on a shaker. The membrane was washed three times with TBST for 10 minutes each time. The secondary antibody was diluted to an appropriate concentration and incubated at room temperature with gentle shaking for 1 h. The membrane was washed three times with TBST for 10 minutes each. The results were detected using chemiluminescence. The AB solution (A solution: B solution = 1:1) was mixed in proportion and incubated in the luminescent solution at room temperature for 3 minutes. The residual luminescent solution was discarded, and the membrane was exposed for image acquisition.
Small Interfering RNA (siRNA) Knockdown of NF-κB
NF-κB siRNA (sense: 5′-CUGGAUGACAUCUUAAACUTT-3’; antisense: 5′-AUGAGGGCUACAUAUUUGGTT -3′) was purchased from GenePharma. The experiment was conducted following the operation steps provided by the manufacturer. When the target cells were ready for transfection, logarithmic phase cells were collected, digested, and counted. Cell density was adjusted, and 4×105 cells were seeded in each well of a 6-well plate and cultured overnight before transfection. The siRNA-Mate transfection reagent was placed at room temperature and gently mixed before use. Subsequently, 100 μL of OPTI-MEM was added to a sterile EP tube, and 10 pmol of siRNA was added to OPTI-MEM and mixed thoroughly. Then, 2 μL of siRNA-Mate transfection reagent was added to the tube and vortexed rapidly for 10 seconds to ensure complete mixing. The siRNA and transfection reagent were left at room temperature for 10 minutes to form a transfection complex. The medium in each well was replaced with 0.9 mL of a fresh medium, and the transfection complex was added and mixed well. The efficiency of protein silencing was detected 24 hours later.
Statistical Analysis
