Abstract
Objective
This study compared the efficacy of different doses of filgotinib (100 mg vs 200 mg) in the treatment of rheumatoid arthritis (RA).
Methods
We conducted a systematic review of 4 electronic databases up to January 2025. Primary outcome measures included ACR20, ACR50, ACR70.
Results
Five RCTs were included in this analysis, in which 2191 patients with RA were evaluated, 100 mg filgotinib was noninferior to 200 mg filgotinib in the efficacy of ACR20 (P = 0.33, RR = 0.96). However, 100 mg filgotinib was inferior to 200 mg filgotinib for RA when using ACR50 (P = 0.04, RR = 0.92) and ACR70 (P = 0.02, RR = 0.86) as outcome measures. At 24 weeks, 100 mg filgotinib was inferior to 200 mg filgotinib at ACR50 (P = 0.07, RR = 0.84), and there was no significant difference between 100 mg and 200 mg filgotinib at ACR70 (P = 0.54, RR =0.88). The 52-week results showed 100 mg filgotinib was inferior to 200 mg filgotinib at ACR70 (P = 0.01, RR = 0.86).
Conclusion
Reduced dose filgotinib (100 mg) may be used as a potential treatment option for RA when treatment is required.

Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease with a global incidence of approximately 0.5% to 1%. 1 It is estimated that there are more than 5 million patients worldwide. 1 The treatment of RA has evolved significantly, transitioning from traditional nonsteroidal anti-inflammatory drugs (NSAIDs) and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) to the era of biological agents and targeted therapies. 1 This progress has notably enhanced the clinical remission rate and quality of life for patients. 1 Among the emerging treatments, Janus kinase (JAK) inhibitors are currently among the most promising drugs.
Filgotinib is an oral, highly selective JAK1 inhibitor that has demonstrated the ability to inhibit JAK1 expression. 2 It is primarily indicated for adult patients with moderate-to-severe active RA who have had an inadequate response or intolerance to traditional synthetic or biologic disease-modifying antirheumatic drugs. 2 By selectively inhibiting the kinase activity of JAK1 and blocking intracellular inflammatory signaling pathways, filgotinib alleviates the symptoms and signs of RA. 2 The choice of dosage is crucial for both efficacy and safety. The standard dose of filgotinib is 200 mg/day. However, for elderly patients (≥65 years) or those with renal insufficiency, a reduced dose of 100 mg/day is recommended to mitigate the risk of adverse events such as infections and malignancies. 3 Although filgotinib is effective, it can also cause side effects. 4
Currently, there are experimental comparisons of the efficacy between 100 mg and 200 mg doses of filgotinib. However, no relevant meta-analysis has been conducted to date. The aim of this study is to perform a meta-analysis to explore the differences in efficacy between these two filgotinib doses in the treatment of RA.
Methodology
Literature Retrieval Strategy
From inception until January 2025, this research searched 4 databases: PubMed, Cochrane Library, scientific Web, and Embase until January 2025. The research used a combination of Medical Subject Headings and free-text terms in English, including “Filgotinib” “JAK kinase inhibitor” and “Rheumatoid Arthritis”. The search strategy was structured as follows: (((Janus Kinase Inhibitor) OR (JAKi)) OR (JAK Inhibitor)) OR (Filgotinib)) AND ((RA) OR (rheumatoid arthritis). Full-text filters were used for the search and manual screening was performed to select relevant literature. References to articles were reviewed to ensure that no omissions were made.
Use Endnote X7 (Thomson Reuters, Toronto, ON, Canada) to manage and remove duplicate records that exist in the retrieved records. This study followed transparent reporting guidelines for systematic reviews and meta-analyses (PRISMA), and we focused on identifying randomized controlled trials (RCTs) comparing 100 mg filgotinib with 200 mg filgotinib for RA. 5 This study is registered with PROSPERO, an international database for registered systematic reviews in health and social care. Detailed meta-analysis protocol can be accessed via PROSPERO at ID: CRD420251017167.