Data were analyzed using GraphPad Prism 9.0 statistical software and expressed as the standard error of the mean (SEM). Between-group differences were compared using an unpaired t-test (2 group results) or one-way ANOVA with multiple comparisons (Bonferroni’s correction) (>2 group results). P < 0.05 was deemed statistically significant.
Results
QU Alleviated Renal Pathological Changes Caused by I/R in Mice
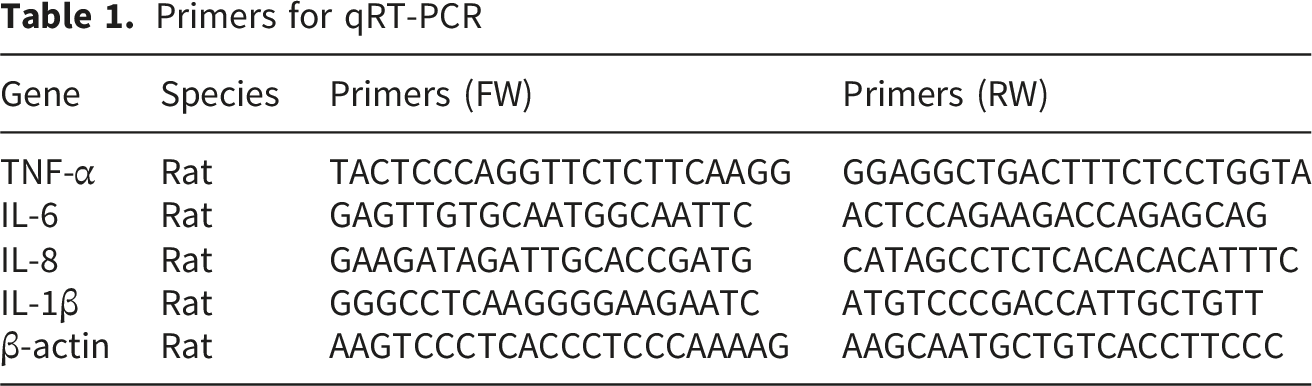
First, we explored the effect of QU on renal I/R injury. H&E staining indicated disordered renal tubules and glomeruli in mice subjected to I/R (Figure 1A). However, mice pretreated with QU one week before the surgery exhibited milder changes compared to mice not pretreated with QU. Similarly, Masson staining showed a large area of fibrotic-like lesions in the renal tissues of I/R mice, while fibrotic changes in the renal tissues of mice pretreated with QU one week before the operation were milder than those of I/R mice (Figure 1B and C). These results suggest that QU alleviated renal I/R injury in mice. QU alleviates renal pathological changes and inflammatory cell infiltration caused by ischemia-reperfusion in mice. (A) H&E staining and (B-C) Masson staining images and quantitative analysis of renal tissues after each treatment. (D-G) Immunohistochemical images and quantitative analysis of CD68 (D-E) and Ly6C (F-G) in renal tissues in each group. Mean ± SEM; n=6 in each group, ***P<0.001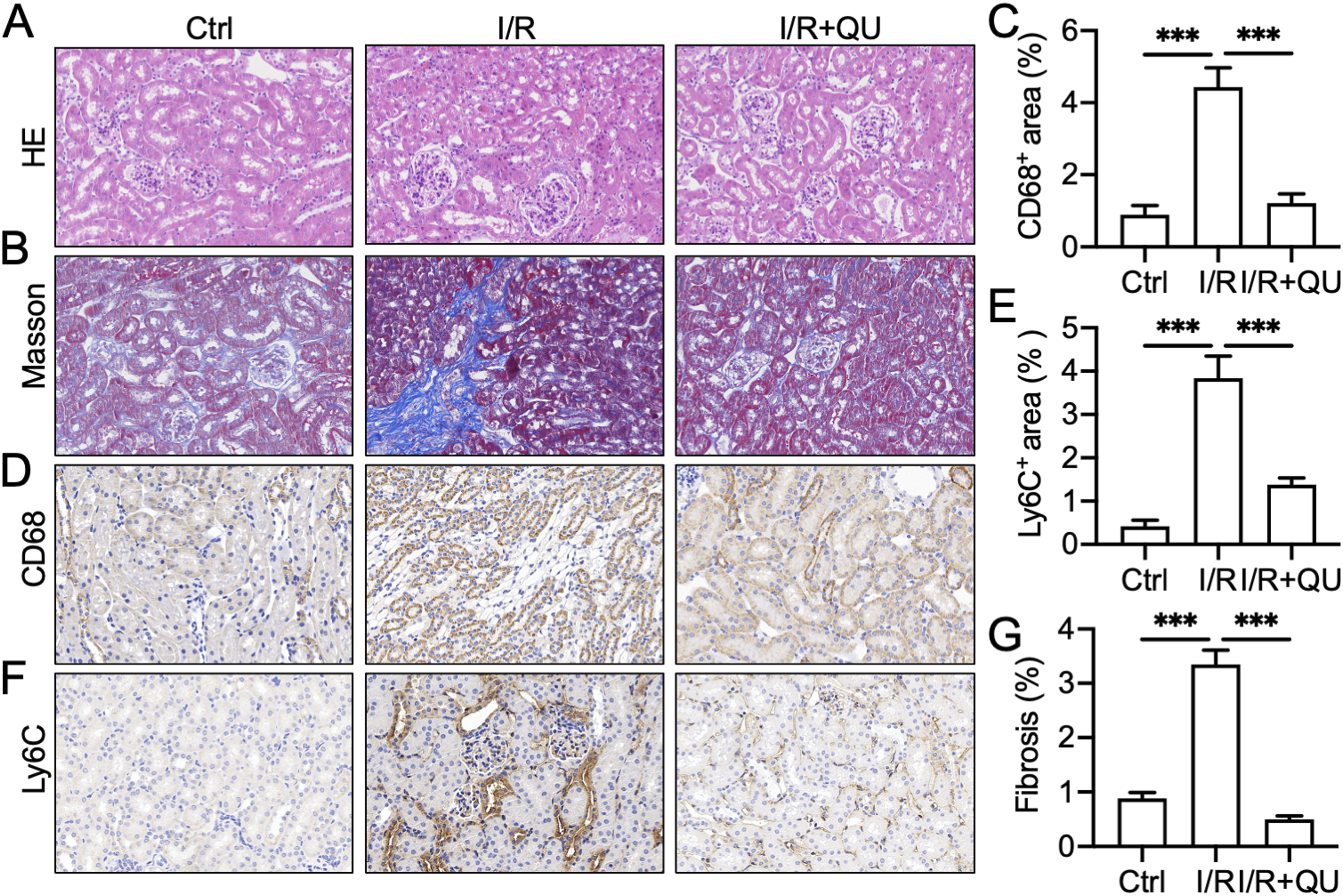
QU Alleviated Renal I/R-Induced Inflammatory Cell Infiltration and H2O2-Induced Inflammatory Responses
Many studies have shown that during renal I/R, imbalances in ion homeostasis, oxidative stress, disruption of mitochondrial outer membrane permeability, and inflammatory responses can lead to tissue damage.
23
Among them, the inflammatory response is one of the main factors that induce cell apoptosis and lead to I/R injury.