Literature screening was performed independently by two investigators according to prespecified inclusion and exclusion criteria. Discrepancies were resolved through discussions with third-party arbitrators when necessary.
Inclusion and Exclusion Criteria
The criteria for study inclusion in this meta-analysis were based on the authors’ prespecified PICOS (participant, intervention, comparison, outcomes, study design) framework. The criteria are as follows:
Participant (P): The study had to include participants with a diagnosis of RA, regardless of race or nationality, who were 18 years of age or older.
Intervention (I): Participants in the intervention group had to receive filgotinib at a dose of 100 mg.
Comparison (C): The control group had to receive 200 mg of filgotinib.
Outcomes (O): The primary outcome measures included, but were not limited to, American College of Rheumatology20 (ACR20), American College of Rheumatology50 (ACR50), American College of Rheumatology70 (ACR70), the simplified disease activity index (SDAI), the clinical disease activity index (CDAI), and functional assessment of chronic illness therapy (FACIT).
Study design (S): The RCT was included in this study.
Exclusion criteria: To improve the relevance and generalizability of the meta-analysis, only the most comprehensive or recent studies were included in this meta-analysis. Non-english language literature, studies without full text or relevant data were excluded.
Data Extraction and Outcomes of Interest
Data for the meta-analysis were extracted according to the authors’ pre-defined structured protocol. Variables extracted from each study included country, authors, year of publication, duration of follow-up, treatment options and efficacy. The efficacy outcomes considered were ACR20, ACR50, ACR70, SDAI, CDAI, FACIT.
ACR20 is defined as a reduction of at least 20% in both swollen and tender joint counts, accompanied by a minimum 20% improvement in three or more of the following five core measures: global assessment of disease activity (e.g., Visual Analogue Scale (VAS)), global disease activity assessed by healthcare providers (e.g., Health Services Administration), patient pain assessment (e.g., VAS), functional disability (e.g., provided by health assistance agencies), and acute phase reactants (erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP)). 6
ACR50 denotes a reduction of at least 50% in swollen and tender joint counts, along with a 50% improvement in at least three of the five core measures.
ACR70 signifies a reduction of at least 70% in swollen and tender joint counts, with a corresponding 70% improvement in at least three of the five core measures.
CDAI is utilized to evaluate the clinical disease activity in RA. This index includes: (1) number of tender joints out of 28; (2) number of swollen joints out of 28; (3) patient’s assessment of disease activity on a scale from 0 to 10; (4) physician’s global assessment of disease activity on a scale from 0 to 10. 7
SDAI extends the CDAI by incorporating the serum hypersensitive CRP level (mg/L) and is employed to assess treatment efficacy in RA. It comprises the number of tender joints out of 28, number of swollen joints out of 28, patient’s assessment of disease activity (0-10 points), physician’s overall assessment (0-10 points), and serum hypersensitive CRP level. 7
FACIT is a scale system used to assess the treatment function of patients with Chronic Illness. It is mainly used to assess the fatigue level of patients and its impact on daily life. The FACIT-F scale uses a Likert 4-point scale, with each item scored from 0 (very tired) to 4 (not tired at all). 1
Literature Quality Evaluation
The quality of the included literature was assessed using the bias assessment tool recommended by the Cochrane Handbook for Systematic Reviews of interventions, version 5.3.5. Several areas covered included, methods of randomization, allocation concealment, blinding of participants, personnel and outcome assessors, completeness of outcome data, selective reporting, and other potential biases. Each domain was categorized according to “Low risk of bias” “High risk of bias” or “Unclear risk of bias”.
Statistical Analysis
Statistical analysis was performed using Review Manager (RevMan, Cochrane Collaboration, version 5.3.5). Dichotomous outcomes are presented as risk ratio (RR) with 95% confidence intervals (CI), continuous variables use weighted mean difference (WMD). Study heterogeneity was assessed using the chi-square test (χ2) and quantified with the I2 statistic. Thresholds of P < 0.05 and I2 > 50% were interpreted as significant heterogeneity, whereas P ≥ 0.05 and I2 ≤50% indicated low heterogeneity. 8 According to the heterogeneity size, a random effects model was used when there is significant heterogeneity and a fixed effect model was used when the heterogeneity is low.
RR > 1 (the 100mg filgotinib is used for the experimental group) indicates outcomes favoring the experimental group, whereas RR < 1 (the 100mg filgotinib is used for the experimental group) indicates outcomes favoring the control group.
Publication bias was assessed using funnel plots. All statistical tests were two-sided and the significance threshold was P < 0.05.
Results
Study Selection
A search of PubMed, the Cochrane Library, Web of Science, and Embase yielded 11871 records. After removing duplicate records, 9213 records remain. Screening by title and abstract excluded 9180 records, leaving 33 articles for full-text assessment. Of these, 17 were excluded for various reasons: 4 were updated RCTs, 3 missed data of interest, 4 could not obtain the full text, and 17 did not meet the inclusion criteria. Ultimately, 5 studies were included in the analysis.9-13 The PRISMA flow diagram depicts the detailed selection process (Figure 1). Flow diagram of the selection process
Study Characteristics
Characteristics of all the Studies Included in the Meta-Analysis

RCT, randomized controlled trial.
Quality Assessment of Included Studies
The quality of the included studies was assessed using the bias assessment tool recommended by the Cochrane Handbook for Systematic Reviews of interventions, version 5.3.5 (SFig. 1 and SFig. 2). Because of the well-documented patient randomization method and random sequence generation, the risk of selection bias was considered low. Adequate descriptions in the study help us identify blinding methods that minimize performance and detection bias. The risk of attrition and reporting bias is also considered low because the criteria for measuring results are clear and data are reported comprehensively. However, because there is insufficient other information to make a definitive assessment, the risk of other potential biases remains unclear.
Meta-Analysis and Subgroup Analyses
ACR Analysis
A total of 5 studies reported on the ACR20 response rate. The pooled analysis revealed that the 100 mg filgotinib group was noninferior to the 200 mg filgotinib group (RR = 0.96, = 0.33). Subgroup analyses stratified by follow-up duration showed no significant differences between the two groups at either 24 weeks (RR = 0.92, P = 0.55) or 52 weeks (RR = 0.96, P = 0.18) (Figure 2). Forest plot of ACR20’s differences in efficacy of 100 mg filgotinib and 200 mg filgotinib in RA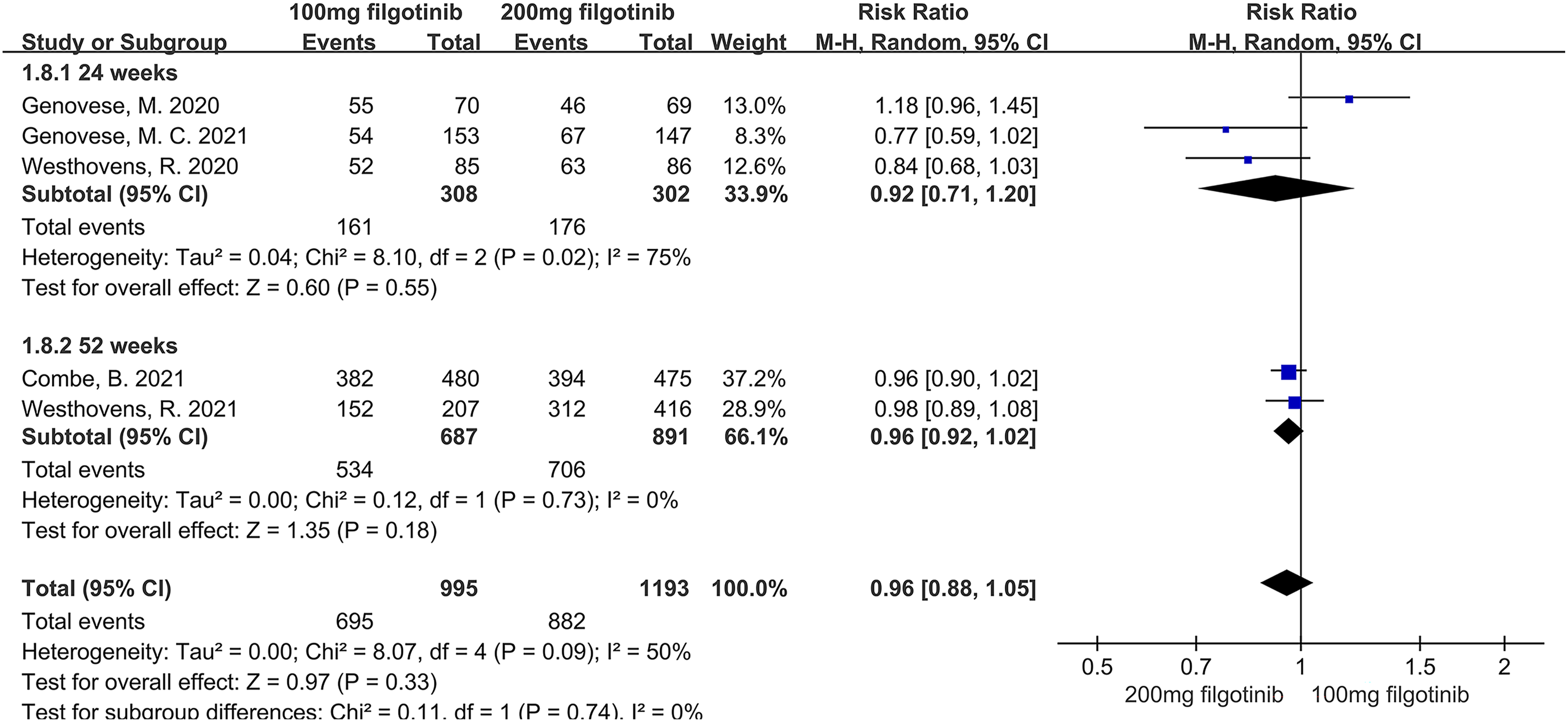
When ACR50 was used as the outcome measure, the pooled analysis indicated that the 100 mg filgotinib group was inferior to the 200 mg filgotinib group (RR = 0.92, P = 0.04). Subgroup analyses by follow-up duration revealed a trend toward lower response rates in the 100 mg group compared to the 200 mg group at 24 weeks (RR = 0.84, P = 0.07), although this difference did not reach statistical significance. At 52 weeks, no significant difference was observed between the two groups (RR = 0.95, P = 0.19) (Figure 3). Forest plot of ACR50’s differences in efficacy of 100 mg filgotinib and 200 mg filgotinib in RA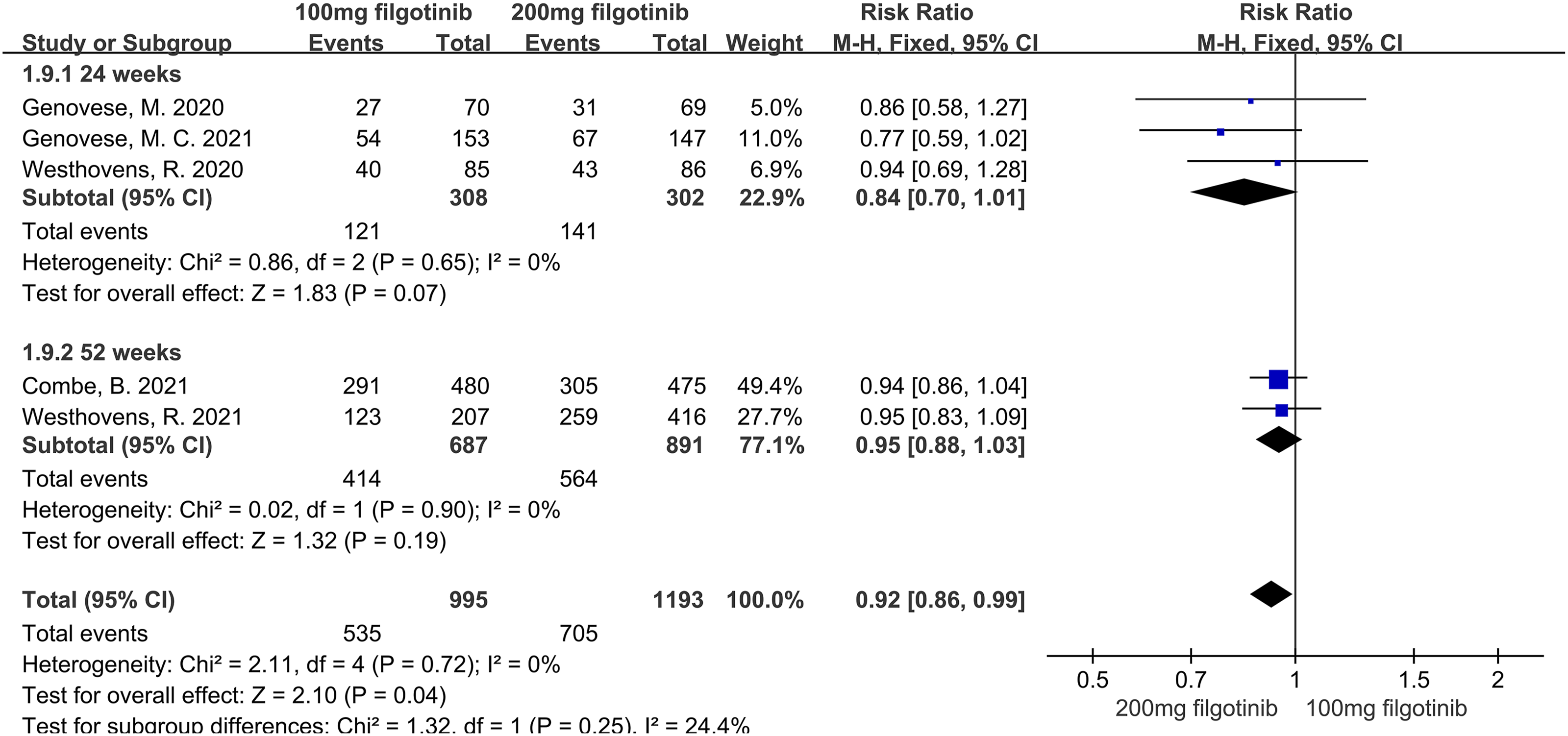
For ACR70, the pooled analysis showed that the 100 mg filgotinib group had a lower response rate compared to the 200 mg filgotinib group (RR = 0.86, P = 0.02). Subgroup analyses demonstrated no significant difference between the groups at 24 weeks (RR = 0.88, P = 0.54). However, at 