3
Compared to the sham operation group, an increased number of inflammatory cells, including monocytes and macrophages, infiltrated into the damaged tissue in I/R mice (Figure 1D–F). Additionally, we detected the expression of inflammatory factors in NRK-52E cells exposed to H2O2. RT-qPCR showed that compared to the control group, the mRNA levels of IL-1β, interleukin-6 (IL-6), interleukin-8 (IL-8), and TNF-α were significantly increased in NRK-52E cells exposed to H2O2. In contrast, QU dose-dependently reduced the mRNA levels of IL-1β, IL-6, IL-8, and TNF-α after H2O2 exposure (Figure 2A–D). The above results indicate that QU alleviated the inflammatory response caused by renal I/R in mice. QU alleviates H2O2-induced inflammatory responses. (A–D) The mRNA expression of IL-1β, IL-6, IL-8, and TNF-α with different treatments in NRK-52E cells incubated with H2O2. Mean ± SEM; n=3 in each group. ###P<0.001, vs DMSO group; *P<0.05, **P<0.01, ***P<0.001, ns, no significance, vs H2O2 group
QU Inhibited NF-κB-Mediated Inflammatory Responses in H2O2-Induced NRK-52E Cells
NF-κB is a transcription factor that binds to antioxidant response elements (AREs) and regulates the expression of genes linked to the clearance of oxidative toxins.
24
To investigate whether QU alleviates inflammation by inhibiting the NF-κB signaling pathway, NRK-52E cells were pretreated with QU for 1 hour and then incubated with H2O2 (100 μM) for 30 minutes. NF-κB phosphorylation and inhibitor of κB-α (IκB-α) degradation were inhibited by QU (Figure 3A). To further clarify whether the anti-inflammatory effect of QU depends on NF-κB, we analyzed the inflammatory response in H2O2-induced cells using small interfering RNA (siRNA) targeting NF-κB p65. We conducted NF-κB p65 siRNA transfection to downregulate the expression of NF-κB in NRK-52E cells (Figure 3B). NF-κB siRNA significantly reversed H2O2-induced increase in the mRNA levels of IL-1β, IL-6, IL-8, and TNF-α. The anti-inflammatory properties of QU were attenuated when NF-κB was knocked down in NRK-52E cells (Figure 3C–F). In conclusion, QU alleviated H2O2-induced inflammatory responses by inhibiting the NF-κB signaling pathway. QU inhibited NF-κB-mediated inflammatory responses in H2O2-induced NRK-52E cells. (A) Western blot analysis of the expression of p-NF-κB and IκB-α in NRK-52E cells incubated with H2O2. (B) Western blot analysis of the expression of NF-κB in NRK-52E cells incubated with siRNA-NF-κB. (A–D) The mRNA expression of IL-1β, IL-6, IL-8, and TNF-α with different treatments in NRK-52E cells incubated with H2O2. Mean ± SEM; n=3 in each group. ##P, ###P<0.001, vs Ctrl group; *P<0.05, ***P<0.001, vs H2O2 group; ns, no significance, vs H2O2+siNF-κB group