52 weeks, the 200 mg filgotinib group was significantly superior to the 100 mg filgotinib group (RR = 0.86, P = 0.01) (Figure 4). Forest plot of ACR70’s differences in efficacy of 100 mg filgotinib and 200 mg filgotinib in RA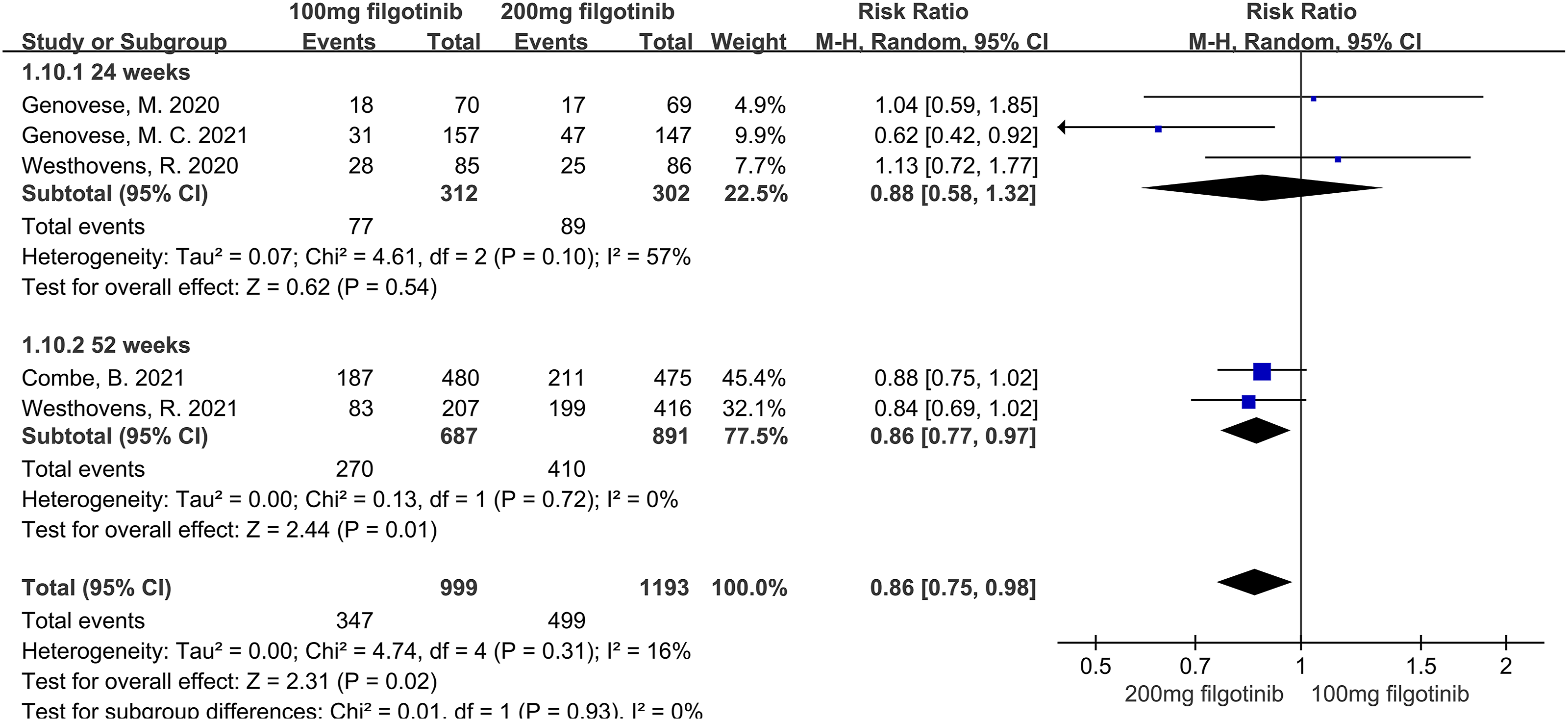
When using the clinical disease activity index (CDAI) as the outcome measure, the pooled analysis showed noninferiority between the 100 mg and 200 mg filgotinib groups (WMD = 0.92, P = 0.24) (SFig. 3). Similarly, when the simplified disease activity index (SDAI) was used, the 100 mg filgotinib group was noninferior to the 200 mg filgotinib group (WMD = 1.04, P = 0.21) (SFig. 4). Additionally, when the functional assessment of chronic illness therapy (FACIT) was used as an outcome measure, the 100 mg filgotinib group was noninferior to the 200 mg filgotinib group (WMD = -0.89, P = 0.18) (SFig. 5).
Publication Bias
For ACR20, ACR50 and ACR70, the funnel plots are drawn and the publication bias is retrieved, all funnel plots are roughly symmetric with no significant publication bias. See SFig. 6, SFig. 7, and SFig. 8 for specific funnel charts.
Discussion
In this study, 5 RCTs were included to compare the efficacy of 100 mg filgotinib and 200 mg filgotinib in the treatment of rheumatoid arthritis (RA). 1 The results demonstrated no significant difference in efficacy between the 100 mg and 200 mg doses based on ACR20, CDAI, SDAI, and FACIT outcomes. However, 200 mg filgotinib exhibited superior efficacy in achieving ACR50 and ACR70.
Filgotinib exerts its therapeutic effects by selectively inhibiting JAK1, thereby disrupting the JAK (Janus tyrosine kinase) - STAT (signal transducer and activator of transcription) signaling pathway, which is crucial for reducing inflammatory responses and immune-mediated tissue damage. 14 The specific mechanisms are as follows: 1. Inhibition of the JAK1 Signaling Pathway: JAK1 is a key member of the JAK family and is involved in the signaling of multiple pro-inflammatory cytokines, including IL-6, IL-12, IL-23, and IFN-γ. Filgotinib reduces inflammation by inhibiting JAK1 and blocking the signaling of these cytokines. 15 2. Regulation of Cytokine Release: Filgotinib decreases the production and release of pro-inflammatory cytokines, such as TNF-α and IL-6. This reduction in cytokine levels alleviates inflammation in joint tissues and relieves joint pain and swelling. 16 3. Amelioration of Structural Joint Injury: By inhibiting inflammatory signaling pathways, filgotinib slows down the destruction of articular cartilage and bone, thereby reducing the risk of structural joint damage. 17
In the context of dose reduction, the lack of significant differences between 100 mg and 200 mg filgotinib for ACR20, CDAI, SDAI, and FACIT may be attributed to the relatively low treatment thresholds of these measures. With a lower dose of filgotinib, comparable efficacy can be achieved across different treatment doses, such as 100 mg and 200 mg. 12 This phenomenon has also been observed with other JAK inhibitors, such as tofacitinib, baricitinib, and upadacitinib. A meta-analysis showed no significant difference between 5 mg and 10 mg doses of tofacitinib at ACR20. 18
For ACR50, 200 mg filgotinib demonstrated superior overall efficacy compared to 100 mg filgotinib in the treatment of RA. This difference was more pronounced at 24 weeks and diminished at 52 weeks, suggesting that lower doses require more time to compensate for efficacy differences. Low-dose filgotinib activates a limited number of JAK-STAT signaling pathways over a limited period, resulting in the release of a limited number of cytokines and failing to achieve substantial improvements in joint injury. In contrast, high-dose filgotinib can rapidly activate a large number of JAK-STAT signaling pathways and release a large number of cytokines, thereby improving joint injury and meeting the requirements of ACR50 in a shorter time. 19 Low-dose filgotinib does not reach peak efficacy in the short term, while high-dose filgotinib is more likely to reach saturation than low-dose filgotinib. This suggests that the superiority of 200 mg filgotinib diminishes with prolonged treatment. 1 At week 52, there was no longer a significant difference between 200 mg and 100 mg filgotinib in the treatment of RA. While the efficacy of filgotinib is positively correlated with dose, doubling the dose (e.g., from 100 mg to 200 mg) does not significantly improve efficacy. 10 This may be related to its pharmacokinetic properties. Pharmacokinetic studies of filgotinib have shown that plasma concentrations at both 100 mg and 200 mg achieve effective treatment levels. 2 Doubling the dose increases exposure, but the diminishing returns in efficacy suggest that the 100 mg dose may be close to a saturation point. 1
When using ACR70 as the outcome measure, 200 mg filgotinib was superior to 100 mg filgotinib in the treatment of RA. There was no significant difference between 100 mg and 200 mg filgotinib at 24 weeks, likely because the requirement for ACR70 is very high, and neither dose could meet this requirement in a short period of time. 20 At 52 weeks, however, the effects of high-dose filgotinib began to emerge. With the activation of a large number of JAK1 signaling pathways, the body released sufficient cytokines to significantly improve joint damage, making 200 mg filgotinib superior to 100 mg filgotinib. 2
Therefore, when using a lower dose, extending the duration of treatment is necessary to improve efficacy. However, a high dose of 200 mg filgotinib is still required to meet higher therapeutic demands. Low-dose 100 mg filgotinib is a suitable option for RA when clinical requirements are modest or when there is intolerance to filgotinib.