QU Alleviated Renal I/R-Induced Inflammatory Response Through the STING/NF-κB Pathway
Reportedly, STING plays a crucial role in I/R injury in different organs. In the STING signaling pathway, NF-κB constitutes the primary downstream molecule that induces the inflammatory response.25,26 Therefore, we determined whether QU inhibited renal I/R-induced NF-κB activation through STING. We detected STING expression and NF-κB activation in the renal tissues of mice. Compared to the sham operation group, NF-κB phosphorylation was enhanced in the renal tissues of I/R mice, while NF-κB phosphorylation was decreased after treatment with QU (Figure 4A). Next, we used STING gene knockout mice to explore the effect of QU. As shown in Figure 4B, homozygous STING gene knockout mice bred were used for subsequent experiments. H&E and Masson staining indicated that STING gene knockout mitigated renal structural damage and collagen accumulation in I/R mice; however, QU did not exhibit protective effects in STING gene knockout mice after renal I/R (Figure 4C–E). Furthermore, STING gene knockout inhibited monocyte and macrophage infiltration in I/R mice (Figure 4F–I), and QU failed to exert the anti-inflammatory effects in STING gene knockout mice. These results indicated that QU alleviated renal I/R-induced inflammatory response by inhibiting STING. QU alleviates renal ischemia-reperfusion-induced inflammatory response through STING-NF-κB pathway. (A) Western blot analysis of the expression of STING, p-NF-κB, and NF-κB in renal tissues. (B) Genotyping of STING knockout mice. He, Heterozygote; KO, STING knockout Homozygote; WT, wildtype. (C) H&E staining and (D-E) Masson staining images and quantitative analysis of renal tissues after each treatment. (F–I) Immunohistochemical images and quantitative analysis of CD68 (F–G) and Ly6c (H–I) in renal tissues in each group. Mean ± SEM; n=6 in each group, ***P<0.001, ns, no significance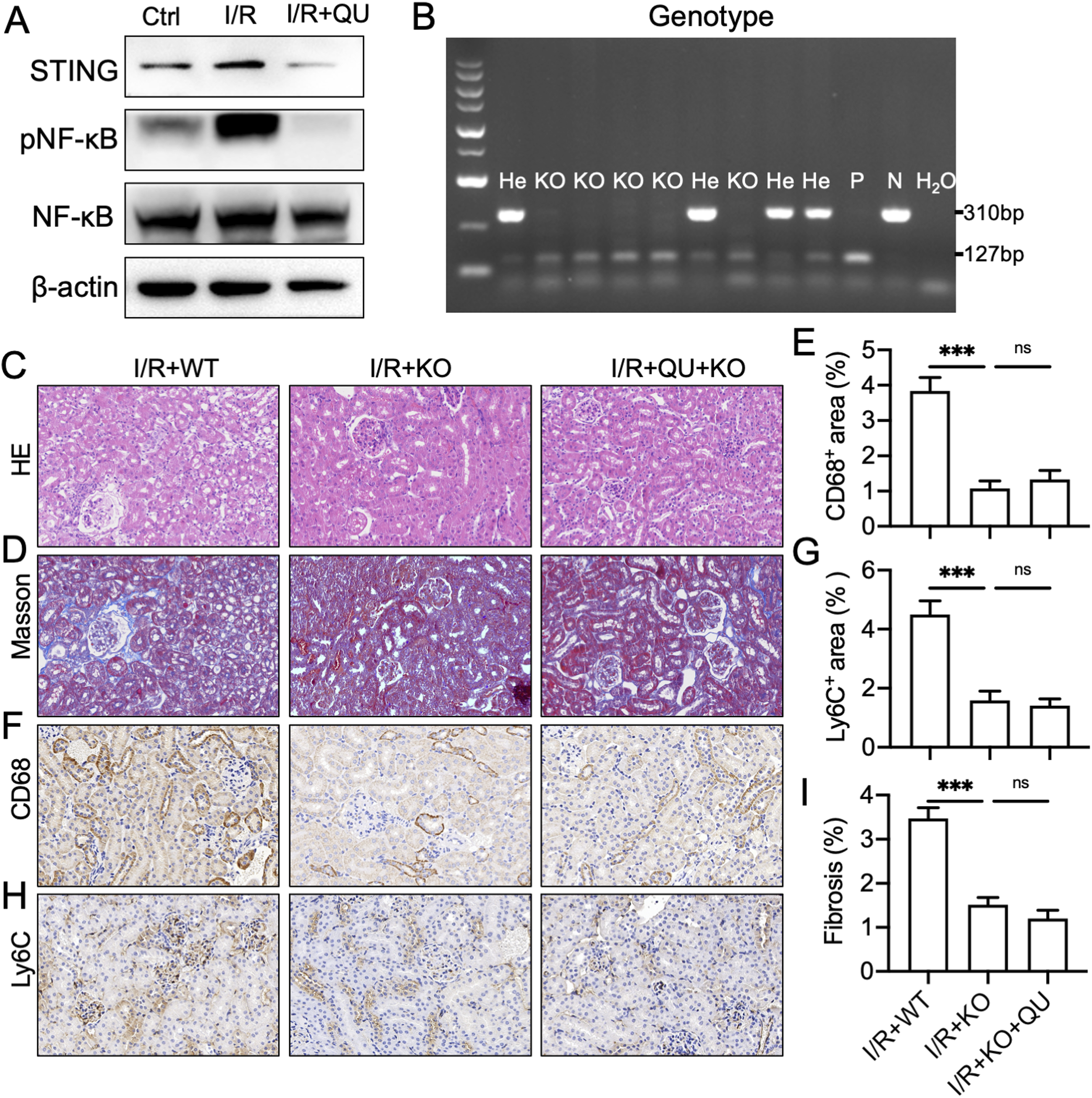
Discussion
I/R injury refers to the condition where tissues or organs experience transient ischemia due to vascular occlusion, and the damage intensifies after the restoration of the blood supply. I/R injury is a common pathophysiological finding in clinical settings. 27 I/R often occurs secondary to kidney transplantation, acute trauma, shock, and cardiac extracorporeal circulation surgery. I/R is an important factor leading to acute kidney injury and renal failure, with extremely high mortality and poor prognosis.1,2 Since the pathogenesis of I/R has not been fully elucidated, there is still a lack of effective drugs and treatment methods in clinical practice. Therefore, identification of effective drugs against renal I/R injury and discovering novel therapeutic targets are of significant scientific and clinical value.