The strength of this study lies in its systematic comparison of the efficacy of 100 mg filgotinib with that of 200 mg filgotinib in the treatment of RA, supported by subgroup analyses. It was also found that 200 mg filgotinib was superior to 100 mg filgotinib with ACR50 as the outcome measure, with a larger difference observed at 24 weeks and a smaller difference at 52 weeks. With ACR70 as the index, 200 mg filgotinib was superior to 100 mg filgotinib, but this difference was not significant at 24 weeks and became more pronounced at 52 weeks. Additionally, because this was a dose analysis of a single agent, the heterogeneity of this study was low, and the credibility is high. This meta-analysis has certain limitations, firstly, the limited number of RCTs included, which may reduce the statistical robustness of the combined effect amount. Secondly, a small number of studies limit our ability to conduct in-depth subgroup analyses to explore the sources of heterogeneity, such as the influence of different population characteristics on the filgotinib effect response of different doses, and the accurate inference of the dose-effect relationship. Furthermore, some individual data on the dose-renal function relationship is not available due to differences in the original study design, which may affect the judgment on the optimal dose choice for specific populations, such as those with mild renal function impairment. Finally, rheumatoid arthritis generally requires long-term medication, and the follow-up time included in this study was relatively short, unable to provide definitive conclusions on the long-term safety of different doses of filgotinib (such as major cardiovascular adverse events, malignancy incidence). Therefore, future long-term studies based on larger samples or real-world data are needed to further validate the above conclusions.
Conclusion
When treating RA, a lower dose of filgotinib (100 mg) requires a longer duration of treatment to achieve optimal efficacy. However, a higher dose of filgotinib (200 mg) is still necessary to meet more stringent therapeutic goals. Nevertheless, when clinical requirements are modest or when patients experience intolerance to filgotinib, a low dose of 100 mg filgotinib remains a viable and suitable option for managing RA.
Supplemental Material
Supplemental material - Comparison of the efficacy of different doses of filgotinib in rheumatoid arthritis: a systematic review and meta-analysis based on the RCTs
Supplemental material for Comparison of the efficacy of different doses of filgotinib in rheumatoid arthritis: a systematic review and meta-analysis based on the RCTs by Xia Ye, Sidong Zhu, Dongying Wang, Lifeng Hu, Yudie Li, Qingping Wu, Jingqi Chen, Bangsheng Chen in Dose-Response.
Supplemental Material
Supplemental material - Comparison of the efficacy of different doses of filgotinib in rheumatoid arthritis: a systematic review and meta-analysis based on the RCTs
Supplemental material for Comparison of the efficacy of different doses of filgotinib in rheumatoid arthritis: a systematic review and meta-analysis based on the RCTs by Xia Ye, Sidong Zhu, Dongying Wang, Lifeng Hu, Yudie Li, Qingping Wu, Jingqi Chen, Bangsheng Chen in Dose-Response.
Footnotes
Acknowledgments
We extend our gratitude to all the researchers who provided essential data for this study.
Consent to Participate
This paper is based on research conducted using global databases and did not involve direct human participation.
Author Contributions
All authors had read and approved the manuscript. Xia Ye and Sidong Zhu writing manuscript; Dongying Wang, Lifeng Hu and Yudie Li performing procedures and data analysis; Qingping Wu and Jingqi Chen contribution to writing the manuscript; Bangsheng Chen contribution to drafting conception and design.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ningbo Natural Science Foundation (No. 2022J039).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets supporting the conclusions of this article are included within the article itself. For additional detailed data regarding this study, please contact the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