Natural components from natural sources, including peptides or natural products, are widely explored as new drugs for disease treatment. In renal I/R injury, the mitochondria-targeted peptide SS-31 (elamipretide) has shown significant nephroprotective effects by stabilizing cardiolipin and preserving mitochondrial function.28,29 Additionally, bioactive peptides, such as the cyclic heptapeptide FZ1, andersonin-W1, and Cy RL-QN15 and so on, exerts the effects in tissue repair and inflammation regulation.25,30-32 Among them, apelin, atrial natriuretic peptide (ANP),33,34 and brain natriuretic peptide (BNP), have been investigated for their effects on renal I/R injury.35-37 Similar to peptides, natural products have gradually been found to possess various biological activities. QU is a natural flavonoid compound widely present in plants. It possesses various biological activities, such as expectorant, antitussive, lipid-lowering, weight-reducing, antioxidant, hypoglycemic, anti-inflammatory, anti-tumor, antibacterial, and cardioprotective properties.38-41 Other studies have shown that QU can alleviate I/R injury in the liver, brain, and heart.42-44 Histopathological analysis indicated that tubular and glomerular structures of renal tissues were significantly damaged and collagen accumulation was increased in the I/R group. However, the pathological changes and fibrotic damage were significantly alleviated in the kidneys of mice in the QU administration group. These results suggest that QU may be a potential drug for treating renal I/R injury. The mechanism underlying I/R injury has not been fully elucidated so far, and the importance of the inflammatory response in I/R injury has been increasingly reported. 45 Studies have shown that QU possesses a potent anti-inflammatory effect46,47; thus, this study focused on the protective effects of QU on renal I/R-induced inflammatory response. In this study, inflammatory cell infiltration was significantly observed after renal I/R injury; however, the number of infiltrating inflammatory cells decreased in mice treated with QU. Moreover, we found that QU downregulated H2O2-induced increase in the expression of inflammatory factors in vitro. These results suggest that QU may inhibit the inflammatory response subsequent to I/R.
Numerous studies have reported that NF-κB inhibition can alleviate renal I/R injury in vivo.48-50 Our results showed that QU inhibits NF-κB phosphorylation induced by renal I/R and H2O2 in vivo and in vitro. Moreover, after silencing NF-κB expression, QU did not affect inflammatory factor expression subsequent to H2O2. Further experiments indicated that QU also inhibits the expression of STING, and the protective effect of QU against renal I/R injury depends on the regulation of the STING/NF-κB signaling pathway. In the present study, we employed a complementary in vitro–in vivo strategy combining rat NRK-52E renal tubular epithelial cells and a mouse model of renal I/R injury to systematically investigate the nephroprotective effects of QU. This cross-species, orthogonal experimental design not only facilitates in-depth mechanistic exploration but also improves the translational potential of our findings.
Limitations
This study had several limitations. First, the sample size for animal experiments was determined based on previous studies and established practices in the field 51 rather than formal statistical power analysis. This may affect the statistical power and generalizability of our findings. Second, while our results demonstrated that QU alleviates renal I/R injury by inhibiting the STING/NF-κB pathway, the molecular mechanism through which QU binds to and inhibits STING remains insufficiently explored. Specifically, our study lacks structural and theoretical evidence regarding the structure-activity relationship between QU and STING, which limits our ability to optimize the chemical structure of QU and enhance its inhibitory efficacy against STING. Future studies employing molecular docking, molecular dynamics simulations, and structure-based drug design are warranted to elucidate the precise binding mode of QU to STING and identify QU derivatives with improved potency and selectivity.
Conclusion
Our study found that QU can effectively alleviate renal I/R injury and inhibit the STING/NF-κB-mediated inflammatory response. This indicates that QU can be a candidate drug for treating renal I/R injury, and targeting the STING-NF-κB-mediated inflammatory response is a feasible therapeutic strategy for treating renal I/R injury.
Footnotes
Acknowledgement
We are grateful to Lishui People’s Hospital for her professional technical support and EditSprings for language editing.
Author Contributions
Funding
This study was supported by the Science and Technology Planning Project of Lishui City (No. 2022GYX58), a Public Welfare Technology Application Research Project, funded by Lishui Municipal Bureau of Science and Technology.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data are included within the article or Supplementary Information or available from the authors on request.
